

高等学校化学学报 ›› 2021, Vol. 42 ›› Issue (4): 1093.doi: 10.7503/cjcu20200597
收稿日期:2020-08-24
出版日期:2021-04-10
发布日期:2021-03-01
通讯作者:
杨辉
E-mail:yanghui2018@tju.edu.cn
基金资助:
ZHANG Jun, LIU Yixuan, DU Xiaohui, YANG Hui( )
)
Received:2020-08-24
Online:2021-04-10
Published:2021-03-01
Contact:
YANG Hui
E-mail:yanghui2018@tju.edu.cn
Supported by:摘要:
随着柔性电子技术和人工智能的蓬勃发展, 将柔性传感器与人工智能、 大数据和5G通讯有机结合, 能够构建以人为中心的智能人机交互系统, 在智能健康监测和生物医疗等领域发挥越来越重要的作用. 作为连接人体信号与网络空间的人机交互界面, 皮肤电极的力学性能直接影响监测信号的灵敏度和稳定性. 从材料的角度出发, 如何同时实现电极的可拉伸性和高黏附性, 让其稳定地贴敷于皮肤表面, 成为皮肤电极进一步应用亟待解决的科学难题和技术挑战. 本文综合评述了近年来高黏附可拉伸高分子材料的研究进展及在人机交互界面的各种应用, 并展望了下一代智能人机交互界面的发展趋势.
中图分类号:
TrendMD:
张鋆, 刘忆旋, 杜晓慧, 杨辉. 基于高黏附可拉伸高分子材料的人机交互界面. 高等学校化学学报, 2021, 42(4): 1093.
ZHANG Jun, LIU Yixuan, DU Xiaohui, YANG Hui. Highly Adhesive and Stretchable Polymers for the Interface of Cyber-human Interaction. Chem. J. Chinese Universities, 2021, 42(4): 1093.
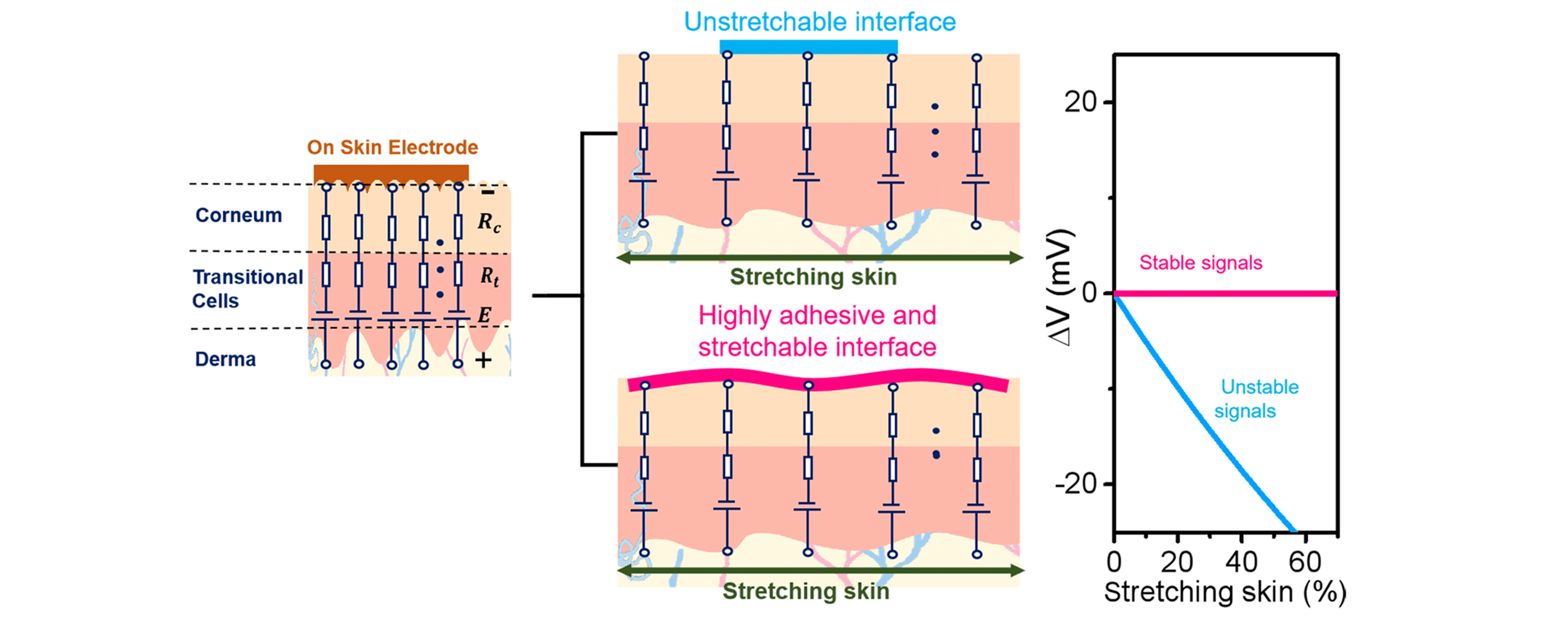
Fig.1 Adhesive and stretchable property of on?skin electrode for enhancing the stability of human signal detection[9]Copyright 2020, American Chemical Society.
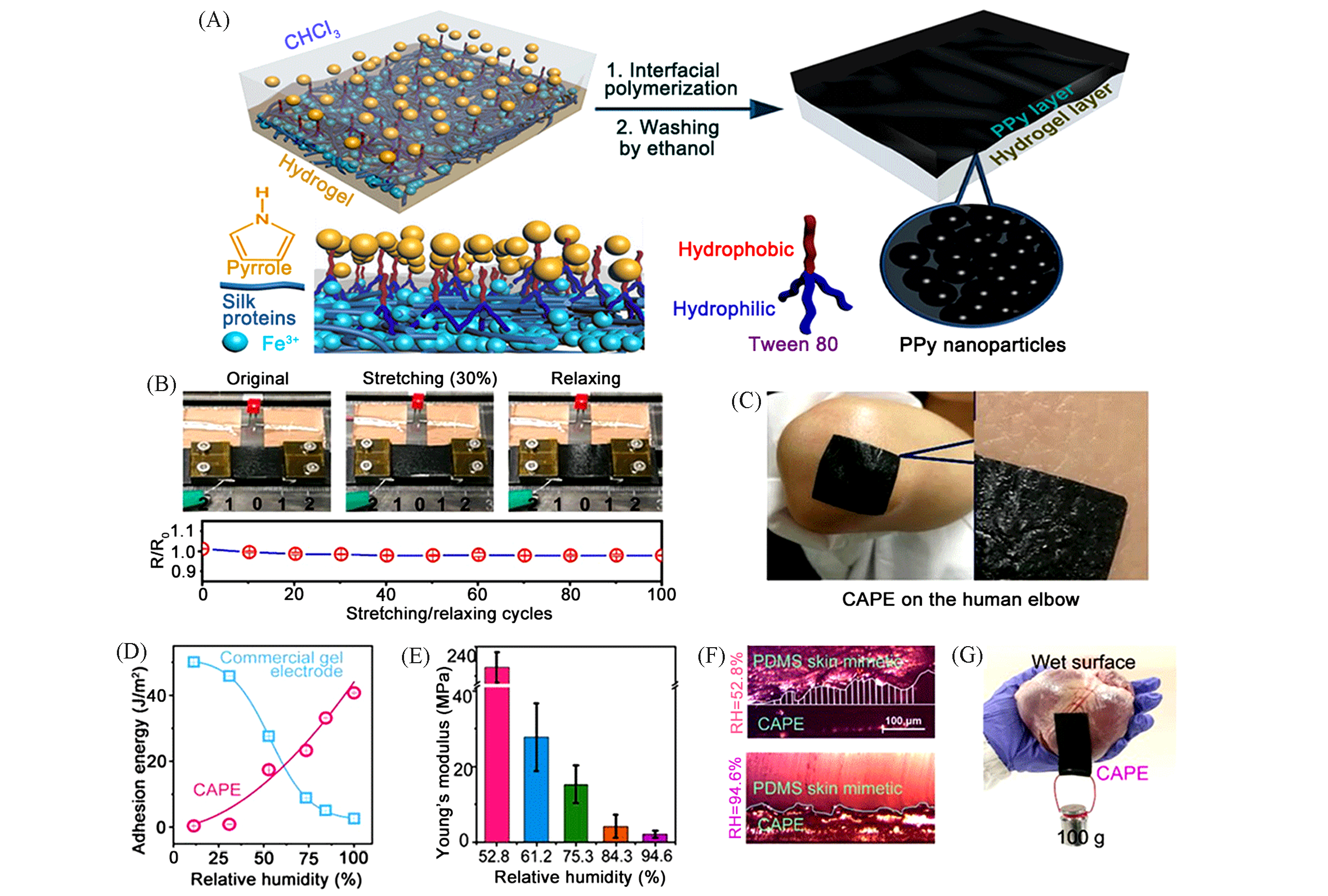
Fig.2 Bio?composite conformal electrodes based on SF and PPy(CAPE)[9](A) The preparation process of CAPE; (B) the stable conductivity of CAPE with 20%(mass fraction) of FeCl3 upon being stretched to 30% and cycle stretched, LED light using CAPE as a stretchable conductor retained its luminescence when stretching and relaxing; (C) the highly conformal property of CAPE on the human elbow; (D) interfacial adhesion energy of CAPE increased with increasing RH from 11% to 94.6%, compared with commercial gel electrodes; (E) Young’s modulus of CAPE decreased with increasing RH; (F) the increase of contact area between CAPE and PDMS like skin with increasing RH(the shade indicates void space); (G) the strong interfacial adhesion of CAPE on the wet surface of a pig heart.Copyright 2020, American Chemical Society.
