

Chem. J. Chinese Universities ›› 2019, Vol. 40 ›› Issue (5): 995.doi: 10.7503/cjcu20180768
• Physical Chemistry • Previous Articles Next Articles
SUN Guodong1, WANG Xue1, JIANG Guoliang1, XU Zhiyong2, LIU Hongmei1,*( )
)
Received:2018-11-15
Online:2019-04-04
Published:2019-04-04
Contact:
LIU Hongmei
E-mail:liuhongmei@lyu.edu.cn
Supported by:CLC Number:
TrendMD:
SUN Guodong,WANG Xue,JIANG Guoliang,XU Zhiyong,LIU Hongmei. Effects of Gas Adsorption on Two Dimensional Metal-hexaiminobenzene Frameworks†[J]. Chem. J. Chinese Universities, 2019, 40(5): 995.
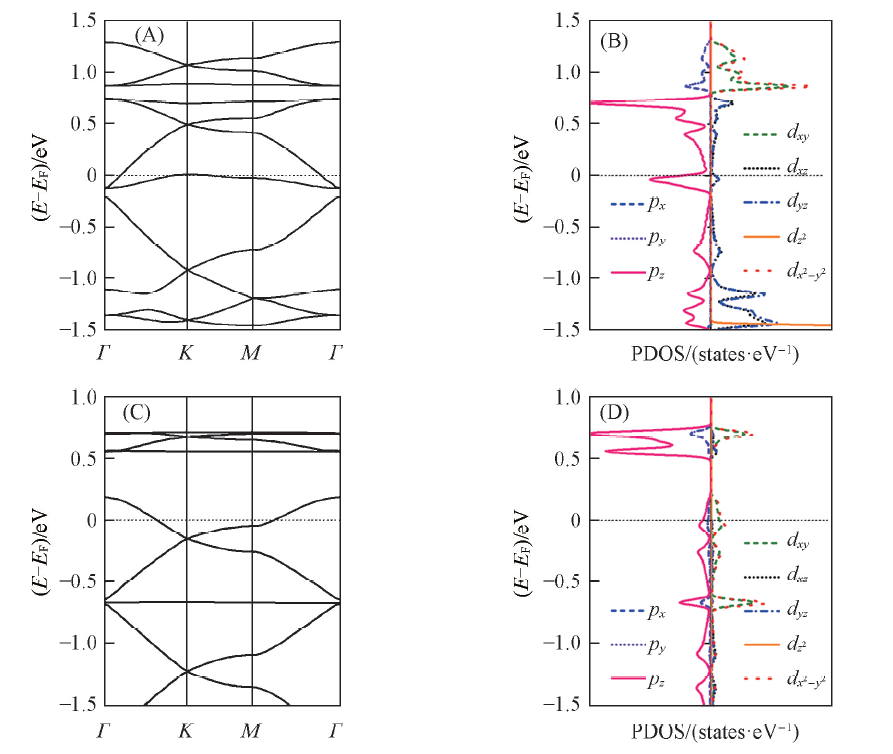
Fig.2 Band structures(A, C) and the projected density of states(PDOS)(B, D) of free-standing Ni3(HIB)2(A, B) and Cu3(HIB)2(C, D) films along the high symmetry directionsThe Fermi level is marked by dashed lines.
