

Chem. J. Chinese Universities ›› 2020, Vol. 41 ›› Issue (4): 623.doi: 10.7503/cjcu20190686
Previous Articles Next Articles
LING Yunyun1,2,LI Li1,LIANG Xiurong1,XIA Yunsheng1,*( )
)
Received:2019-12-18
Online:2020-04-10
Published:2020-02-17
Contact:
Yunsheng XIA
E-mail:xiayuns@mail.ahnu.edu.cn
Supported by:CLC Number:
TrendMD:
LING Yunyun,LI Li,LIANG Xiurong,XIA Yunsheng. Glutathione Sensing: from Colorimetry to Single Particle Spectroscopy Based on Gold NP@MnO2 Nanosheets Supraparticles[J]. Chem. J. Chinese Universities, 2020, 41(4): 623.
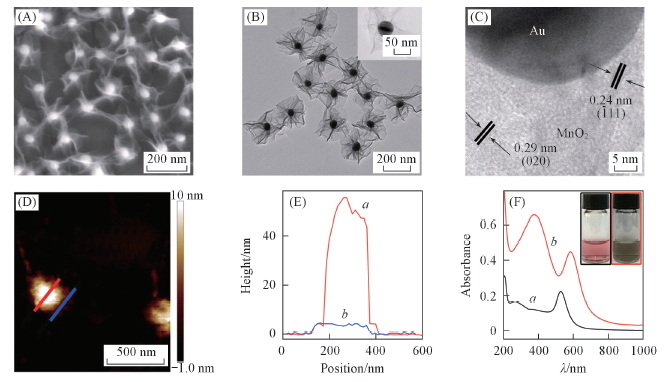
Fig.1 Characterizations of the AMNS-SPs^SEM(A), TEM(B), HRTEM(C) and AFM(D, E) images of the AMNS-SPs. The curves a and b in Fig.1(E) represents the height profiles along the red and blue lines in Fig.1(D), respectively. (F) UV-Vis absorption spectra of AuNPs(a) and AMNS-SPs(b), and the insets are the corresponding photo images.
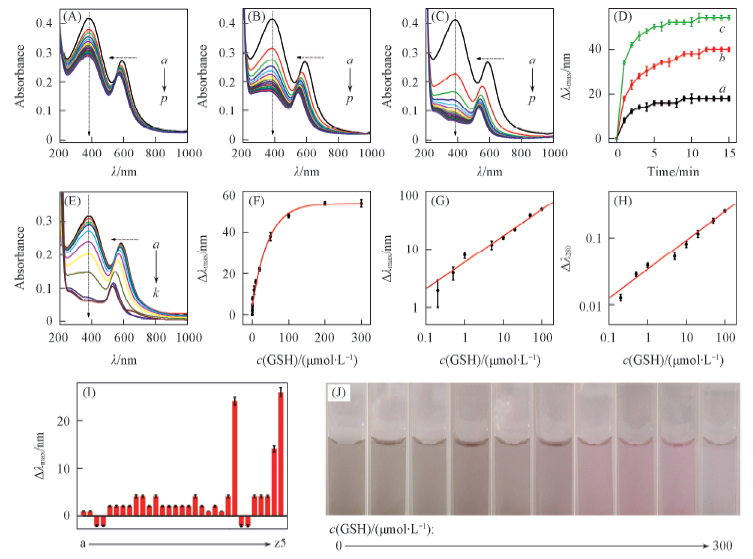
Fig.3 GSH sensing in bulk solution with AMNS-SPsTime-dependent absorption spectra of the AMNS-SPs in 10 (A), 50 (B), 100 (C) μmol/L of GSH, respectively. Time of curves a—p: 0—15 min. (D) Etching dynamics of the AMNS-SPs by 10(a), 50(b) and 100(c) μmol/L GSH. (E) c(GSH)/(μmol·L-1): a. 0; b. 0.2; c. 0.5; d. 1; e. 5; f. 10; g. 20; h. 50; i. 100; j. 200; k. 300. Absorption spectra of the AMNS-SPs in the presence of various concentrations of GSH. Dynamic range[0—300 μmol/L, (F)] and linear range [0.2—100 μmol/L, (G)] of the LSPR peak shift(Δλ) in different concentrations of