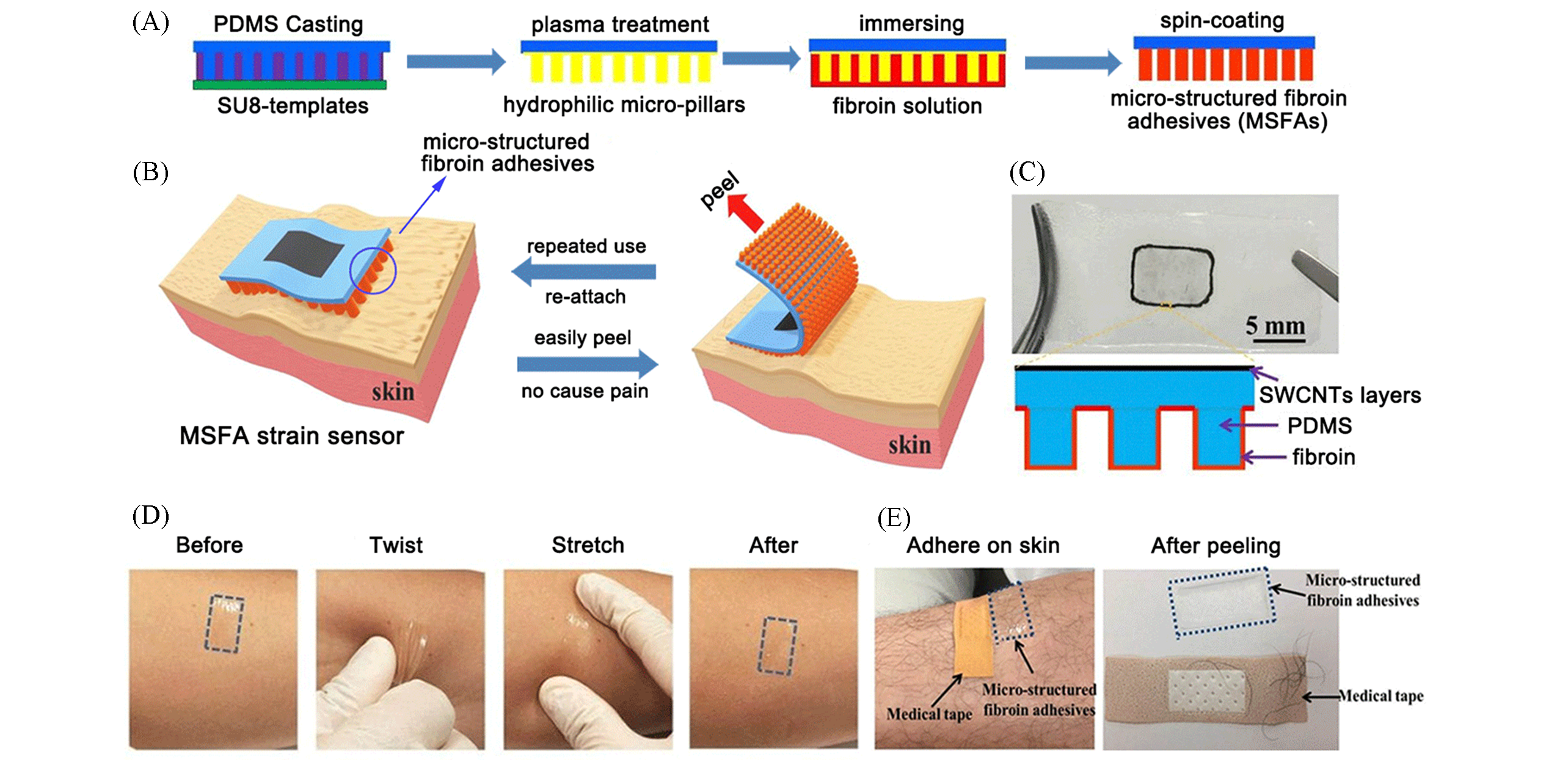
Fig.3 Micro?structured silk fibroin adhesives for flexible skin sensors[45](A) Fabrication process of micro?structured fibroin adhesive(MSFA): preparation of the PDMS micro?pillars film, plasma treatment and silk fibroin coating; (B) MSFA strain sensor can be easily peeled off from and re?attached to the skin for repeating use; (C) a photo of the flexible strain sensor with the MSFA(upper) and the cross section schematic of the sensor(lower); (D) photos of an MSFA before and after twisting, compression and stretching on human skin; (E) commercial medical tape can lead to hair stripping during its removal process while the MSFA did not strip any hair during the peeling process.Copyright 2020, American Chemical Society.
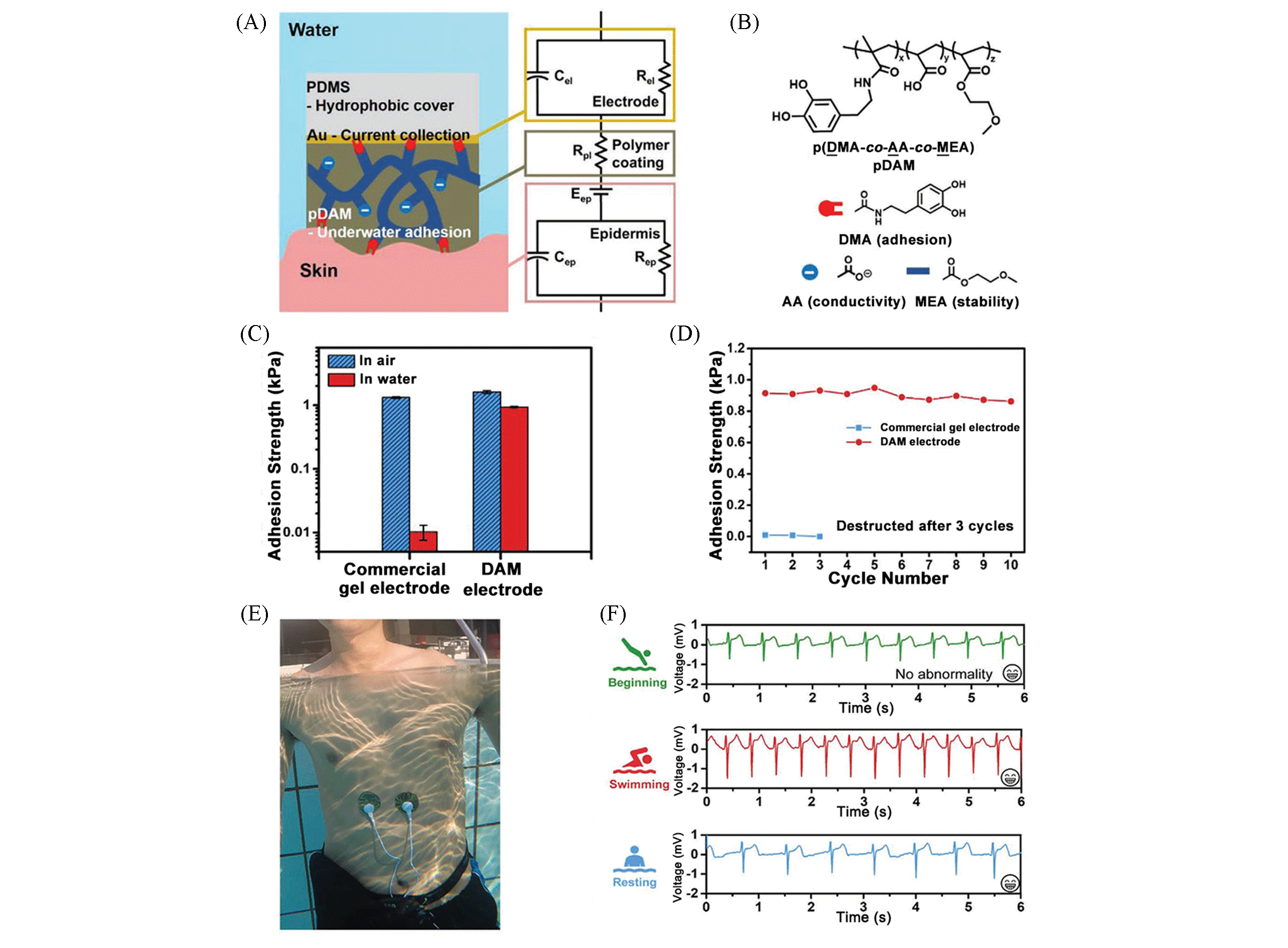
Fig.4 Water?resistant conformal hybrid electrodes based on pDAM polymer[49](A) Schematic showing the water?resistant pDAM polymer coating bridging the Au/PDMS electrode and skin(left), and the corresponding circuit(right); (B) chemical structures of pDAM, and the corresponding symbols used in (A); (C) adhesion strength tested on PDMS?skin model in air and in water; (D) adhesion strength during underwater paste and peel off cycles; (E) photo of the device with DAM electrodes in water; (F) ECG signals recorded by the device in water before swimming, during swimming, and in water after swimming and resting. Copyright 2020, Wiley?VCH.
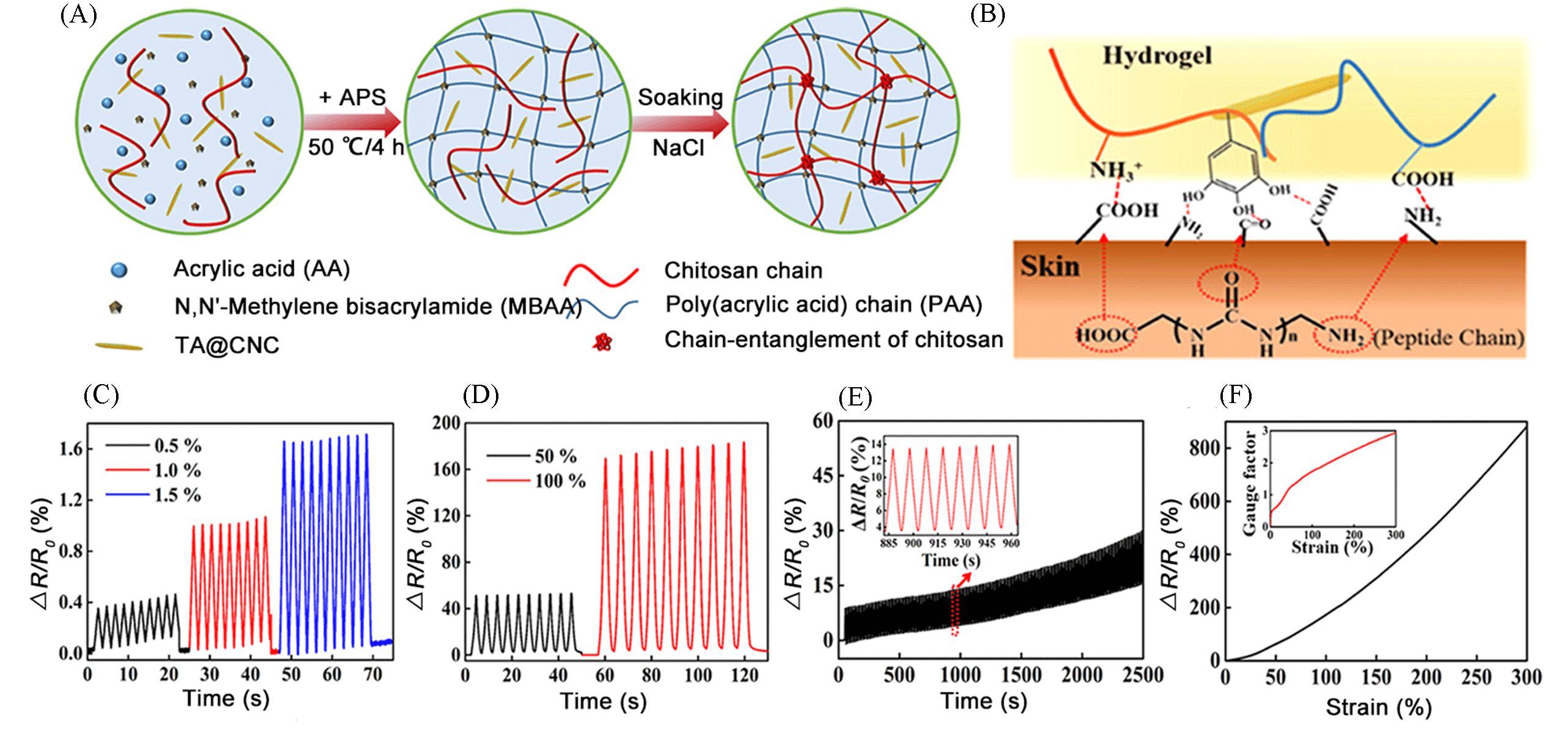
Fig.5 Chitosan/poly(acrylic acid) double?network nanocomposite hydrogels for flexible sensors[54](A) The preparation process of CS/PAA/TA@CNC hydrogels; (B) schematic illustration of possible adhesion mechanism; (C, D) time?dependent relative resistance changes of the sensors at small strains(0.5%, 1.0%, and 1.5%)(C, D) and large strains(50 and 100%) under the applied voltage of 5 V(D); (E) stability of relative resistance changes of the sensors by repeatedly applying 20% strain for sensors under applied voltage of 5 V; (F) relative resistance change of sensor as a function of the strain, and the effects of various applied strain on the gauge factor.Copyright 2019, American Chemical Society.
