

Chem. J. Chinese Universities ›› 2020, Vol. 41 ›› Issue (4): 591.doi: 10.7503/cjcu20190651
• Review • Previous Articles Next Articles
HAN Fangjie1,2,DAI Mengjiao1,2,LIANG Zhishan3,SONG Zhongqian1,HAN Dongxue1,2,3,*( ),NIU Li1,2,3
),NIU Li1,2,3
Received:2019-12-11
Online:2020-04-10
Published:2020-02-07
Contact:
Dongxue HAN
E-mail:dxhan@gzhu.edu.cn
Supported by:CLC Number:
TrendMD:
HAN Fangjie, DAI Mengjiao, LIANG Zhishan, SONG Zhongqian, HAN Dongxue, NIU Li. Research Progress of Photoelectrochemical Technology Applied in Antioxidant Analysis †[J]. Chem. J. Chinese Universities, 2020, 41(4): 591.
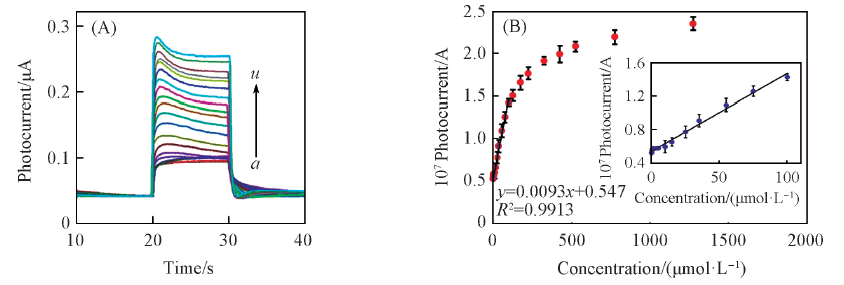
Fig.3 Photocurrent responses of the g-C3N4/ITO electrode to different concentrations of AA(A) and corresponding calibration curve(B) in 0.1 mol/L PBS(pH=7.4)[62](A) c(AA)/(μmol·L-1): a. 0; b. 0.25; c. 0.5; d. 1; e. 3; f. 5; g. 10; h. 15; i. 25; j. 35; k. 55; l. 75; m. 100; n. 125; o. 175; p. 225; q. 325; r. 425; s. 525; t. 775; u. 1275. Inset of (B) shows better illustration of linear part in calibration curve. Error bars correspond to the standard deviations from three independent measurements with RSD equal to 5.2%.Copyright 2018, Elsevier.
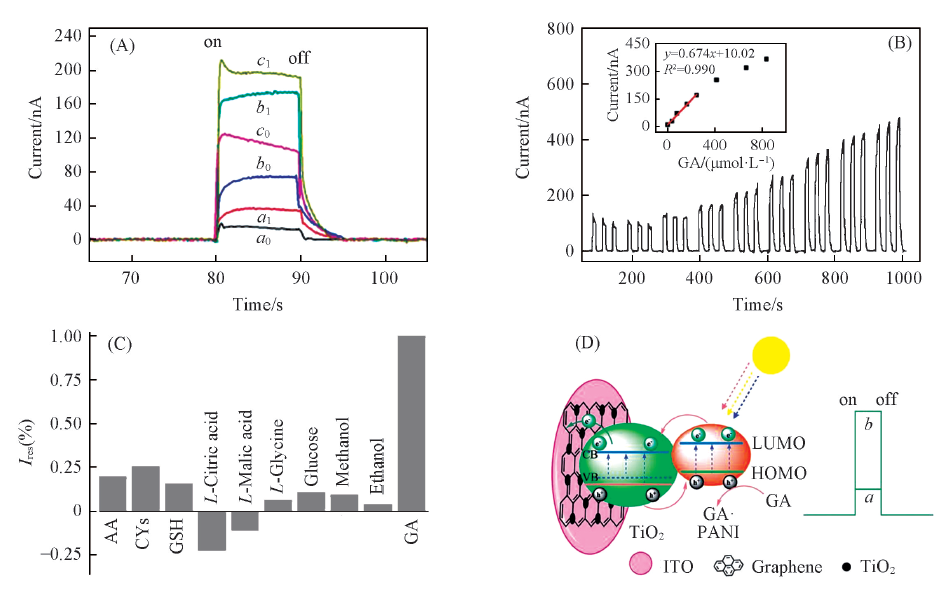
Fig.4 Photocurrent responses of TiO2(a0, a1), rGO-TiO2(b0, b1) and PANI-rGO-TiO2(c0, c1) modified ITO without(a0—c0) and with(a1—c1) 166.8 mmol/L GA(A); photocurrent responses of PANI-rGO-TiO2 modified ITO with different concentrations of GA(B); photocurrent response of a PANI-rGO-TiO2 modified ITO electrode upon the addition of 166.8 mmol/L each of AA, GA, 33.36 mmol/L each of GSH, CYs, L-citric acid, L-malic acid, 0.167 mol/L each of ethanol, methanol, glucose, L-glycine in 0.1 mol/L PBS(pH=7.4) at 0 V under 420 nm light excitation(C) and illustration of photoelectro-chemical process for GA oxidation at PANI-rGO-TiO2 modified ITO(D)[39](B) The photoelectrochemical sensors were applied at 0 V under 420 nm light excitation in 0.1 mol/L PBS(pH=7.4). Inset in (B) is the linear calibration curve. ^Copyright 2013, Royal Society of Chemistry.
