

Chem. J. Chinese Universities ›› 2017, Vol. 38 ›› Issue (12): 2198.doi: 10.7503/cjcu20170205
• Organic Chemistry • Previous Articles Next Articles
SUN Liwen1, ZHANG Yunxiao1,2, CAI Tao1,*( ), SHANG Wangji1,2, LIU Dan1, TONG Dingyi1, LIU Shenggao1
), SHANG Wangji1,2, LIU Dan1, TONG Dingyi1, LIU Shenggao1
Received:2017-04-06
Online:2017-12-10
Published:2017-11-21
Contact:
CAI Tao
E-mail:caitao@nimte.ac.cn
Supported by:CLC Number:
TrendMD:
SUN Liwen, ZHANG Yunxiao, CAI Tao, SHANG Wangji, LIU Dan, TONG Dingyi, LIU Shenggao. Imidazolium-bis(salicylato)borate Ionic Liquids as Lubricant Additives: the Effect of Alkyl Chain Lengths on the Tribological Performance†[J]. Chem. J. Chinese Universities, 2017, 38(12): 2198.
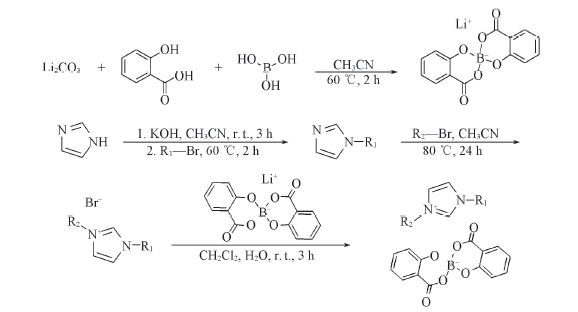
Scheme 1 Synthesis routes of D-CnImBScB(n=4, 6, 8, 10) and CnImC8BScB(n=4, 6, 10)D-C4ImBScB: R1=R2=butyl; D-C6ImBScB: R1=R2=hexyl; D-C8ImBScB: R1=R2=octyl; D-C10ImBScB: R1=R2=decyl; C4ImC8BScB: R1=butyl, R2=octyl; C6ImC8BScB: R1=hexyl, R2=octyl; C10ImC8BScB: R1=decyl, R2=octyl.
| Compd. | 1H NMR(400 MHz, CDCl3), δ |
|---|---|
| D-C4ImBScB | 0.831(t, J=4.0 Hz, 6H, CH3), 1.159—1.252(m, 4H, CH2), 1.654—1. 729(m, 4H, CH2), 4.052(t, J=7.4 Hz, 4H, CH2), 6.874(dd, J=8.4, 13.2 Hz, 4H, C6H4), 7.129(s, 2H, C6H4), 7.387(t, J=7.6 Hz, 2H, C6H4), 7.858(d, J=7.6, 8.0 Hz, 2H, Imi—CH—CH—), 9.274(s, 1H, Imi—CH) |
| D-C6ImBScB | 0.813(t, J=6.8 Hz, 6H, CH3), 1.185[s, 12H, (CH2)3], 1.717(t, J=6.4 Hz, 4H, CH2), 4.056(t, J=7.6 Hz, 4H, CH2), 6.846—6.898(m, 4H, C6H4), 7.015(d, J=2.4 Hz, 2H, C6H4), 7.360—7.408(m, 2H, C6H4), 7.865(d, J=1.2 Hz, 2H, Imi—CH—CH—), 9.307(s, 1H, Imi—CH) |
| D-C8ImBScB | 0.855(t, J=6.8 Hz, 6H, CH3), 1.194—1.271[m, 20H, (CH2)5], 1.739(t, J=5.6 Hz, 4H, CH2), 4.087(t, J=7.6 Hz, 4H, CH2), 6.845—6.922(m, 4H, C6H4), 7.082(d, J=1.6 Hz, 2H, C6H4), 7.367—7.410(m, 2H, C6H4), 7.859(dd, J=1.6, 8.0 Hz, 2H, Imi—CH—CH—), 9.363(s, 1H, Imi—CH) |
| D-C10ImBScB | 0.871(t, J=6.8 Hz, 6H, CH3), 1.186—1.298[m, 28H, (CH2)7], 1.713—1.763(m, 4H, CH2), 4.075(t, J=7.2 Hz, 4H, CH2), 6.837—6.901(m, 4H, C6H4), 7.103(d, J=1.6 Hz, 2H, C6H4), 7.353—7.396(m, 2H, C6H4), 7.850(dd, J=1.6, 7.8 Hz, 2H, Imi—CH—CH—), 9.394(s, 1H, Imi—CH) |
