

高等学校化学学报 ›› 2023, Vol. 44 ›› Issue (3): 20220346.doi: 10.7503/cjcu20220346
收稿日期:2022-05-16
出版日期:2023-03-10
发布日期:2023-03-14
通讯作者:
张艳
E-mail:njuzy@nju.edu.cn
基金资助:
KONG Hao, XU Feiyang, WANG Yixiang, ZHANG Yan( )
)
Received:2022-05-16
Online:2023-03-10
Published:2023-03-14
Contact:
ZHANG Yan
E-mail:njuzy@nju.edu.cn
Supported by:摘要:
CRISPR技术是目前基因编辑领域的一大研究热点, 已在疾病治疗、 作物改良等领域得到广泛的应用, CRISPR-Cas9系统是其中研究最为深入的一种类型. 如何降低Cas9/sgRNA复合物在细胞内作用的脱靶性是CRISPR-Cas9技术发展面临的主要挑战之一. 利用光或活性小分子诱发的小分子化学反应工具构建CRISPR-Cas9功能调控体系, 通过对sgRNA, Cas9或Cas9/sgRNA复合物的功能进行调控, 可以在细胞乃至活体水平上一定程度实现对CRISPR-Cas9作用的时间或空间特异性的操纵, 大大降低非特异性基因编辑作用发生的概率, 同时小分子对原体系的干扰较小, 因此小分子化学反应逐渐成为操纵CRISPR-Cas9体系的一种重要的研究工具. 本文总结和介绍了小分子反应工具用于CRISPR-Cas9功能调控系统构建的主要研究进展, 并对其未来的发展进行了展望.
中图分类号:
TrendMD:
孔好, 徐菲洋, 王依香, 张艳. 基于小分子化学反应工具构建CRISPR-Cas9功能调控体系的研究进展. 高等学校化学学报, 2023, 44(3): 20220346.
KONG Hao, XU Feiyang, WANG Yixiang, ZHANG Yan. Research Progress of CRISPR-Cas9 Functional Regulation System Based on Small Molecule Reaction Tools. Chem. J. Chinese Universities, 2023, 44(3): 20220346.
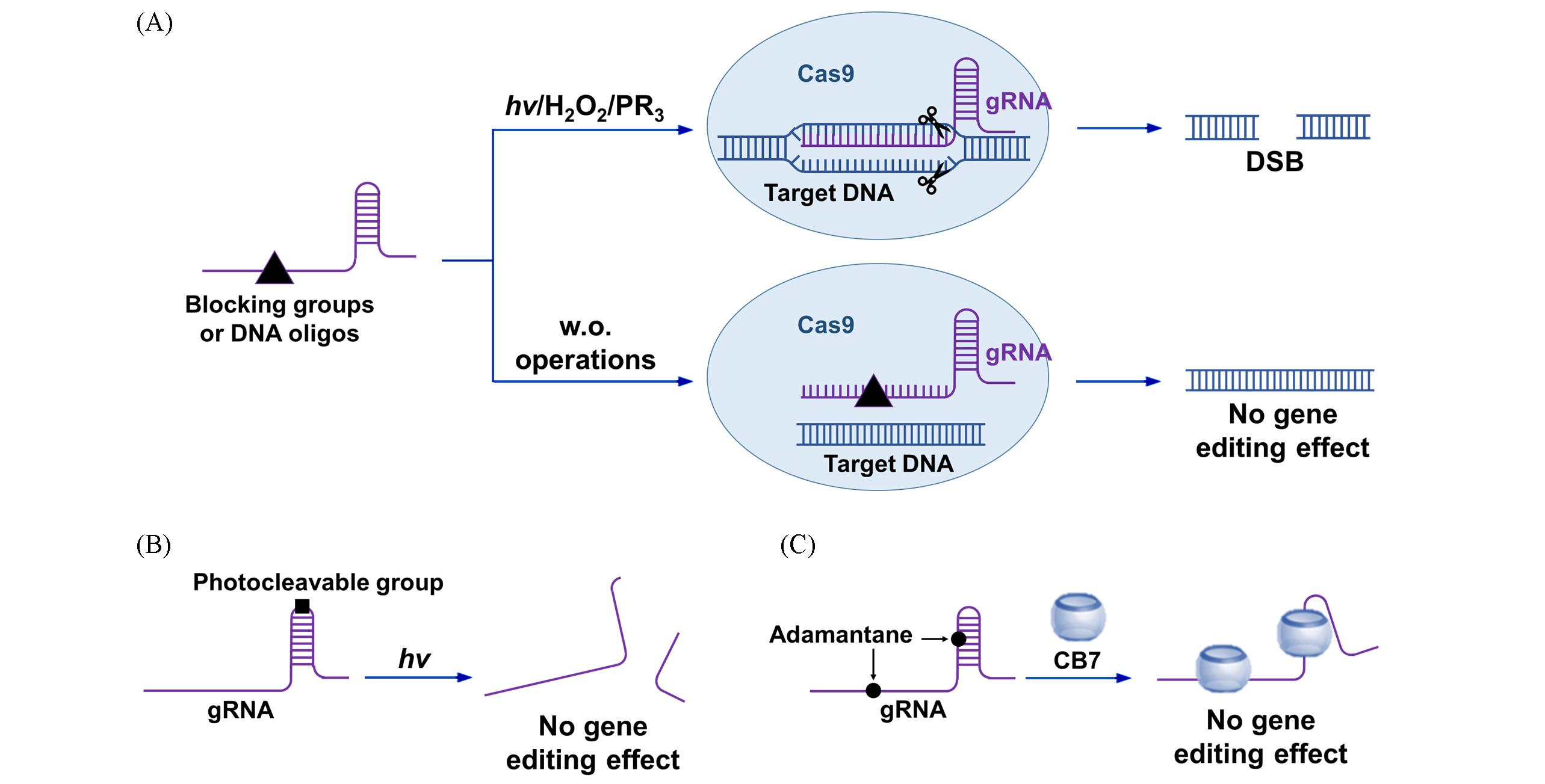
Fig.2 Schematic diagram of sgRNA regulation systems(A) Schematic illustration of sgRNA functional activation induced by light or small molecules; (B) schematic illustration of photo-induced sgRNA inactivation system; (C) schematic illustration of the sgRNA regulation system in small molecules induced gene editing inactivation through host-guest recognition.
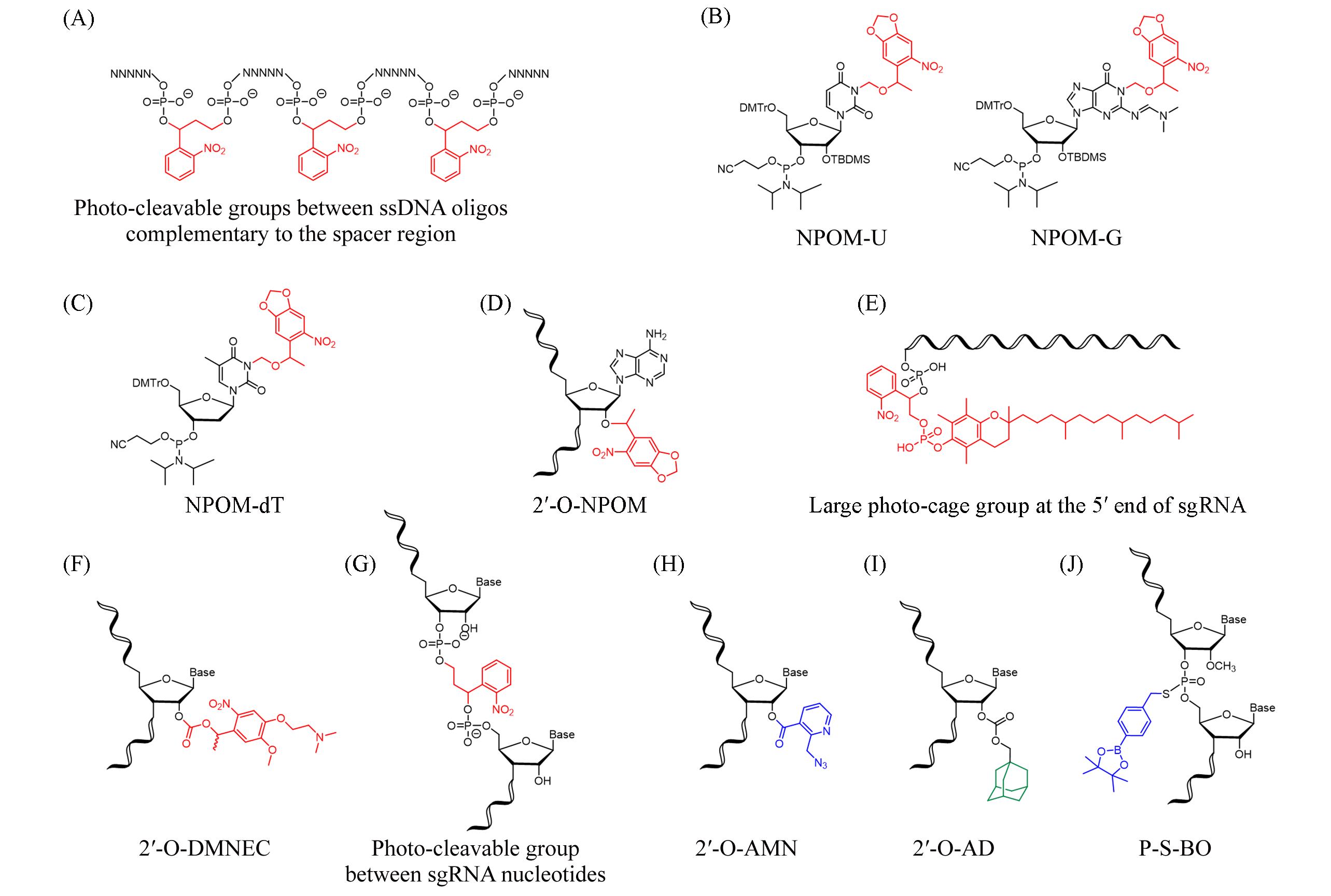