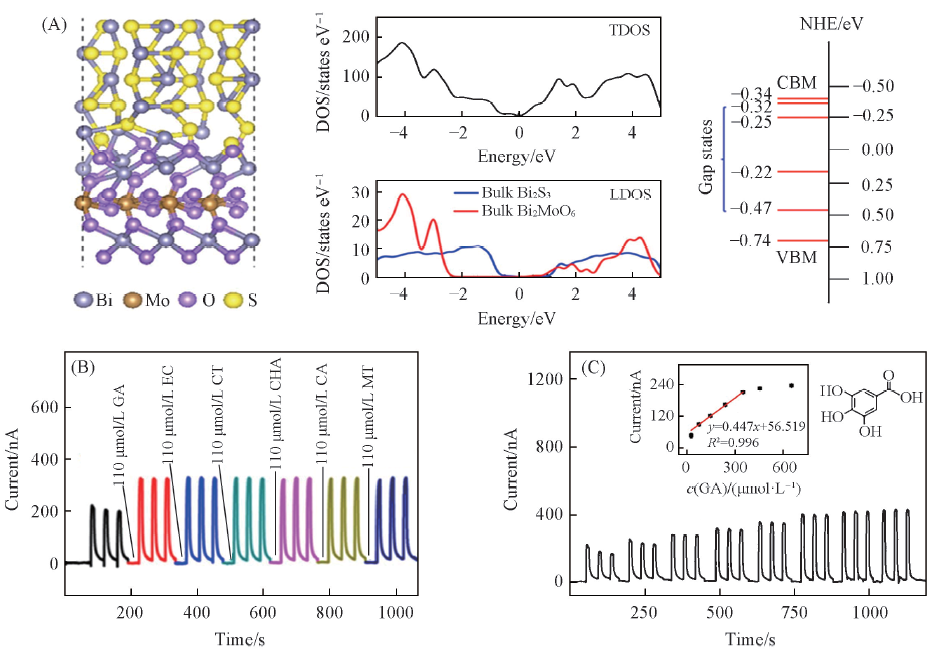
Fig.5 Schematic illustration of the surface contact region between Bi2MoO6 and Bi2S3, TDOS and LDOS of the Bi2MoO6/Bi2S3 heterostructures, the energy levels for valence and conduction bands of Bi2MoO6/Bi2S3(A); photocurrents of GA, EC, CT, CHA, CA and MT on Bi2MoO6/Bi2S3(B) and photocurrent responses of Bi2MoO6/Bi2S3 modified ITO electrode upon different concentrations of GA(C)[42]Inset of (C) is the corresponding liner calibration. The photoelectrochemical sensor was applied at 0 V under 470 nm irradiation in 0.1 mol/L PBS(pH=7.4). Copyright 2017, Elsevier.
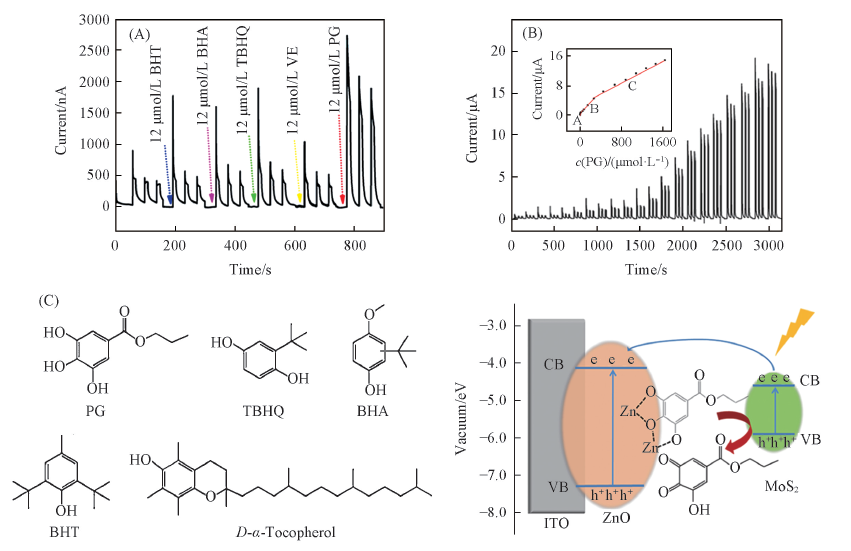
Fig.6 Photocurrents of 12 μmol/L PG, TBHQ, BHA, BHT and VE on MoS2/ZnO heterostructures(A); photocurrent responses of MoS2/ZnO heterostructures modified ITO electrodes upon different concentrations of PG(B) and chemical structures of five antioxidants and proposed mechanism of the MoS2/ZnO-based photoelectrochemical sensor for the detection of PG(C)[66] (B) The inset is the corresponding linear calibration. The preceding photoelectrochemical experiments were applied at 0 V(vs. Ag/AgCl) under 470 nm irradiation in 0.1 mol/L PBS(pH=7.4). Copyright 2019, American Chemical Society.