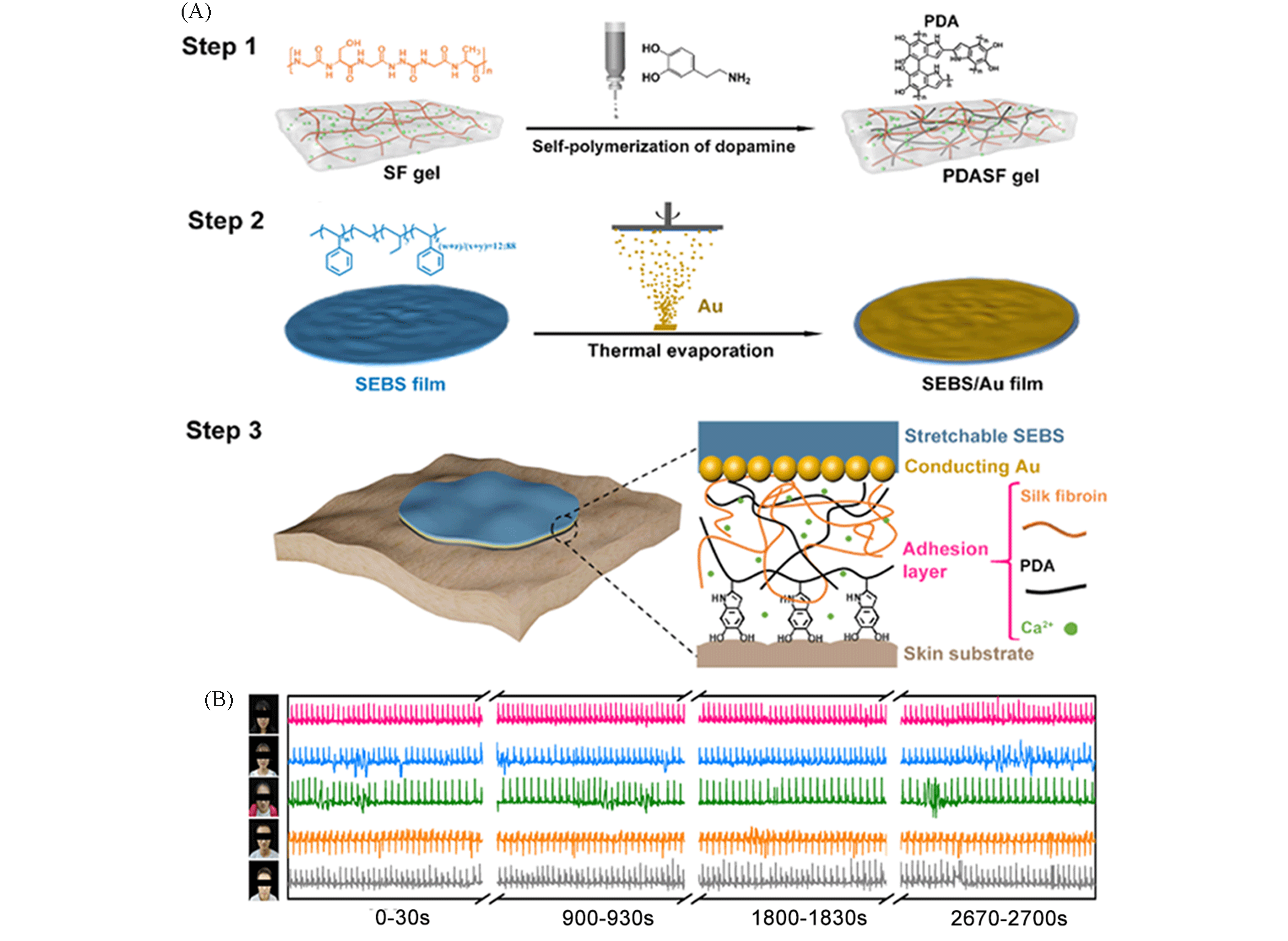
Fig.6 Highly adhesive and stretchable AWS electrodes based on polydopamine and silk fibroin[55](A) Design strategy for the highly skin?adhesive and washable AWS electrodes. (B) the demonstration of the universality of AWS electrodes through sensing different ambulatory ECG signals of five volunteers.Copyright 2020, the Royal Society of Chemistry.
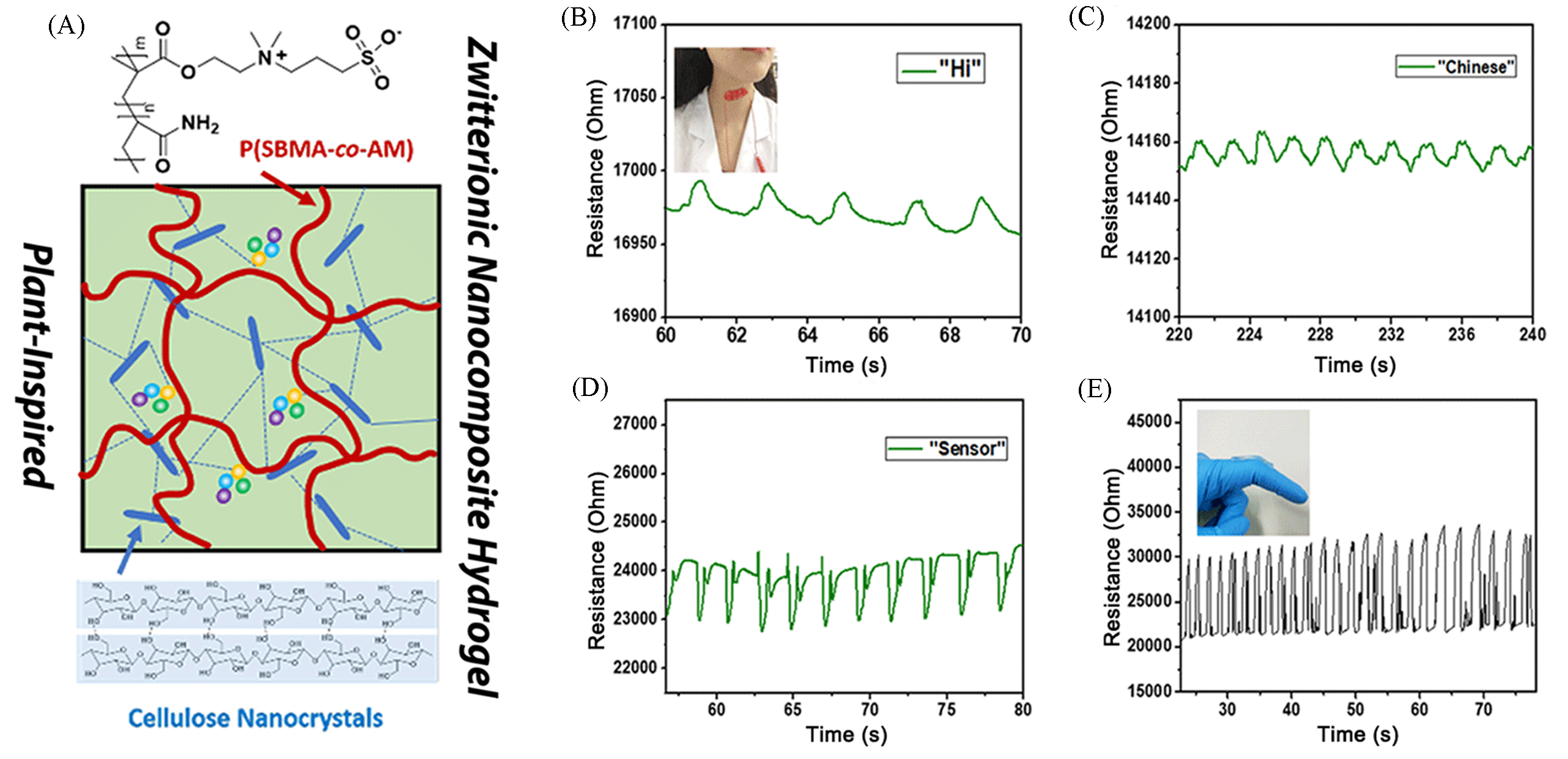
Fig.7 Highly stretchable and adhesive zwitterionic nanocomposite hydrogel[61](A) Schematic illustration of CNC@P(SBMA?co?AM) zwitterionic nanocomposite hydrogels. (B—E) Photographs of the hydrogel?based pressure sensor during monitoring tiny physical motions: pronounce distinction of “Hi”(B), “Chinese”(C), and “ensor”(D) and continuous finger bending(E).Copyright 2019, American Chemical Society.
