

Chem. J. Chinese Universities ›› 2020, Vol. 41 ›› Issue (4): 716.doi: 10.7503/cjcu20190504
• Organic Chemistry • Previous Articles Next Articles
LI Kangming1,LI Yansai1,YI Yangjie1,XU Leitao1,YE Jiao1,OU Xiaoming2,LI Jianming2,HU Aixi1,*( )
)
Received:2019-09-27
Online:2020-04-10
Published:2020-02-07
Contact:
Aixi HU
E-mail:axhu@hnu.edu.cn
Supported by:CLC Number:
TrendMD:
LI Kangming, LI Yansai, YI Yangjie, XU Leitao, YE Jiao, OU Xiaoming, LI Jianming, HU Aixi. Design, Synthesis and Biological Activity of 5-Pyrazole Carboxamides †[J]. Chem. J. Chinese Universities, 2020, 41(4): 716.
| Compd. | Reaction time/h | Feature | m. p./℃ | Yield(%) | HRMS(calcd.), m/z* |
|---|---|---|---|---|---|
| A1 | 4.0 | Grey solid | 148—149 | 68.8 | 364.1012(364.1310) |
| A2 | 1.0 | White solid | 149—150 | 66.0 | 378.1209(378.1507) |
| A3 | 1.0 | White solid | 157—158 | 59.5 | 392.1405(392.1707) |
| A4 | 0.5 | Yellow solid | 138—139 | 61.4 | 440.0810(440.1714) |
| A5 | 0.5 | White solid | 173—174 | 32.7 | 522.1002(522.0701) |
| A6 | 8.0 | Yellow solid | 250—252 | 48.2 | 491.0907(491.0421) |
| Compd. | Reaction time/h | Feature | m. p./℃ | Yield(%) | HRMS(calcd.), m/z* |
|---|---|---|---|---|---|
| A1 | 4.0 | Grey solid | 148—149 | 68.8 | 364.1012(364.1310) |
| A2 | 1.0 | White solid | 149—150 | 66.0 | 378.1209(378.1507) |
| A3 | 1.0 | White solid | 157—158 | 59.5 | 392.1405(392.1707) |
| A4 | 0.5 | Yellow solid | 138—139 | 61.4 | 440.0810(440.1714) |
| A5 | 0.5 | White solid | 173—174 | 32.7 | 522.1002(522.0701) |
| A6 | 8.0 | Yellow solid | 250—252 | 48.2 | 491.0907(491.0421) |
| Compd. | Reaction time/h | Feature | m. p./℃ | Yield(%) | HRMS(calcd.), m/z* |
|---|---|---|---|---|---|
| B1 | 4 | White solid | 177—179 | 28.1 | 447.0534(447.1229) |
| B2 | 10 | White solid | 185—187 | 30.9 | 461.0247(461.1349) |
| B3 | 10 | White solid | 135—138 | 31.6 | 560.0925(560.0139) |
| B4 | 10 | White solid | 218—220 | 52.4 | 574.0826(574.0229) |
| Compd. | Reaction time/h | Feature | m. p./℃ | Yield(%) | HRMS(calcd.), m/z* |
|---|---|---|---|---|---|
| B1 | 4 | White solid | 177—179 | 28.1 | 447.0534(447.1229) |
| B2 | 10 | White solid | 185—187 | 30.9 | 461.0247(461.1349) |
| B3 | 10 | White solid | 135—138 | 31.6 | 560.0925(560.0139) |
| B4 | 10 | White solid | 218—220 | 52.4 | 574.0826(574.0229) |
| Compd. | Reaction time/h | Feature | m. p./℃ | Yield(%) | HRMS(calcd.), m/z* |
|---|---|---|---|---|---|
| C1 | 8 | White solid | 126—128 | 43.6 | 327.1037(327.0640) |
| C2 | 16 | White solid | 151—152 | 69.4 | 356.0103(356.0902) |
| C3 | 4 | White solid | 126—128 | 62.0 | 357.0416(357.0717) |
| C4 | 4 | Grey solid | 164—166 | 44.2 | 394.0312(394.1113) |
| C5 | 4 | Grey solid | 177—179 | 33.5 | 432.0509(432.9812) |