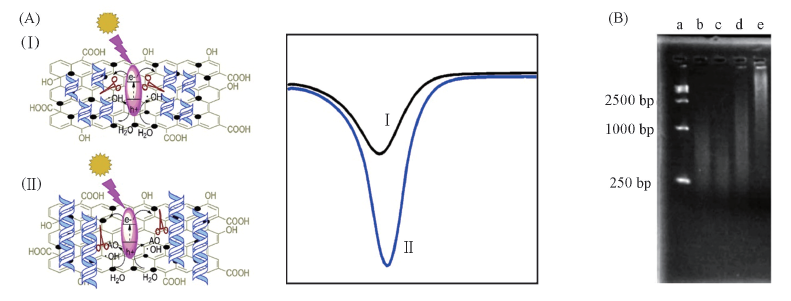
Fig.7 Schematic illustration of photoelectrochemical process for antioxidant capacity sensor designed with GO-TiO2 composites as source of OH radicals and DNA as a molecular probe(A) and agarose gel electrophoresis of different types of DNA(B) [40] Lane a: DNA markers; lanes b and c: DNA illuminated for 20 and 30 min with GOB-TiO2; lane d: after addition of gallic acid, DNA was illuminated 30 min with GOB-TiO2; lane e: DNA was illuminated 30 min without GOB-TiO2. Copyright 2013, Royal Society of Chemistry.
| Sample | DNA sensor for GA/(mg·g-1) | F-C method for GA/(mg·g-1) | DPPH method for trolox/(mg·g-1) |
|---|---|---|---|
| B1 | 47.45 | 60.29 | 141.20 |
| B2 | 66.61 | 62.94 | 201.10 |
| B3 | 41.86 | 78.40 | 191.50 |
| B4 | 24.34 | 18.55 | 49.90 |
| B5 | 38.39 | 39.82 | 86.30 |
| Sample | DNA sensor for GA/(mg·g-1) | F-C method for GA/(mg·g-1) | DPPH method for trolox/(mg·g-1) |
|---|---|---|---|
| B1 | 47.45 | 60.29 | 141.20 |
| B2 | 66.61 | 62.94 | 201.10 |
| B3 | 41.86 | 78.40 | 191.50 |
| B4 | 24.34 | 18.55 | 49.90 |
| B5 | 38.39 | 39.82 | 86.30 |
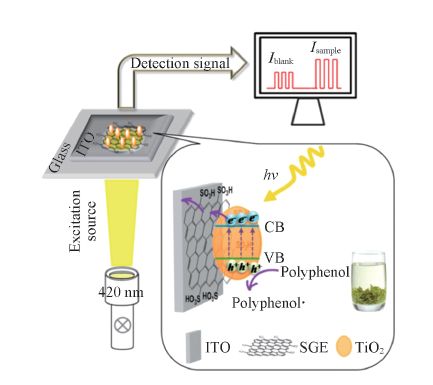
Fig.8 Schematic illustration of photoelectrochemical process for tea polyphenols oxidation at SGE-TiO2 modified ITO electrode[37] Copyright 2014, American Chemistry Society.
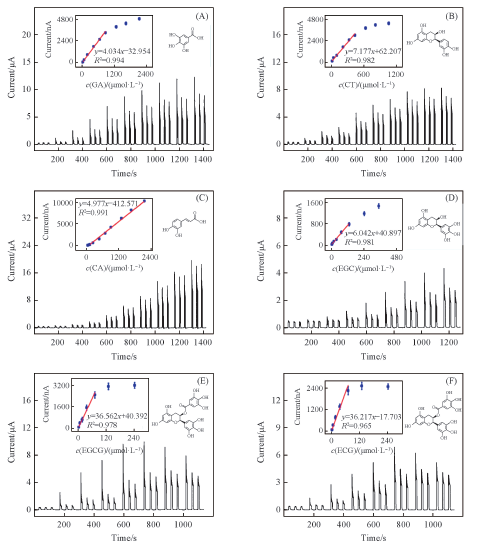
Fig.9 Photocurrent responses of SGE-TiO2 modified ITO electrode upon different concentrations of GA(A), CT(B), CA(C), EGC(D), EGCG(E) and ECG(F), respectively [37]Inset in each graph is the corresponding liner calibration curve. The photoelectrochemical sensors were applied at 0 V under 420 nm light excitation in 0.1 mol/L PBS(pH=7.4).Copyright 2014, American Chemistry Society.
