

高等学校化学学报 ›› 2021, Vol. 42 ›› Issue (4): 1005.doi: 10.7503/cjcu20200643
邹俊彦1, 张焱焱2, 陈石1, 邵怀宇1, 汤育欣1,2( )
)
收稿日期:2020-09-02
出版日期:2021-04-10
发布日期:2021-04-08
通讯作者:
汤育欣
E-mail:yxtang@fzu.edu.cn
基金资助:
ZOU Junyan1, ZHANG Yanyan2, CHEN Shi1, SHAO Huaiyu1, TANG Yuxin1,2( )
)
Received:2020-09-02
Online:2021-04-10
Published:2021-04-08
Contact:
TANG Yuxin
E-mail:yxtang@fzu.edu.cn
Supported by:摘要:
传统的锂金属电池存在电解液易泄漏、 易燃等安全隐患, 因此开发不燃性全固态电解质对于解决锂金属电池安全问题至关重要, 而如何有效降低固体电解质与电极之间的界面电阻是发展高性能全固态锂金属电池的关键. 针对如何优化全固态锂金属电池表界面的问题, 本文综述了全固态锂金属电池电极和电解质表面修饰的最新研究进展, 对提高界面接触和降低界面电阻的传统方法进行了探讨, 分析并点评了新型的表面修饰技术, 为进一步提高全固态锂金属电池的综合性能提供新思路. 最后, 对全固态锂金属电池的研究前景进行了展望.
中图分类号:
TrendMD:
邹俊彦, 张焱焱, 陈石, 邵怀宇, 汤育欣. 全固态锂金属电池表界面化学的研究进展. 高等学校化学学报, 2021, 42(4): 1005.
ZOU Junyan, ZHANG Yanyan, CHEN Shi, SHAO Huaiyu, TANG Yuxin. Recent Development on Surface-interface Chemistry of All-solid-state Lithium Batteries. Chem. J. Chinese Universities, 2021, 42(4): 1005.
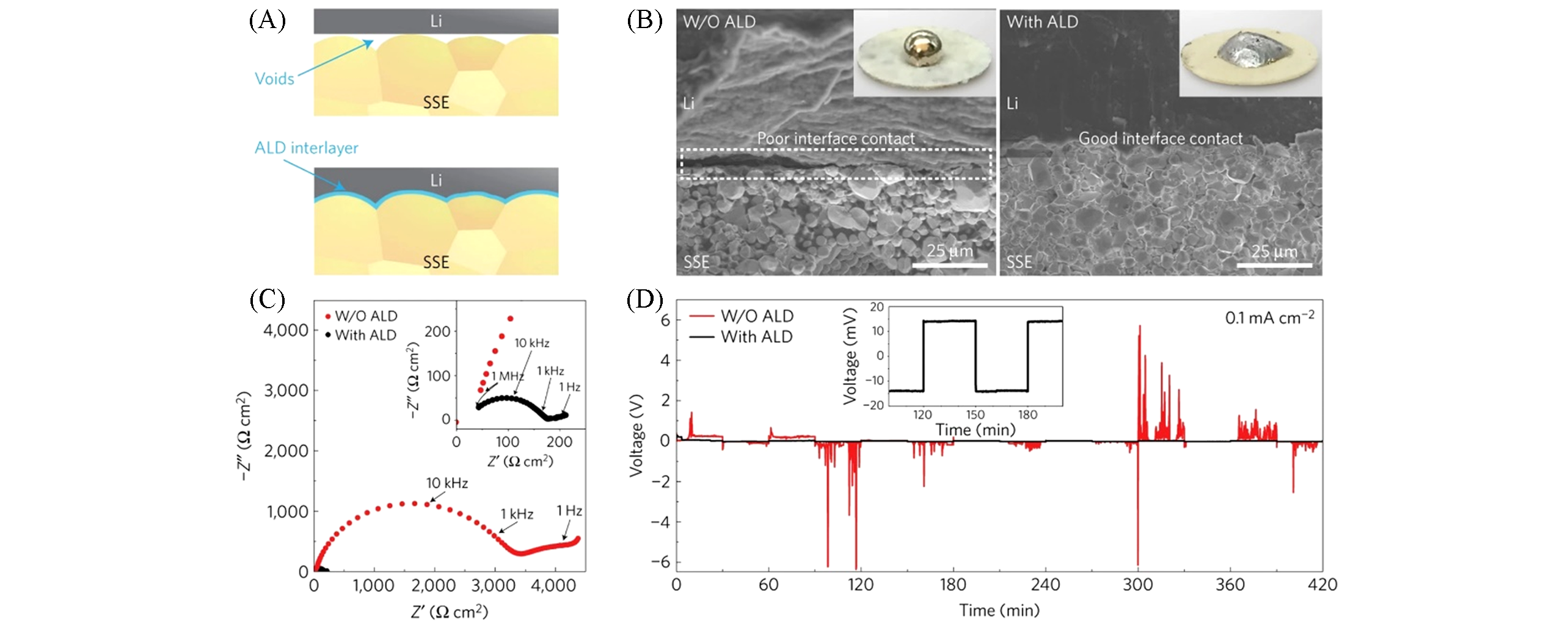
Fig.1 Characterizations of garnet solid?state electrolyte/Li metal interface[9](A) Schematic of the wetting behaviour of garnet surface with molten Li. (B) SEM images of the garnet solid-state electrolyte/Li metal interface. Without ALD-Al2O3 coating, garnet has a poor interfacial contact with Li metal even on heating. With the help of ALD-Al2O3 coating on garnet, Li metal can uniformly bond with garnet at the interface on heating. Insets are photos of molten Li metal on top of the garnet surface clearly demonstrating classical wetting behaviour for the ALD-treated garnet surface. (C) Comparison of EIS profiles of the symmetric Li non-blocking garnet cells. Inset shows the enlarged impedance curve of the ALD-treated garnet cell. (D) Comparison of dc. cycling for symmetric cells of Li/bare garnet/Li(black curve) and Li/ALD-treated garnet/Li(red curve) at a current density of 0.1 mA/cm2. The inset is the magnified curve of the ALD-treated cell. Copyright 2017, Springer Nature.