| C6 | 5 | White solid | 92—94 | 58.6 | 357.0615(357.0718) |
| C7 | 4 | Yellow solid | 166—168 | 35.6 | 447.0609(447.1213) |
| C8 | 10 | White solid | 174—176 | 42.6 | 461.0913(461.1319) |
| C9 | 4 | White solid | 220—222 | 57.3 | 470.0309(469.9612) |
| C10 | 6 | White solid | 222—224 | 52.1 | 507.0629(507.0031) |
| C11 | 4 | White solid | 147—150 | 40.7 | 470.0425(469.9617) |
| C12 | 10 | White solid | 158—160 | 50.2 | 560.0523(560.0121) |
| C13 | 10 | White solid | 131—133 | 63.4 | 574.0009(574.0213) |
| Compd. | Reaction time/h | Feature | m. p./℃ | Yield(%) | HRMS(calcd.), m/z* |
|---|---|---|---|---|---|
| C1 | 8 | White solid | 126—128 | 43.6 | 327.1037(327.0640) |
| C2 | 16 | White solid | 151—152 | 69.4 | 356.0103(356.0902) |
| C3 | 4 | White solid | 126—128 | 62.0 | 357.0416(357.0717) |
| C4 | 4 | Grey solid | 164—166 | 44.2 | 394.0312(394.1113) |
| C5 | 4 | Grey solid | 177—179 | 33.5 | 432.0509(432.9812) |
| C6 | 5 | White solid | 92—94 | 58.6 | 357.0615(357.0718) |
| C7 | 4 | Yellow solid | 166—168 | 35.6 | 447.0609(447.1213) |
| C8 | 10 | White solid | 174—176 | 42.6 | 461.0913(461.1319) |
| C9 | 4 | White solid | 220—222 | 57.3 | 470.0309(469.9612) |
| C10 | 6 | White solid | 222—224 | 52.1 | 507.0629(507.0031) |
| C11 | 4 | White solid | 147—150 | 40.7 | 470.0425(469.9617) |
| C12 | 10 | White solid | 158—160 | 50.2 | 560.0523(560.0121) |
| C13 | 10 | White solid | 131—133 | 63.4 | 574.0009(574.0213) |
| Compd. | 1H NMR(400 MHz, CDCl3), δ | 13C NMR(100 MHz, CDCl3), δ |
|---|---|---|
| A1 | 8.28(s, 1H, NH), 7.05(s, 1H, benzofuran-H), 7.00(s, 1H, benzofuran-H), 4.16(s, 3H, NCH3), 3.89(s, 3H, OCH3), 3.04(s, 2H, CH2), 2.67(q, J=7.6 Hz, 2H, CH2), 1.51(s, 6H, 2CH3), 1.26(t, J=7.6 Hz, 3H, CH3) | 156.42, 149.64, 144.90, 144.30, 131.29, 129.89, 128.13, 110.45, 107.36, 104.92, 87.94, 56.02, 43.41, 40.78, 28.19, 19.29, 12.82 |
| A2 | 8.25(s, 1H, NH), 7.02(s, 2H, benzofuran-H), 4.14(d, J=6.8 Hz, 2H, CH2), 4.16(s, 3H, CH3), 3.03(s, 2H, CH2), 2.67(q, J=7.5 Hz, 2H, CH2), 1.51(s, 6H, 2CH3), 1.44(t, J=7.0 Hz, 3H, CH3), 1.27(t, J=7.5 Hz, 3H, CH3) | 156.38, 149.62, 145.28, 143.50, 131.33, 129.78, 128.42, 110.45, 107.33, 106.51, 87.74, 64.66, 43.47, 40.77, 28.18, 19.30, 14.85, 12.83 |
| A3 | 8.25(s, 1H, NH), 7.01(s, 2H, benzofuran-H), 4.17(s, 3H, NCH3), 4.03(t, J=6.9 Hz, 2H, OCH2), 3.02(s, 2H, CH2), 2.67(q, J=7.6 Hz, 2H, CH2), 1.90—1.80(m, 2H, CH2), 1.51(s, 6H, 2 CH3), 1.27(t, J=7.6 Hz, 3H, CH3), 1.02(t, J=7.4 Hz, 3H, CH3) | |
| Compd. | 1H NMR(400 MHz, CDCl3), δ | 13C NMR(100 MHz, CDCl3), δ |
| A4 | 8.19(s, 1H, NH), 7.45(d, J=7.5 Hz, 2H, Ph-H), 7.38—7.27(m, 3H, Ph-H), 7.08(s, 1H, benzofuran-H), 6.99(s, 1H, benzofuran-H), 5.19(s, 2H, OCH2), 4.15(s, 3H, NCH3), 3.03(s, 2H, CH2), 2.66(q, J=7.6 Hz, 2H, CH2), 1.52(s, 6H, 2CH3), 1.26(t, J=7.6 Hz, 3H, CH3) | 156.45, 149.72, 145.69, 143.24, 137.13, 131.42, 129.88, 128.92, 128.59, 128.01, 127.73, 111.25, 107.99, 107.45, 87.99, 71.43, 43.49, 40.84, 28.29, 19.40, 12.95 |