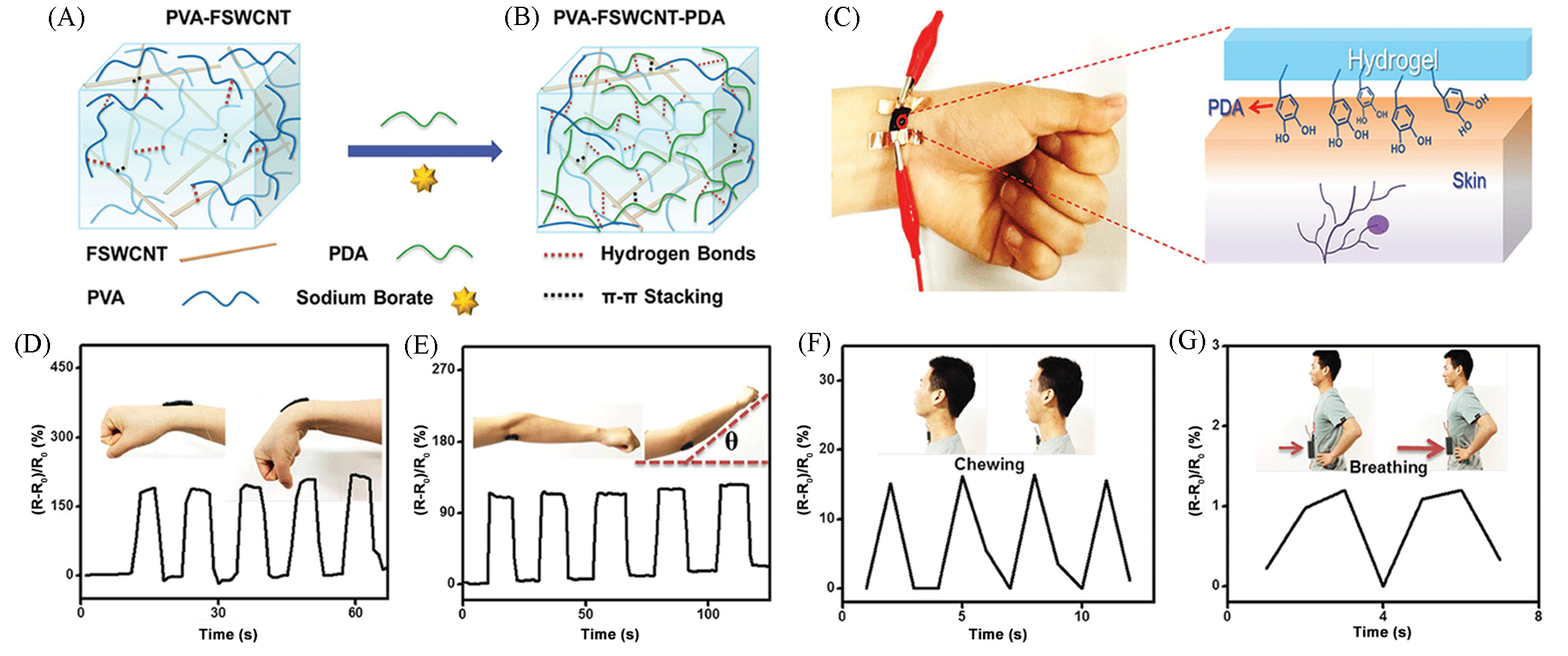
Fig.8 Wearable, healable and adhesive mussel?inspired conductive hybrid hydrogel[70](A, B) Schematic representation for the fabrication of the PVA?FSWCNT?PDA hydrogel; (C) the PVA?FSWCNT?PDA hydrogel strain sensors could be self?adhered on the wrist; (D―G) the recorded resistance variations of the PVA?FSWCNT?PDA hydrogel strain sensors in response to the opisthenar(D) and elbow(E), chewing(F) and breathing(G).Copyright 2017, Wiley?VCH.
| Type | Strategy | Material | Mechanical performance | Adhesive strength | Biocompati bility | Ref. |
|---|---|---|---|---|---|---|
Silk | Interfacial polymerization | SF and PPy | Tensile strain=300% | Adhesion energy≈40 J/m2; (94.6% RH) to PDMS?skin model | Good | [ |
| Plasticized silk protein | SF with 30% CaCl2 | Tensile strain>400% | Good | [ | ||
| SF with 20% CaCl2 | Peel forces≈10 N/m | Good | [ | |||
Micro?pillars structure | SF and PDMS | Adhesion energy≈4.83 J/m2 to glass | Good | [ | ||
| Polydopamine | DOPA?based copolymer | P(DMA?co?AA?coMEA) | ≈1 kPa to wet PDMS?skin model | Good | [ | |
| Nanoclay confined dopaminepolymerization | PDA?clay?PAM hydrogel | Tensile strain=4800% | ≈28.5 kPa to porcine skin | Good | [ | |
Microgel?tackified hydrogel | PAAc?PAM?PDA | Tensile strain=660% | ≈15 kPa to various substrates | Acceptable | [ | |
| Double?network nanocomposite hydrogels | CS/PAA/TA@CNC | Tensile strain=800% | ≈20 kPa to glass | Good | [ | |
| Self?polymerization of dopamine in silk fibroin | PDA and SF | Tensile strain=320% | ≈9.87 MPa to wet PDMS?skin model(100% RH) | Good | [ | |
| Zwitterionic nanocomposite hydrogels | Free radical polymerization | P(SBMA?co?HEMA) | Tensile strain=2000% | Lap shear adhesion≈17.6 kPa to glass | Acceptable | [ |
| CNC@P(SBMA?co?AM) | Tensile strain=1127% | Lap shear adhesion≈10.5 kPa to porcine skin | Good | [ | ||
| Dry?crosslin?king mechanism | Dry double?sided tape | Gelatin or chitosan and cross?linked PAAc?NHS ester | Fracture toughness=1000 J/m2, fracture strain >16 | Lap shear adhesion≈120 kPa to wet porcine skin | Good | [ |
Self?Healing hydrogels | Covalent and non? covalent interactions | Cross?linked PAA chains and DHA polymer | Tensile strain=800% | ≈12.6 kPa to porcine skin | Acceptable | [ |
| Nanocomposite network hydrogels | PVA?FSWCNT?PDA | ≈5.2 kPa to porcine skin | Good | [ |
Table 1 Summary of highly adhesive and stretchable polymers using different strategies
| Type | Strategy | Material | Mechanical performance | Adhesive strength | Biocompati bility | Ref. |
|---|---|---|---|---|---|---|
Silk | Interfacial polymerization | SF and PPy | Tensile strain=300% | Adhesion energy≈40 J/m2; (94.6% RH) to PDMS?skin model | Good | [ |
| Plasticized silk protein | SF with 30% CaCl2 | Tensile strain>400% | Good | [ | ||
| SF with 20% CaCl2 | Peel forces≈10 N/m | Good | [ | |||
Micro?pillars structure | SF and PDMS | Adhesion energy≈4.83 J/m2 to glass | Good | [ | ||
| Polydopamine | DOPA?based copolymer | P(DMA?co?AA?coMEA) | ≈1 kPa to wet PDMS?skin model | Good | [ | |
| Nanoclay confined dopaminepolymerization | PDA?clay?PAM hydrogel | Tensile strain=4800% | ≈28.5 kPa to porcine skin | Good | [ | |
Microgel?tackified hydrogel | PAAc?PAM?PDA | Tensile strain=660% | ≈15 kPa to various substrates | Acceptable | [ | |
| Double?network nanocomposite hydrogels | CS/PAA/TA@CNC | Tensile strain=800% | ≈20 kPa to glass | Good | [ | |
| Self?polymerization of dopamine in silk fibroin | PDA and SF | Tensile strain=320% | ≈9.87 MPa to wet PDMS?skin model(100% RH) | Good | [ | |
| Zwitterionic nanocomposite hydrogels | Free radical polymerization | P(SBMA?co?HEMA) | Tensile strain=2000% | Lap shear adhesion≈17.6 kPa to glass | Acceptable | [ |
| CNC@P(SBMA?co?AM) | Tensile strain=1127% | Lap shear adhesion≈10.5 kPa to porcine skin | Good | [ | ||
| Dry?crosslin?king mechanism | Dry double?sided tape | Gelatin or chitosan and cross?linked PAAc?NHS ester | Fracture toughness=1000 J/m2, fracture strain >16 | Lap shear adhesion≈120 kPa to wet porcine skin | Good | [ |
Self?Healing hydrogels | Covalent and non? covalent interactions | Cross?linked PAA chains and DHA polymer | Tensile strain=800% | ≈12.6 kPa to porcine skin | Acceptable | [ |
| Nanocomposite network hydrogels | PVA?FSWCNT?PDA | ≈5.2 kPa to porcine skin | Good | [ |
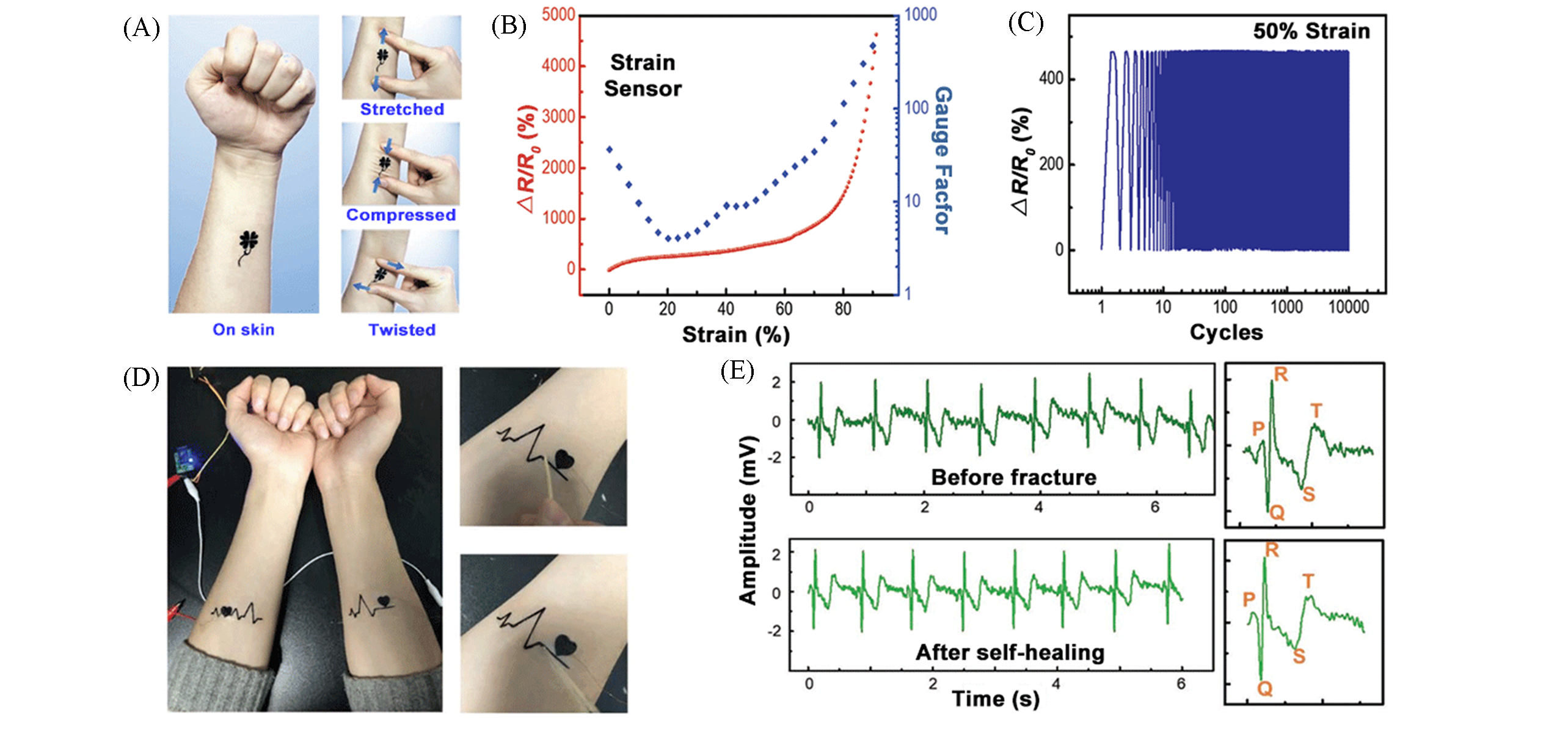
Fig.9 Gr/SF/Ca2+?based E?Tattoos[77](A) An E?tattoo attached to the forearm in different states: stretched, compressed, and twisted; (B) relative resistance changes as a function of applied tensile strain; (C) long?term stability of stretchable strain sensors upon exposure to 10000 tensile strain cycles at an applied strain of 50%; (D) a self?healed E?tattoo attached on the forearms for ECG measurements; (E) relative resistance changes of pristine and healed ECG sensors.Copyright 2019, Wiley?VCH.