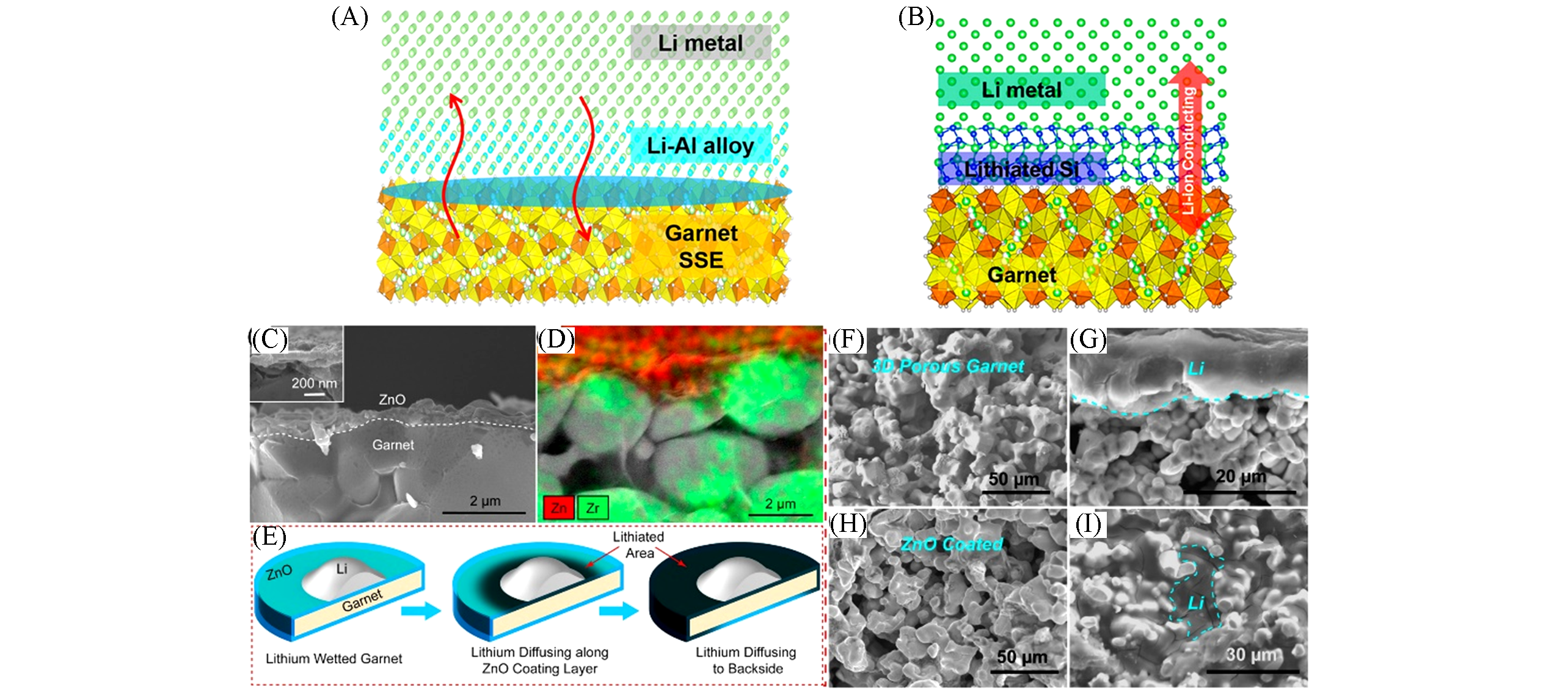
Fig.2 The reaction between Al and Li promotes enhanced molten Li infusion onto the garnet’s rough surface, whereas the formation of a Li?Al alloy fills the gap between the garnet solid electrolyte and the Li metal to improve interfacial contact and enhance Li+ transport(A)[11], schematic view of the superlithiophilic garnet?Li metal interface enabled by the in situ?formed, lithiated Si layer(B)[12], cross?section SEM image(C) and elemental mapping(D) of the garnet electrolyte coated with a 50 nm ALD ZnO layer, schematic of the lithium diffusion process along the ZnO coating layer on the garnet surface(E), the cross?section SEM images of the Pristine(F) and the lithium infiltrated porous garnet with a porosity of 60%—70%(G), the cross? section SEM image of the porous garnet coated with conformal ZnO surface layer using ALD process(H) and lithium infiltrated porous garnet with ZnO surface treatment, where almost all pores have been filled with lithium metal(I)[10]The inset of (C) is a cross-section SEM image of the garnet/ZnO interface at higher magnification. The lithium metal area has been marked with a cyan dashed line. (A) Copyright 2017, American Association for the Advancement of Science; (B) Copyright 2016, American Chemical Society; (C—I) Copyright 2017, American Chemical Societry.

Fig.3 Images demonstrating the effectiveness of the Li?Sn alloy welding process on garnet SSE pellets[15]In less than 10 s, the polished garnet pellet was successfully coated with uniform Li?Sn alloy.Copyright 2018, Wiley?VCH.