| C4ImC8BScB | 0.834(q, J=7.4 Hz, 6H, CH3), 1.165—1.256[m, 12H, (CH2)5 and CH2], 1.665(dt, J=7.6, 15.4 Hz, 4H, CH2), 4.039(t, J=7.2 Hz, 4H, CH2), 6.865(t, J=8.0 Hz, 4H, C6H4), 7.144(s, 2H, C6H4), 7.379(t, J=7.8 Hz, 2H, C6H4), 7.857(d, J=7.6 Hz, 2H, Imi—CH—CH—), 9.260(s, 1H, Imi—CH) |
| C6ImC8BScB | 0.789—0.863(m, 6H, CH3), 1.165—1.242[m, 16H, (CH2)5 and (CH2)3], 1.684—1.736(m, 4H, CH2), 4.037(t, J=7.4 Hz, 4H, CH2), 6.864(t, J=8.6 Hz, 4H, C6H4), 7.139(s, 2H, C6H4), 7.357—7.399(m, 2H, C6H4), 7.855(dd, J=2.8, 9.4 Hz, 2H, Imi—CH—CH—), 9.257(s, 1H, Imi—CH) |
| C10ImC8BScB | 0.862(q, J=7.2 Hz, 6H, CH3), 1.186—1.279[m, 24H, (CH2)5 and (CH2)7], 1.713—1.763(m, 4H, CH2), 4.073(t, J=7.4 Hz, 4H, CH2), 6.844—6.913(m, 4H, C6H4), 7.092(s, 2H, C6H4), 7.384(t, J=7.8 Hz, 2H, C6H4), 7.857(d, J=7.6 Hz, 2H, Imi—CH—CH—), 9.331(s, 1H, Imi—CH) |
Table 1 1H NMR data for D-CnImBScB(n=4, 6, 8, 10) and CnImC8BScB(n=4, 6, 10)
| Compd. | 1H NMR(400 MHz, CDCl3), δ |
|---|---|
| D-C4ImBScB | 0.831(t, J=4.0 Hz, 6H, CH3), 1.159—1.252(m, 4H, CH2), 1.654—1. 729(m, 4H, CH2), 4.052(t, J=7.4 Hz, 4H, CH2), 6.874(dd, J=8.4, 13.2 Hz, 4H, C6H4), 7.129(s, 2H, C6H4), 7.387(t, J=7.6 Hz, 2H, C6H4), 7.858(d, J=7.6, 8.0 Hz, 2H, Imi—CH—CH—), 9.274(s, 1H, Imi—CH) |
| D-C6ImBScB | 0.813(t, J=6.8 Hz, 6H, CH3), 1.185[s, 12H, (CH2)3], 1.717(t, J=6.4 Hz, 4H, CH2), 4.056(t, J=7.6 Hz, 4H, CH2), 6.846—6.898(m, 4H, C6H4), 7.015(d, J=2.4 Hz, 2H, C6H4), 7.360—7.408(m, 2H, C6H4), 7.865(d, J=1.2 Hz, 2H, Imi—CH—CH—), 9.307(s, 1H, Imi—CH) |
| D-C8ImBScB | 0.855(t, J=6.8 Hz, 6H, CH3), 1.194—1.271[m, 20H, (CH2)5], 1.739(t, J=5.6 Hz, 4H, CH2), 4.087(t, J=7.6 Hz, 4H, CH2), 6.845—6.922(m, 4H, C6H4), 7.082(d, J=1.6 Hz, 2H, C6H4), 7.367—7.410(m, 2H, C6H4), 7.859(dd, J=1.6, 8.0 Hz, 2H, Imi—CH—CH—), 9.363(s, 1H, Imi—CH) |
| D-C10ImBScB | 0.871(t, J=6.8 Hz, 6H, CH3), 1.186—1.298[m, 28H, (CH2)7], 1.713—1.763(m, 4H, CH2), 4.075(t, J=7.2 Hz, 4H, CH2), 6.837—6.901(m, 4H, C6H4), 7.103(d, J=1.6 Hz, 2H, C6H4), 7.353—7.396(m, 2H, C6H4), 7.850(dd, J=1.6, 7.8 Hz, 2H, Imi—CH—CH—), 9.394(s, 1H, Imi—CH) |