Fig.3 Schematic diagram of nucleoside monomers modified by different chemical groups in the sgRNA functional regulation systems based on chemical reactionsThe red labeled groups are photo-cleavable groups, the blue labeled groups can be induced to break bonds by small molecules, and the green labeled groups can participate in the host-guest recognition.
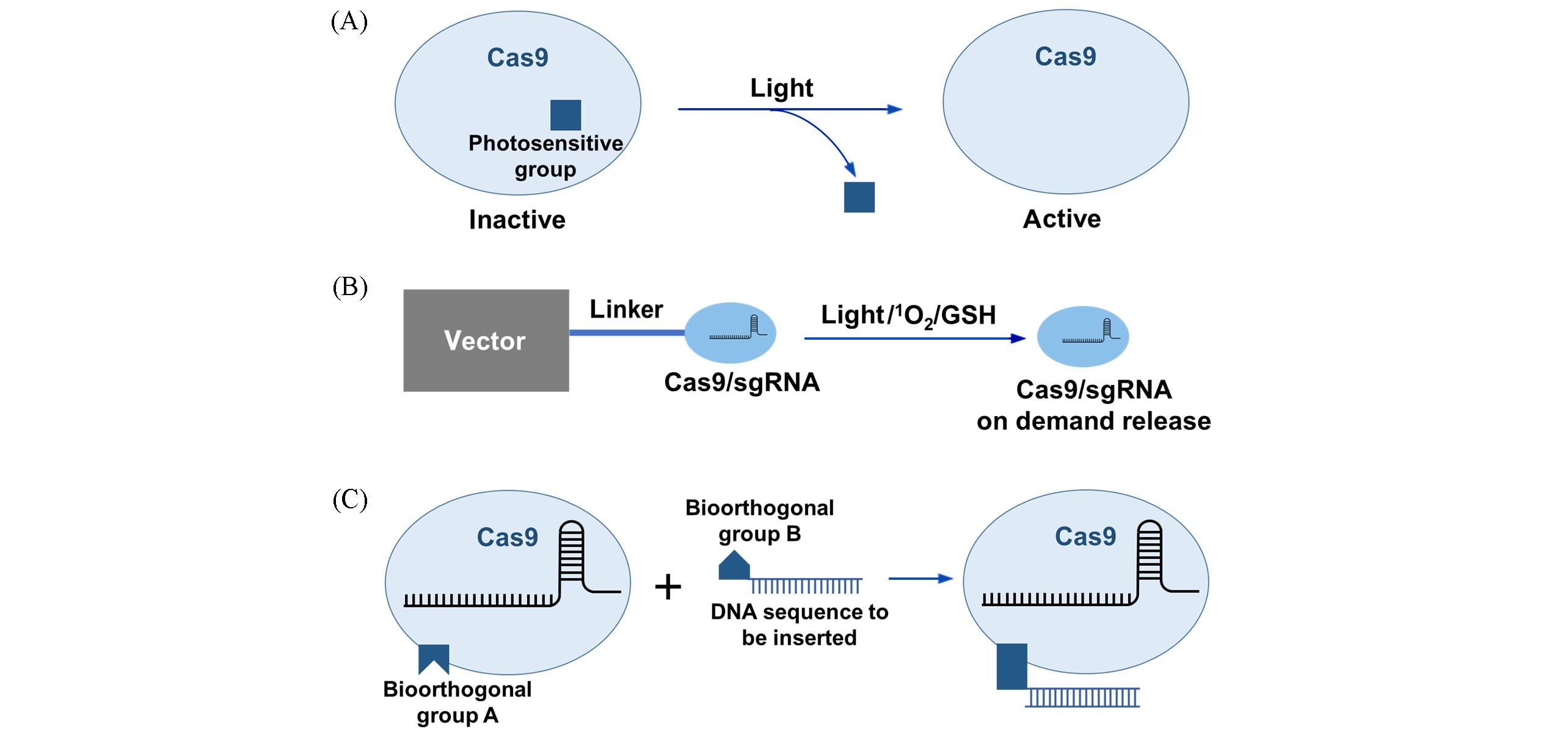
Fig.4 Schematic diagram of Cas9 or Cas9/sgRNA regulation systems(A) Schematic illustration of photo-induced Cas9 protein activation system; (B) schematic illustration of light- or active substance-induced Cas9/sgRNA release systems; (C) schematic illustration of CRISPR optimization systems based on bioorthogonal reactions.