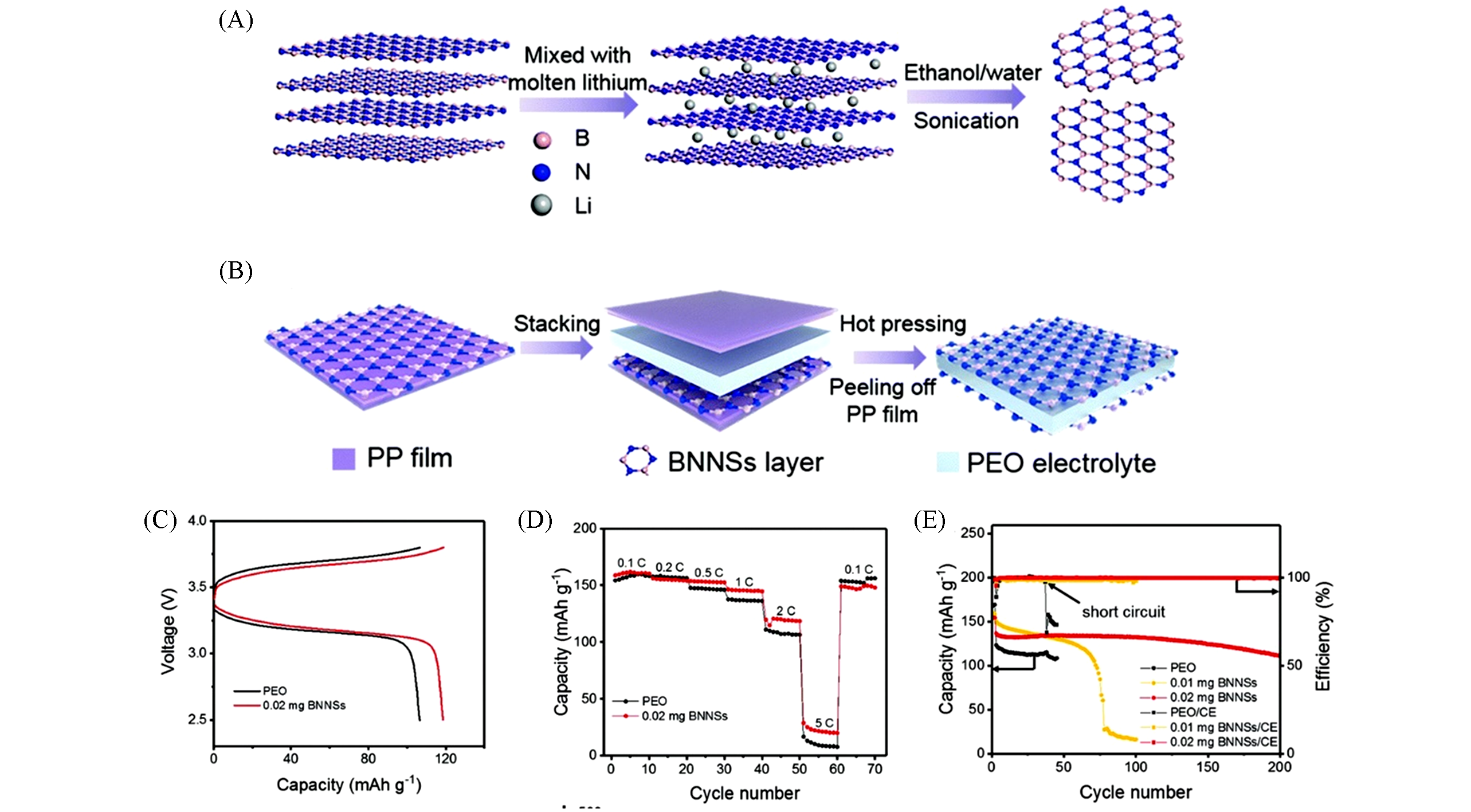
Fig.5 Schematic illustrations of the BNNSs exfoliation process(A) and the transfer process(B); typical charge?discharge voltage profiles of Li/LFP cells with PEO and BNNSs?coated PEO electrolytes at 2 C(C); rate performance comparison of the Li/PEO/LFP cell with the Li/BNNSs?coated PEO/LFP cell(D) and plots of specific discharge capacity and coulombic efficiency versus cycle number of the Li/PEO/LFP cell and Li/BNNSs?coated PEO/LFP cell cycling at 2 C(E)[31]Copyright 2019, Royal Society Chemistry.
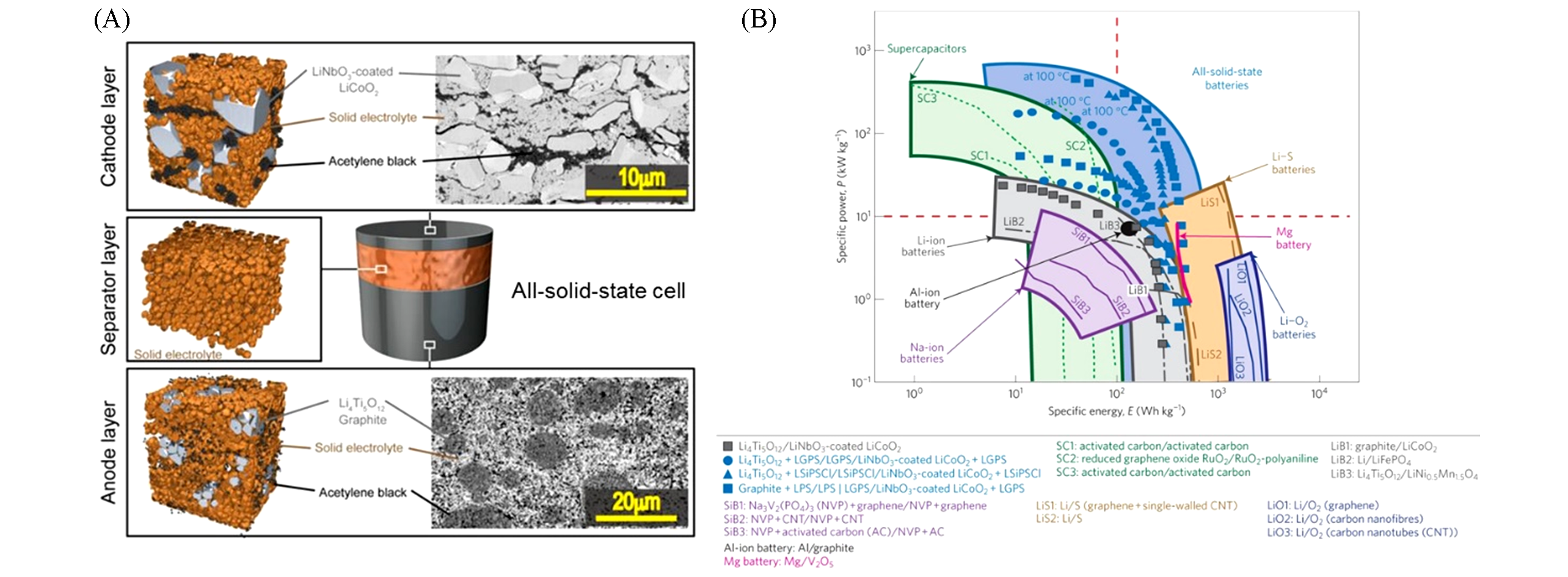
Fig.6 Schematic diagram of the all?solid?state electrochemical cell prepared herein, along with SEM images of the electrode layers(A); the Ragone plots of the cells prepared in this study and previously reported batteries and capacitors(B)[39]The red dashed line indicates the specific energy E=102 Wh/kg and specifice power P=10 kW/kg. The devices powered by liquid electrolytes show the inverse relationship between specific energy and power. The prepared all-solid-state cells simultaneously achieved high energy and power(E>102 Wh/kg and P>10 kW/kg), which is di-cult to achieve for conventional devices. Copyright 2016, Elsevier.