| A5 | 8.32(s, 1H, NH), 8.07(d, J=8.4 Hz, 1H, C6H3), 7.54(s, 1H, benzofuran-H), 7.37(d, J=8.4 Hz, 2H, C6H3), 7.26(s, 1H, benzofuran-H), 4.16(s, 3H, NCH3), 3.09(s, 2H, CH2), 2.67(q, J=7.5 Hz, 2H, CH2), 1.49(s, 6H, 2CH3), 1.26(t, J=7.5 Hz, 3H, CH3) | 162.03, 156.55, 149.80, 147.93, 136.04, 133.67, 133.47, 131.39, 130.52, 129.67, 127.24, 116.42, 114.81, 107.62, 89.30, 43.36, 40.97, 28.18, 19.42, 12.96 |
| A6 | 8.47—7.39(m, 3H, pyridine-H), 6.91(s, 1H, benzofuran-H), 6.83(s, 1H, pyrazole-H), 6.78(s, 1H, benzofuran-H), 4.01(q, J=7.0 Hz, 2H, CH2), 2.94(s, 2H, CH2), 1.47(s, 6H, 2CH3), 1.37(t, J=7.0 Hz, 3H, CH3) | 155.50, 148.85, 146.66, 145.22, 143.34, 139.99, 139.52, 129.40, 129.20, 128.27, 127.98, 125.80, 110.66, 110.14, 106.42, 87.80, 64.49, 43.38, 28.15, 14.79 |
| B1 | 10.11(s, 1H, NH), 7.27—7.03(m, 3H, benzofuran-H+thiazole-H), 4.21(s, 3H, NCH3), 3.95(s, 3H, OCH3), 3.07(s, 2H, CH2), 2.68(q, J=7.6 Hz, 2H, CH2), 1.54(s, 6H, 2CH3), 1.27(t, J=7.5 Hz, 3H, CH3) | 156.08, 155.58, 150.89, 150.07, 147.58, 144.54, 129.15, 128.41, 127.39, 115.47, 109.30, 108.94, 106.20, 88.18, 55.95, 43.26, 41.14, 28.25, 19.22, 12.71 |
| B2 | 10.38(s, 1H, NH), 7.30(s, 1H, benzofuran-H), 7.28(s, 1H, benzofuran-H), 7.02(s, 1H, thiazole-H), 4.22(t, 2H, J=7.2 Hz, CH2), 4.21(s, 3H, CH3), 3.06(s, 2H, CH2), 2.69(q, J=7.6 Hz, 2H, CH2), 1.54(s, 6H, 2CH3), 1.47(t, J=7.0 Hz, 3H, CH3), 1.28(t, J=7.6 Hz, 3H, CH3) | 156.10, 155.62, 154.05, 150.97, 150.12, 148.07, 143.79, 129.23, 128.74, 127.29, 115.55, 111.16, 106.14, 88.02, 64.59, 43.36, 41.17, 28.28, 19.21, 14.95, 12.69 |
| B3 | 8.54—7.44(m, 3H, pyridine-H), 6.98(s, 1H, pyrazole-H), 7.19—6.86(m, 3H, benzofuran-H+thiazole-H), 3.90(s, 3H, CH3), 3.06(s, 2H, CH2), 1.52(s, 6H, 2CH3) | 157.53, 154.97, 150.65, 148.37, 147.80, 146.87, 144.58, 139.56, 137.26, 128.86, 128.51, 128.05, 126.92, 125.89, 115.56, 111.40, 109.32, 106.60, 88.27, 55.83, 43.17, 28.23 |
| B4 | 10.83(s, 1H, NH), 8.55—7.43(m, 3H, pyridine-H), 6.97(s, 1H, pyrazole-H), 7.18—6.76(m, 3H, benzofuran-H+ thiazole-H), 4.14(q, J=7.0 Hz, 2H, CH2), 3.04(s, 2H, CH2), 1.50(s, 6H, 2CH3), 1.43(t, J=7.0 Hz, 3H, CH3) | 157.61, 155.02, 150.71, 148.41, 148.16, 146.90, 143.80, 139.54, 137.24, 128.87, 128.77, 128.02, 126.87, 125.88, 115.55, 111.38, 111.06, 106.54, 88.08, 64.45, 43.22, 28.22, 14.92 |
| C1 | 10.00(s, 1H, NH), 4.22(s, 3H, NCH3), 2.69(t, 2H, J=7.6 Hz, CH2), 2.67(s, 3H, CH3), 2.54(s, 3H, COCH3), 1.27(t, J=7.6 Hz, 3H, CH3) | 190.46, 157.35, 155.72, 155.51, 150.25, 128.64, 126.17, 109.39, 41.26, 30.43, 19.17, 18.09, 12.65 |