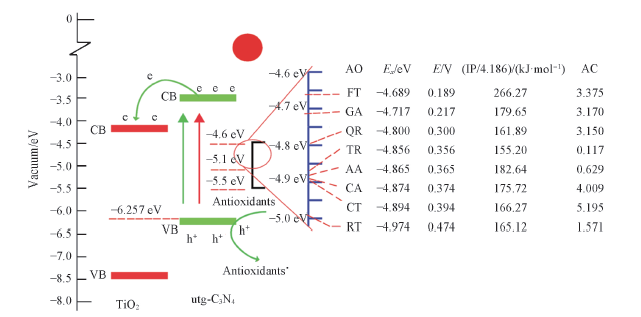
Fig.10 Mechanism of the photoelectrochemical sensor for the detection of the antioxidant capacity[38]AO: antioxidant; Ex(eV): the redox potential of the antioxidants with respect to a vacuum; E(V): the redox potential of the antioxidants(vs. Ag/AgCl); IP(kJ/mol): ionization potential of the antioxidants; AC: antioxidant capacity obtained from the slope of the standard calibration curve of each antioxidant. Copyright 2014, Royal Society of Chemistry.
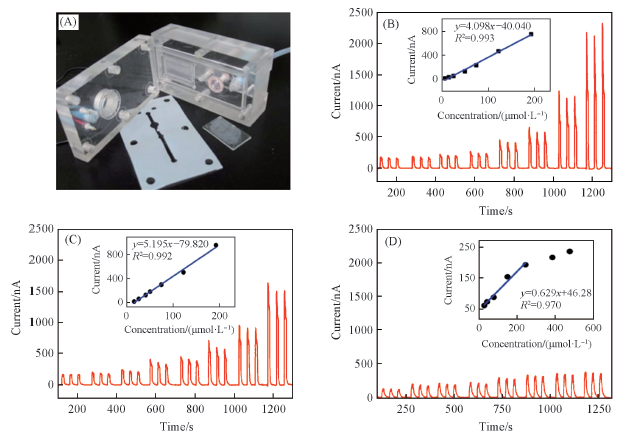
Fig.11 Photograph of the thin layer photoelectrochemical flow cell(A), concentration-dependent photocurrent of CA(B), CT(C) and AA(D)[38]Insets in (B)—(D) are the linear curves of CA, CT and AA, respectively. Copyright 2014, Royal Society of Chemistry.
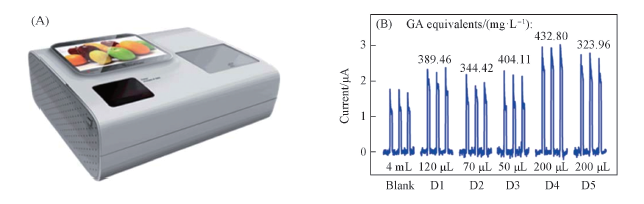
Fig.12 Photograph of the integrated PEC platform(A) and photocurrent responses of the BiMo0.015V0.985O4 modified ITO electrode in the PEC platform without(blank: PBS solution) and with a certain volume of juice into 4 mL PBS(0.1 mol/L, pH=7.4)(B)[35]Copyright 2015, Royal Society of Chemistry.
| Practical sample | Antioxidant capacity/(mg·L-1) | ||
|---|---|---|---|
| PEC sensor | F-C method | DPPH method | |
| Mango | 356.38±6.80 | 369.73±2.63 | 267.61±1.46 |
| Grape | 229.40±1.13 | 247.71±0.61 | 172.73±2.92 |
| Apple | 257.45±4.95 | 252.01±2.43 | 176.86±2.53 |
| Mangosteen | 265.54±6.63 | 308.44±5.27 | 182.01±3.86 |
| Pitaye | 104.18±3.14 | 129.27±0.38 | 83.76±0.73 |
| Orange | 445.59±9.12 | 468.67±1.52 | 281.66±1.82 |
| Leechee | 540.48±1.01 | 564.39±3.04 | 325.50±3.16 |
| Kiwi fruit | 601.12±5.38 | 611.73±4.56 | 344.83±11.38 |
| Practical sample | Antioxidant capacity/(mg·L-1) | ||
|---|---|---|---|
| PEC sensor | F-C method | DPPH method | |
| Mango | 356.38±6.80 | 369.73±2.63 | 267.61±1.46 |
| Grape | 229.40±1.13 | 247.71±0.61 | 172.73±2.92 |
| Apple | 257.45±4.95 | 252.01±2.43 | 176.86±2.53 |
| Mangosteen | 265.54±6.63 | 308.44±5.27 | 182.01±3.86 |
| Pitaye | 104.18±3.14 | 129.27±0.38 | 83.76±0.73 |
| Orange | 445.59±9.12 | 468.67±1.52 | 281.66±1.82 |
| Leechee | 540.48±1.01 | 564.39±3.04 | 325.50±3.16 |
| Kiwi fruit | 601.12±5.38 | 611.73±4.56 | 344.83±11.38 |
| Practical sample | Antioxidant capacity/(mg·g-1 or mg·L-1) | ||
|---|---|---|---|
| PEC sensor | F-C method | DPPH method | |
| T1 | 18.78±0.80 | 35.82±0.25 | 54.84±0.24 |
| T2 | 73.33±1.72 | 106.56±0.53 | 144.03±0.42 |
| T3 | 108.63±1.91 | 120.87±0.70 | 161.18±0.85 |
| T4 | 52.11±1.51 | 71.60±0.23 | 106.90±0.73 |