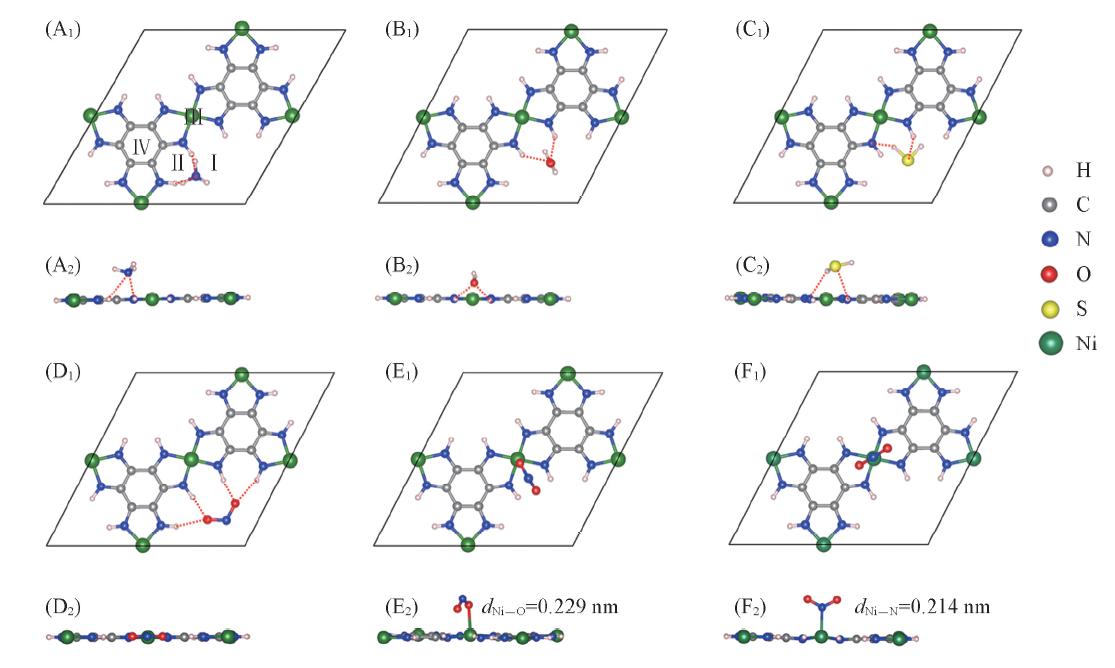
Fig.3 Top(A1—F1) and side(A2—F2) views of the adsorption structures of four molecules on Ni3(HIB)2 surfaceFour adsorption sites are denoted byⅠ, Ⅱ, Ⅲ, and Ⅳ in (A1). ⅢNi—O and ⅢNi—N denote that metal Ni binds with O or N atom in NO2 molecule. The hydrogen bonds are noted by red dotted lines. (A1, A2) NH3; (B1, B2) H2O; (C1, C2) H2S; (D1, D2) NO2; (E1, E2) NO2(ⅢNi—O); (F1, F2) NO2(ⅢNi—N).
| Species | dH—X/nm | Eads/eV | ΔQ/e |
|---|---|---|---|
| NH3(Ⅱ) | 0.247 | -0.35 | -0.011 |
| H2O(Ⅰ) | 0.229 | -0.36 | 0.005 |
| H2S(Ⅰ) | 0.259 | -0.27 | 0.008 |
| NO2(Ⅰ) | 0.179 | -1.72 | 0.847 |
| NO2(ⅢNi—O) | — | -0.83 | 0.544 |
| NO2(ⅢNi—N) | — | -0.73 | 0.468 |
Table 1 Distance dH—X between the hydrogen atom and the adsorbed atoms X(X=N for NH3, O for H2O, O for NO2)*
| Species | dH—X/nm | Eads/eV | ΔQ/e |
|---|---|---|---|
| NH3(Ⅱ) | 0.247 | -0.35 | -0.011 |
| H2O(Ⅰ) | 0.229 | -0.36 | 0.005 |
| H2S(Ⅰ) | 0.259 | -0.27 | 0.008 |
| NO2(Ⅰ) | 0.179 | -1.72 | 0.847 |
| NO2(ⅢNi—O) | — | -0.83 | 0.544 |
| NO2(ⅢNi—N) | — | -0.73 | 0.468 |
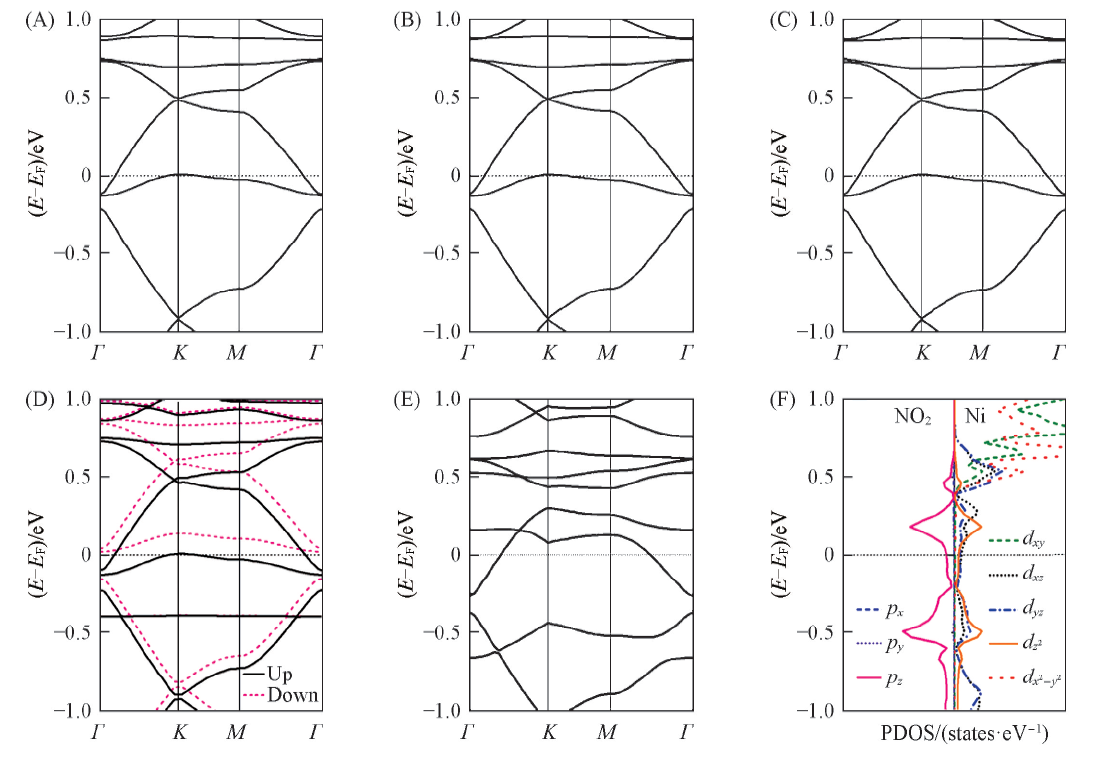
Fig.4 Band structures(A—E) of the gas-adsorbed Ni3(HIB)2 films and PDOS of NO2-adsorbed Ni3(HIB)2 film with ⅢNi—O configuration(F) (A) NH3; (B) H2O; (C) H2S; (D) NO2(Ⅰ); (E) NO2(ⅢNi—O). The Fermi level is marked by dashed lines.
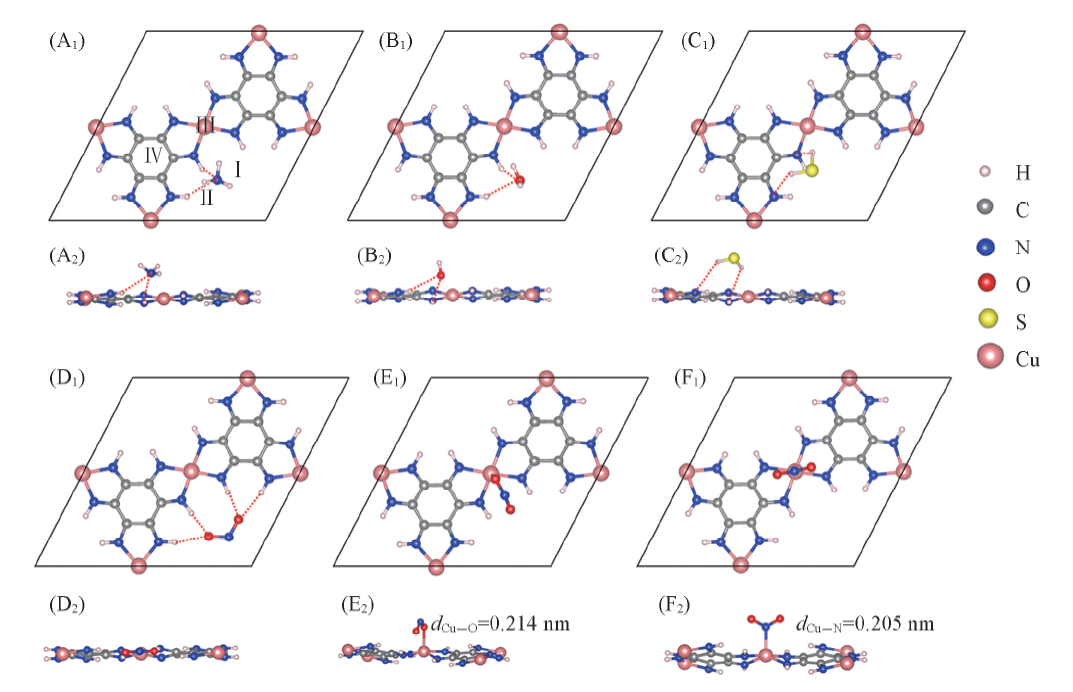
Fig.5 Top(A1—F1) and side(A2—F2) views of the adsorption structures of four molecules on Cu3(HIB)2 surface(A1, A2) NH3; (B1, B2) H2O; (C1, C2) H2S; (D1, D2) NO2; (E1, E2) NO2(ⅢCu—O) ; (F1, F2) NO2(ⅢCu—N). Four adsorption sites are denoted by Ⅰ, Ⅱ, Ⅲ, and Ⅳ in (A). ⅢCu—O and ⅢCu—N denote the adsorption configurations that Cu binds with O or Ni atom in NO2 molecule. The hydrogen bonds are noted by red dotted lines.