| C4ImC8BScB | 0.834(q, J=7.4 Hz, 6H, CH3), 1.165—1.256[m, 12H, (CH2)5 and CH2], 1.665(dt, J=7.6, 15.4 Hz, 4H, CH2), 4.039(t, J=7.2 Hz, 4H, CH2), 6.865(t, J=8.0 Hz, 4H, C6H4), 7.144(s, 2H, C6H4), 7.379(t, J=7.8 Hz, 2H, C6H4), 7.857(d, J=7.6 Hz, 2H, Imi—CH—CH—), 9.260(s, 1H, Imi—CH) |
| C6ImC8BScB | 0.789—0.863(m, 6H, CH3), 1.165—1.242[m, 16H, (CH2)5 and (CH2)3], 1.684—1.736(m, 4H, CH2), 4.037(t, J=7.4 Hz, 4H, CH2), 6.864(t, J=8.6 Hz, 4H, C6H4), 7.139(s, 2H, C6H4), 7.357—7.399(m, 2H, C6H4), 7.855(dd, J=2.8, 9.4 Hz, 2H, Imi—CH—CH—), 9.257(s, 1H, Imi—CH) |
| C10ImC8BScB | 0.862(q, J=7.2 Hz, 6H, CH3), 1.186—1.279[m, 24H, (CH2)5 and (CH2)7], 1.713—1.763(m, 4H, CH2), 4.073(t, J=7.4 Hz, 4H, CH2), 6.844—6.913(m, 4H, C6H4), 7.092(s, 2H, C6H4), 7.384(t, J=7.8 Hz, 2H, C6H4), 7.857(d, J=7.6 Hz, 2H, Imi—CH—CH—), 9.331(s, 1H, Imi—CH) |
| Temp./℃ | Kinematic viscosity/(mm2·s-1) | |||||||
|---|---|---|---|---|---|---|---|---|
| PEG200 | D-C4ImBScB | D-C6ImBScB | D-C8ImBScB | D-C10ImBScB | C4ImC8BScB | C6ImC8BScB | C10ImC8BScB | |
| 40 | 22.99 | 24.09 | 24.41 | 24.05 | 23.83 | 24.56 | 24.24 | 24.28 |
| 100 | 4.20 | 4.18 | 4.22 | 4.19 | 4.21 | 4.16 | 4.21 | 4.26 |
Table 2 Kinematic viscosity of blends of ionic liquid(0.9 mmol/20 g PEG) with PEG
| Temp./℃ | Kinematic viscosity/(mm2·s-1) | |||||||
|---|---|---|---|---|---|---|---|---|
| PEG200 | D-C4ImBScB | D-C6ImBScB | D-C8ImBScB | D-C10ImBScB | C4ImC8BScB | C6ImC8BScB | C10ImC8BScB | |
| 40 | 22.99 | 24.09 | 24.41 | 24.05 | 23.83 | 24.56 | 24.24 | 24.28 |
| 100 | 4.20 | 4.18 | 4.22 | 4.19 | 4.21 | 4.16 | 4.21 | 4.26 |
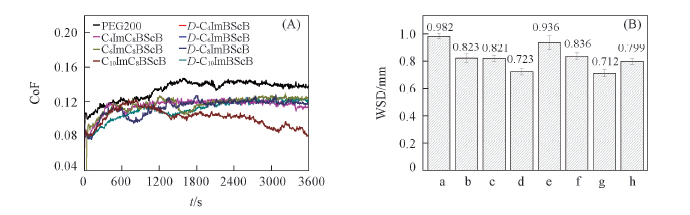
Fig.2 Coefficient of friction with contact time(A) and average wear scar diameter(B) of PEG200 and D-CnImBScB(n=4, 6, 8, 10) or CnImC8BScB(n=4, 6, 10) individually blended in PEG200a. PEG200; b. D-C4ImBScB; c. D-C6ImBScB; d. D-C8ImBScB; e. D-C10ImBScB; f. C4ImC8BScB; g. C6ImC8BScB; h. C10ImC8BScB.