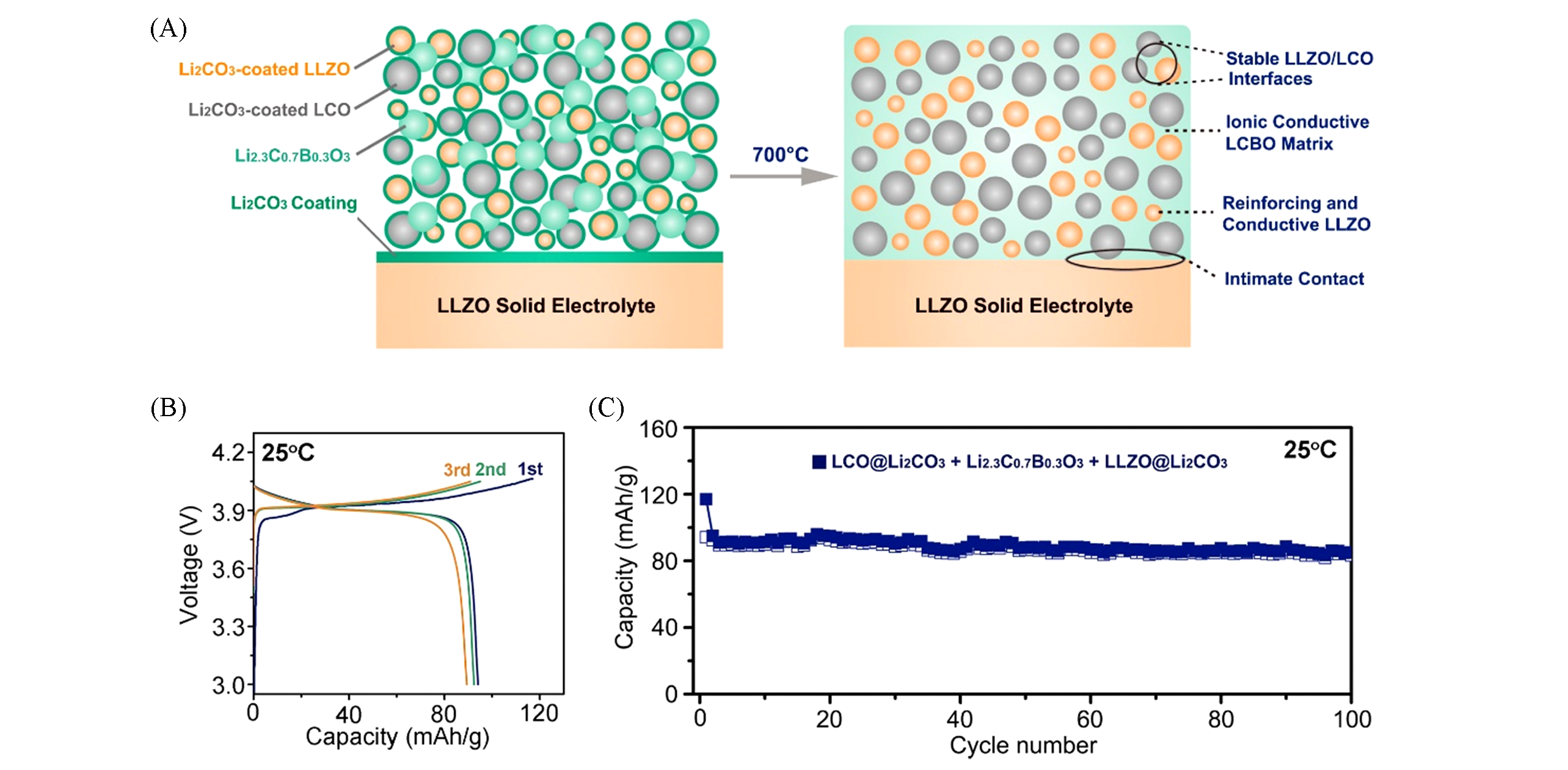
Fig.7 Schematics of the interphase?engineered all?ceramic cathode/electrolyte(A); charge/discharge profiles of the interphase?engineered all?ceramic Li/LLZO/LCO cell for the first three cycles at 0.05 C at 25 ℃(B) and cycling performance of the interphase?engineered all?ceramic Li/LLZO/LCO cell at 0.05 C at 25 °C(C)[42]A thin layer of Li2CO3(3 nm) was artificially coated on LCO because the spontaneously formed Li2CO3 on LCO is too thin. The Li2CO3-coated LCO(LCO@Li2CO3) was then mixed with a spontaneously Li2CO3-coated LLZO(LLZO@Li2CO3), and Li2.3C0.7B0.3O3 solder to make a cathode composite. The cathode composite was then coated on an LLZO ceramic pellet, which was also spon- taneously coated by Li2CO3. After sintering at 700 ℃, Li2.3C0.7B0.3O3 will melt, and react with the Li2CO3 coatings on both LLZO and LCO to form Li2.3-xC0.7+xB0.3-xO3(LCBO) interphase. Copyright 2018, Elsevier.