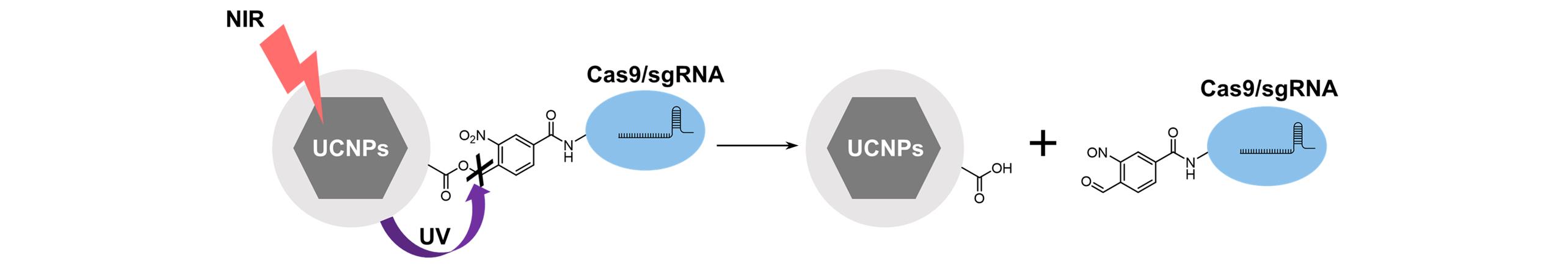
Fig.5 Schematic diagram of the light⁃controlled Cas9/sgRNA release strategyThe UCNPs can convert NIR light(980 nm) into local ultraviolet light for the cleavage of photo-sensitive molecules, thereby resulting in on-demand release of CRISPR-Cas9 .
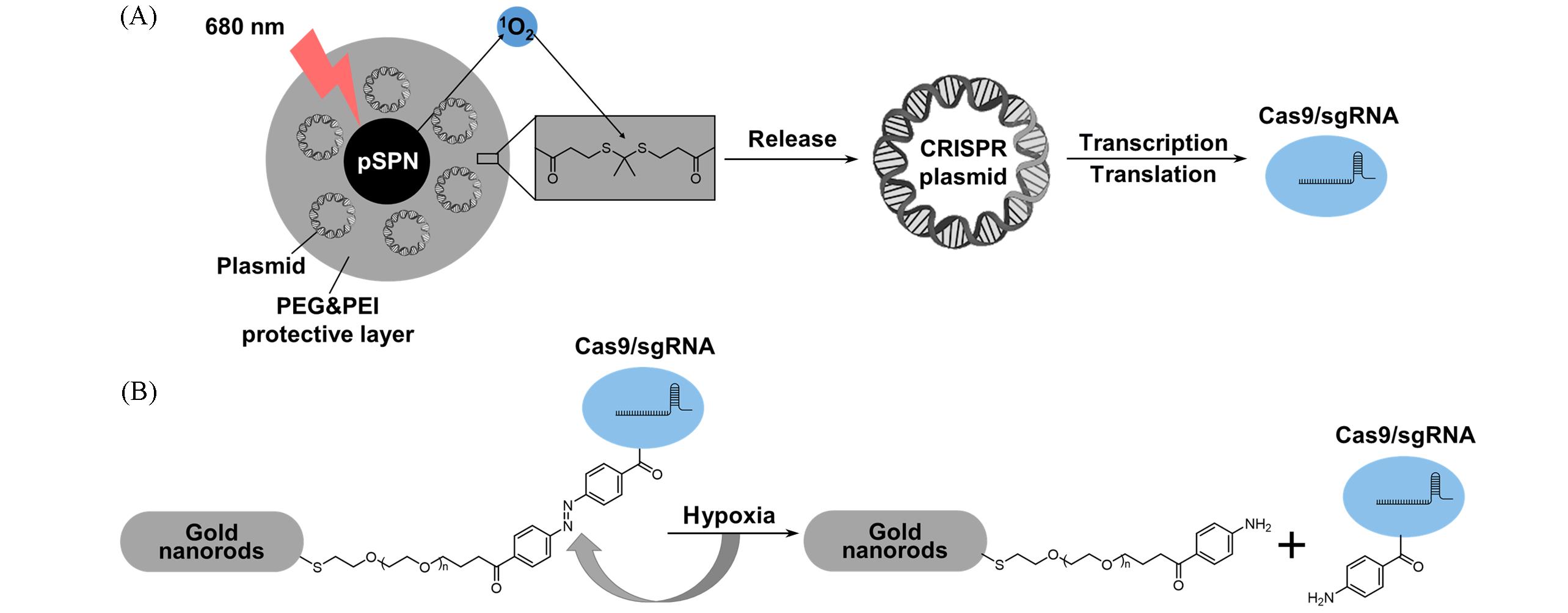
Fig.6 Schematic diagram of the active substance⁃induced Cas9/sgRNA release strategies(A) The photolabile semiconducting polymer nanotransducer(pSPN) comprises a 1O2-generating backbone grafted with polyethylenimine brushes through 1O2-cleavable linkers. NIR photoirradiation spontaneously triggers the cleavage of gene vectors from pSPN, resulting in the release of CRISPR/Cas9 plasmids and subsequently initiating gene editing; (B) gold nanorods are modified with azobenzene-4,4′- dicarboxylic acid(p-AZO) to achieve on-demand release of CRISPR-Cas9 using hypoxia-responsive azo bonds.
| 1 | Doudna J. A., Charpentier E., Science, 2014, 346, 1258096 |
| 2 | Knott G. J., Doudna J. A., Science, 2018, 361, 866—869 |
| 3 | Capecchi M. R., Science, 1989, 244, 1288—1292 |
| 4 | Rudin N., Sugarman E., Haber J. E., Genetics, 1989, 122, 519—534 |
| 5 | Rouet P., Smih F., Jasin M., Mol. Cell. Biol., 1994, 14, 8096—8106 |
| 6 | Klug A., Rhodes D., Cold Spring Harb. Symp. Quant. Biol., 1987, 52, 473—482 |
| 7 | Kim Y. G., Cha J., Chandrasegaran S., Proc. Natl. Acad. Sci. USA, 1996, 93, 1156—1160 |
| 8 | Boch J., Scholze H., Schornack S., Landgraf A., Hahn S., Kay S., Lahaye T., Nickstadt A., Bonas U., Science, 2009, 326, 1509—1512 |
| 9 | Moscou M. J., Bogdanove A. J., Science, 2009, 326, 1501 |
| 10 | Makarova K. S., Wolf Y. I., Iranzo J., Shmakov S. A., Alkhnbashi O. S., Brouns S. J. J., Charpentier E., Cheng D., Haft D. H., Horvath P., Moineau S., Mojica F. J. M., Scott D., Shah S. A., Siksnys V., Terns M. P., Venclovas C., White M. F., Yakunin A. F., Yan W., Zhang F., Garrett R. A., Backofen R., van der Oost J., Barrangou R., Koonin E. V., Nat. Rev. Microbiol., 2020, 18, 67—83 |
| 11 | Doudna J. A., Charpentier E., Science, 2014, 346, 1258096 |
| 12 | Hsu P. D., Lander E. S., Zhang F., Cell, 2014, 157, 1262—1278 |