| D1 | 387.19±0.62 | 420.30±1.07 | 203.64±2.39 |
| D2 | 345.31±1.24 | 386.24±6.77 | 193.31±2.30 |
| D3 | 396.16±4.34 | 437.52±1.32 | 318.36±7.59 |
| D4 | 429.88±4.24 | 459.78±2.97 | 325.78±2.39 |
| D5 | 316.78±0.98 | 337.46±2.21 | 185.21±1.53 |
| D6 | 393.10±0.98 | 435.85±1.14 | 250.63±4.08 |
| D7 | 457.50±3.01 | 492.08±1.14 | 566.88±3.28 |
| D8 | 712.10±13.04 | 770.78±1.02 | 731.51±1.55 |
| D9 | 641.11±9.59 | 701.33±3.70 | 728.08±1.37 |
| D10 | 436.60±4.37 | 487.50±0.41 | 522.16±4.07 |
| D11 | 296.41±0.76 | 310.27±1.70 | 164.06±2.06 |
| D12 | 352.02±5.05 | 388.18±0.62 | 198.06±2.06 |
| D13 | 391.22±1.42 | 432.17±1.03 | 232.09±4.07 |
| Practical sample | Antioxidant capacity/(mg·g-1 or mg·L-1) | ||
| PEC sensor | F-C method | DPPH method | |
| D14 | 176.01±0.76 | 137.02±0.52 | 45.29±1.63 |
| D15 | 58.07±0.36 | 34.43±0.19 | 27.96±0.73 |
| D16 | 59.29±0.27 | 40.32±0.18 | 31.07±0.48 |
| Practical sample | Antioxidant capacity/(mg·g-1 or mg·L-1) | ||
|---|---|---|---|
| PEC sensor | F-C method | DPPH method | |
| T1 | 18.78±0.80 | 35.82±0.25 | 54.84±0.24 |
| T2 | 73.33±1.72 | 106.56±0.53 | 144.03±0.42 |
| T3 | 108.63±1.91 | 120.87±0.70 | 161.18±0.85 |
| T4 | 52.11±1.51 | 71.60±0.23 | 106.90±0.73 |
| D1 | 387.19±0.62 | 420.30±1.07 | 203.64±2.39 |
| D2 | 345.31±1.24 | 386.24±6.77 | 193.31±2.30 |
| D3 | 396.16±4.34 | 437.52±1.32 | 318.36±7.59 |
| D4 | 429.88±4.24 | 459.78±2.97 | 325.78±2.39 |
| D5 | 316.78±0.98 | 337.46±2.21 | 185.21±1.53 |
| D6 | 393.10±0.98 | 435.85±1.14 | 250.63±4.08 |
| D7 | 457.50±3.01 | 492.08±1.14 | 566.88±3.28 |
| D8 | 712.10±13.04 | 770.78±1.02 | 731.51±1.55 |
| D9 | 641.11±9.59 | 701.33±3.70 | 728.08±1.37 |
| D10 | 436.60±4.37 | 487.50±0.41 | 522.16±4.07 |
| D11 | 296.41±0.76 | 310.27±1.70 | 164.06±2.06 |
| D12 | 352.02±5.05 | 388.18±0.62 | 198.06±2.06 |
| D13 | 391.22±1.42 | 432.17±1.03 | 232.09±4.07 |
| Practical sample | Antioxidant capacity/(mg·g-1 or mg·L-1) | ||
| PEC sensor | F-C method | DPPH method | |
| D14 | 176.01±0.76 | 137.02±0.52 | 45.29±1.63 |
| D15 | 58.07±0.36 | 34.43±0.19 | 27.96±0.73 |
| D16 | 59.29±0.27 | 40.32±0.18 | 31.07±0.48 |

Fig.13 Inside(A), the whole instrument(B), one part of the two-EPCS(C), scheme of microfluidic chip(D) and profile of concentration vs. photocurrent of GA on the microfluidic chip(E)[41](1) The counter electrode(d=8.5 mm); (2) the working electrode(d=10.0 mm); (3) the inlet; (4) the outlet; (5) the lead wire of counter electrode; (6) the lead wire of working electrode. Copyright 2015, Elsevier.