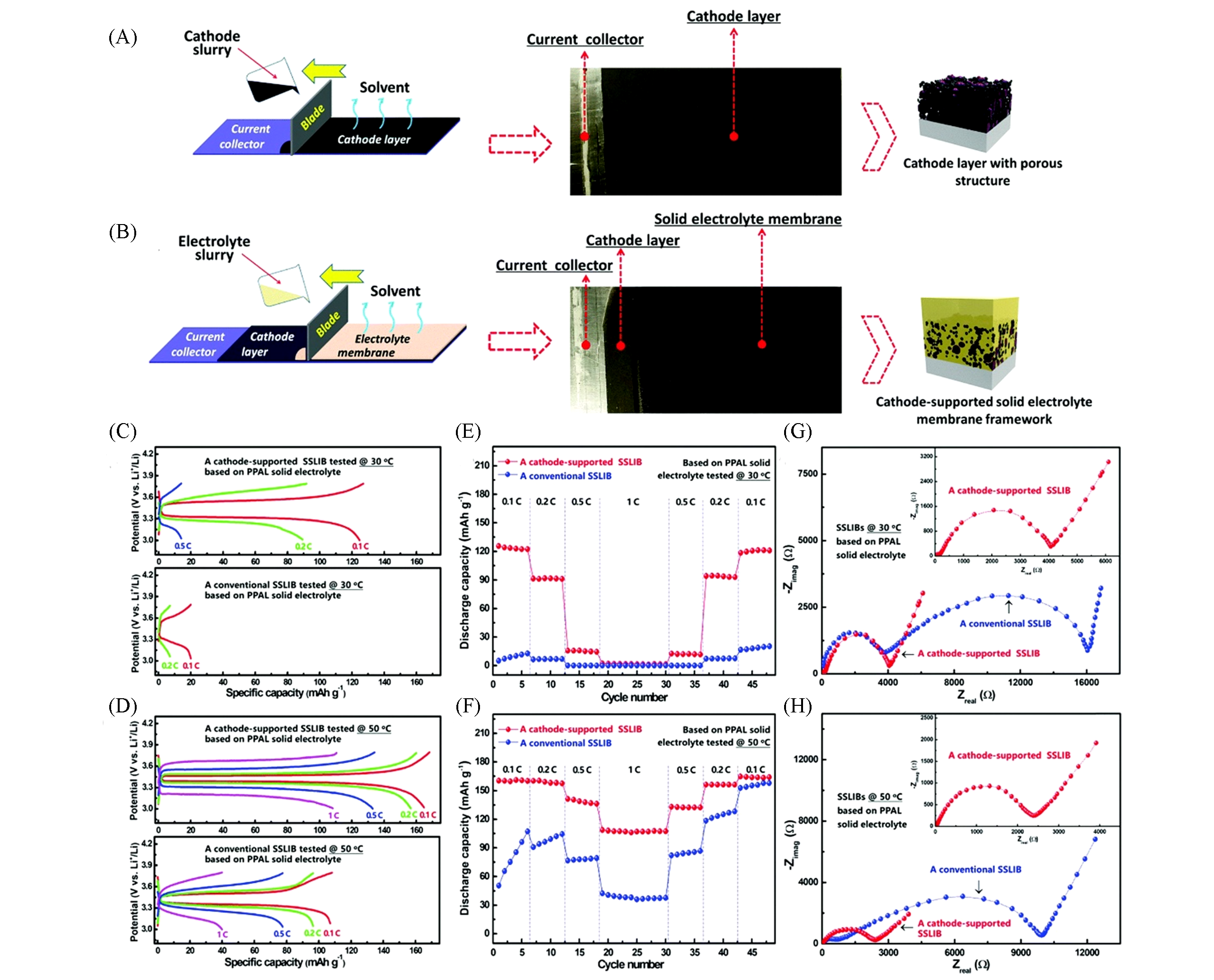
Fig.8 Fabrication of the cathode?supported solid electrolyte membrane framework[43](A) Schematic of the cathode electrode preparation by tape casting, the cathode slurry was tape cast on the aluminium current collector. (B) Schematic of the cathode-supported solid electrolyte membrane preparation by tape casting, the electrolyte slurry was tape cast onto the dry cathode tape showing compact structure after drying. Typical charge-discharge curves of as assembled cathode-supported and conventional SSLIBs based on the PPAL solid electrolyte tested at 30 ℃(C) and 50 ℃(D). Discharge capa-cities of conventional and cathode-supported SSLIBs based on the PPAL solid electrolyte tested at 30 ℃(E) and 50 ℃(F) as a function of cycle number at various rates. Electrochemical impedance plots of a conventional SSLIB and a cathode-supported SSLIB based on the PPAL solid electrolyte at 30 ℃(G) and 50 ℃(H). Copyright 2019, Royal Society Chemistry.