| 13 | Stadtmauer E. A., Fraietta J. A., Davis M. M., Cohen A. D., Weber K. L., Lancaster E., Mangan P. A., Kulikovskaya I., Gupta M., Chen F., Tian L. F., Gonzalez V. E., Xu J., Jung I. Y., Melenhorst J. J., Plesa G., Shea J., Matlawski T., Cervini A., Gaymon A. L., Desjardins S., Lamontagne A., Salas-Mckee J., Fesnak A., Siegel D. L., Levine B. L., Jadlowsky J. K., Young R. M., Chew A., Hwang W. T., Hexner E. O., Carreno B. M., Nobles C. L., Bushman F. D., Parker K. R., Qi Y. Y., Satpathy A. T., Chang H.Y., Zhao Y. B., Lacey S. F., June C. H., Science, 2020, 367, eaba7365 |
| 14 | Zhang M., Eshraghian E. A., Al Jammal O., Zhang Z. B., Zhu X., Biomed. Pharmacother., 2021, 133, 111007 |
| 15 | Zhang D., Zhang D. Q., Zhang Z. Y., Unver T., Zhang B. H., J. Adv. Res., 2021, 29, 207—211 |
| 16 | Zhu H. C., Li C., Gao C. X., Nat. Rev. Mol. Cell Biol., 2020, 21, 661—677 |
| 17 | Musunuru K., Chadwick A. C., Mizoguchi T., Garcia S. P., DeNizio J. E., Reiss C. W., Wang K., Iyer S., Dutta C., Clendaniel V., Amaonye M., Beach A., Berth K., Biswas S., Braun M. C., Chen H. M., Colace T. V., Ganey J. D., Gangopadhyay S. A., Garrity R., Kasiewicz L. N., Lavoie J., Madsen J. A., Matsumoto Y., Mazzola A. M., Nasrullah Y. S., Nneji J., Ren H., Sanjeev A., Shay M., Stahley M. R., Fan S. H. Y., Tam Y. K., Gaudelli N. M., Ciaramella G., Stolz L. E., Malyala P., Cheng C. J., Rajeev K. G., Rohde E., Bellinger A. M., Kathiresan S., Nature, 2021, 593, 429—434 |
| 18 | Dawson T. M., Golde T. E., Lagier⁃Tourenne C., Nat. Neurosci., 2018, 21, 1370—1379 |
| 19 | Maresch R., Mueller S., Veltkamp C., Oellinger R., Friedrich M., Heid I., Steiger K., Weber J., Engleitner T., Barenboim M., Klein S., Louzada S., Banerjee R., Strong A., Stauber T., Gross N., Geumann U., Lange S., Ringelhan M., Varela I., Unger K., Yang F. T., Schmid R. M., Vassiliou G. S., Braren R., Schneider N., Heikenwalder M., Bradley A., Saur D., Rad R., Nat. Commun., 2016, 7, 10770 |
| 20 | Kaneko T., Tanaka S., Biochem. Bioph. Res. Co., 2020, 527, 1039—1042 |
| 21 | Yan Q., Zhang Q., Yang H., Zou Q., Tang C., Fan N., Lai L., Cell Regen., 2014, 3, 11 |
| 22 | Vilarinor M., Rashid S. T., Suchy F. P., McNabb B., van der Meulen T., Fine E. J., Ahsan S. D., Mursaliyev N., Sebastiano V., Diab S. S., Huising M. O., Nakauchi H., Ross P. J., Sci. Rep., 2020, 10, 7500 |