| [1] | Sohal R S .,Weindruch R., Science, 1996, ( 273), 59— 63 |
| [2] | Harman D ., Proceedings of the National Academy of Sciences of the United States of America-Biological Sciences, 1981, ( 78), 7124— 7128 |
| [3] | Halliwell B ., J. Neurochem., 1992, ( 59), 1609— 1623 |
| [4] | Perera R. M., Bardeesy N., Nature, 2011, ( 475), 43— 44 |
| [5] | Mittler R ., Trends Plant Sci., 2002, ( 7), 405— 410 |
| [6] | Velioglu Y. S., Mazza G., Gao L., Oomah B. D., J. Agric. Food Chem., 1998, ( 46), 4113— 4117 |
| [7] | Eberhardt M. V ., Lee C. Y., Liu R. H., Nature, 2000, ( 405), 903— 904 |
| [8] | Bedner M., Duewer D . L.,Anal. Chem., 2011, ( 83), 6169— 6176 |
| [9] | Merken H M., .Beecher G. R., J. Agric. Food Chem., 2000, ( 48), 577— 599 |
| [10] | Pukalskas A ., van Beek T. A.,de Waard P., J. Chromatogr. A, 2005, ( 1074), 81— 88 |
| [11] | Prior R L .,Wu X. L.,Schaich K., J. Agric. Food Chem., 2005, ( 53), 4290— 4302 |
| [12] | Ozyurek M., Guclu K., Apak R ., TRAC-Trends in Anal. Chem., 2011, ( 30), 652— 664 |
| [13] | Ozyurek M., Gungor N., Baki S., Guclu K., Apak R ., Anal. Chem., 2012, ( 84), 8052— 8059 |
| [14] | Kilmartin P. A., Zou H. L.,Waterhouse A. L., J. Agric. Food Chem., 2001, ( 49), 1957— 1965 |
| [15] | Crevillen A. G ., Avila M.,Pumera M.,Gonzalez M. C.,Escarpa A., Anal. Chem., 2007, ( 79), 7408— 7415 |
| [16] | Liu J., Su B., Lagger G., Tacchini P., Girault H. H., Anal. Chem., 2006, ( 78), 6879— 6884 |
| [17] | Liu J F .,Roussel C.,Lagger G.,Tacchini P.,Girault H. H., Anal. Chem., 2005, ( 77), 7687— 7694 |
| [18] | Koleva ,II, Niederlander H. A. G.,Van Beek T. A., Anal. Chem., 2000, ( 72), 2323— 2328 |
| [19] | Tsao R., Yang R., Christopher J., Zhu Y., Zhu.H H ., J. Agric. Food Chem., 2003, ( 51), 6347— 6353 |
| [20] | Li H., Li J., Xu Q., Hu X ., Anal. Chem., 2011, ( 83), 9681— 9686 |
| [21] | Pardo-Yissar V., Katz E., Wasserman J., Willner I., J. Am. Chem. Soc., 2003, (125),622—623 |
| 22 | Sheeney-Haj-Khia L., Basnar B.,Willner I Angew. Chem. Int. Ed., 2005, ( 44), 78— 83 |
| [23] | Xin Y. M., Li Z. Z., Wu W. L., Fu B. H., Wu H. J., Zhang Z. H., Biosens. Bioelectron., 2017, 87, 396— 403 |
| [24] | Li X., Yu J. G., Jaroniec M., Chem. Soc. Rev., 2016, 45, 2603— 2636 |
| [25] | Wang Q., Ruan Y., Zhao W., Lin P., Xu J., Chen H ., Anal. Chem., 2018, ( 90), 3759— 3765 |
| [26] | Yu L., Zhu Y., Liu Y., Qu P., Xu M., Shen Q., Zhao W., Anal. Chem., 2018, 90, 10803— 10811 |
| [27] | Zhan W W .,Kuang Q., Zhou J. Z.,Kong X. J.,Xie Z. X.,Zheng L. S., J. Am. Chem. Soc., 2013, ( 135), 1926— 1933 |
| [28] | Zhang .S S.,Zhang S. Q.,Peng B. Y.,Wang H. J.,Yu H.,Wang H. H.,Peng F., Electrochem. Commun., 2014, ( 40), 24— 27 |
| [29] | Tang J., Kong B., Wang Y.C .,Xu M.,Wang Y. L.,Wu H., Zheng G. F., Nano Letter, 2013, ( 13), 5350— 5354 |
| [30] | Tu W. W ., Dong Y. T., Lei J. P., Ju H. X., Anal. Chem., 2010, ( 82), 8711— 8716 |
| [31] | Zhao W W., .Ma Z. Y., Yan D. Y., Xu J. J., Chen H. Y.,Anal. Chem., 2012, ( 84), 10518— 10521 |
| [32] | Y Li .J ., Ma M. J., Zhu J. J., Anal. Chem., 2012, ( 84), 10492— 10499 |
| [33] | Zhao .W W ., Yu P. P., Shan Y., Wang J., Xu J. J., Chen H. Y., Anal. Chem., 2012, ( 84), 5892— 5897 |
| [34] | W Zhao .W., Tian C. Y., Xu J. J.,Chen H. Y., Chem. Commun., 2012, ( 48), 895 |
| [35] | Wang L N., .Han D. X., Ni S., MW. G., Wang W., Niu L.,Chem. Sci., 2015, ( 6), 6632— 6638 |
| [36] | Ma W.G, Wang L. N., Zhang N., Han D. X., Dong X. D., Niu L ., Anal. Chem., 2015, ( 87), 4844— 4850 |
| [37] | Wang L.N ., W. G. Ma, S. Y. Gan, D. X. Han, Q. X. Zhang, L. Niu, Anal. Chem., 2014, ( 86), 10171— 10178 |