| Species | dH—X/nm | Eads/eV | ΔQ/e |
|---|---|---|---|
| NH3(Ⅰ) | 0.249 | -0.32 | -0.010 |
| H2O(Ⅱ) | 0.245 | -0.34 | 0.008 |
| H2S(Ⅱ) | 0.279 | -0.27 | 0.010 |
| NO2(Ⅰ) | 0.183 | -1.41 | 0.806 |
| NO2(ⅢCu—O) | — | -0.71 | 0.573 |
| NO2(ⅢCu—N) | — | -0.65 | 0.504 |
Table 2 Distance dH—X between the hydrogen atom and the adsorbed atoms X(X=N for NH3, O for H2O, O for NO2)*
| Species | dH—X/nm | Eads/eV | ΔQ/e |
|---|---|---|---|
| NH3(Ⅰ) | 0.249 | -0.32 | -0.010 |
| H2O(Ⅱ) | 0.245 | -0.34 | 0.008 |
| H2S(Ⅱ) | 0.279 | -0.27 | 0.010 |
| NO2(Ⅰ) | 0.183 | -1.41 | 0.806 |
| NO2(ⅢCu—O) | — | -0.71 | 0.573 |
| NO2(ⅢCu—N) | — | -0.65 | 0.504 |
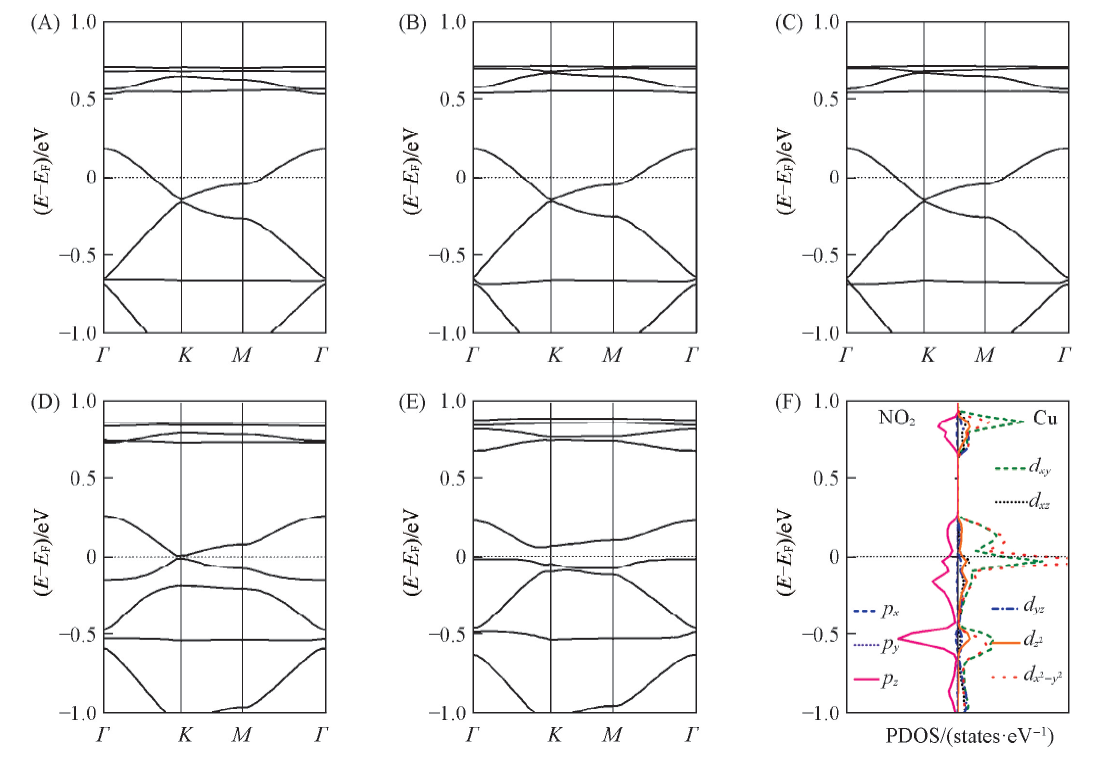
Fig.6 Band structures(A—E) of the gas-adsorbed Cu3(HIB)2 film and PDOS(F) of NO2-adsorbed Cu3(HIB)2 film with ⅢCu—O configuration (A) NH3; (B) H2O; (C) H2S; (D) NO2(Ⅰ); (E) NO2(ⅢCu—O); (F) NO2(ⅢCu—O). The Fermi level is marked by dashed lines.
| [1] | Kitagawa S., Kitaura R., Noro S. I., Angew. Chem. Int. Ed.,2004, 43, 2334—2375 |
| [2] | Zhu Q. L., Xu Q., Chem. Soc. Rev.,2014, 43, 5468—5512 |
| [3] | Xiao J. D., Li D. D., Jiang H. L., Sci. Sin. Chim.,2018, 48, 1058—1075 |
| (肖娟定, 李丹丹, 江海龙. 中国科学: 化学, 2018, 48, 1058—1075) | |
| [4] | Murray L. J., Dinc$\check{a}$ M., Long J. R., Chem. Soc. Rev.,2009, 38, 1294—1314 |
| [5] | Wu X. J., Zheng J., Li J., Cai W. Q., Acta Phys. Chim. Sin.,2013, 29, 2207—2214 |
| (吴选军, 郑佶, 李江, 蔡卫权. 物理化学学报, 2013, 29, 2207—2214) | |