| 23 | Fujii W., Kawasaki K., Sugiura K., Naito K., Nucleic Acids Res., 2013, 41, e187 |
| 24 | Ryu J. Y., Won E. J., Lee H. A. R., Kim J. H., Hui E., Kim H. P., Yoon T. J., Biomaterials, 2020, 232, 119736 |
| 25 | Zhang S., Shen J., Li D., Cheng Y., Theranostics, 2021, 11, 614—648 |
| 26 | Xu C. F., Chen G. J., Luo Y. L., Zhang Y., Zhao G., Lu Z. D., Czarna A., Gu Z., Wang J., Adv. Drug. Deliv. Rev., 2021, 168, 3—29 |
| 27 | Chuang Y. F., Phipps A. J., Lin F. L., Hecht V., Hewitt A. W., Wang P. Y., Liu G. S., Cell. Mol. Life Sci., 2021, 78, 2683—2708 |
| 28 | Pattanayak V., Lin S., Guilinger J. P., Ma E., Doudna J. A., Liu D. R., Nat. Biotechnol., 2013, 31, 839—843 |
| 29 | Zischewski J., Fischer R., Bortesi L., Biotechnol. Adv., 2017, 35, 95—104 |
| 30 | Wang Y., Cheng X., Shan Q. W., Zhang Y., Liu J. X., Gao C. X., Qiu J. L., Nat. Biotechnol., 2014, 32, 947—951 |
| 31 | Fu Y. F., Sander J. D., Reyon D., Cascio V. M., Joung J. K., Nat. Biotechnol., 2014, 32, 279—284 |
| 32 | Cromwell C. R., Sung K., Park J., Krysler A. R., Jovel J., Kim S. K., Hubbard B. P., Nat. Commun., 2018, 9, 1448 |
| 33 | Brown W., Zhou W., Deiters A., ChemBioChem, 2021, 22, 63—72 |
| 34 | Gangopadhyay S. A., Cox K. J., Manna D., Lim D., Maji B., Zhou Q., Choudhary A., Biochemistry, 2019, 58, 234—244 |
| 35 | Tang W., Hu J. H., Liu D. R., Nat. Commun., 2017, 8, 15939 |
| 36 | Lin J., Wang W. J., Wang Y., Liu Y., Xu L., J. Am. Chem. Soc., 2021, 143, 19834—19843 |
| 37 | Kundert K., Lucas J. E., Watters K. E., Fellmann C., Ng A. H., Heineike B. M., Fitzsimmons C. M., Oakes B. L., Qu J., Prasad N., Rosenberg O. S., Savage D. F., El⁃Samad H., Doudna J. A., Kortemme T., Nat. Commun., 2019, 10, 2127 |
| 38 | Jain S., Xun G. Abesteh S., Ho S., Lingamaneni M., Martin T. A., Tasan I., Yang C., Zhao H., ACS Synth. Biol., 2021, 10, 1320—1327 |
| 39 | Manna D., Maji B., Gangopadhyay S. A., Cox K. J., Zhou Q., Law B. K., Mazitschek R., Choudhary A., Angew. Chem. Int. Ed., 2019, 58, 6285—6289 |
| 40 | Nihongaki Y., Kawano F., Nakajima T., Sato M., Nat. Biotechnol., 2015, 33, 755—760 |