| C2 | 4.21(s, 3H, NCH3), 3.96—3.97[2s, (2.25+0.75)H, OCH3], 2.69(t, 2H, J=7.6 Hz, CH2), 2.46—2.51[2s, (0.75+2.25)H, CH3], 2.25, 2.31[2s, (2.25+0.75) H, CH3], 1.27(t, J=7.6 Hz, 3H, CH3) | |
| C3 | 10.06(s, 1H, NH), 4.32(q, J=6.9 Hz, 2H, OCH2), 4.20(s, 3H, NCH3), 2.69—2.65(m, 5H, CH3+CH2), 1.36(t, J=7.0 Hz, 3H, CH3), 1.26(t, J=7.4 Hz, 3H, CH3) | 162.52, 157.92, 156.72, 155.70, 150.22, 128.72, 116.74, 109.31, 60.96, 41.25, 19.18, 17.10, 14.34, 12.67 |
| C4 | 9.88(s, 1H, NH), 8.27(s, 1H, 1,2,4-triazole-H), 8.11(s, 1H, 1,2,4-triazole-H), 4.18(s, 3H, NCH3), 2.69(q, J=7.5 Hz, 2H, CH2), 1.27(t, J=7.6 Hz, 3H, CH3), 1.16(s, 9H, 3CH3) | 156.73, 155.70, 152.53, 152.48, 150.22, 147.36, 128.68, 119.78, 109.18, 41.19, 35.95, 29.58, 19.20, 12.70 |
| C5 | 10.16(s, 1H, NH), 7.82(d, 1H, C6H2), 7.50(d, 1H, C6H2), 7.26(s, 1H, thiazole-H), 4.20(s, 3H, NCH3), 2.68(q, J=7.6 Hz, 2H, CH2), 1.27(t, J=7.6 Hz, 3H, CH3) | 158.02, 155.64(d), 155.55, 150.17, 144.75, 132.77(d), 131.91, 128.90, 126.87(d), 120.87(d), 12.67, 19.21, 41.15, 109.14, 114.53, 118.55(d) |
| C6 | 9.98(s, 1H, NH), 6.89(s, 1H, thiazole-H), 4.20(s, 5H, COCH2+NCH3), 3.73—3.70(m, 2H, CH2), 2.71—2.61(m, 2H, CH2), 1.32—1.22(m, 6H, CH3+CH3) | 170.17, 156.39, 155.51, 150.10, 144.01, 129.08, 111.54, 109.03, 61.13, 41.12, 37.01, 19.1, 14.18, 12.68 |
| Compd. | 1H NMR(400 MHz, CDCl3), δ | 13C NMR(100 MHz, CDCl3), δ |
| C7 | 10.06(s, 1H, NH), 7.89(d, J=8.5 Hz, 1H, benzofuran-H), 7.57(s, 1H, thiazole-H), 6.48(d, J=8.5 Hz, 1H, benzofuran-H), 4.22(s, 3H, NCH3), 3.86(s, 3H, OCH3), 3.00(s, 2H, CH2), 2.68(q, J=7.3 Hz, 2H, CH2), 1.55(s, 6H, 2CH3), 1.27(t, J=7.4 Hz, 3H, CH3) | 157.30, 156.15, 155.52, 155.15, 150.04, 146.24, 129.31, 127.67, 114.18, 111.09, 109.72, 108.87, 102.80, 88.27, 55.41, 41.14, 40.08, 28.67, 19.23, 12.73 |
| C8 | 10.02(s, 1H, NH), 7.88(d, J=8.6 Hz, 1H, benzofuran-H), 7.57(s, 1H, thiazole-H), 6.47(d, J=8.7 Hz, 1H, benzofuran-H), 4.22(s, 3H, CH3), 4.09(q, J=7.0 Hz, 2H, CH2), 3.01(s, 2H, CH3), 2.69(q, J=7.6 Hz, 2H, CH2), 1.55(s, 6H, 2CH3), 1.41(t, J=7.0 Hz, 3H, CH3), 1.28(t, J=7.6 Hz, 3H, CH3) | 157.33, 155.59, 155.56, 155.20, 150.04, 146.31, 129.38, 127.57, 114.4, 110.86, 109.61, 108.88, 103.77, 88.19, 63.59, 41.10, 40.19, 28.69, 19.23, 14.96, 12.72 |
| C9 | 8.58—7.48(m, 3H, pyridine-H), 6.95(s, 1H, pyrazole-H), 4.27(q, J=7.0 Hz, 2H, CH2), 2.61(s, 3H, CH3), 1.31(t, J=7.1 Hz, 3H, CH3) | 162.25, 158.79, 155.90, 155.24, 148.22, 147.00, 140.00, 137.25, 128.98, 128.35, 126.08, 116.73, 111.84, 61.02, 16.92, 14.27 |
| C10 | 8.47(s, 1H, 1,2,4-triazole-H), 8.09(s, 1H, 1,2,4-triazole-H), 8.77—7.52(m, 3H, pyridine-H), 6.86(s, 1H, pyrazole-H), 1.10(s, 9H, 3CH3) | |
| C11 | 8.54—7.45(m, 3H, pyridine-H), 6.91(s, 1H, pyrazole-H), 6.82(s, 1H, thiazole-H), 4.18(q, J=7.1 Hz, 2H, CH2), 3.67(s, 2H, CH2), 1.26(t, J=7.0 Hz, 3H, CH3) | 170.21, 157.07, 154.83, 148.35, 146.99, 143.50, 139.67, 137.67, 128.91, 128.23, 126.02, 111.72, 111.44, 61.25, 36.90, 14.15 |
