

Chem. J. Chinese Universities ›› 2019, Vol. 40 ›› Issue (1): 130.doi: 10.7503/cjcu20180453
• Physical Chemistry • Previous Articles Next Articles
ZHANG Guanghua1,*( ), DONG Qiuchen1, ZHANG Wanbin2, WANG Shuang1
), DONG Qiuchen1, ZHANG Wanbin2, WANG Shuang1
Received:2018-06-22
Online:2019-01-10
Published:2018-12-17
Contact:
ZHANG Guanghua
E-mail:zhanggh@sust.edu.cn
Supported by:CLC Number:
TrendMD:
ZHANG Guanghua,DONG Qiuchen,ZHANG Wanbin,WANG Shuang. Corrosion Inhibition of Q235 Steel by Octyl Dimethyl Benzyl Quaternary Ammonium Salt Ionic Liquid†[J]. Chem. J. Chinese Universities, 2019, 40(1): 130.
| Temperature/℃ | c(ODBA)/ (g·L-1) | V/ (mg·cm-2·h-1) | θ | η(%) | Temperature/℃ | c(ODBA)/ (g·L-1) | V/ (mg·cm-2·h-1) | θ | η(%) |
|---|---|---|---|---|---|---|---|---|---|
| 30 | 0 | 1.6509 | | —— | 40 | 0.2 | 0.1655 | 0.93 | 92.88 |
| 0.08 | 0.1547 | 0.91 | 90.63 | 0.3 | 0.1424 | 0.94 | 93.87 | ||
| 0.1 | 0.1126 | 0.93 | 93.18 | 0.5 | 0.1068 | 0.95 | 95.41 | ||
| 0.2 | 0.0738 | 0.96 | 95.53 | 50 | 0 | 3.6317 | —— | —— | |
| 0.3 | 0.0601 | 0.96 | 96.36 | 0.08 | 0.6018 | 0.83 | 83.43 | ||
| 0.5 | 0.0476 | 0.97 | 97.11 | 0.1 | 0.5287 | 0.11 | 85.44 | ||
| 40 | 0 | 2.3250 | —— | —— | 0.2 | 0.3924 | 0.33 | 89.20 | |
| 0.08 | 0.3057 | 0.87 | 86.85 | 0.3 | 0.2985 | 0.51 | 91.78 | ||
| 0.1 | 0.2614 | 0.89 | 88.77 | 0.5 | 0.2512 | 0.84 | 93.08 |
Table 1 Corrosion rate of mild steel and inhibition efficiency of various concentrations of ODBA in 1 mol/L HCl at different temperature obtained from mass loss measurements
| Temperature/℃ | c(ODBA)/ (g·L-1) | V/ (mg·cm-2·h-1) | θ | η(%) | Temperature/℃ | c(ODBA)/ (g·L-1) | V/ (mg·cm-2·h-1) | θ | η(%) |
|---|---|---|---|---|---|---|---|---|---|
| 30 | 0 | 1.6509 | | —— | 40 | 0.2 | 0.1655 | 0.93 | 92.88 |
| 0.08 | 0.1547 | 0.91 | 90.63 | 0.3 | 0.1424 | 0.94 | 93.87 | ||
| 0.1 | 0.1126 | 0.93 | 93.18 | 0.5 | 0.1068 | 0.95 | 95.41 | ||
| 0.2 | 0.0738 | 0.96 | 95.53 | 50 | 0 | 3.6317 | —— | —— | |
| 0.3 | 0.0601 | 0.96 | 96.36 | 0.08 | 0.6018 | 0.83 | 83.43 | ||
| 0.5 | 0.0476 | 0.97 | 97.11 | 0.1 | 0.5287 | 0.11 | 85.44 | ||
| 40 | 0 | 2.3250 | —— | —— | 0.2 | 0.3924 | 0.33 | 89.20 | |
| 0.08 | 0.3057 | 0.87 | 86.85 | 0.3 | 0.2985 | 0.51 | 91.78 | ||
| 0.1 | 0.2614 | 0.89 | 88.77 | 0.5 | 0.2512 | 0.84 | 93.08 |
| c(ODBA)/(g·L-1) | Ecorr/mV | icorr/(mA·cm-2) | βa/(mV·dec-1) | βc/(mV·dec-1) | η(%) |
|---|---|---|---|---|---|
| 0 | -419.1 | 1.8757 | 83 | -140 | —— |
| 0.08 | -395.7 | 0.3845 | 523 | -172 | 79.50 |
| 0.1 | -394.9 | 0.2721 | 614 | -171 | 85.49 |
| 0.2 | -390.3 | 0.2010 | 605 | -169 | 89.28 |
| 0.3 | -383.7 | 0.1315 | 508 | -187 | 92.99 |
| 0.5 | -385.3 | 0.0236 | 637 | -174 | 98.74 |