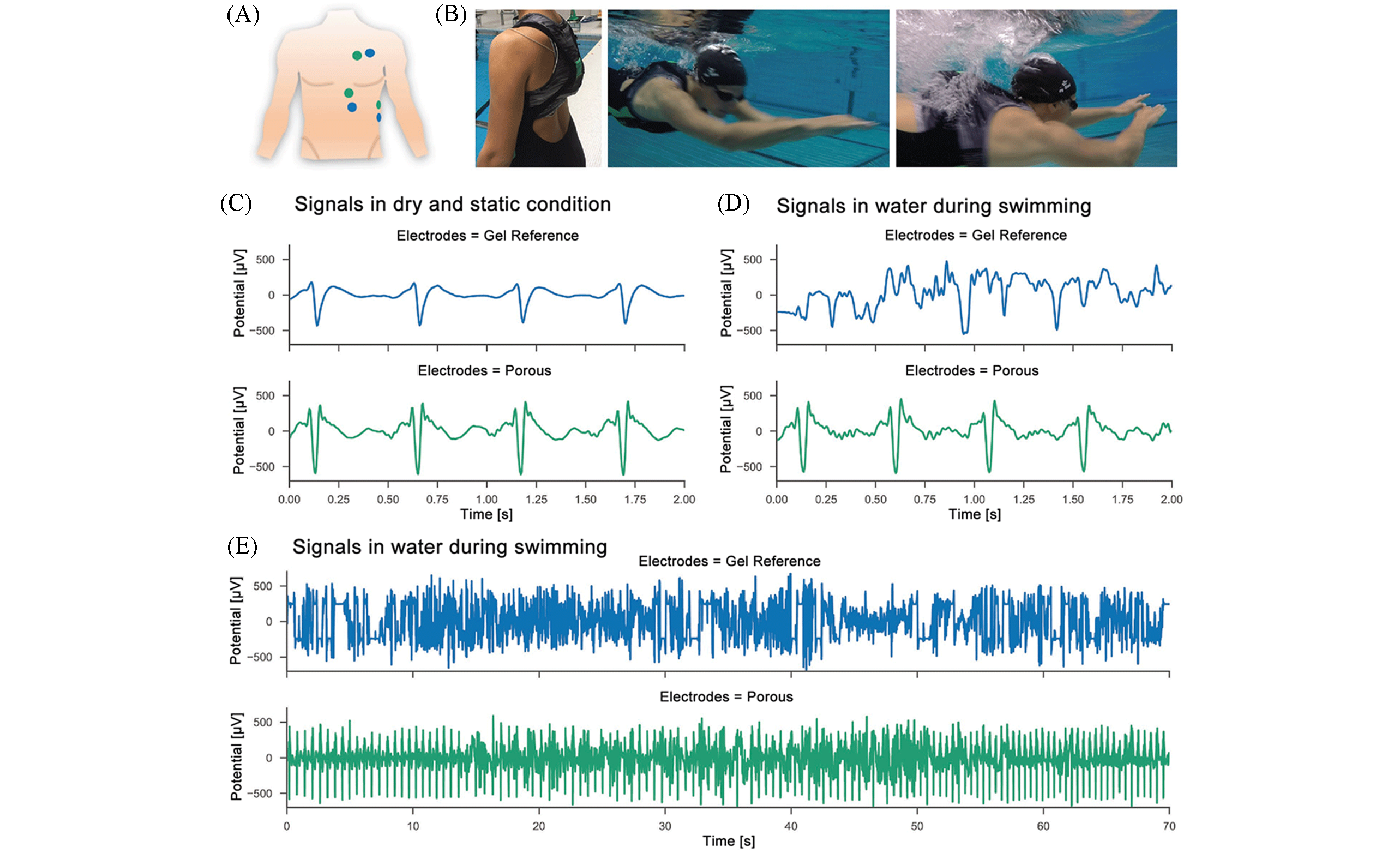
Fig.10 ECG recordings by soft polymer electrodes and the gel reference electrodes during swimming[81](A) Electrode placement of the soft polymer electrodes(green) and the gel reference electrodes(blue); (B) fixation of the recording device using textiles and the swimmer in action under water; (C) simultaneous ECG measurements in dry and static condition before swimming for both electrode types; (D) ECG recordings during swimming showed good signal quality for soft polymer electrodes; (E) ECG recordings for 70 s while swimming lanes with breaststrokes.Copyright 2018, Wiley?VCH.
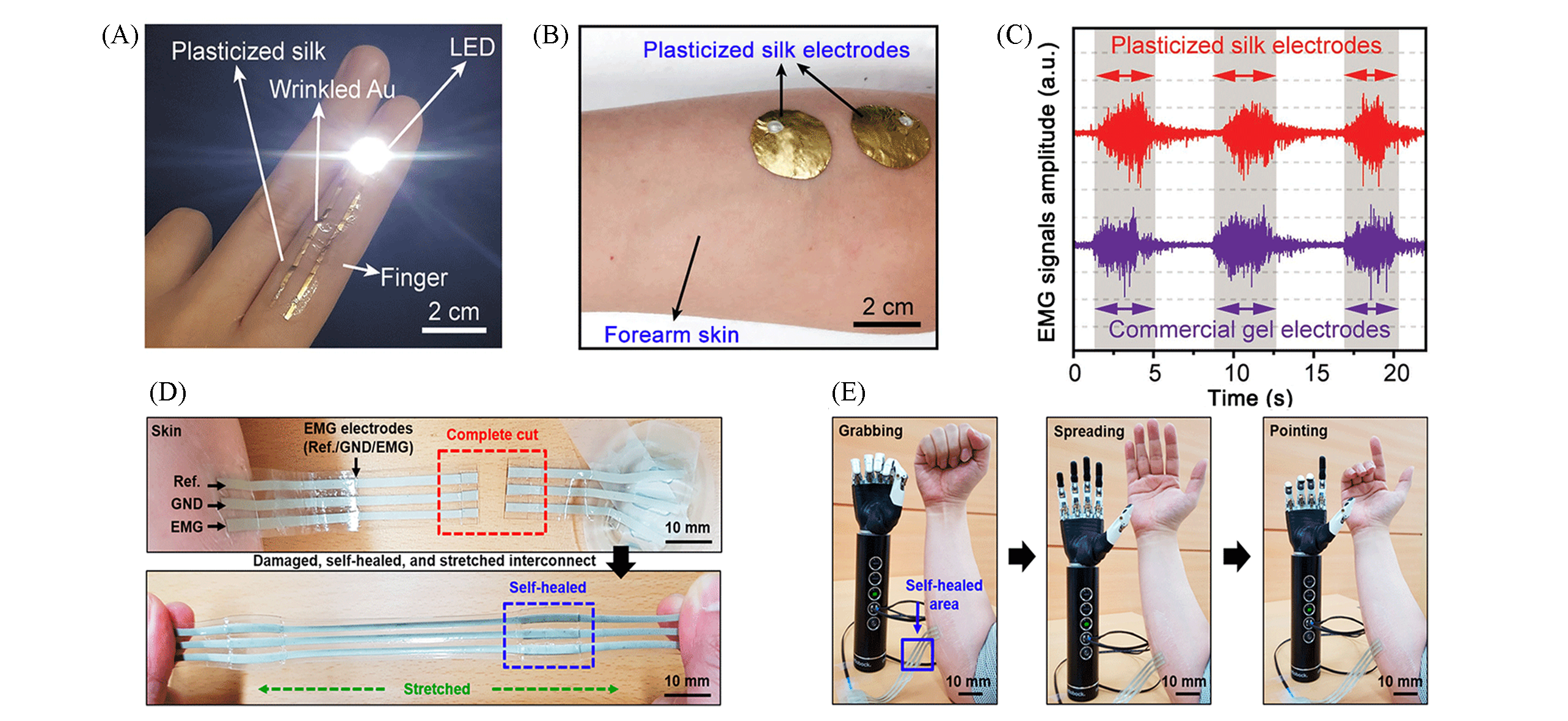
Fig.11 High conformability of plasticized silk electrodes(A—C)[41] and an ultrastretchable and self?Healable nanocomposite conductor(D, E)[82] for ambulatory EMG monitoring(A) The silk electrodes conformably attached on a finger skin; the conductance of the electrodes is as high as an LED can be operated in 2.7 V; (B) An EMG measurement setup using plasticized silk electrodes laminated on a human forearm; (C) EMG signals obtained by the plasticized silk electrodes and commercial gel electrodes; (D, E) robust interactive human?robot interface based on stretchable and self?healable interconnect: (D) photographs of the self?healable interconnect that is sequentially cut, self?healed, and stretched; (E) demonstration of human?robot interface using the self?healed interconnect after process (D).