| 41 | Taiariol L., Chaix C., Farre C., Moreau E., Chem. Rev., 2022, 122, 340—384 |
| 42 | Wang J., Wang X., Fan X., Chen P. R., ACS Cent. Sci., 2021, 7, 929—943 |
| 43 | Scinto S. L., Bilodeau D. A., Hincapie R., Lee W., Nguyen S. S., Xu M., Am Ende C. W., Finn M. G., Lang K., Lin Q., Pezacki J. P., Prescher J. A., Robillard M. S., Fox J. M., Nat. Rev. Methods Primers, 2021, 1, 30—84 |
| 44 | Kumar G. S., Lin Q., Chem. Rev., 2021, 121, 6991—7031 |
| 45 | He K., Chou E. T., Begay S., Anderson E. M., van Brabant Smith A., ChemBioChem, 2016, 17, 1809—1812 |
| 46 | Taemaitree L., Shivalingam A., El-Sagheer A. H., Brown T., Nat. Commun., 2019, 10, 1610—1617 |
| 47 | Hüll K., Morstein J., Trauner D., Chem. Rev., 2018, 118, 10710—10747 |
| 48 | Li J., Kong, H., Zhu C., Zhang Y., Chem. Sci., 2020, 11, 3390—3396 |
| 49 | Jain P. K., Ramanan V., Schepers A. G., Dalvie N. S., Panda A., Fleming H. E., Bhatia S. N., Angew. Chem. Int. Ed., 2016, 55, 12440—12444 |
| 50 | Zhou W., Brown W., Bardhan A., Delaney M., Ilk A. S., Rauen R. R., Kahn S. I., Tsang M., Deiters A., Angew. Chem. Int. Ed., 2020, 59, 8998—9003 |
| 51 | Moroz⁃Omori E. V., Satyapertiwi D., Ramel M. C., Hogset H., Sunyovszki I. K., Liu Z., Wojciechowski J. P., Zhang Y., Grigsby C. L., Brito L., Bugeon L., Dallman M. J., Stevens M. M., ACS Cent. Sci., 2020, 6, 695—703 |
| 52 | Liu Y., Zou R.S., He S., Nihongaki Y., Li X., Razavi S., Wu B., Ha T., Science, 2020, 368, 1265—1269 |
| 53 | Wang Y., Liu Y., Xie F., Lin J., Xu L., Chem. Sci., 2020, 11, 11478—11484 |
| 54 | Zhang Y., Ling X., Su X., Zhang S., Wang J., Zhang P., Feng W., Zhu Y. Y., Liu T., Tang X., Angew. Chem. Int. Ed., 2020, 59, 20895—20899 |
| 55 | Wang S., Wei L., Wang J. Q., Ji H., Xiong W., Liu J., Yin P., Tian T., Zhou X., ACS Chem. Biol., 2020, 15, 1455—1463 |
| 56 | Zhang D., Liu L., Jin S., Tota E., Li Z., Piao X., Zhang X., Fu X. D., Devaraj N. K., J. Am. Chem. Soc., 2022, 144, 4487—4495 |
| 57 | Carlson⁃Stevermer J., Kelso R., Kadina A., Joshi S., Rossi N., Walker J., Stoner R., Maures T., Nat. Commun., 2020, 11, 5041—5047 |
| 58 | Habibian M., McKinlay C., Blake T. R., Kietrys A. M., Waymouth R. M., Wender P. A., Kool E. T., Chem. Sci., 2019, 11, 1011—1016 |