| [38] | Ma W.G ., D Han. X., Zhou M., Sun H., Wang L. N., Dong X. D., Niu L., Chem. Sci., 2014, ( 5), 3946— 3951 |
| [39] | Ma W G.. Han D. X., Gan S. Y., Zhang N., Liu S. W., Wu T. S., Zhang Q. X., Dong X. D., Niu L., Chem. Commun., 2013, ( 49), 7842— 7844 |
| [40] | W Ma. G ., Han D. X., Zhang N., Li F. H., Wu T. S., Dong X. D., Niu L., Analyst, 2013, ( 138), 2335— 2342 |
| [41] | Han D.X ., Ma W. G., Wang L. N., Ni S., Zhang N., Wang W., Dong X. D., Niu L., Biosens. Bioelectron., 2015, ( 75), 458— 464 |
| [42] | Wang L., Liu Z., Wang D., Ni S., Han D., Wang W., Niu L ., Biosens. Bioelectron. 2017, ( 94), 107— 114 |
| [43] | Jiang D., Du X., Zhou L., Li H., Wang K ., Anal. Chem., 2017, ( 89), 4525— 4531 |
| [44] | Zhang Z.J., Wang W. Z, Wang, L., Sun S. M., ACS Appl. Mater. Interf., 2012, ( 4), 593— 597 |
| [45] | Li H P. .,Liu J Y., Hou W. G., Du ., Zhang R., Appl. Catal. B: Environ., 2014, ( 160), 89— 97 |
| [46] | Zhao K., Yan X., Gu Y., Kang Z., Bai Z., Cao S., Liu Y., Zhang X., Zhang Y ., Small, 2016, 12( 2), 245— 251 |
| [47] | Kang Z., Yan X., Wang Y., Bai Z., L., Zhang Z., Lin P., Zhang X., Yuan H., Zhang X., Zhang Y., Scientific Reports, 2015, 5, 7882— 7888 |
| [48] | Li Z., Zhang J., Li Y., Zhao S., Zhang P., Zhang Y., Bi J., Liu G., Yue Z., Biosens. Bioelectron., 2018, 99, 251— 258 |
| [49] | Bouayed J., Bohn T ., Oxidative Medicine and Cellular Longevity, 2010, ( 3), 228— 237 |
| [50] | Kang Q., Wang X., Ma X., Kong L., Zhang P., Shen D., Sens. Actuators B, 2016, 230, 231— 241 |
| [51] | Monteiro T., Tanaka A., Damos F., Luz R ., Food Chem., 2017, ( 227) 16— 21 |
| [52] | Monteiro T., Neto S., Damos F., Luz R., J. Electroanal. Chem., 2016, 774, 36— 41 |
| [53] | Watjen W., Michels G., Steffan B.R .,Niering P., Chovolou Y., Kampkotter A., Tran-Thi Q H., Proksch P., Kahl R., .,Journal of Nutrition, 2005, ( 135), 525— 531 |
| [54] | Galati G., Lin A., Sultan A M., O’Brien P J., .,Free Radical Biol. Med., 2006, ( 40), 570— 580 |
| [55] | Robaszkiewicz A., Balcerczyk A., Bartosz G ., Cell Biol. Int., 2007, ( 31), 1245— 1250 |
| [56] | De Marchi U., Biasutto L., Garbisa S., Toninello A., Zoratti M., Biochim. Biophys. Acta-Bioenerg., 2009, 1787, 1425— 1432 |
| [57] | Lima D. R. S., M. Cossenza, Garcia C. G., Portugal C. C., Marques F. F. d. C., Paes-de-Carvalho R., Pereira Netto A. D., Anal. Methods, 2016, 8, 5441— 5447 |
| [58] | Liu J., Chen Y., Wang W., Feng J., Liang M., Ma S., Chen X., J. Agric. Food Chem., 2016, 64, 371— 380 |
| [59] | Jothi L., Neogi S., Jaganathan S. K., Nageswaran G., Biosens. Bioelectron., 2018, 105, 236— 242 |
| [60] | Noroozifar M., Khorasani-Motlagh M., Akbari R., Bemanadi Parizi M., Biosens. Bioelectron., 2011, 28, 56— 63 |
| [61] | Chen H., Li W., Zhao P., Nie Z., Yao S ., Electrochimica Acta, 2015, ( 178), 407— 413 |
| [62] | Mazhabi R. M .,Ge L.,Q Jiang.,H Wang X.,.,Biosens. Bioelectron., 2018, ( 107), 54— 61 |
| [63] | Naczk M., Shahidi F ., J. Pharm. Biomed. Anal., 2006, ( 41), 1523— 1542 |
| [64] | Cui M., Huang J., Wang Y., Wu Y., Luo X., Biosens. Bioelectron., 2015, 68, 563— 569 |
| [65] | Andre C., Castanheira I., Cruz J., Paseiro P., Sanches-Silva A ., Trends Food Sci. Technol. 2010, ( 21), 229— 246 |
| [66] | Han F. J., Song Z. Q., Nawaz M. H., Dai M. J., Han D. F., Han L. P., Fan Y. Y., Xu J. N., Han D. X., and Niu L., Anal. Chem., 2019, 91, 10657— 10662 |
| [67] | Vikraman A., Rasheed Z., Rajith L., Lonappan L., Krishnapillai K., Food Anal. Methods, 2013, 6, 775— 780 |