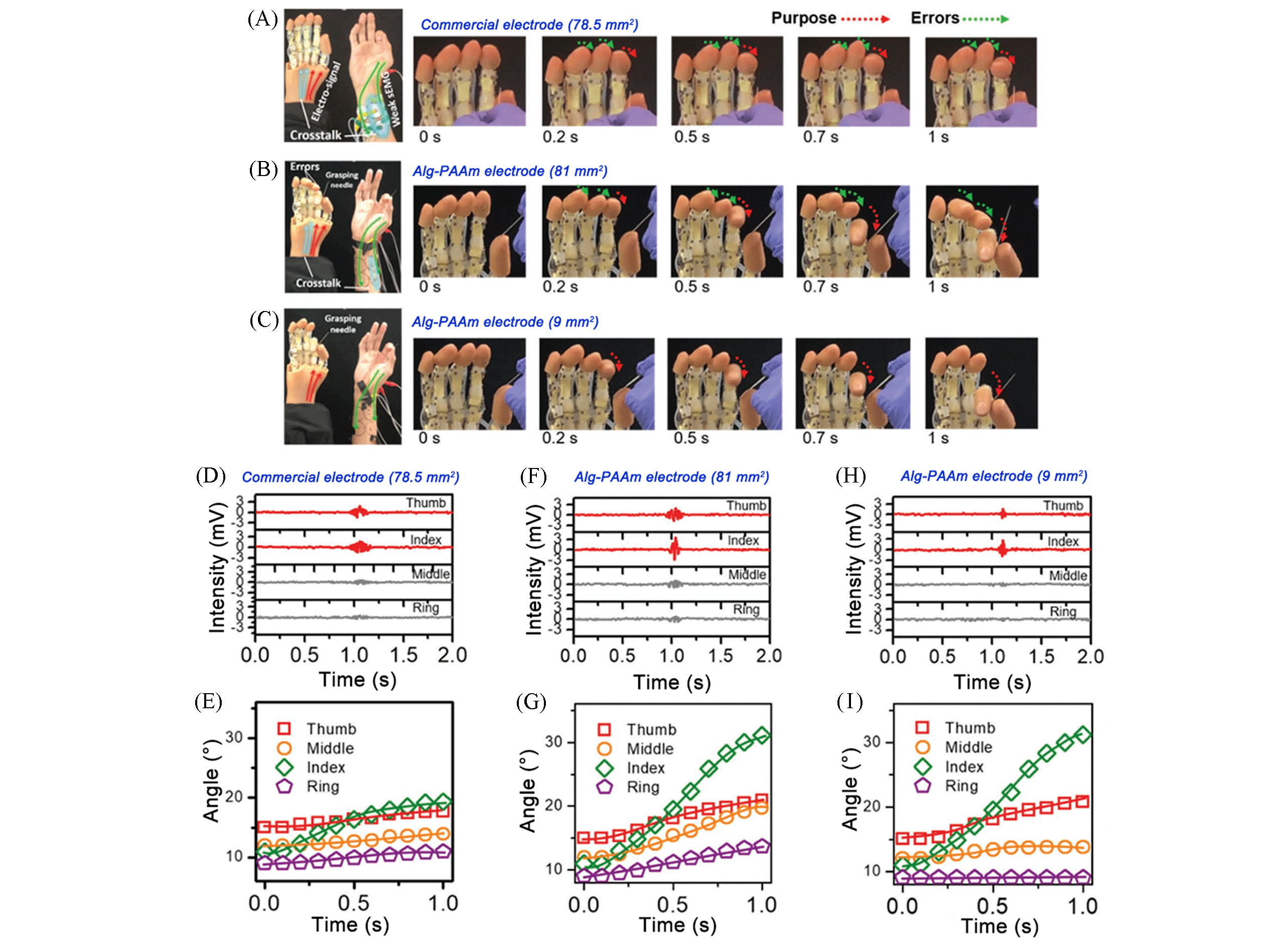
Fig.12 Driving the prosthetic hand to grasp a needle based on commercial and Alg?PAAm compliant electrodes and correlative sEMG signals[83](A—C) Needle grasping experiments based on commercial electrodes(A), 81 mm2 Alg?PAAm electrod(B), and 9 mm2 Alg?PAAm electrode(C); (D—I) sEMG data(D, F, H) and bending angles(E, G, I) obtained from four different fingers of the human hand while grasping a needle based on the commercial electrode(D, E), the 81 mm2 Alg?PAAm electrode(F, G), and the 9 mm2 Alg?PAAm electrode(H, I).Copyright 2020, Wiley?VCH.
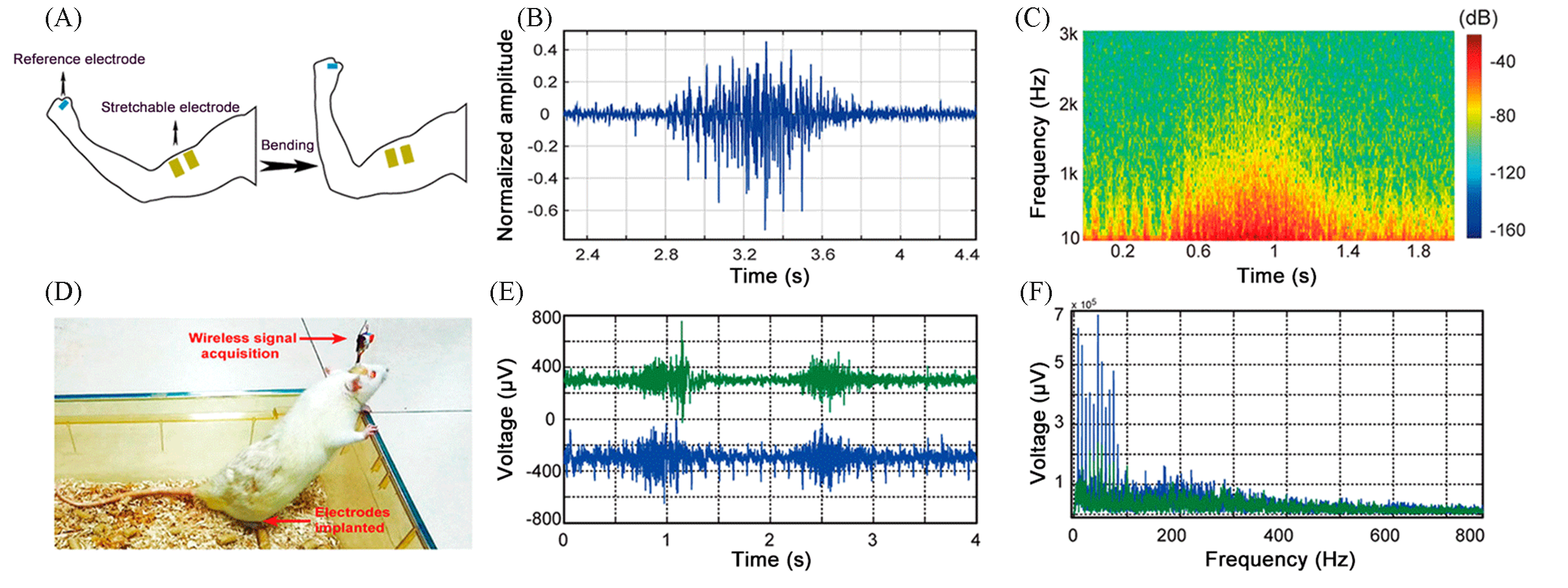
Fig.13 High?adhesion property of the electrode and on?skin and in vivo EMG detection[86](A) Sketch of the on?skin EMG detection; (B) typical EMG signal detected; (C) energy spectrum analysis of the EMG signal in (B); (D) side view of the rat implanted with SPM?2?based stretchable thin film gold electrode; (E) typical subcutaneous EMG signal; (F) spectrum analysis of the EMG signal in (E).Copyright 2018, American Chemical Society.
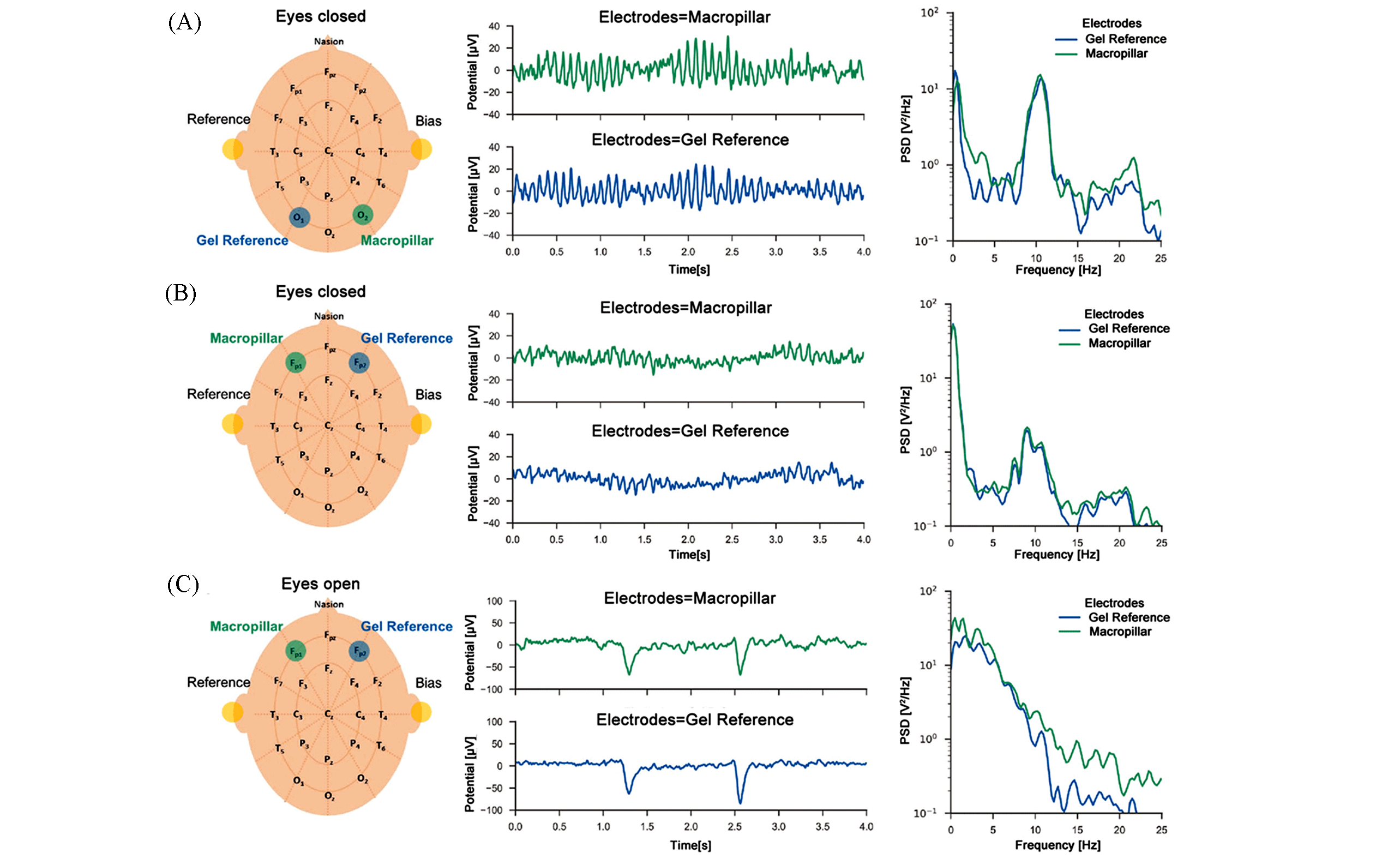
Fig.14 Proof?of?principle EEG measurements using soft pillar electrodes[81](A) The power spectral density shows the characteristic α wave peak around 10 Hz; (B) α waves could also be detected from the forehead when the eyes are closed; (C) the α waves disappear when the eyes are kept open.Copyright 2018, Wiley?VCH.