GSH, respectively. (H) Linear range(0.2—100μmol/L) of ΔA380 in different concentrations of GSH. (I) a. PBS(pH=7.0); b. PBS(pH=7.4); c. Tris(pH=7.0); d. Tris(pH=7.4); e. KCl; f. K2CO3; g. NaCl; h. Na2SO4; i. MgSO4; j. MnSO4; k. alanine; l. arginine; m. lysine; n. serin; o. threonin; p. valine; q. histidine; r. glycine; s. leucine; t. glutamic acid; u. tyrosine; v. aspartic acid; w. tryptophan; x. cysteine; y. BSA; z. HSA; z1. fructose; z2 maltose; z3 glucose; z4 AA; z5 GSH. The responses of the AMNS-SPs to various biomolecules[the concentrations of various electrolytes(columns a—j) are 10 mmol/L. The concentrations of various amino acids(columns k—x) are 10, 10, 1.0, 10, 10, 10, 10, 10, 10, 0.25, 0.25, 0.25, 0.25 and 0.025 mmol/L, respectively. Both the concentrations of bovine serum albumin(BSA) and human serum albumin(HSA) are 1 mg/mL. The concentrations of fructose, maltose, glucose, AA and GSH(columns z1—z5) are 10, 10, 10, 0.025, and 0.025 mmol/L, respectively. (J) Photos of the AMNS-SPs etched by GSH. From left to right, the concentrations of GSH are 0, 0.5, 1, 5, 10, 20, 50, 100, 200, 300 μmol/L, respectively. The reaction time is 15 min.
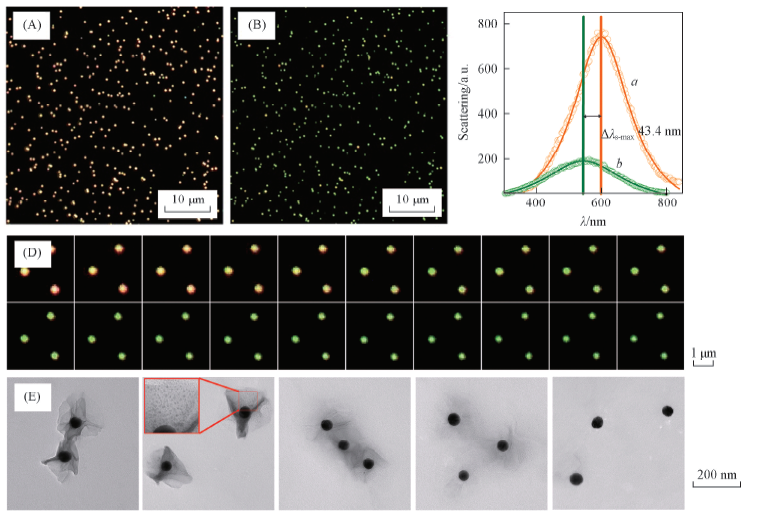
Fig.4 Etching process of the AMNS-SPs by GSH molecules(0.3 μmol/L)Dark-field images of the AMNS-SPs before(A) and after(B) the etching process, respectively. (C) Scattering spectra of a single AMNS-SP before(a) and after(b) the etching process. (D) Time-dependent dark-field images of the AMNS-SPs. (E) TEM images of the AMNS-SPs during the etching process.