Table 2 Electrochemical parameters for the corrosion of mild steel in 1 mol/L HCl solution containing different concentrations of ODBA at 30 ℃
| c(ODBA)/(g·L-1) | Ecorr/mV | icorr/(mA·cm-2) | βa/(mV·dec-1) | βc/(mV·dec-1) | η(%) |
|---|---|---|---|---|---|
| 0 | -419.1 | 1.8757 | 83 | -140 | —— |
| 0.08 | -395.7 | 0.3845 | 523 | -172 | 79.50 |
| 0.1 | -394.9 | 0.2721 | 614 | -171 | 85.49 |
| 0.2 | -390.3 | 0.2010 | 605 | -169 | 89.28 |
| 0.3 | -383.7 | 0.1315 | 508 | -187 | 92.99 |
| 0.5 | -385.3 | 0.0236 | 637 | -174 | 98.74 |
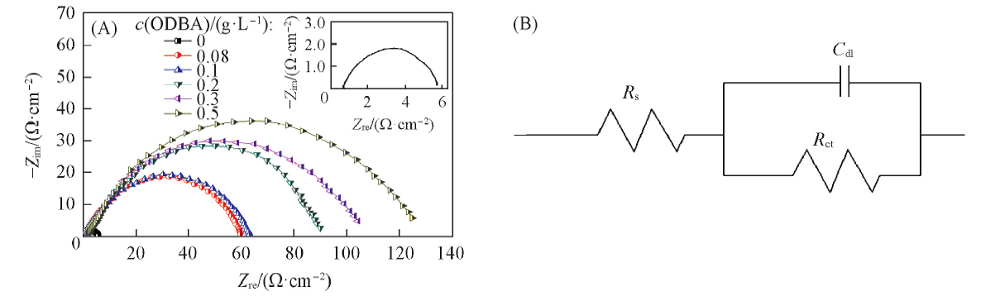
Fig.6 Nyquist plots(A) and equivalent circuit model(B) for mild steel in 1 mol/L HCl solution in the absence and in the presence of different concentrations of ODBA at 30 ℃The inset shows the enlarged plot of blank system.
| c(ODBA)/(g·L-1) | Rs/(Ω·cm2) | Rct/(Ω·cm2) | Cdl /(μF·cm-2) | n | η(%) |
|---|---|---|---|---|---|
| 0 | 0.63 | 5.7 | 543.3 | 0.80 | —— |
| 0.08 | 0.76 | 59.7 | 148.3 | 0.71 | 90.45 |
| 0.1 | 0.56 | 63.2 | 192.6 | 0.69 | 90.98 |
| 0.2 | 0.74 | 90.1 | 224.5 | 0.66 | 93.67 |
| 0.3 | 1.24 | 104.3 | 329.8 | 0.65 | 94.74 |
| 0.5 | 1.50 | 124.8 | 127.5 | 0.65 | 95.43 |
Table 3 Impedance parameters for mild steel in 1 mol/L HCl in the absence and in the presence of different concentrations of ODBA at 30 ℃
| c(ODBA)/(g·L-1) | Rs/(Ω·cm2) | Rct/(Ω·cm2) | Cdl /(μF·cm-2) | n | η(%) |
|---|---|---|---|---|---|
| 0 | 0.63 | 5.7 | 543.3 | 0.80 | —— |
| 0.08 | 0.76 | 59.7 | 148.3 | 0.71 | 90.45 |
| 0.1 | 0.56 | 63.2 | 192.6 | 0.69 | 90.98 |
| 0.2 | 0.74 | 90.1 | 224.5 | 0.66 | 93.67 |
| 0.3 | 1.24 | 104.3 | 329.8 | 0.65 | 94.74 |
| 0.5 | 1.50 | 124.8 | 127.5 | 0.65 | 95.43 |
| Temperature/℃ | Kads/(L·mol-1) | Δ | Δ | Δ |
|---|---|---|---|---|
| 30 | 42409 | -36.96 | —— | 18.38 |
| 40 | 25265 | -36.84 | -31.39 | 17.41 |
| 50 | 19936 | -37.37 | 18.51 |
Table 4 Adsorption parameters obtained from mass loss measurements for the studied compound in 1 mol/L HCl at different temperatures
| Temperature/℃ | Kads/(L·mol-1) | Δ | Δ | Δ |
|---|---|---|---|---|
| 30 | 42409 | -36.96 | —— | 18.38 |
| 40 | 25265 | -36.84 | -31.39 | 17.41 |
| 50 | 19936 | -37.37 | 18.51 |
| c(ODBA)/(g·L-1) | Ea/(kJ·mol-1) | ΔHa/(kJ·mol-1) | ΔSa/(kJ·mol-1) |
|---|---|---|---|
| 0 | 9.93 | -30.16 | -334.58 |
| 0.3 | 20.19 | -64.05 | -466.88 |
Table 5 Calculated thermodynamic parameters of adsorption from mass loss measurements
| c(ODBA)/(g·L-1) | Ea/(kJ·mol-1) | ΔHa/(kJ·mol-1) | ΔSa/(kJ·mol-1) |
|---|---|---|---|
| 0 | 9.93 | -30.16 | -334.58 |
| 0.3 | 20.19 | -64.05 | -466.88 |
| [1] | Wang X., Yang H., Wang F., Corros. Sci.,2010, 52(4), 1268—1276 |
| [2] | Guo R., Li Y. P., Tu R. X., Song B., Guo Y., Chem. J. Chinese Universities,2018, 39(5), 1018—1025 |