| [6] | Li J. R., Kuppler R. J., Zhou H. C., Chem. Soc. Rev.,2009, 38, 1477—1504 |
| [7] | Du T., Long Y., Tang Q., Li S. L., Liu L. Y., Chem. J. Chinese Universities,2017, 38(2), 225—230 |
| (杜涛, 龙渊, 汤琦, 李生璐, 刘丽影. 高等学校化学学报,2017, 38(2), 225—230) | |
| [8] | Liu X., Luo J., Chen X., Yang Y., Yang S., Chem. Res. Chinese Universities,2017, 33(2), 268—273 |
| [9] | Clough A. J., Yoo J. W., Mecklenburg M. H., Marinescu S. C., J. Am. Chem. Soc.,2015, 137, 118—121 |
| [10] | Downes C. A., Marinescu S. C., J. Am. Chem. Soc.,2015, 137, 13740—13743 |
| [11] | Huang G., Chen Y. Z., Jiang H. L., Acta Chim. Sinica,2016, 74, 113—129 |
| (黄岗, 陈玉贞, 江海龙. 化学学报, 2016, 74, 113—129) | |
| [12] | Liu Y., Liu B., Zhou Q., Zhang T., Wu W., Chem. Res. Chinese Universities,2017, 33(6), 971—978 |
| [13] | Chen B., Yang Y., Zapata F., Lin G., Qian G., Lobkovsky E. B., Adv. Mater.,2007, 19, 1693—1696 |
| [14] | Kambe T., Sakamoto R., Hoshiko K., Takada K., Miyachi M., Ryu J. H., Sasaki S., Kim J., Nakazato K., Takata M., Nishihara H., J. Am. Chem. Soc.,2013, 135, 2462—2465 |
| [15] | Kambe T., Sakamoto R., Kusamoto T., Pal T., Fukui N., Hoshiko K., Shimojima T., Wang Z., Hirahara T., Ishizaka K., Hasegawa S., Liu F., Nishihara H., J. Am. Chem. Soc.,2014, 136, 14357—14360 |
| [16] | Downes C. A., Clough A. J., Chen K., Yoo J. W., Marinescu S. C., ACS Appl. Mater. Interfaces,2018, 10, 1719—1727 |
| [17] | Pal T., Kambe T., Kusamoto T., Foo M. L., Matsuoka R., Sakamoto R., Nishihara H., ChemPlusChem,2015, 80, 1255—1258 |
| [18] | Cui J., Xu Z., Chem. Comm.,2014, 50, 3986—3988 |