| C12 | 7.69(d, J=8.6 Hz, 1H, benzofuran), 8.55—7.43(m, 3H, pyridine-H), 7.40(s, 1H, thiazole-H), 6.92(s, 1H, pyrazole-H), 6.47(d, J=8.7 Hz, 1H, benzofuran-H), 3.84(s, 3H, OCH3), 2.98(s, 2H, CH2), 1.54(s, 6H, 2CH3) | 157.71, 157.13, 156.37, 155.90, 148.62, 146.87, 144.74, 139.49, 138.17, 129.00, 128.08, 127.23, 125.84, 114.38, 111.49, 110.02, 109.05, 103.08, 88.55, 55.42, 40.02, 28.64 |
| C13 | 10.74(s, 1H, NH), 7.65(d, J=8.6 Hz, 1H, benzofuran-H), 8.55—7.43(m, 3H, pyridine-H), 7.39(s, 1H, thiazole-H), 6.85(s, 1H, pyrazole-H), 6.43(d, J=8.7 Hz, 1H, benzofuran-H), 4.07(q, J=7.0 Hz, 2H, CH2), 2.99(s, 2H, CH2), 1.54(s, 6H, 2CH3), 1.40(t, J=7.0 Hz, 3H, CH3) | 157.85, 157.10, 156.09, 155.85, 148.65, 146.85, 146.48, 139.49, 138.45, 129.03, 128.12, 127.10, 125.83, 114.62, 111.51, 109.66, 108.69, 104.05, 88.54, 63.63, 40.12, 28.66, 14.93 |
| Compd. | 1H NMR(400 MHz, CDCl3), δ | 13C NMR(100 MHz, CDCl3), δ |
|---|---|---|
| A1 | 8.28(s, 1H, NH), 7.05(s, 1H, benzofuran-H), 7.00(s, 1H, benzofuran-H), 4.16(s, 3H, NCH3), 3.89(s, 3H, OCH3), 3.04(s, 2H, CH2), 2.67(q, J=7.6 Hz, 2H, CH2), 1.51(s, 6H, 2CH3), 1.26(t, J=7.6 Hz, 3H, CH3) | 156.42, 149.64, 144.90, 144.30, 131.29, 129.89, 128.13, 110.45, 107.36, 104.92, 87.94, 56.02, 43.41, 40.78, 28.19, 19.29, 12.82 |
| A2 | 8.25(s, 1H, NH), 7.02(s, 2H, benzofuran-H), 4.14(d, J=6.8 Hz, 2H, CH2), 4.16(s, 3H, CH3), 3.03(s, 2H, CH2), 2.67(q, J=7.5 Hz, 2H, CH2), 1.51(s, 6H, 2CH3), 1.44(t, J=7.0 Hz, 3H, CH3), 1.27(t, J=7.5 Hz, 3H, CH3) | 156.38, 149.62, 145.28, 143.50, 131.33, 129.78, 128.42, 110.45, 107.33, 106.51, 87.74, 64.66, 43.47, 40.77, 28.18, 19.30, 14.85, 12.83 |
| A3 | 8.25(s, 1H, NH), 7.01(s, 2H, benzofuran-H), 4.17(s, 3H, NCH3), 4.03(t, J=6.9 Hz, 2H, OCH2), 3.02(s, 2H, CH2), 2.67(q, J=7.6 Hz, 2H, CH2), 1.90—1.80(m, 2H, CH2), 1.51(s, 6H, 2 CH3), 1.27(t, J=7.6 Hz, 3H, CH3), 1.02(t, J=7.4 Hz, 3H, CH3) | |
| Compd. | 1H NMR(400 MHz, CDCl3), δ | 13C NMR(100 MHz, CDCl3), δ |
| A4 | 8.19(s, 1H, NH), 7.45(d, J=7.5 Hz, 2H, Ph-H), 7.38—7.27(m, 3H, Ph-H), 7.08(s, 1H, benzofuran-H), 6.99(s, 1H, benzofuran-H), 5.19(s, 2H, OCH2), 4.15(s, 3H, NCH3), 3.03(s, 2H, CH2), 2.66(q, J=7.6 Hz, 2H, CH2), 1.52(s, 6H, 2CH3), 1.26(t, J=7.6 Hz, 3H, CH3) | 156.45, 149.72, 145.69, 143.24, 137.13, 131.42, 129.88, 128.92, 128.59, 128.01, 127.73, 111.25, 107.99, 107.45, 87.99, 71.43, 43.49, 40.84, 28.29, 19.40, 12.95 |
| A5 | 8.32(s, 1H, NH), 8.07(d, J=8.4 Hz, 1H, C6H3), 7.54(s, 1H, benzofuran-H), 7.37(d, J=8.4 Hz, 2H, C6H3), 7.26(s, 1H, benzofuran-H), 4.16(s, 3H, NCH3), 3.09(s, 2H, CH2), 2.67(q, J=7.5 Hz, 2H, CH2), 1.49(s, 6H, 2CH3), 1.26(t, J=7.5 Hz, 3H, CH3) | 162.03, 156.55, 149.80, 147.93, 136.04, 133.67, 133.47, 131.39, 130.52, 129.67, 127.24, 116.42, 114.81, 107.62, 89.30, 43.36, 40.97, 28.18, 19.42, 12.96 |