| (郭睿, 李云鹏, 土瑞香, 宋博, 郭煜. 高等学校化学学报, 2018, 39(5), 1018—1025) | |
| [3] | Zhang S., Tao Z., Li W., Hou B., Appl. Surf. Sci.,2009, 255(15), 6757—6763 |
| [4] | Donnelly B., Downie T. C., Grzeskowiak R., Hamburg H. R., Short D., Corros. Sci.,1978, 18(2), 109—116 |
| [5] | Trabanelli G., Corrosion, 2012, 47(6), 410—419 |
| [6] | Abdallah M., Meghed H. E., Sobhi M., Mater. Chem. Phys.,2009, 118(1), 111—117 |
| [7] | Hosseini M. G., Ehteshamzadeh M., Shahrabi T., Electrochim. Acta,2007, 52(11), 3680—3685 |
| [8] | Wang L., Corros. Sci., 2006, 48(3), 608—616 |
| [9] | Verma C., Ebenso E. E., Quraishi M. A., J. Mol. Liq.,2017, 233, 403—414 |
| [10] | Likhanova N. V., Domínguez-Aguilar M. A., Olivares-Xometl O., Nava-Entzana N., Arce E., Dorantes H., Corros. Sci.,2010, 52(6), 2088—2097 |
| [11] | Javadiana S., Yousefia A., Neshati J., Appl. Surf. Sci.,2013, 285, 674—681 |
| [12] | Godec R. F., Ind. Eng. Chem.Res., 2010, 49, 6407—6415 |
| [13] | Yousefi A., Javadian S., Neshati J., Ind. Eng. Chem. Res.,2014, 53(13), 5475—5489 |
| [14] | Wang Z. Q., Gong Y. L., Jing C., Huang H. J., Li H. R., Zhang S. T., Gao F., Corros. Sci.,2016, 113, 64—77 |
| [15] | Hanza A. P., Naderi R., Kowsari E., Sayebani M., Corros. Sci.,2016, 107, 96—106 |
| [16] | , Yildirim D., Tüken T., Corros. Sci.,2017, 120, 184—193 |
| [17] | Zarrouk A., Zarrok H., Ramli Y., Bouachrine M., Hammouti B., Sahibed-Dine A., Bentiss F., J. Mol. Liq.,2016, 222, 239—252 |
| [18] | Zhao J., Chen G., Electrochim. Acta,2012, 69(5), 247—255 |
| [19] | Hegazy M. A., Corros. Sci., 2009, 51(11), 2610—2618 |
| [20] | Li J. B., Fu L. P., Ye Z. R., Tang M. J., Zeng B., Qiu Z. C., Fine Chem.,2018, 35(5), 872—878, 900 |
| (李建波, 符罗坪, 叶正荣, 唐明进, 曾波, 裘智超. 精细化工, 2018, 35(5), 872—878, 900) | |
| [21] | Zhao T., Mu G., Corros. Sci.,1999, 41(10), 1937—1944 |
| [22] | Bouklah M., Benchat N., Hammouti B., Aouniti A., Kertit S., Mater.,2006, 60(15), 1901—1905 |
| [23] | Ahamad I., Prasad R., Quraishi M. A., Corros. Sci.,2010, 52(4), 1472—1481 |
| [24] | Amar H., Benzakour J., Derja A., Villemin D., Moreau B., Braisaz T., Appl. Surf. Sci.,2006, 252(18), 6162—6172 |
| [25] | Khaled K. F., Hackerman N., Electrochim. Acta,2003, 48(19), 2715—2723 |
| [26] | Guo R., Ma L., Wang Y. Y., Li Y. P., Tu R. X., Song B., Guo Y., Chem. Pro.,2018, 37(3), 1145—1151 |
| (郭睿, 马兰, 王映月, 李云鹏, 土瑞香, 宋博, 郭煜. 化工进展, 2018, 37(3), 1145—1151) | |
| [27] | Jeyaprabha C., Sathiyanarayanan S., Venkatachari G., Appl. Surf. Sci.,2005, 246(1), 108—116 |
| [28] | Atia A. A., Saleh M. M., J. Appl. Electrochem.,2003, 33(2), 171—177 |
| [29] | Hegazy M. A., Abdallah M., Ahmed H., Corros. Sci.,2010, 52(9), 2897—2904 |