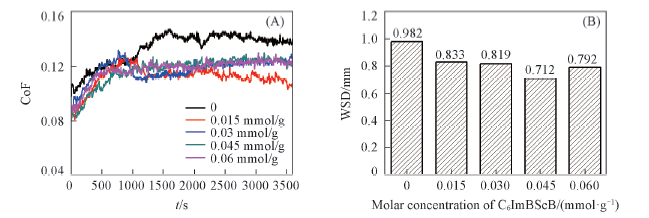
Fig.3 Coefficient of friction with contact time(A) and average wear scar diameter(B) of PEG200 and C6ImC8BScB blended in PEG200 with 0.015, 0.03, 0.045, 0.06 mmol/g PEG200, respectively

Fig.5 Elements distribution of worn area lubricated with PEG200(A) and C6ImC8BScB blends with PEG200(B)(A1) Total worn area with PEG 200; (A2) carbon; (A3) oxygen; (B1) total worn area of C6ImC8BScB blends with PEG200;(B2) carbon; (B3) oxygen; (B4) nitrogen; (B5) boron.
| Oil sample | Element | Mass fraction(%) | Atomic fraction(%) |
|---|---|---|---|
| PEG200 | C | 6.81 | 23.81 |
| O | 3.12 | 8.18 | |
| Si | 0.27 | 0.41 | |
| Cr | 1.72 | 1.39 | |
| Fe | 88.07 | 66.21 | |
| PEG200 and C6ImC8BScB | C | 8.12 | 25.69 |
| O | 2.87 | 6.81 | |
| Si | 0.23 | 0.31 | |
| Cr | 1.63 | 1.19 | |
| Fe | 84.63 | 57.57 | |
| B | 1.99 | 7.01 | |
| N | 0.53 | 1.43 |
Table 3 EDX element analysis of tribofilms formed by PEG200 and PEG200 with C6ImC8BScB
| Oil sample | Element | Mass fraction(%) | Atomic fraction(%) |
|---|---|---|---|
| PEG200 | C | 6.81 | 23.81 |
| O | 3.12 | 8.18 | |
| Si | 0.27 | 0.41 | |
| Cr | 1.72 | 1.39 | |
| Fe | 88.07 | 66.21 | |
| PEG200 and C6ImC8BScB | C | 8.12 | 25.69 |
| O | 2.87 | 6.81 | |
| Si | 0.23 | 0.31 | |
| Cr | 1.63 | 1.19 | |
| Fe | 84.63 | 57.57 | |
| B | 1.99 | 7.01 | |
| N | 0.53 | 1.43 |
| [1] | Holmberg K., Andersson P., Erdemir A., Tribol. Int., 2012, 47, 221—234 |
| [2] | Eswaraiah V., Sankaranarayanan V., Ramaprabhu S., ACS Appl. Mater. Interfaces, 2011, 3(11), 4221—4227 |
| [3] | Jaiswal V., Kalyani Rastogi R. B., Kumar R., J. Mater. Chem. A, 2014, 2(27), 10424—10434 |
| [4] | Rastogi R. B., Maurya J. L., Jaiswal V., Tribol. T., 2013, 56(4), 592—606 |
| [5] | Zhang Y. W., Zeng X. Q., Wu H., Li Z. P., Ren T. H., Zhao Y. D., Tribol. Lett., 2014, 53(3), 533—542 |
| [6] | Yan J. C., Zeng X. Q., van der Heide E., Ren T. H., Zhao Y. D., RSC Adv., 2014, 4(40), 20940—20947 |
| [7] | Pu J. B., Wan S. H., Zhao W. J., Mo Y. F., Zhang X. Q., Wang L. P., Xue Q. J., J. Phys. Chem. C, 2011, 115(27), 13275—13284 |
| [8] | Ding J., Desikan V., Han X. X., Xiao T. L., Ding R. F., Jenks W. S., Armstrong D. W., Org. Lett., 2005, 7(2), 335—337 |
| [9] | Wang B., Luo H. Y., Zhang Y. H., Sun Y. D., Liu C. J., Chem. J. Chinese Universities, 2015, 36(8), 1530—1534 |
| (王斌, 罗海燕, 张永红, 孙亚栋, 刘晨江. 高等学校化学学报, 2015, 36(8), 1530—1534) | |
| [10] | Lu Y., Moganty S. S., Schaefer J. L., Archer L. A., J. Mater. Chem., 2012, 22(9), 4066—4072 |
| [11] | Xue F., Ma R., Sun Y. D., Chem. J. Chinese Universities, 2015, 36(7), 1298—1303 |
| (薛飞, 麻荣, 孙亚栋. 高等学校化学学报, 2015, 36(7), 1298—1303) | |
| [12] | Zhu L., Zhao Q., Wu X., Zhao G., Wang X., Tribol. Int., 2016, 97, 192—199 |