| 59 | Wang S. R., Wu L. Y., Huang H. Y., Xiong W., Liu J., Wei L., Yin P., Tian T., Zhou X., Nat. Commun., 2020, 11, 91—101 |
| 60 | Xiong W., Liu X., Qi Q., Ji H., Liu F., Zhong C., Liu S., Tian T., Zhou X., Nucleic Acids Res., 2022, 50, 1241—1255 |
| 61 | Gu C., Xiao L., Shang J., Xu X., He L., Xiang Y., Chem. Sci., 2021, 12, 9934—9945 |
| 62 | Hemphill J., Borchardt E. K., Brown K., Asokan A., Deiters A., J. Am. Chem. Soc., 2015, 137, 5642—5645 |
| 63 | Pan Y. C., Yang J. J., Luan X. W., Liu X. L., Li X. Q., Yang J., Huang T., Sun L., Wang Y. Z., Lin Y. H., Song Y. J., Sci. Adv., 2019, 5, eaav7199 |
| 64 | Lyu Y., He S., Li J., Jiang Y., Sun H., Miao Y., Pu K., Angew. Chem. Int. Ed., 2019, 58, 18197—18201 |
| 65 | Li X., Pan Y., Chen C., Gao Y., Liu X., Yang K., Luan X., Zhou D., Zeng F., Han X., Song Y., Angew. Chem. Int. Ed., 2021, 60, 21200—21204 |
| 66 | Kwiatkowski S., Knap B., Przystupski D., Saczko J., Kedzierska E., Knap⁃Czop K., Kotlinska J., Michel O., Kotowski K., Kulbacka J., Biomed. Pharmacother., 2018, 106, 1098—1107 |
| 67 | Li X., Kwon N., Guo T., Liu Z., Yoon J., Angew. Chem. Int. Ed., 2018, 57, 11522—11531 |
| 68 | Mfouo⁃Tynga I. S., Dias L. D., Inada N. M., Kurachi C., Photodiagnosis Photodyn. Ther., 2021, 34, 102091 |
| 69 | He M., Li J., Han H., Borges C. A., Neiman G., Roise J. J., Hadaczek P., Mendonsa R., Holm V. R., Wilson R. C., Bankiewicz K., Zhang Y., Sadlowski C. M., Healy K., Riley L. W., Murthy N., Chem. Sci., 2020, 11, 8973—8980 |
| 70 | Ling X., Xie B., Gao X., Chang L., Zheng W., Chen H., Huang Y., Tan L., Li M., Liu T., Sci. Adv., 2020, 6, eaaz0051 |
| 71 | Ling X., Chang L., Chen H., Gao X., Yin J., Zuo Y., Huang Y., Zhang B., Hu J., Liu T., Mol. Cell, 2021, 81, 1—10 |
| [1] | 胡玉灿, 曹朝辉, 郑灵刚, 沈俊涛, 赵维, 戴磊. CRISPR⁃Cas基因编辑技术在微生物组工程中的应用[J]. 高等学校化学学报, 2023, 44(3): 20220362. |
| [2] | 盛劲菡, 郑琪臻, 汪铭. CRISPR/Cas9基因编辑非病毒递送系统[J]. 高等学校化学学报, 2023, 44(3): 20220344. |
| [3] | 常丽颖, 凌鑫宇, 陈和祺, 王雪, 刘涛. 基因编辑在线粒体疾病中的应用[J]. 高等学校化学学报, 2023, 44(3): 20220363. |
| [4] | 肖珩, 李永奎, 邢曦雯. 化学调控CRISPR/Cas9基因编辑技术的研究进展[J]. 高等学校化学学报, 2023, 44(3): 20220410. |
| [5] | 刘红, 江敬红, 段志娟, 徐仕军, 黄福建, 夏帆. 光控CRISPR技术的研究进展[J]. 高等学校化学学报, 2021, 42(11): 3321. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||