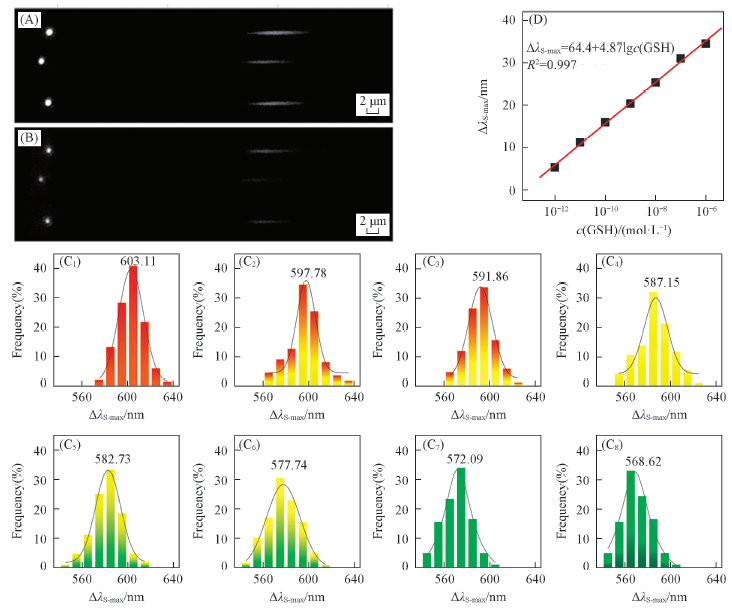
Fig.5 GSH sensing with single particle dark-field spectral imagingTypical single particle spectral images before(A) and after(B) adding GSH. (C) Distribution of λs-max after adding 0 pmol/L(C1), 1 pmol/L(C2), 10 pmol/L(C3), 100 pmol/L(C4), 1 nmol/L(C5), 10 nmol/L(C6), 100 nmol/L(C7) and 1 μmol/L(C8) GSH. 100 of the SPs are counted in each case. (D) Δλs-max vs. GSH concentration.
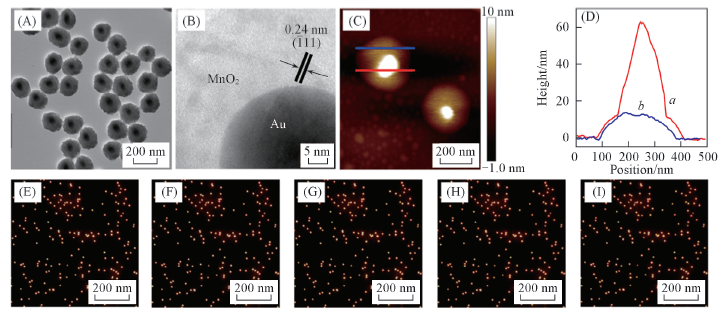
Fig.6 Characterizations of the TAMNS-SPs and their etching process by GSH molecules(0.3 μmol/L)TEM(A), HRTEM(B) and AFM(C, D) images of the TAMNS-SPs. The curves a and b in Fig.6(D) represents the height profiles along the red and blue lines in Fig.6(C), respectively. The time-dependent dark-field images of the TAMNS-SPs, time/min: (E) 0; (F) 5; (G) 10; (H) 15; (I) 20.
| [1] | Meister A., Anderson M. E., Annu. Rev. Biochem., 1983, 52( 1), 711— 760 |
| [2] | Sies H., Free Radicals Biol. Med., 1999, 27( 9/10), 916— 921 |
| [3] | Deneke S. M., Fanburg B. L., Am. J. Physiol., 1989, 257( 4), L163— L173 |
| [4] | Wu G., Fang Y., Yang S., Lupton J. R., Turner N. D., J. Nutr., 2004, 134( 3), 489— 492 |
| [5] | Smirnova G. V., Oktyabrsky O. N ., Biochemistry(Moscow), 2005, 70( 11), 1199— 1211 |
| [6] | Forman H., Zhang H., Rinna A., Mol. Aspects Med., 2009, 30( 1/2), 1— 12 |
| [7] | Tietze F., Anal. Biochem., 1969, 27( 3), 502— 522 |
| [8] | Harfield J. C., Batchelor-McAuley C., Compton R. G ., Analyst, 2012, 137( 10), 2285— 2296 |
| [9] | Chen X., Zhou Y., Peng X., Yoon J., Chem. Soc. Rev., 2010, 39( 6), 2120— 2135 |
| [10] | Reed D., Babson J., Beatty P., Brodie A., Ellis W., Potter D., Anal. Biochem., 1980, 106( 1), 55— 62 |
| [11] | Miao P., Liu L., Nie Y., Li G., Biosens. Bioelectron., 2009, 24( 11), 3347— 3351 |
| [12] | Umezawa K., Yoshida M., Kamiya M., Yamasoba T., Urano Y., Nat. Chem., 2017, 9( 3), 279— 286 |
| [13] | Jiang X., Chen J., Bajic A., Zhang C., Song X., Carroll S. L., Cai Z., Tang M., Xue M., Cheng N., Schaaf C. P., Li F., MacKenzie K. R., Ferreon A. M., Xia F., Wang M. C., Maletic-Savati M., Wang J., Nat. Commun., 2017, 8( 1), 1— 13 |
| [14] | Lee S., Li J., Zhou X., Yin J., Yoon J., Coord. Chem. Rev., 2018, 366, 29— 68 |
| [15] | Ling Y., Zhang D., Cui X., Wei M., Zhang T., Wang J., Xiao L., Xia Y., Angew. Chem. Int. Ed., 2019, 58( 31), 10542— 10546 |
| [16] | Herszage J., dos Santos Afonso M., Luther G. W., Environ. Sci. Technol., 2003, 37( 15), 3332— 3338 |
| [17] | Deng R., Xie X., Vendrell M., Chang Y., Liu X., J. Am. Chem. Soc., 2011, 133( 50), 20168— 20171 |