| [13] | Zhenglin T., Shaohui L., Crit. Rev. Solid State, 2014, 18, 119—139 |
| [14] | Yang D. W., Li L., Wang Q., Wang X. C., Li Q. Y., Chem. J. Chinese Universities, 2016, 37(1), 94—99 |
| (杨冬伟, 李露, 王琴, 王晓春, 李青远. 高等学校化学学报, 2016, 37(1), 94—99) | |
| [15] | Qu J., Luo H., Chi M., Ma C., Blau P. J., Dai S., Viola M. B., Tribol. Int., 2014, 71, 88—97 |
| [16] | Zhou F., Liang Y., Liu W., Chem. Soc. Rev., 2009, 38(9), 2590—2599 |
| [17] | Pejakovic V., Igartua A., Kalin M., Lubr. Sci., 2015, 27(8), 489—503 |
| [18] | Mahrova M., Pagano F., Pejakovic V., Valea A., Kalin M., Igartua A., Tojo E., Tribol. Int., 2015, 82, 245—254 |
| [19] | Dilasari B., Jung Y., Sohn J., Kim S., Kwon K., Int. J. Electrochem. Sci., 2016, 11(2), 1482—1495 |
| [20] | Mu L., Shi Y., Ji T., Chen L., Yuan R., Wang H., Zhu J., ACS Appl. Mater. Interfaces, 2016, 8(7), 4977—4984 |
| [21] | Gusain R., Dhingra S., Khatri O. P., Ind. Eng. Chem. Res., 2016, 55(4), 856—865 |
| [22] | Khatri P. K., Joshi C., Thakre G. D., Jain S. L., New J. Chem., 2016, 40(6), 5294—5299 |
| [23] | Gusain R., Gupta P., Saran S., Khatri O. P., ACS Appl. Mater. Interfaces, 2014, 6(17), 15318—15328 |
| [24] | Gusain R., Singh R., Sivakumar K. L. N., Khatri O. P., RSC Adv., 2014, 4(3), 1293—1301 |
| [25] | Gusain R., Khatri O. P., RSC Adv., 2015, 5(32), 25287—25294 |
| [26] | Shah F. U., Glavatskih S., Macfarlane D. R., Somers A., Forsyth M., Antzutkin O. N., Phys. Chem. Chem. Phys., 2011, 13(28), 12865—12873 |
| [27] | Pejakov NTomastik C., Dörr N., Kalin M., Tribol. Int., 2016, 97, 234—243 |
| [28] | Rohini R., Lee C. K., Lu J., J. Chin. Chem. Soc., 2013, 60, 1—10 |
| [29] | Sharma V., Doerr N., Erdemir A., Aswath P. B., RSC Adv., 2016, 6(58), 53148—53161 |
| [30] | Fan M. J., Wang X. L., Yang D. S., Wang D. M., Van Y. Y., Zhang C. Y., Liu X. C., Tribol. Int.2015, 92, 344—352 |
| [31] | Zhou F., Liang Y., Liu W., Chem. Soc. Rev., 2009, 38, 2590—2599 |
| [32] | Rashi G., Piyush G., Sandeep S., Om P. K., RSC Adv., 2014, 4, 1293—1301 |
| [1] | CUI Wei, ZHAO Deyin, BAI Wenxuan, ZHANG Xiaodong, YU Jiang. CO2 Absorption in Composite of Aprotic Solvent and Iron-based Ionic Liquid [J]. Chem. J. Chinese Universities, 2022, 43(8): 20220120. |
| [2] | PENG Kuilin, LI Guilin, JIANG Chongyang, ZENG Shaojuan, ZHANG Xiangping. Research Progress for the Role of Electrolytes in the CO2 Electrochemical Reduction [J]. Chem. J. Chinese Universities, 2022, 43(7): 20220238. |
| [3] | JI Shuangqi, JIN Zhao, GUAN Wenna, PAN Xiangyu, GUAN Tong. Preparation and Chromatographic Performance of Mixed-mode Silica Stationary Phase Modified by Double Cationic Ionic Liquid and Octadecyl Group [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220008. |