| 1 | Mun J., Wang G. J. N., Oh J. Y., Katsumata T., Lee F. L., Kang J., Wu H. C., Lissel F., Rondeau⁃Gagné S., Tok J. B. H., Bao Z. N., Adv. Funct. Mater., 2018, 28(43), 1804222 |
| 2 | Baik S., Lee H. J., Kim D. W., Kim J. W., Lee Y., Pang C., Adv. Mater., 2019, 31(34), 1803309 |
| 3 | Wang C. F., Wang C. H., Huang Z. L., Xu S., Adv. Mater., 2018, 30, 1801368 |
| 4 | Kim S. Y., Choo Y., Bilodeau A., Yuen M. C., Kaufman G., Shah D. S., Osuji C. O., Kramer⁃Bottiglio R., Sci. Robot., 2020, 5, eaay3604 |
| 5 | Wang T., Wang M., Yang L., Li Z. Y., Loh X. J., Chen X. D., Adv. Mater., 2020, 32, 1905522 |
| 6 | Yao S. S., Swetha P., Zhu Y., Adv. Healthcare Mater., 2017, 7(1), 1700889 |
| 7 | Pan S. W., Liu Z. Y., Wang M., Jiang Y., Luo Y. F., Wan C. J., Qi D. P., Wang C. X., Ge X., Chen X. D., Adv. Mater., 2019, 31(35), 1903130 |
| 8 | Jiang Y., Liu Z. Y., Wang C. X., Chen X. D., Acc. Chem. Res., 2019, 52(1), 82—90 |
| 9 | Yang H., Ji S. B., Chaturvedi I., Xia H. R., Wang T., Chen G., Pan L., Wan C. J., Qi D. P., Ong Y. S., Chen X. D., ACS Materials Lett., 2020, 2, 478—484 |
| 10 | Wan C. J., Chen G., Fu Y. M., Wang M., Matsuhisa N., Pan S. W., Pan L., Yang H., Wan Q., Zhu L. Q., Chen X. D., Adv. Mater., 2018, 30(30), 1801291 |
| 11 | Wang T., Qi D. P., Yang H., Liu Z. Y., Wang M., Leow W. R., Chen G., Yu J. C., He K., Cheng H. W., Wu Y. L., Zhang H., Chen X. D., Adv. Mater., 2018, 31(1), 1803883 |
| 12 | Ameri S. K., Ho R., Jang H., Li T., Wang Y. H., Wang L., Schnyer D. M., Akinwande D., Lu N. S., ACS Nano, 2017, 11, 7634—7641 |
| 13 | He F. L.,You X. Y., Gong H., Yang Y., Bai T., Wang W. G., Guo W. X., Liu X. Y., Ye M. D., ACS Appl. Mater. Interfaces, 2020, 12(5), 6442—6450 |
| 14 | Yang H., Qi D. P., Liu Z. Y., Chandran B. K., Wang T., Yu J. C., Chen X. D., Adv. Mater., 2016, 28(41), 9175—9181 |
| 15 | Liu Y. X., Liu J., Chen S. H., Lei T., Kim Y., Niu S. M., Wang H. L., Wang X., Foudeh A. M., Tok J. B. H., Bao Z. N., Nat. Biomed, Eng., 2019, 3, 58—68 |
| 16 | Xu J., Wang S. H., Wang G. J. N., Zhu C. X., Luo S. C., Jin L. H., Gu X. D., Chen S. C., Feig V. R., To J. W. F., Gagné S. R., Park J., Schroeder B. C., Lu C., Oh J. Y., Wang Y. M., Kim Y. H., Yan H., Sinclair R., Zhou D. S., Xue G., Murrmann B., Linder C., Cai W., Tok J. B. H., Chung J. W., Bao Z. N., Science, 2017, 355, 59—64 |
| 17 | Rogers J. A., Chen X. D., Feng X., Adv. Mater., 2020, 32(15), 1905590 |
| 18 | Jiang Y., Liu Z. Y., Matsuhisa N., Qi D. P., Leow W. R., Yang H., Yu J. C., Chen G., Liu Y. Q., Wan C. J., Liu Z. J., Chen X. D., Adv. Mater., 2018, 30(12), 1706589 |
| 19 | Drotlef D. M., Amjadi M., Yunusa M., Sitti M., Adv. Mater., 2017, 29, 1701353 |
| 20 | Chen X. D., Small Methods, 2017, 1, 1600029 |
| 21 | Pang C., Koo J. H., Nguyen A., Caves J. M., Kim M. G., Chortos A., Kim K., Wang P. J., Tok J. B. H., Bao Z. N., Adv. Mater., 2014, 27(4), 634—640 |
| 22 | Qi D. P., Liu Z. Y., Yan L., Jiang Y., Leow W. R., Pal M., Pan S. W., Yang H., Wang Y., Zhang X. Q., Yu J. C., Li B., Wang W., Chen X. D., Adv. Mater., 2017, 29(27), 1702800 |
| 23 | Huang X., Liu Y. H., Chen K., Shin W. J., Lu C. J., Kong G. W., Patnaik D., Lee S. H., Cortes J. F., Rogers J. A., Small, 2014, 10(15), 3083—3090 |
| 24 | Pan S. W., Liu Z. Y., Wang M., Jiang Y., Luo Y. F., Wan C. J., Qi D. P., Wang C. X., Ge X., Chen X. D., Adv. Mater., 2019, 31(35), 1903130 |
| 25 | Liu Z. Y., Qi D. P., Guo P. Z., Liu Y., Zhu B. W., Yang H., Liu Y. Q., Li B., Zhang C. G., Yu J. C., Liedberg B., Chen X. D., Adv. Mater., 2015, 27(40), 6230—6237 |
| 26 | Chen Y. P., Meng J. X., Gu Z., Wan X. Z., Jiang L., Wang S. T., Adv. Funct. Mater., 2020, 30(5), 1905287 |
| 27 | Lee S. M., Byeon H. J., Lee J. H., Baek D. H., Lee K. H., Hong J. S., Lee S. H., Sci. Rep. UK, 2015, 4, 6074 |
| 28 | Liu Z. Y., Wang X. T., Qi D. P., Xu C., Yu J. C., Liu Y. Q., Jiang Y., Liedberg B., Chen X. D., Adv. Mater., 2016, 29(2), 1603382 |
| 29 | Wang Z., Guo L. F., Xiao H. Y., Cong H., Wang S. T., Mater. Horiz, 2020,7, 282—288 |
| 30 | Zhang Q., Shi C. Y., Qu D. H., Long Y. T., Feringa B. L., Tian H., Sci. Adv., 2018, 4, eaat8192 |
| 31 | Qi D. P., Liu Z. Y., Leow W. R., Chen X. D., MRS Bulletin, 2017, 42(2), 103—107 |
| 32 | Wang C. Y., Xia K. L., Zhang Y. Y., Kaplan D. L., Acc. Chem. Res., 2019, 52(10), 2916—2927 |
| 33 | Ha M., Lim S., Ko H., J. Mater. Chem. B, 2018, 6, 4043—4064 |
| 34 | Liu Y. J., Cao W. T., Ma M. G., Wan P. B., ACS Appl. Mater. Interfaces, 2017, 9(30), 25559—25570 |
| 35 | Long X. T., Guan J., Chen X., Shao Z. Z., Chem. J. Chinese Universities, 2018, 39(1), 1—11(龙星潼, 管娟, 陈新, 邵正中. 高等学校化学学报, 2018, 39(1), 1—11) |
| 36 | Burke K. A., Roberts D. C., Kaplan D. L., Biomacromolecules, 2016, 17(1), 237—245 |
| 37 | Johnston E. R., Miyagi Y., Chuah J. A., Numata K., Serban M. A., ACS Biomater. Sci. Eng., 2018, 4(8), 2815—2824 |
| 38 | Chen X. D., Rogers J. A., Lacour S. P., Hu W. P., Kim D. H., Chem. Soc. Rev., 2019, 48, 1431—1433 |
| 39 | Chu B., Burnett W., Chung J W., Bao Z. N., Nature, 2017, 549, 328—330 |
| 40 | Niu Q. Q., Peng Q. F., Lu L., Fan S. N., Shao H. L., Zhang H. H., Wu R. L., Hsiao B. S., Zhang Y. P., ACS Nano, 2018, 12(12), 11860—11870 |
| 41 | Chen G., Matsuhisa N., Liu Z. Y., Qi D. P., Cai P. Q., Jiang Y., Wan C. J., Cui Y. J., Leow W. R., Liu Z. J., Gong S. X., Zhang K. Q., Cheng Y., Chen X. D., Adv. Mater., 2018, 30(21), 1800129 |
| 42 | Jo M., Min K., Roy B., Kim S., Lee S., Park J. Y., ACS Nano, 2018, 12, 5637—5645 |
| 43 | Gong S., Yap L. W., Zhu B. W., Cheng W. L., Adv. Mater., 2019, 32(15), 1902278 |
| 44 | Wang D., Xu H. Z., Chen S. H., Lou X. F., Tan J. F., Xu Y., Skin Wound Care, 2019, 32, 176—182 |
| 45 | Liu X. J., Liu J., Wang J. L., Wang T., Jiang Y., Hu J. Q., Liu Z. J., Chen X. D., Yu J., ACS Appl. Mater. Interfaces, 2020, 12(5), 5601—5609 |
| 46 | Li Y. R., Cao Y., Nanoscale Adv., 2019, 1, 4246 |
| 47 | Sha X. Y., Zhang C. X., Wang Y. L., Zhou Y. F., Chem. J. Chinese Universities, 2020, 41(7), 1477—1483(沙心易, 张常旭, 汪羽翎, 周永丰. 高等学校化学学报, 2020, 41(7), 1477—1483) |
| 48 | Meng X. F., Zhai Z. W., Guo A. J., Chem. J. Chinese Universities, 2018, 39(10), 2245—2252(孟秀峰, 翟志伟, 郭爱军. 高等学校化学学报, 2018, 39(10), 2245—2252) |
| 49 | Ji S. B., Wan C. J., Wang T., Liu X. J., Li Q. S., Chen G., Wang J. W., Liu Z. Y., Yang H., Chen X. D., Adv. Mater., 2020, 32(26), 2001496 |
| 50 | Zhang C., Wu B. H., Zhou Y. S., Zhou F., Liu W. M., Wang Z. K., Chem. Soc. Rev., 2020, 49(11), 3605—3637 |