| [18] | Fan H., Yan G., Zhao Z., Hu X., Zhang W., Liu H., Fu X., Fu T., Zhang X., Tan W., Angew. Chem. Int. Ed., 2016, 55( 18), 5477— 5482 |
| [19] | Huang Z., Cai Q., Ding D., Ge J., Hu Y., Yang J., Zhang L., Li Z., Sens. Actuators B: Chem., 2017, 242, 355— 361 |
| [20] | Chen J., Meng H., Tian Y., Yang R., Du D., Li Z., Qu L., Lin Y., Nanoscale Horiz., 2019, 4( 2), 321— 338 |
| [21] | Xia H., Xiahou Y., Zhang P., Ding W., Wang D ., Langmuir, 2016, 32( 23), 5870— 5880 |
| [22] | Kim D., Hwang Y., Yoon C., Yoon H., Chang K., Lee G., Lee S., Yi G., Phys. Chem. Chem. Phys., 2015, 17( 32), 20786— 20794 |
| [23] | Qi F., Han Y., Ye Z., Liu H., Wei L., Xiao L., Anal. Chem., 2018, 90( 18), 11146— 11153 |
| [24] | Truong T. T., Liu Y., Ren Y., Trahey L., Sun Y., ACS Nano, 2012, 6( 9), 8067— 8077 |
| [25] | Sun Y., Wang L., Liu Y., Ren Y ., Small, 2015, 11( 3), 300— 305 |
| [26] | Ma Z., Shao G., Fan Y., Wang G., Song J., Shen D., ACS Appl. Mater. Interfaces, 2016, 8( 14), 9050— 9058 |
| [27] | Instruments M ., Sample Dispersion and Refractive Index Guide, MAN0396 1.0, Malvern Instruments Ltd., Malvern, UK, 2007, 1— 18 |
| [28] | Kai K., Yoshida Y., Kageyama H., Saito G., Ishigaki T., Furukawa Y., Kawamata J., J. Am. Chem. Soc., 2008, 130( 47), 15938— 15943 |
| [29] | Yi X., Chen L., Zhong X., Gao R., Qian Y., Wu F., Song G., Chai Z., Liu Z., Yang K., Nano Res., 2016, 9( 11), 3267— 3278 |
| [30] | Liu B., Mosa I. M., Song W., Zheng H., Kuo C., Rusling J. F., Suib S. L., He J., J. Mater. Chem. A, 2016, 4( 17), 6447— 6455 |
| [31] | Lin X., Uzayisenga V., Li J., Fang P., Wu D., Ren B., Tian Z., J. Raman Spectrosc., 2012, 43( 1), 40— 45 |
| [32] | Fan W., Bu W., Shen B., He Q., Cui Z., Liu Y., Zheng X., Zhao K., Shi J., Adv. Mater., 2015, 27( 28), 4155— 4161 |
| [1] | YUAN Chunling, YAO Xiaotiao, XU Yuanjin, QIN Xiu, SHI Rui, CHENG Shiqi, WANG Yilin. Colorimetry/Ratio Fluorimetry Determination of Glucose with Bifunctional Carbon Dots [J]. Chem. J. Chinese Universities, 2021, 42(8): 2428. |
| [2] | LIU Zhaoyang, SONG Yongxin, WANG Tianshu, SHAN Guiye. Preparation of Histidine Modified Prussian Blue and Its Interaction with Silver Ions† [J]. Chem. J. Chinese Universities, 2020, 41(7): 1492. |
| [3] | ZHENG Shan, LIU Yang, CHEN Piaopiao, XING Yichen, HUANG Chaobiao. Novel Glutathione Photoelectrochemical Sensor Based on PbS QDs/TiO2 NPs † [J]. Chem. J. Chinese Universities, 2019, 40(9): 1866. |