| [30] | Okafor P. C., Zheng Y., Corros. Sci.,2009, 51(4), 850—859 |
| [31] | Xu Q. J., Li C. X., Zhou G. D., Zhu L. J., Lin C. J., Acta Phys. Chim. Sin.,2009, 25(1), 86—90 |
| (徐群杰, 李春香, 周国定, 朱律均, 林昌健. 物理化学学报, 2009, 25(1), 86—90) | |
| [32] | Lian B. J., Shi Z. M., Xu H., Zhao Q. F., Wang M. L., Jiang Y. Y., Hu S. Q., Surf. Tech.,2015, 44(12), 19—26 |
| (廉兵杰, 石泽民, 徐慧, 赵起锋, 王木立, 姜云瑛, 胡松青. 表面技术, 2015, 44(12), 19—26) | |
| [33] | Liu Z., Li B. R., Pan Y. X., Shi K., Wang W. C., Chem. J. Chinese Universities,2017, 38(4), 669—677 |
| (刘志, 李炳睿, 潘艳雄, 石凯, 王伟财. 高等学校化学学报, 2017, 38(4), 669—677) | |
| [34] | Zhang W., Ma R., Liu H., Liu Y., Li S., Niu L., J. Mol. Liq.,2016, 222, 671—679 |
| [35] | Solmaz R., Kardaş G., Yazıcı B., Erbil M., Colloids Surf. A: Physicochem. Eng. Aspects,2008, 312(1), 7—17 |
| [36] | Wang X., Yang H., Wang F.,Corros. Sci.,2011, 53(1), 113—121 |
| [37] | Halliburton W. D., J. Appl. Electrochem., 2001, 31(11), 1221—1226 |
| [38] | Zarrouk A., Zarrok H., Ramli Y., Bouachrine M., Hammouti B., Sahibed-Dine A., J. Mol. Liq.,2016, 222, 239—252 |
| [39] | Li X. H., Deng S. D., Mu G. N., Fu H., Yang F. Z., Corros. Sci.,2008, 50(2), 420—430 |
| [40] | Abdallah M., Corros. Sci., 2002, 44(4), 717—728 |
| [41] | Hegazy M. A., Badawi A. M., Rehim S. S. A. E., Kamel W. M., Corros. Sci.,2013, 69(2), 110—122 |
| [42] | Hu S. Q., Hu J. C., Gao Y. J., Jia X. L., Guo W. Y., CIESC J.,2011, 62(1), 147—155 |
| (胡松青, 胡建春, 高元军, 贾晓林, 郭文跃. 化工学报, 2011, 62(1), 147—155) | |
| [43] | Tebbji K., Faska N., Tounsi A., Oudda H., Benkaddour M., Hammouti B., Mater. Chem. Phys.,2007, 106(2), 260—267 |
| [44] | Oguzie E. E., Njoku V. O., Enenebeaku C. K., Akalezi C. O., Obi C., Corros.Sci. ,2008, 50(12), 3480—3486 |
| [1] | CUI Wei, ZHAO Deyin, BAI Wenxuan, ZHANG Xiaodong, YU Jiang. CO2 Absorption in Composite of Aprotic Solvent and Iron-based Ionic Liquid [J]. Chem. J. Chinese Universities, 2022, 43(8): 20220120. |
| [2] | PENG Kuilin, LI Guilin, JIANG Chongyang, ZENG Shaojuan, ZHANG Xiangping. Research Progress for the Role of Electrolytes in the CO2 Electrochemical Reduction [J]. Chem. J. Chinese Universities, 2022, 43(7): 20220238. |
| [3] | JIANG Hongbin, DAI Wenchen, ZHANG Rao, XU Xiaochen, CHEN Jie, YANG Guang, YANG Fenglin. Research on Co3O4/UiO-66@α-Al2O3 Ceramic Membrane Separation and Catalytic Spraying Industry VOCs Waste Gas [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220025. |
| [4] | HAO Honglei, MENG Fanyu, LI Ruoyu, LI Yingqiu, JIA Mingjun, ZHANG Wenxiang, YUAN Xiaoling. Biomass Derived Nitrogen Doped Porous Carbon Materials as Adsorbents for Removal of Methylene Blue in Water [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220055. |