| [68] | Ni S., Han F. J., Wang W., Han D. F., Bao Y., Han D. X., Wang H. Y., Niu L., Sen. Actuators B, 2018, 259, 963— 971 |
| [1] | LI Yulong, XIE Fating, GUAN Yan, LIU Jiali, ZHANG Guiqun, YAO Chao, YANG Tong, YANG Yunhui, HU Rong. A Ratiometric Electrochemical Sensor Based on Silver Ion Interaction with DNA for the Detection of Silver Ion [J]. Chem. J. Chinese Universities, 2022, 43(8): 20220202. |
| [2] | WANG Junyang, LIU Zheng, ZHANG Qian, SUN Chunyan, LI Hongxia. Application of DNA Silver Nanoclusters in the Fluorescence Biosensors based on Functional Nucleic Acids [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220010. |
| [3] | WEI Chuangyu, CHEN Yanli, JIANG Jianzhuang. Fabrication of Electrochemical Sensor for Dopamine and Uric Acid Based on a Novel Dimeric Phthalocyanine-involved Quintuple-decker Modified Indium Tin Oxide Electrode [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210582. |
| [4] | ZHAO Lingyun, HUANG Hanxiong, LUO Duyu, SU Fengchun. Effect of Flexibility of Composites on Performances of Sensors with Micro-structured Inverted Pyramid Arrays [J]. Chem. J. Chinese Universities, 2021, 42(9): 2953. |
| [5] | HUANG Luoyi, WENG Yueyue, HUANG Xuhui, WANG Chaojie. Theoretical Study on the Structures and Properties of Flavonoids in Plantain [J]. Chem. J. Chinese Universities, 2021, 42(9): 2752. |
| [6] | HUANG Chibao, KANG Shuai, PAN Qi, LYU Guoling. Carbazole-derived Dicyanostilbene Two-photon Fluorescence Probe for Lipid Raft [J]. Chem. J. Chinese Universities, 2021, 42(8): 2443. |
| [7] | PAN Xiaojun, BAO Rongrong, PAN Caofeng. Research Progress of Flexible Tactile Sensors Applied to Wearable Electronics [J]. Chem. J. Chinese Universities, 2021, 42(8): 2359. |
| [8] | CAI Yaqian, ZHANG Jiahuai, LIU Fangzhe, LI Haichao, SHI Jianping, GUAN Shuang. Protein-based Hydrogel Assisted by Hofmeister Effect for Strain Sensor [J]. Chem. J. Chinese Universities, 2021, 42(8): 2609. |
| [9] | HU Haocheng, LI Wenli, ZHANG Jianing, LIU Yubo. Extraction, Structure Characterization and Biological Activities of Oligosaccharides from Auricularia heimuer [J]. Chem. J. Chinese Universities, 2021, 42(8): 2465. |
| [10] | WU Yangyi, CHEN Jianping, Ai Yijing, WANG Qingxiang, GAO Fei, GAO Feng. Synthesis of 2-(2-Hydroxy-3-methoxyphenyl)-C60 and Its Application for Sensing of Cauliflower Mosaic Virus 35S Promotor [J]. Chem. J. Chinese Universities, 2021, 42(6): 1754. |
| [11] | YANG Yiran, YAO Hua, YAN Jianghong, SUN Zhiheng, ZHANG Yu, FANG Xueqing, LI Xuwen, JIN Yon⁃Ri. Chemical Constituents of New Steroidal Saponins from Allium chinense G. Don [J]. Chem. J. Chinese Universities, 2021, 42(6): 1742. |
| [12] | XU Mengyi, HUANG Xuewen, LI Xiaojie, WEI Wei, LIU Xiaoya. Fabrication of Biosensor Based on “Beads-on-a-String” Shaped Composite Nano-assembly Modified Screen Printed Electrode [J]. Chem. J. Chinese Universities, 2021, 42(6): 1768. |
| [13] | YANG Ruiqi, YU Xin, LIU Hong. Scientific Study of Photocatalytic Material Based on Sn3O4 [J]. Chem. J. Chinese Universities, 2021, 42(5): 1340. |
| [14] | WANG Jie, LI Ying, SHAO Liang, BAI Yang, MA Zhonglei, MA Jianzhong. Preparation and Properties of Poly(vinyl alcohol)/polypyrrole Composite Conductive Hydrogel Strain Sensor [J]. Chem. J. Chinese Universities, 2021, 42(3): 929. |
| [15] | SHA Huiwen, MA Weiting, ZHOU Xiaojuan, SONG Weixing. One-step Preparation and Applications of Laser Induced Three-dimensional Reticular Graphene [J]. Chem. J. Chinese Universities, 2021, 42(2): 607. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||