| [4] | XIONG Qin,YANG Wuye,YANG Shaojie,LIU Jing,YANG Shihua,CHEN Wanchao,LI Long,DU Yiping. Rapid Detection of Trace Organophosphorus Pesticides by Membrane Enrichment Combined with Membrane Image Colorimetric† [J]. Chem. J. Chinese Universities, 2019, 40(6): 1135. |
| [5] | XIA Yuting, JIANG Bo, WU Qing, HU Qinghong, YUAN Zeli. Synthesis and Application of Colorimetric Probe for the Simultaneous Detection of Sulphion and Homocysteine/Cysteine in the Serum Samples† [J]. Chem. J. Chinese Universities, 2018, 39(8): 1647. |
| [6] | ZHANG Lingyue, CHI Yanan, SHAN Guiye, CHEN Yanwei, LIU Na. Preparation of GSH-Modified Au Nanorods and Their Interaction with Copper Ions† [J]. Chem. J. Chinese Universities, 2016, 37(7): 1239. |
| [7] | HUO Danqun, FU Beibei, LI Junjie, YANG Mei, SHEN Caihong, LUO Huibo, HOU Changjun. New Method for Identifying Different Teas Based on Liquid Colorimetric Sensor† [J]. Chem. J. Chinese Universities, 2016, 37(5): 873. |
| [8] | WU Shuo, XING Panpan, SONG Jie, ZHAO Yanqiu. Synthesis of Triphenylamine Functional Dye for Highly Sensitive and Ultra-low Potential Photoelectrochemical Sensing of Glutathione† [J]. Chem. J. Chinese Universities, 2016, 37(4): 619. |
| [9] | YANG Runjie, TANG Yao, ZHU Weiping. Ratiometric Fluorescent Probe for the Detection of Glutathione in Living Cells† [J]. Chem. J. Chinese Universities, 2016, 37(4): 643. |
| [10] | HU Jie, WANG Yong, NI Yongnian. Colorimetric Detection of Tin(Ⅱ) Ions Based on Layered Molybdenum Disulfide Nanosheets† [J]. Chem. J. Chinese Universities, 2016, 37(3): 448. |
| [11] | SHEN Wen, SHAO Xueguang, CAI Wensheng. Inclusion Mechanism of Cyclodextrins with Glutathione† [J]. Chem. J. Chinese Universities, 2016, 37(10): 1809. |
| [12] | FENG Juanjuan, ZHAO Yiman, WANG Haiyan. Colorimetric Detection of Dopamine Based on Silver Nanoparticles† [J]. Chem. J. Chinese Universities, 2015, 36(7): 1269. |
| [13] | GUO Xiao, YU Yang, CHEN Long, WANG Mingshuo, JIN Mengmeng, LIU Guangna, ZHANG Li, LI Tao, GAO Zhuang, WEI Jingyan. Catalytic Activity of Seleno-hGSTZ1c-1c Based on Site-directed Mutagenesis† [J]. Chem. J. Chinese Universities, 2014, 35(10): 2109. |
| [14] | LV Bing-Cong, DONG Seng, XUE Qiao, ZONG Hui, LV Shao-Wu, LUO Gui-Min. New Derivative of Tellurium Containing Cyclodextrin(6-AnSeCD) with Glutathione Peroxidase(GPx) Activity [J]. Chem. J. Chinese Universities, 2013, 34(9): 2146. |
| [15] | WANG Qing, LIU Wei, YANG Xiao-Hai, WANG Ke-Min, LIU Pei, HE Lei-Liang. High Sensitive Glutathione and Cysteine Detection by Au Nanoparticles Enhanced Electrochemical Biosensor [J]. Chem. J. Chinese Universities, 2013, 34(8): 1845. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||