| [19] | Huang X., Sheng P., Tu Z., Zhang F., Wang J., Geng H., Zou Y., Di C. A., Yi Y., Sun Y., Xu W., Zhu D., Nature Commun.,2015, 6, 7408 |
| [20] | Campbell M. G., Sheberla D., Liu S. F., Swager T. M., Dinc$\check{a}$ M., Angew. Chem. Int. Ed.,2015, 54, 4349—4352 |
| [21] | Dou J. H., Sun L., Ge Y., Li W., Hendon C. H., Li J., Gul S., Yano J., Stach E. A., Dinc$\check{a}$ M., J. Am. Chem. Soc.,2017, 139, 13608—13611 |
| [22] | Lahiri N., Lotfizadeh N., Tsuchikawa R., Deshpande V. V., Louie J., J. Am. Chem. Soc.,2017, 139, 19—22 |
| [23] | Campbell M. G., Liu S. F., Swager T. M., Dinc$\check{a}$ M., J. Am. Chem. Soc.,2015, 43, 13780—13783 |
| [24] | Smith M. K., Jensen K. E., Pivak P. A., Mirica K. A., Chem. Mater.,2016, 28, 5264—5268 |
| [25] | Yassine O., Shekhah O., Assen A. H., Belmabkhout Y., Salama K. N., Eddaoudi M., Angew. Chem. Int. Ed.,2016, 55, 15879—15883 |
| [26] | Chakravarty C., Mandal B., Sarkar P., J. Phys. Chem. C,2016, 120, 28307—28319 |
| [27] | Liu H., Li X., Chen L., Wang X., Pan H., Zhang X., Zhao M., J. Phys. Chem. C,2016, 120, 3846—3852 |
| [28] | Liu H., Li X., Shi C., Wang D., Chen L., He Y., Zhao J., Phys. Chem. Chem. Phys.,2018, 20, 16939—16948 |
| [29] | Kresse G., Hafner J., Phys. Rev. B,1993, 47, 558—561 |
| [30] | Perdew J. P., Burke K., Ernzerhof M., Phys. Rev. Lett.,1996, 77, 3865—3868 |
| [31] | Grimme S., Antony J., Ehrlich S., Krieg H., J. Chem. Phys.,2010, 132, 154104 |
| [32] | Jiang W., Liu Z., Mei J. W., Cui B., Liu F., Nanoscale,2019, 11, 955—961 |
| [1] | ZHAO Yingzhe, ZHANG Jianling. Applications of Metal-organic Framework-based Material in Carbon Dioxide Photocatalytic Conversion [J]. Chem. J. Chinese Universities, 2022, 43(7): 20220223. |