| A6 | 8.47—7.39(m, 3H, pyridine-H), 6.91(s, 1H, benzofuran-H), 6.83(s, 1H, pyrazole-H), 6.78(s, 1H, benzofuran-H), 4.01(q, J=7.0 Hz, 2H, CH2), 2.94(s, 2H, CH2), 1.47(s, 6H, 2CH3), 1.37(t, J=7.0 Hz, 3H, CH3) | 155.50, 148.85, 146.66, 145.22, 143.34, 139.99, 139.52, 129.40, 129.20, 128.27, 127.98, 125.80, 110.66, 110.14, 106.42, 87.80, 64.49, 43.38, 28.15, 14.79 |
| B1 | 10.11(s, 1H, NH), 7.27—7.03(m, 3H, benzofuran-H+thiazole-H), 4.21(s, 3H, NCH3), 3.95(s, 3H, OCH3), 3.07(s, 2H, CH2), 2.68(q, J=7.6 Hz, 2H, CH2), 1.54(s, 6H, 2CH3), 1.27(t, J=7.5 Hz, 3H, CH3) | 156.08, 155.58, 150.89, 150.07, 147.58, 144.54, 129.15, 128.41, 127.39, 115.47, 109.30, 108.94, 106.20, 88.18, 55.95, 43.26, 41.14, 28.25, 19.22, 12.71 |
| B2 | 10.38(s, 1H, NH), 7.30(s, 1H, benzofuran-H), 7.28(s, 1H, benzofuran-H), 7.02(s, 1H, thiazole-H), 4.22(t, 2H, J=7.2 Hz, CH2), 4.21(s, 3H, CH3), 3.06(s, 2H, CH2), 2.69(q, J=7.6 Hz, 2H, CH2), 1.54(s, 6H, 2CH3), 1.47(t, J=7.0 Hz, 3H, CH3), 1.28(t, J=7.6 Hz, 3H, CH3) | 156.10, 155.62, 154.05, 150.97, 150.12, 148.07, 143.79, 129.23, 128.74, 127.29, 115.55, 111.16, 106.14, 88.02, 64.59, 43.36, 41.17, 28.28, 19.21, 14.95, 12.69 |
| B3 | 8.54—7.44(m, 3H, pyridine-H), 6.98(s, 1H, pyrazole-H), 7.19—6.86(m, 3H, benzofuran-H+thiazole-H), 3.90(s, 3H, CH3), 3.06(s, 2H, CH2), 1.52(s, 6H, 2CH3) | 157.53, 154.97, 150.65, 148.37, 147.80, 146.87, 144.58, 139.56, 137.26, 128.86, 128.51, 128.05, 126.92, 125.89, 115.56, 111.40, 109.32, 106.60, 88.27, 55.83, 43.17, 28.23 |
| B4 | 10.83(s, 1H, NH), 8.55—7.43(m, 3H, pyridine-H), 6.97(s, 1H, pyrazole-H), 7.18—6.76(m, 3H, benzofuran-H+ thiazole-H), 4.14(q, J=7.0 Hz, 2H, CH2), 3.04(s, 2H, CH2), 1.50(s, 6H, 2CH3), 1.43(t, J=7.0 Hz, 3H, CH3) | 157.61, 155.02, 150.71, 148.41, 148.16, 146.90, 143.80, 139.54, 137.24, 128.87, 128.77, 128.02, 126.87, 125.88, 115.55, 111.38, 111.06, 106.54, 88.08, 64.45, 43.22, 28.22, 14.92 |
| C1 | 10.00(s, 1H, NH), 4.22(s, 3H, NCH3), 2.69(t, 2H, J=7.6 Hz, CH2), 2.67(s, 3H, CH3), 2.54(s, 3H, COCH3), 1.27(t, J=7.6 Hz, 3H, CH3) | 190.46, 157.35, 155.72, 155.51, 150.25, 128.64, 126.17, 109.39, 41.26, 30.43, 19.17, 18.09, 12.65 |
| C2 | 4.21(s, 3H, NCH3), 3.96—3.97[2s, (2.25+0.75)H, OCH3], 2.69(t, 2H, J=7.6 Hz, CH2), 2.46—2.51[2s, (0.75+2.25)H, CH3], 2.25, 2.31[2s, (2.25+0.75) H, CH3], 1.27(t, J=7.6 Hz, 3H, CH3) | |
| C3 | 10.06(s, 1H, NH), 4.32(q, J=6.9 Hz, 2H, OCH2), 4.20(s, 3H, NCH3), 2.69—2.65(m, 5H, CH3+CH2), 1.36(t, J=7.0 Hz, 3H, CH3), 1.26(t, J=7.4 Hz, 3H, CH3) | 162.52, 157.92, 156.72, 155.70, 150.22, 128.72, 116.74, 109.31, 60.96, 41.25, 19.18, 17.10, 14.34, 12.67 |