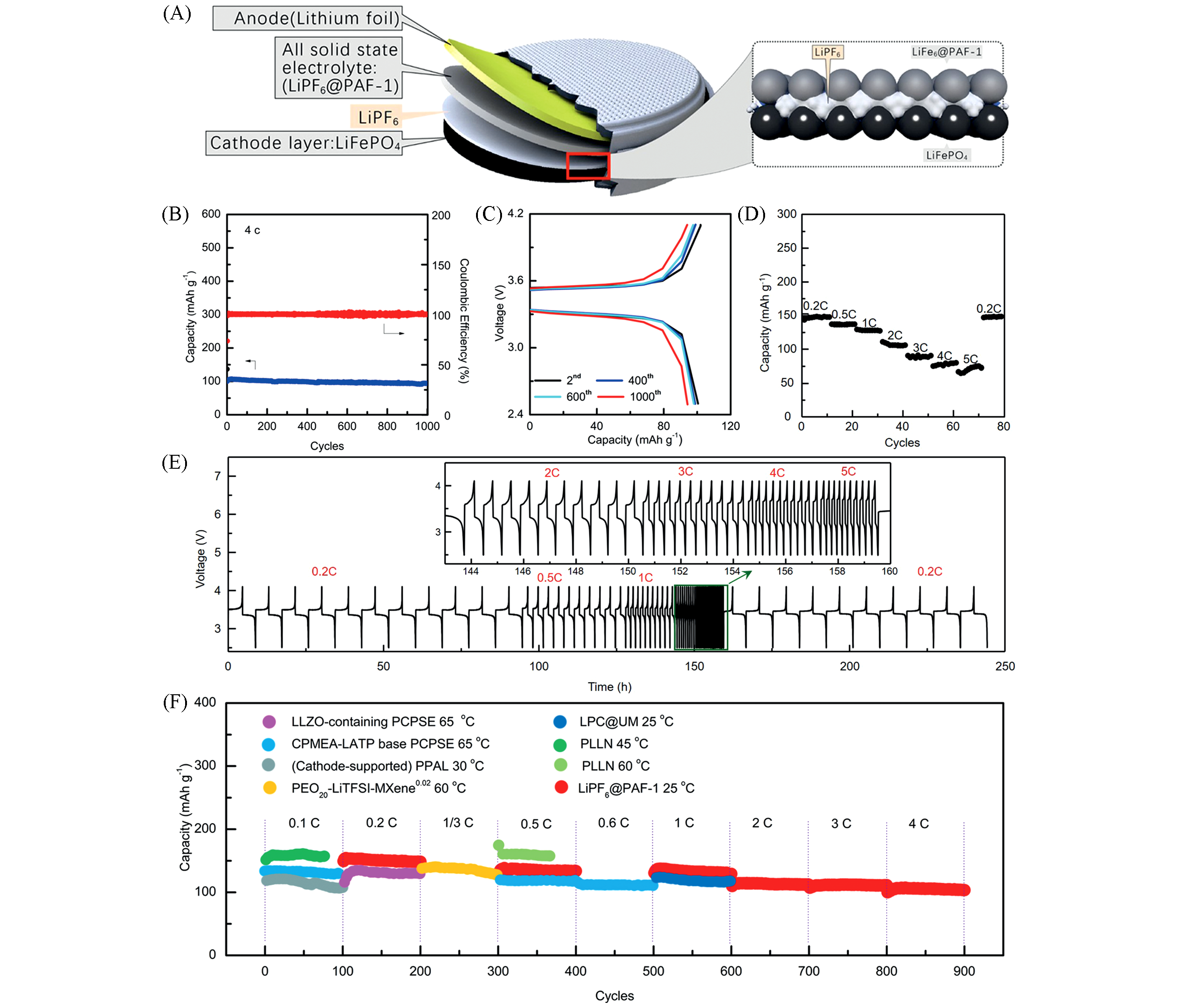
Fig.9 Schematic and characterization of the LiFePO4 /LiPF6@PAF?1 /Li cell[44](A) Schematic diagram for the LiFePO4/LiPF6@PAF-1/Li cell; (B) cycling stability and Coulombic efficiency of the LiFePO4/ LiPF6@PAF-1/Li cell at a current density of 4 C; (C) voltage profiles of the LiFePO4/LiPF6@PAF-1/Li cell at a current density of 4 C; (D) rate capabilities of the LiFePO4/LiPF6@PAF-1/Li cell; (E) voltage profiles versus time of LiFePO4/LiPF6@PAF-1/Li cell at different current densities; (F) cycling performance of Li/LiFePO4 cells with other SSEs and LiPF6@PAF-1 in long term cycles(first 100 cycles).Copyright 2020, Wiley-VCH.
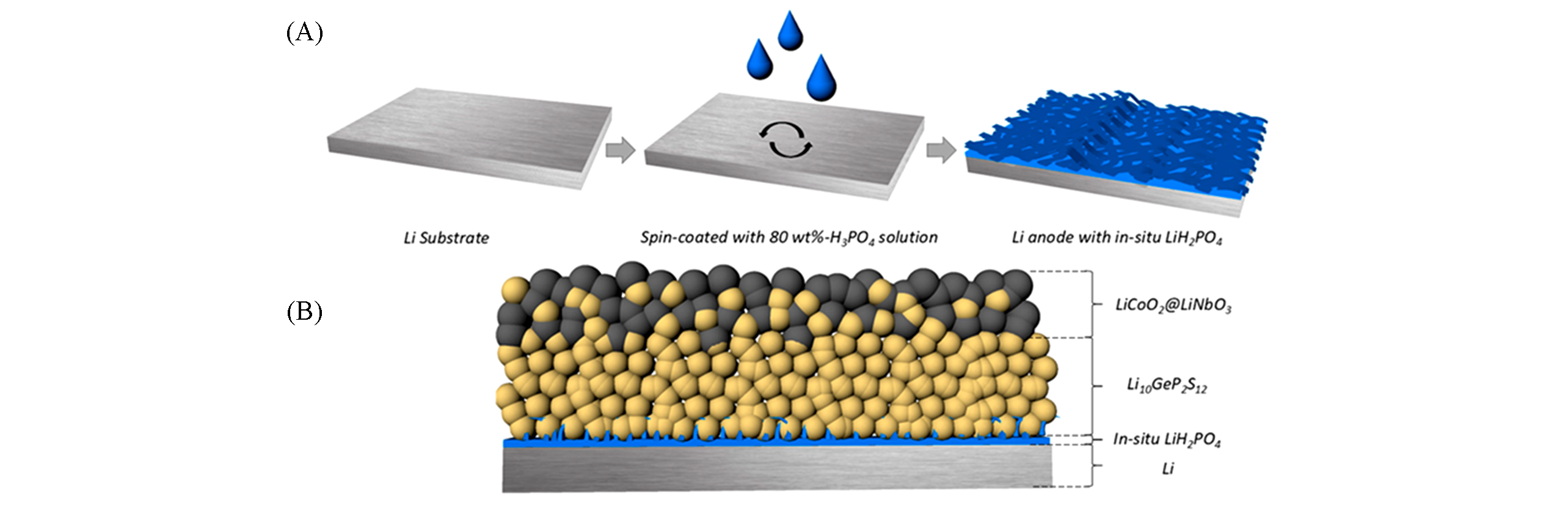
Fig.10 Schematic of the preparation process of in situ LiH2PO4 protective layer(A) and the LCO/LGPS/LiH2PO4?Li ASSLIB with optimized structure(B)[52]Copyright 2018, American Chemical Society.