| [2] | LU Cong, LI Zhenhua, LIU Jinlu, HUA Jia, LI Guanghua, SHI Zhan, FENG Shouhua. Synthesis, Structure and Fluorescence Detection Properties of a New Lanthanide Metal-Organic Framework Material [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220037. |
| [3] | XIA Tian, WAN Jiawei, YU Ranbo. Progress of the Structure-property Correlation of Heteroatomic Coordination Structured Carbon-based Single-atom Electrocatalysts [J]. Chem. J. Chinese Universities, 2022, 43(5): 20220162. |
| [4] | TIAN Xueqin, MO Zheng, DING Xin, WU Pengyan, WANG Yu, WANG Jian. A Squaramide-containing Luminescent Metal-organic Framework as a High Selective Sensor for Histidine [J]. Chem. J. Chinese Universities, 2022, 43(2): 20210589. |
| [5] | WANG Zumin, MENG Cheng, YU Ranbo. Doping Regulation in Transition Metal Phosphides for Hydrogen Evolution Catalysts [J]. Chem. J. Chinese Universities, 2022, 43(11): 20220544. |
| [6] | XING Peiqi, LU Tong, LI Guanghua, WANG Liyan. Controllable Syntheses of Two Cd(II) Metal-organic Frameworks Possessing Related Structures [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220218. |
| [7] | LI Shurong, WANG Lin, CHEN Yuzhen, JIANG Hailong. Research Progress of Metal⁃organic Frameworks on Liquid Phase Catalytic Chemical Hydrogen Production [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210575. |
| [8] | ZHANG Chi, SUN Fuxing, ZHU Guangshan. Synthesis, N2 Adsorption and Mixed-matrix Membrane Performance of Bimetal Isostructural CAU-21 [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210578. |