| C4 | 9.88(s, 1H, NH), 8.27(s, 1H, 1,2,4-triazole-H), 8.11(s, 1H, 1,2,4-triazole-H), 4.18(s, 3H, NCH3), 2.69(q, J=7.5 Hz, 2H, CH2), 1.27(t, J=7.6 Hz, 3H, CH3), 1.16(s, 9H, 3CH3) | 156.73, 155.70, 152.53, 152.48, 150.22, 147.36, 128.68, 119.78, 109.18, 41.19, 35.95, 29.58, 19.20, 12.70 |
| C5 | 10.16(s, 1H, NH), 7.82(d, 1H, C6H2), 7.50(d, 1H, C6H2), 7.26(s, 1H, thiazole-H), 4.20(s, 3H, NCH3), 2.68(q, J=7.6 Hz, 2H, CH2), 1.27(t, J=7.6 Hz, 3H, CH3) | 158.02, 155.64(d), 155.55, 150.17, 144.75, 132.77(d), 131.91, 128.90, 126.87(d), 120.87(d), 12.67, 19.21, 41.15, 109.14, 114.53, 118.55(d) |
| C6 | 9.98(s, 1H, NH), 6.89(s, 1H, thiazole-H), 4.20(s, 5H, COCH2+NCH3), 3.73—3.70(m, 2H, CH2), 2.71—2.61(m, 2H, CH2), 1.32—1.22(m, 6H, CH3+CH3) | 170.17, 156.39, 155.51, 150.10, 144.01, 129.08, 111.54, 109.03, 61.13, 41.12, 37.01, 19.1, 14.18, 12.68 |
| Compd. | 1H NMR(400 MHz, CDCl3), δ | 13C NMR(100 MHz, CDCl3), δ |
| C7 | 10.06(s, 1H, NH), 7.89(d, J=8.5 Hz, 1H, benzofuran-H), 7.57(s, 1H, thiazole-H), 6.48(d, J=8.5 Hz, 1H, benzofuran-H), 4.22(s, 3H, NCH3), 3.86(s, 3H, OCH3), 3.00(s, 2H, CH2), 2.68(q, J=7.3 Hz, 2H, CH2), 1.55(s, 6H, 2CH3), 1.27(t, J=7.4 Hz, 3H, CH3) | 157.30, 156.15, 155.52, 155.15, 150.04, 146.24, 129.31, 127.67, 114.18, 111.09, 109.72, 108.87, 102.80, 88.27, 55.41, 41.14, 40.08, 28.67, 19.23, 12.73 |
| C8 | 10.02(s, 1H, NH), 7.88(d, J=8.6 Hz, 1H, benzofuran-H), 7.57(s, 1H, thiazole-H), 6.47(d, J=8.7 Hz, 1H, benzofuran-H), 4.22(s, 3H, CH3), 4.09(q, J=7.0 Hz, 2H, CH2), 3.01(s, 2H, CH3), 2.69(q, J=7.6 Hz, 2H, CH2), 1.55(s, 6H, 2CH3), 1.41(t, J=7.0 Hz, 3H, CH3), 1.28(t, J=7.6 Hz, 3H, CH3) | 157.33, 155.59, 155.56, 155.20, 150.04, 146.31, 129.38, 127.57, 114.4, 110.86, 109.61, 108.88, 103.77, 88.19, 63.59, 41.10, 40.19, 28.69, 19.23, 14.96, 12.72 |
| C9 | 8.58—7.48(m, 3H, pyridine-H), 6.95(s, 1H, pyrazole-H), 4.27(q, J=7.0 Hz, 2H, CH2), 2.61(s, 3H, CH3), 1.31(t, J=7.1 Hz, 3H, CH3) | 162.25, 158.79, 155.90, 155.24, 148.22, 147.00, 140.00, 137.25, 128.98, 128.35, 126.08, 116.73, 111.84, 61.02, 16.92, 14.27 |
| C10 | 8.47(s, 1H, 1,2,4-triazole-H), 8.09(s, 1H, 1,2,4-triazole-H), 8.77—7.52(m, 3H, pyridine-H), 6.86(s, 1H, pyrazole-H), 1.10(s, 9H, 3CH3) | |
| C11 | 8.54—7.45(m, 3H, pyridine-H), 6.91(s, 1H, pyrazole-H), 6.82(s, 1H, thiazole-H), 4.18(q, J=7.1 Hz, 2H, CH2), 3.67(s, 2H, CH2), 1.26(t, J=7.0 Hz, 3H, CH3) | 170.21, 157.07, 154.83, 148.35, 146.99, 143.50, 139.67, 137.67, 128.91, 128.23, 126.02, 111.72, 111.44, 61.25, 36.90, 14.15 |
| C12 | 7.69(d, J=8.6 Hz, 1H, benzofuran), 8.55—7.43(m, 3H, pyridine-H), 7.40(s, 1H, thiazole-H), 6.92(s, 1H, pyrazole-H), 6.47(d, J=8.7 Hz, 1H, benzofuran-H), 3.84(s, 3H, OCH3), 2.98(s, 2H, CH2), 1.54(s, 6H, 2CH3) | 157.71, 157.13, 156.37, 155.90, 148.62, 146.87, 144.74, 139.49, 138.17, 129.00, 128.08, 127.23, 125.84, 114.38, 111.49, 110.02, 109.05, 103.08, 88.55, 55.42, 40.02, 28.64 |