| 51 | Han L., Lu X., Liu K. Z., Wang K. F., Fang L. M., Weng L. T., Zhang H. P., Tang Y. H., Ren F. Z., Zhao C. C., Sun G. X., Liang R., Li Z. J., ACS Nano, 2017, 11, 2561—2574 |
| 52 | He X. Y., Liu L. Q., Han H. M., Shi W. Y., Yang W., Lu X. Q., Macromolecules, 2019, 52(1), 72—80 |
| 53 | Qiao H. Y., Qi P. F., Zhang X. H., Wang L. N., Tan Y. Q., Luan Z. H., Xia Y. Z., Li Y. H., Sui K. Y., ACS Appl. Mater. Interfaces, 2019, 11, 7755—7763 |
| 54 | Cui C., Shao C. Y., Meng L., Yang J., ACS Appl. Mater. Interfaces, 2019, 11, 39228—39237 |
| 55 | Du X. H., Niu Z. K., Li R. J., Yang H., Hu W. P., J. Mater. Chem. C, 2020, 8, 12257 |
| 56 | Danko M., Kronekova Z., Mrlik M., Osicka J., Yousaf A. B., Durackova A., Tkac J., Kasak P., Langmuir, 2019, 35(5), 1391—1403 |
| 57 | Peng X., Liu H. L., Yin Q., Wu J. C., Chen P. Z., Zhang G. Z., Liu G. M., Wu C. Z., Xie Y., Nat. Commun., 2016, 7, 11782 |
| 58 | Lee C. J., Wu H. Y., Hu Y., Young M., Wang H. F., Lynch D., Xu F. J., Cong H. B., Cheng G., ACS Appl. Mater. Interfaces, 2018, 10, 5845—5852 |
| 59 | Xiao S. W., Zhang M. Z., He X. M., Huang L., Zhang Y. X., Ren B. P., Zhong M. Q., Chang Y., Yang J. T., Zheng J., ACS Appl. Mater. Interfaces, 2018, 10(25), 21642—21653 |
| 60 | Wang L. F., Gao G. R., Zhou Y., Xu T., Chen J., Wang R., Zhang R., Fu J., ACS Appl. Mater. Interfaces, 2019, 11(3), 3506—3515 |
| 61 | Yang B. W., Yuan W. Z., ACS Appl. Mater. Interfaces, 2019, 11(43), 40620—40628 |
| 62 | Singla S., Amarpuri G., Dhopatkar N., Blackledge T. A., Dhinojwala A., Nat. Commun., 2018, 9, 1890 |
| 63 | Bai S. M., Zhang X. L., Cai P. Q., Huang X. W., Huang Y. Q., Liu R., Zhang M. Y., Song J. B., Chen X. D., Yang H. H., Nanoscale Horiz., 2019, 4, 1333—1341 |
| 64 | Yuk H., Varela C. E., Nabzdyk C. S., Mao X. Y., Padera R. F., Roche E. T., Zhao X. H., Nature, 2019, 575, 169—174 |
| 65 | Yuk H., Lu B. Y., Zhao X. H., Chem. Soc. Rev., 2019, 48, 1642—1667 |
| 66 | Pan S. W., Zhang F. L., Cai P. Q., Wang M., He K., Luo Y. F., Li Z., Chen G., Ji S. B., Liu Z. H., Loh X. J., Chen X. D., Adv. Funct. Mater., 2020, 30, 1909540 |
| 67 | Zhou Y., Wan C. J., Yang Y. S., Yang H., Wang S. C., Dai Z. D., Ji K. J., Jiang H., Chen X. D., Long Y., Adv. Funct. Mater., 2018, 29(1), 1806220 |
| 68 | Hou J. X., Li C., Guan Y., Zhang Y. J., Zhu X. X., Polym. Chem., 2015, 6, 2204—2213 |
| 69 | Zhang Z., Gao Z. L., Wang Y. T., Guo L. X., Yin C. H., Zhang X. L., Hao J. C., Zhang G. M., Chen L. S., Macromolecules, 2019, 52(6), 2531—2541 |
| 70 | Liao M. H., Wan P. B., Wen J. R., Gong M., Wu X. X., Wang Y. G., Shi R., Zhang L. Q., Adv. Funct. Mater., 2017, 27, 1703852 |
| 71 | Liu Y. H., Pharr., Salvatore G. A., ACS Nano, 2017, 11, 9614—9635 |
| 72 | Wang T., Yang H., Qi D. P., Liu Z. Y., Cai P. Q., Zhang H., Chen X. D., Small, 2018, 14(11), 1702933 |
| 73 | Lin C. T., Chuang C. H., Cao Z. H., Singh A. K., Hung C. S., Yu Y. H., Nascimben M., Liu Y. T., King J. T., Su T. P., Wang S. J., IEEE Access, 2017, 5, 10612—10621 |
| 74 | Zheng W. L., Lu B. L., J. Neural Eng., 2017, 14, 1741—2552 |
| 75 | He K., Liu Z. Y., Wan C. J., Jiang Y., Wang T., Wang M., Zhang F. L., Liu Y. Q., Pan L., Xiao M., Yang H., Chen X. D., Adv. Mater., 2020, 32(24), 2001130 |
| 76 | Majumder S., Mondal T., Deen M. J., Sensors, 2017, 17, 130 |
| 77 | Wang Q., Ling S. J., Wang H. M., Lu H. J., Zhang Y. Y., Adv. Funct. Mater., 2019, 29, 1808695 |
| 78 | Son D., Kang J., Vardoulis O., Kim Y., Matsuhisa N., Oh J. Y., To J. W., Mun J., Katsumata T., Liu Y. X., McGuire A. F., Krason M., Molina⁃Lopez F., Ham J., Kraft U., Lee Y., Yun Y. J., Tok J. B. H., Bao Z. N., Nat. Nanotechnol., 2018, 13, 1057—1065 |
| 79 | Tottman A. C., Alsweiler J. M., Bloomfield F. H., Harding J. E., Arch. Dis. Child Fetal Neonatal Ed., 2018, 103(3), F277—F279 |
| 80 | Chung H. U., Kim B. H., Lee J. Y., Lee J. Y., Xie Z. Q., Ibler E. M., Lee K. H., Banks A., Jeong J. Y., Kim J., Ogle C., Grande D., Yu Y., Jang H., Pourya A., Ryu D., Kwak J. W., Namkoong M., Park J. B., Lee Y., Kim D. H., Ryu A., Jeong J., You K., Ji B. W., Liu Z. J., Hou Q. Z., Feng X., Deng A., Xu Y. S., Jang K. I., Kim J., Zhang Y. H., Ghaffari R., Rand C. M., Schau M., Hamvas A., Weese⁃Mayer D. E., Huang Y., Lee S. M., Lee C. H., Shanbhag N. R., Paller A. S., Xu S., Rogers J. A., Science, 2019, 363, eaau0780 |
| 81 | Stauffer F., Thielen M., Sauter C., Chardonnens S., Bachmann S., Tybrandt K., Peters C., Hierold C., Vörös J., Adv. Healthcare Mater., 2018, 7(7), 1700994 |
| 82 | Kim S. H., Seo H., Kang J., Hong J., Seong D., Kim H. J., Kim J., Mun J., Youn I., Kim J., Kim Y. C., Seok H. K., Lee C., Tok J. B. H., Bao Z. N., Son D., ACS Nano, 2019, 13(6), 6531—6539 |
| 83 | Pan L., Cai P. Q., Mei L., Cheng Y., Zeng Y., Wang M., Wang T., Jiang Y., Ji B. H., Li D. C., Chen X. D., Adv. Mater., 2020, 32(31), 2003723 |
| 84 | Zhang X., Zhu J., Tang Y. D., Zhao H., Ying T. T., Yuan Y., Li S. T., World Neurosurgery, 2017, 102, 85—90 |
| 85 | Marasco P. D., Kuiken T. A., J. Neurol., 2010, 30(47), 16008—16014 |
| 86 | Yan X. Z., Liu Z. Y., Zhang Q. H., Lopez J., Wang H., Wu H. C., Niu S. M., Yan H. P., Wang S. H., Lei T., Li J. H., Qi D. P., Huang P. G., Huang J. P., Zhang Y., Wang Y. Y., Li G. L., Jeffery B. H. T., Chen X. D., Bao Z. N., J. Am. Chem. Soc., 2018, 140, 5280—5289 |
| 87 | Ferrari L. M., Sudha S., Tarantino S., Esposti R., Bolzoni F., Cavallari P., Cipriani C., Mattoli V., Greco F., Adv. Sci., 2018, 5, 1700771 |
| 88 | Won S. M., Song E., Zhao J. N., Li J. H., Rivnay J., Rogers J. A., Adv. Mater., 2018, 30(30), 1800534 |
| 89 | Heikenfeld J., Jajack A., Rogers J., Gutruf P., Tian L., Pan T., Li R., Khine M., Kim J., Wang J., Kim J., Lab Chip, 2018, 18, 217—248 |
| 90 | Tian L. M., Zimmerman B., Akhtar A., Yu K. J., Moore M., Wu J., Larsen R. J., Lee J. W., Li J. H., Liu Y. H., Metzger B., Qu S. B., Guo X. G., Mathewson K. E., Fan J. A., Cornman J., Fatina M., Xie Z. Q., Ma Y. J., Zhang J., Zhang Y. H., Dolcos F., Fabiani M., Gratton G., Bretl T., Hargrove L. J., Braun P. V., Huang Y. G., Rogers J. A., Nat. Biomed. Eng., 2019, 3, 194—205 |
| [1] | 潘晓君, 鲍容容, 潘曹峰. 可穿戴柔性触觉传感器的研究进展[J]. 高等学校化学学报, 2021, 42(8): 2359. |
| [2] | 姬少博, 陈晓东. 柔性电子学中的表界面化学[J]. 高等学校化学学报, 2021, 42(4): 1074. |
| [3] | 盛卉, 薛斌, 秦猛, 王炜, 曹毅. 可拉伸超韧水凝胶的制备和应用[J]. 高等学校化学学报, 2020, 41(6): 1194. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||