| [5] | JI Shuangqi, JIN Zhao, GUAN Wenna, PAN Xiangyu, GUAN Tong. Preparation and Chromatographic Performance of Mixed-mode Silica Stationary Phase Modified by Double Cationic Ionic Liquid and Octadecyl Group [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220008. |
| [6] | WANG Hongning, HUANG Li, QING Jiang, MA Tengzhou, JIANG Wei, HUANG Weiqiu, CHEN Ruoyu. Activation of Biochar from Cattail and the VOCs Adsorption Application [J]. Chem. J. Chinese Universities, 2022, 43(4): 20210824. |
| [7] | CHEN Xiaolu, YUAN Zhenyan, ZHONG Yingchun, REN Hao. Preparation of Triphenylamine Based PAF-106s via Mechanical Ball Milling and C2 Hydrocarbons Adsorption Property [J]. Chem. J. Chinese Universities, 2022, 43(3): 20210771. |
| [8] | MENG Xianglong, YANG Ge, GUO Hailing, LIU Chenguang, CHAI Yongming, WANG Chunzheng, GUO Yongmei. Synthesis of Nano-zeolite and Its Adsorption Performance for Hydrogen Sulfide [J]. Chem. J. Chinese Universities, 2022, 43(3): 20210687. |
| [9] | CHANG Sihui, CHEN Tao, ZHAO Liming, QIU Yongjun. Thermal Degradation Mechanism of Bio-based Polybutylactam Plasticized by Ionic Liquids [J]. Chem. J. Chinese Universities, 2022, 43(11): 20220353. |
| [10] | TAN Lejian, ZHONG Xuanshu, WANG Jin, LIU Zongjian, ZHANG Aiying, YE Lin, FENG Zengguo. Low Critical Dissolution Temperature Behavior of β⁃Cyclodextrin and Its Application in the Preparation of β⁃Cyclodextrin Sheet Crystal with Ordered Nano⁃channel [J]. Chem. J. Chinese Universities, 2022, 43(11): 20220405. |
| [11] | ZHENG Meiqi, MAO Fangqi, KONG Xianggui, DUAN Xue. Layered Double Hydroxides as Sorbent for Remediation of Radioactive Wastewater [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220456. |
| [12] | TIAN Xiaokang, ZHANG Qingsong, YANG Shulin, BAI Jie, CHEN Bingjie, PAN Jie, CHEN Li, WEI Yen. Porous Materials Inspired by Microbial Fermentation: Preparation Method and Application [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220216. |
| [13] | MA Jianxin, LIU Xiaodong, XU Na, LIU Guocheng, WANG Xiuli. A Multi-functional Zn(II) Coordination Polymer with Luminescence Sensing, Amperometric Sensing, and Dye Adsorption Performance [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210585. |
| [14] | ZHANG Chi, SUN Fuxing, ZHU Guangshan. Synthesis, N2 Adsorption and Mixed-matrix Membrane Performance of Bimetal Isostructural CAU-21 [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210578. |
| [15] | LIU Changhui, LIANG Guojun, LI Yanlu, CHENG Xiufeng, ZHAO Xian. Density Functional Theory Study of NH3 Adsorption on Boron Nanotubes [J]. Chem. J. Chinese Universities, 2021, 42(7): 2263. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||