| 1 | Zhang S., Wang S. F., Ling S. G., Gao J., Wu J. Y., Xiao R. J., Li H., Chen L. Q., Energy Storage Science and Technology, 2014, 3(4), 376—394(张舒, 王少飞, 凌仕刚, 高健, 吴娇杨, 肖睿娟, 李泓, 陈立泉. 储能科学与技术, 2014, 3(4), 376— 394) |
| 2 | Chen M., Xiao J., Hua W., Hu Z., Wang W., Gu Q., Tang Y., Chou S., Liu H., Dou S., Angew. Chem. Int. Ed., 2020, 59, 12076—12083 |
| 3 | Chen H., Tu H., Hu C., Liu Y., Dong D., Sun Y., Dai Y., Wang S., Qian H., Lin Z., Chen L., J. Am. Chem. Soc., 2018, 140, 896—899 |
| 4 | Huo H., Wu B., Zhang T., Zheng X., Ge L., Xu T., Guo X., Sun X., Energy Stor. Mater., 2019, 18, 59—67 |
| 5 | Zhang Q., Yao X. Y., Zhang H. Z., Zhang L. Q., Xu X. X., Energy Storage Science and Technology, 2016, 5(5), 659—667(张强, 姚霞银, 张洪周, 张联齐, 许晓雄. 储能科学与技术, 2016, 5(5), 659—667) |
| 6 | Thangadurai V., Narayanan S., Pinzaru D., Chem. Soc. Rev., 2014, 43, 4714—4727 |
| 7 | Cox E. G., Structural Inorganic Chemistry, 4th Ed., Clarendon Press, Oxford, 1951, Vol. 168 |
| 8 | Zheng B. Z., Wang H. C., Ma J. L., Gong Z. L., Yang Y., Scientia Sinica Chimica, 2017, 47(5), 579—593(郑碧珠, 王红春, 马嘉林, 龚正良, 杨勇. 中国科学: 化学, 2017, 47(5), 579—593) |
| 9 | Han X., Gong Y., Fu K., He X., Hitz G. T., Dai J., Pearse A., Liu B., Wang H., Rubloff G., Mo Y., Thangadurai V., Wachsman E. D., Hu L., Nat. Mater., 2017, 16, 572—579 |
| 10 | Wang C., Gong Y., Liu B., Fu K., Yao Y., Hitz E., Li Y., Dai J., Xu S., Luo W., Wachsman E. D., Hu L., Nano Lett., 2017, 17, 565—571 |
| 11 | Fu K. K., Gong Y., Liu B., Zhu Y., Xu S., Yao Y., Luo W., Wang C., Lacey S. D., Dai J., Sci. Adv., 2017, 3, e1601659 |
| 12 | Luo W., Gong Y., Zhu Y., Fu K. K., Dai J., Lacey S. D., Wang C., Liu B. Han X., Mo Y., Wachsman E. D., Hu L., J. Am. Chem. Soc., 2016, 138, 12258—12262 |
| 13 | Tsai C. L., Roddatis V., Chandran C. V., Ma Q., Uhlenbruck S., Bram M., Heitjans P., Guillon O., ACS Appl. Mater. Interfaces, 2016, 8, 10617—10626 |
| 14 | Wakasugi J., Munakata H., Kanamura K., J. Electrochem. Soc., 2017, 164, A1022—A1025 |
| 15 | Wang C., Xie H., Zhang L., Gong Y., Pastel G., Dai J., Liu B., Wachsman E. D., Hu L., Adv. Energy Mater., 2018, 8, 1701963 |
| 16 | Sharafi A., Kazyak E., Davis A. L., Yu S., Thompson T., Siegel D. J., Dasgupta N. P., Sakamoto J., Chem. Mater., 2017, 29, 7961—7968 |
| 17 | Li Y., Chen X., Dolocan A., Cui Z., Xin S., Xue L., Xu H., Park K., Goodenough J. B., J. Am. Chem. Soc., 2018, 140, 6448— 6455 |
| 18 | Wang C., Xie H., Ping W., Dai J., Feng G., Yao Y., He S., Weaver J., Wang H., Gaskell K., Hu L., Energy Storage Mater., 2019, 17, 234—241 |
| 19 | Wu J. F., Pu B., W., Wang D., Shi S. Q., Zhao N., Guo X., Guo X., ACS Appl. Mater. Interfaces, 2019, 11, 898—905 |
| 20 | Zhang Z. Z., Synthesis and Modification of NASICON⁃typed Na3Zr2Si2PO12 Solid State Electrolytes, Zhejiang Sci⁃Tech University, Hangzhou, 2013(章志珍. NASICON结构固态电解质Na3Zr2Si2PO12的制备与改性, 杭州: 浙江理工大学, 2013) |
| 21 | Zhou W., Gao H., Goodenough J. B., Advanced Energy Materials, 2016, 6(1), 1501802 |
| 22 | Fenton D. E., Parker J. M., Wright P. V., Polymer, 1973, 14, 589 |
| 23 | Takada K., Acta Materialia, 2013, 61, 759—770 |
| 24 | Sun Y., Liu N., Cui Y., Nature Energy, 2016, 1, 16071 |
| 25 | Fang H., Jena P., ACS Appl. Mater. Interfaces, 2018, 12, 89—103 |
| 26 | Park S. S., Tulchinsky Y., Dinca M., J. Am. Chem. Soc., 2017, 139, 13260—13263 |
| 27 | Tao C., Gao M. H., Yin B. H., Li B., Huang Y. P., Xu G., Bao J. J., Electrochim. Acta, 2017, 257, 31—39 |
| 28 | Thiam A., Martinez⁃Cisneros C., Molméret Y., Iojoiu C., Sanchez J. Y., Electrochim. Acta, 2019, 302, 338—343 |
| 29 | Xie H., Yang C., Fu K. K., Yao Y., Jiang F., Hitz E., Liu B., Wang S., Hu L., Adv. Energy Mater., 2018, 8, 1703474 |
| 30 | Chen L., Li Y., Li S., Fan L., Nan C., Goodenough J. B., Nano Energy, 2018, 46, 176—184 |
| 31 | Shen B., Zhang T., Yin Y., Zhu Z., Lu L., Ma C., Zhou F., Yao H., Chem. Comm., 2019, 55, 7703—7706 |
| 32 | Shen B., Surface Coated Polyethylene Oxide Solid Electrolyte: Fabrication and Performance Study, University of Science and Technology of China, Hefei, 2019(沈豹. 聚氧乙烯固态电解质的表面修饰, 合肥: 中国科学技术大学, 2019) |