| [9] | SHI Xiaofan, ZHU Jian, BAI Tianyu, FU Zixuan, ZHANG Jijie, BU Xianhe. Research Status and Progress of MOFs with Application in Photoelectrochemical Water-splitting [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210613. |
| [10] | WU Ji, ZHANG Hao, LUO Yuhui, GENG Wuyue, LAN Yaqian. A Microporous Cationic Ga(III)-MOF with Fluorescence Properties for Selective sensing Fe3+ Ion and Nitroaromatic Compounds [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210617. |
| [11] | LI Wen, QIAO Junyi, LIU Xinyao, LIU Yunling. Zirconium-based Metal-Organic Framework with Naphthalene for Fluorescent Detection of Nitroaromatic Explosives in Water [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210654. |
| [12] | WANG Jie, HUO Haiyan, WANG Yang, ZHANG Zhong, LIU Shuxia. General Strategy for In situ Synthesis of NENU-n Series Polyoxometalate-based MOFs on Copper Foil [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210557. |
| [13] | HAN Zongsu, YU Xiaoyong, MIN Hui, SHI Wei, CHENG Peng. A Rare Earth Metal-Organic Framework with H6TTAB Ligand [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210342. |
| [14] | MO Zongwen, ZHANG Xuewen, ZHOU Haolong, ZHOU Dongdong, ZHANG Jiepeng. Guest-responses of A Porous Coordination Polymer Based on Synergistic Hydrogen Bonds [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210576. |
| [15] | LIU Xueguang, YANG Xiaoshan, MA Jingjing, LIU Weisheng. Separating Methyl Blue Selectively from the Mixture of Dyes by Europium Metal-organic Frameworks [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210715. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||