| [4] | CHANG Sihui, CHEN Tao, ZHAO Liming, QIU Yongjun. Thermal Degradation Mechanism of Bio-based Polybutylactam Plasticized by Ionic Liquids [J]. Chem. J. Chinese Universities, 2022, 43(11): 20220353. |
| [5] | WAN Ren, SONG Fan, PENG Changjun, LIU Honglai. Group Contribution Method for Infinite Dilution Molar Conductivity of Unconventional Ions in Water [J]. Chem. J. Chinese Universities, 2021, 42(12): 3672. |
| [6] | WANG Man, WANG Xin, ZHOU Jing, GAO Guohua. Efficient Synthesis of Dimethyl Carbonate via Transesterification of Methanol and Ethylene Carbonate Catalyzed by Poly(ionic liquid)s [J]. Chem. J. Chinese Universities, 2021, 42(12): 3701. |
| [7] | ZHOU Molin, JIANG Xin, YI Ting, YANG Xiangguang, ZHANG Yibo. Improvement of Interface Stability Between Sulfide Solid Electrolyte Li10GeP2S12 and Lithium Metal [J]. Chem. J. Chinese Universities, 2020, 41(8): 1810. |
| [8] | GAO Chong,YU Fengli,XIE Congxia,YU Shitao. Baeyer-Villiger Oxidation of Cyclic Ketones Catalyzed by Amino Alcohol Heteropoly Acid Ionic Liquid † [J]. Chem. J. Chinese Universities, 2020, 41(5): 1101. |
| [9] | GAO Naiwei, MA Qiang, HE Yonglin, WANG Yapei. Green Electronic Devices Based on Ionic Liquids † [J]. Chem. J. Chinese Universities, 2020, 41(5): 901. |
| [10] | CHENG Shifu,HU Hao,CHEN Bihua,WU Haihong,GAO Guohua,HE Mingyuan. Preparation and Electrochemical Performance of Porous Carbons Prepared from Binary Ionic Liquids † [J]. Chem. J. Chinese Universities, 2020, 41(5): 1048. |
| [11] | PIAO Huilan,MA Pinyi,QIN Zucheng,JIANG Yanxiao,SUN Ying,WANG Xinghua,SONG Daqian. Determination of Triazine Herbicides from Fruit Juice Samples Using Effervescence Assisted Microextraction Method Based on Acidic Ionic Liquid Packed Syringe [J]. Chem. J. Chinese Universities, 2020, 41(2): 228. |
| [12] | ZHANG Li,QIAN Mingchao,LIU Xueke,Gao Shuaitao,YU Jiang,XIE Haishen,WANG Hongbin,SUN Fengjiang,SU Xianghong. Dynamic Study of Oxidative Desulfurization by Iron-based Ionic Liquids/NHD † [J]. Chem. J. Chinese Universities, 2020, 41(2): 317. |
| [13] | WANG Nan,YAO Kaisheng,ZHAO Chenchen,LI Tianjin,LU Weiwei. Ionic Liquid-assisted Synthesis of AuPd Nanosponges and Their Catalytic Performance † [J]. Chem. J. Chinese Universities, 2020, 41(1): 62. |
| [14] | LIU Xiaozhou, GUAN Xinyu, FANG Qianrong, JIN Yongri. Three-dimensional Covalent Organic Frameworks Synthesized by Room Temperature Ionic Liquid Method† [J]. Chem. J. Chinese Universities, 2019, 40(7): 1341. |
| [15] | LI Chenguang, HUA Er, LIU Tianxia. Tribological Behaviour of Protic Ionic Liquid Composed of 2-Ethylhexylethylenediaminium Cation and Trifluoromethanesulfonate Anion as Liquid Paraffin Additive† [J]. Chem. J. Chinese Universities, 2019, 40(7): 1411. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||