| 33 | Wenzel S., Randau S., Leichtweiss T., Weber D. A., Sann J., Zeier W.G., Janek J., Chem. Mater., 2016, 28, 2400—2407 |
| 34 | Schwobel A., Hausbrand R., Jaegermann W., Solid State Ionics, 2015, 273, 51—54 |
| 35 | Tang Y., Zhang Y., Malyi O. I., Bucher N., Xia H., Xi S., Zhu Z., Lv Z., Li W., Wei J., Srinivasan M., Borgna A., Antonietti M., Du Y., Chen X., Adv. Mater., 2018, 30, 1802200 |
| 36 | Li H., Mo Y., Pei N., Xu X., Huang X., Chen L., J. Phys. Chem. B, 2000, 104(35), 8477—8480 |
| 37 | Yao X. Y., Huang B. X., Yin J. Y., Peng G., Huang Z., Gao C., Liu D., Xu X. X., Chinese Physics B, 2016, 25(1), 018802 |
| 38 | Okumura T., Nakatsutsumi T., Ina T., Orikasa Y., Arai H., Fukutsuka T., Iriyama Y., Uruga T., Tanida H., Uchimoto Y., Ogumi Z., J. Mater. Chem., 2011, 21(27), 10051—10060 |
| 39 | Kato Y., Hori S., Saito T., Suzuki K., Hirayama M., Mitsui A., Yonemura M., Iba H., Kanno R., Nat. Energy, 2016, 1, 16030 |
| 40 | Fan Q., Yang S., Liu J., Liu H., Lin K., Liu R., Hong C., Liu L., Chen Y., An K., Liu P., Shi Z., Yang Y., J. Power Sources, 2019, 421, 91—99 |
| 41 | Park K., Yu B. C., Jung J. W., Li Y., Zhou W., Gao H., Son S., Goodenough J. B., Chem. Mater., 2016, 28, 8051— 8059 |
| 42 | Han F., Yue J., Chen C., Zhao N., Fan X., Ma Z., Gao T., Wang F., Guo X., Wang C., Joule, 2018, 2, 497—508 |
| 43 | Chen X., He W., Ding L. X., Wang S., Wang H., Energy Environ. Sci., 2019, 12, 938—944 |
| 44 | Zou J., Trewin A., Ben T., Qiu S., Angew. Chem., 2020, 132, 779—784 |
| 45 | Zhang Y., Tang Y., Deng J., Leow W. R., Xia H., Zhu Z., Lv Z., Wei J., Li W., Persson C., Malyi O. I., Antonietti M., Chen X., ACS Mater. Lett., 2019, 1, 519—525 |
| 46 | Zhou W., Wang S., Li Y., Xin S., Manthiram A., Goodenough J. B., J. Am. Chem. Soc., 2016, 138, 9385—9388 |
| 47 | Chen X., He W., Ding L. X., Wang S., Wang H., Energy Environ. Sci., 2019, 12, 938—944 |
| 48 | Pan Q., Zheng Y., Kota S., Huang W., Wang S., Li C. Y., Nanoscale Adv., 2019, 1, 395—402 |
| 49 | Wan Z., Lei D., Yang W., Liu C., Shi K., Hao X., Shen L., Lv W., Li B., Yang Q., Kang F., He Y., Adv. Funct. Mater., 2018, 28, 1805301 |
| 50 | Lei Y. Q., New Energy Material, Tianjin University Press, Tianjin, 2000(雷永泉. 新能源材料, 天津: 天津大学出版社, 2000) |
| 51 | Ogawa M., Kanda R., Yoshida K., Uemura T., Harada K., J. Power Sources, 2012, 205(205), 487—490 |
| 52 | Zhang Z., Chen S., Yang J., Wang J., Yao L., Yao X., Cui P., Xu X., ACS Appl. Mater. Interfaces, 2018, 10(3), 2556— 2565 |
| 53 | Cai W., Yan C., Yao Y., Xu L., Xu R., Jiang L., Huang J., Zhang Q., Small Structures, 2020, 1, 2000010 |
| [1] | 侯从聪, 王惠颖, 李婷婷, 张志明, 常春蕊, 安立宝. N-CNTs/NiCo-LDH复合材料的制备及电化学性能[J]. 高等学校化学学报, 2022, 43(10): 20220351. |
| [2] | 王增强, 孙一翎, 钱正芳, 王任衡. 基于表界面反应及优化的锂金属电池研究进展[J]. 高等学校化学学报, 2021, 42(4): 1017. |
| [3] | 周墨林, 蒋欣, 易婷, 杨向光, 张一波. 硫化物固态电解质Li10GeP2S12与锂金属间界面稳定性的改善研究[J]. 高等学校化学学报, 2020, 41(8): 1810. |
| [4] | 刘奔, 张行颖, 陈韶云, 胡成龙. 一维有序聚苯胺纳米阵列的制备及电化学储能性能[J]. 高等学校化学学报, 2019, 40(3): 498. |
| [5] | 李龙, 胡红利, 丁书江. 鳞状CoMn2O4/石墨烯复合材料的制备及在超级电容器中的应用[J]. 高等学校化学学报, 2018, 39(9): 2010. |
| [6] | 乜广弟, 朱云, 田地, 王策. 静电纺丝纳米纤维基超级电容器电极材料的研究进展[J]. 高等学校化学学报, 2018, 39(7): 1349. |
| [7] | 王存国, 潘璇, 张雷, 朱孟康, 李德凯, 刁玲博, 李伟彦. 高容量锂离子电池核壳型硅/碳复合电极材料的制备与性能[J]. 高等学校化学学报, 2015, 36(2): 368. |
| [8] | 陈春阳, 于飞, 周慧明, 陈君红, 马杰. 三维石墨烯凝胶电极的制备及在电容去离子中的应用[J]. 高等学校化学学报, 2015, 36(12): 2516. |
| [9] | 付昱, 孙立, 田春贵, 林海波. 炭/多壁碳纳米管复合材料的制备与超级电容性能[J]. 高等学校化学学报, 2013, 34(10): 2389. |
| [10] | 吴娟, 吴雪艳, 魏霄, 王开学, 陈接胜. 聚苯胺/介孔碳纳米线复合电极材料的制备和性能[J]. 高等学校化学学报, 2012, 33(07): 1540. |
| [11] | 王焕磊 高秋明. 多孔碳材料的模板法制备、活化处理及储能应用[J]. 高等学校化学学报, 2011, 32(3): 462. |
| [12] | 张金娜, 赵庆良, 尤世界, 张国栋. 生物阴极微生物燃料电池不同阴极材料产电特性[J]. 高等学校化学学报, 2010, 31(1): 162. |
| [13] | 王美涵, 张连中, 孙立贤, 张耀, 徐芬, 谭志诚, 袁华堂, 张涛. Mg1.75Al0.25Ni1-xCrx(0 ≤x≤ 0.2)合金的制备及其性能[J]. 高等学校化学学报, 2005, 26(10): 1877. |
| [14] | 夏熙, 李娟, 李清文. 纳米MnO2的固相合成及其电化学性能研究(Ⅱ)纳米V-MnO2的电化学性能[J]. 高等学校化学学报, 1999, 20(10): 1584. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||