| C13 | 10.74(s, 1H, NH), 7.65(d, J=8.6 Hz, 1H, benzofuran-H), 8.55—7.43(m, 3H, pyridine-H), 7.39(s, 1H, thiazole-H), 6.85(s, 1H, pyrazole-H), 6.43(d, J=8.7 Hz, 1H, benzofuran-H), 4.07(q, J=7.0 Hz, 2H, CH2), 2.99(s, 2H, CH2), 1.54(s, 6H, 2CH3), 1.40(t, J=7.0 Hz, 3H, CH3) | 157.85, 157.10, 156.09, 155.85, 148.65, 146.85, 146.48, 139.49, 138.45, 129.03, 128.12, 127.10, 125.83, 114.62, 111.51, 109.66, 108.69, 104.05, 88.54, 63.63, 40.12, 28.66, 14.93 |
| Compd. | Inhibition rate(%) | Mortality rate(%) | |||
|---|---|---|---|---|---|
| Aa | Gz | Ss | Ms | Af | |
| A1 | 45.00 | 40.00 | 56.30 | 100 | 50.68 |
| A2 | 30.00 | 14.30 | 45.80 | 0 | 22.93 |
| A3 | 50.00 | 17.10 | 33.30 | 0 | 49.69 |
| A4 | 30.00 | 11.40 | 54.20 | 0 | 19.64 |
| A5 | 45.00 | 22.90 | 31.30 | 0 | 58.91 |
| A6 | 0 | 0 | | 0 | 54.16 |
| B1 | 6.82 | 40.58 | 12.98 | 100 | 18.21 |
| B2 | 0.00 | 5.80 | | 0 | 62.32 |
| B3 | 7.50 | 23.19 | | 0 | 100 |
| B4 | 0.00 | 0 | | 0 | 100 |
| C1 | 6.82 | 14.49 | 9.87 | 0 | 41.85 |
| C2 | | | | | |
| C3 | 22.50 | 28.99 | | 0 | 18.27 |
| C4 | 13.64 | 40.58 | 42.36 | 4.55 | 12.66 |
| C5 | 0.00 | 47.83 | 25.41 | 5.56 | 19.93 |
| C6 | 15.91 | 33.33 | 42.81 | 0 | 36.09 |
| C7 | 0.00 | 53.62 | 37.68 | 3.85 | 16.90 |
| C8 | 0.00 | 8.70 | | 0 | 47.00 |
| C9 | 0.00 | 11.59 | | 0 | 95.54 |
| C10 | 5.00 | 10.14 | | 0 | 67.21 |
| C11 | 7.50 | 0 | | 0 | 56.04 |
| C12 | | | | | |
| C13 | | | | | |
| Boscalid | | 37.00 | 100 | | |
| Azoxystrobin | | | 93.50 | | |
| Dimethachlon | 100 | | | | |
| Chlorantraniliprole | | | | 100 | |
| Imidacloprid | | | | | 100 |
| Compd. | Inhibition rate(%) | Mortality rate(%) | |||
|---|---|---|---|---|---|
| Aa | Gz | Ss | Ms | Af | |
| A1 | 45.00 | 40.00 | 56.30 | 100 | 50.68 |
| A2 | 30.00 | 14.30 | 45.80 | 0 | 22.93 |
| A3 | 50.00 | 17.10 | 33.30 | 0 | 49.69 |
| A4 | 30.00 | 11.40 | 54.20 | 0 | 19.64 |
| A5 | 45.00 | 22.90 | 31.30 | 0 | 58.91 |
| A6 | 0 | 0 | | 0 | 54.16 |
| B1 | 6.82 | 40.58 | 12.98 | 100 | 18.21 |
| B2 | 0.00 | 5.80 | | 0 | 62.32 |
| B3 | 7.50 | 23.19 | | 0 | 100 |
| B4 | 0.00 | 0 | | 0 | 100 |
| C1 | 6.82 | 14.49 | 9.87 | 0 | 41.85 |
| C2 | | | | | |
| C3 | 22.50 | 28.99 | | 0 | 18.27 |
| C4 | 13.64 | 40.58 | 42.36 | 4.55 | 12.66 |
| C5 | 0.00 | 47.83 | 25.41 | 5.56 | 19.93 |
| C6 | 15.91 | 33.33 | 42.81 | 0 | 36.09 |
| C7 | 0.00 | 53.62 | 37.68 | 3.85 | 16.90 |
| C8 | 0.00 | 8.70 | | 0 | 47.00 |
| C9 | 0.00 | 11.59 | | 0 | 95.54 |
| C10 | 5.00 | 10.14 | | 0 | 67.21 |
| C11 | 7.50 | 0 | | 0 | 56.04 |
| C12 | | | | | |
| C13 | | | | | |
| Boscalid | | 37.00 | 100 | | |
| Azoxystrobin | | | 93.50 | | |
| Dimethachlon | 100 | | | | |
| Chlorantraniliprole | | | | 100 | |
| Imidacloprid | | | | | 100 |
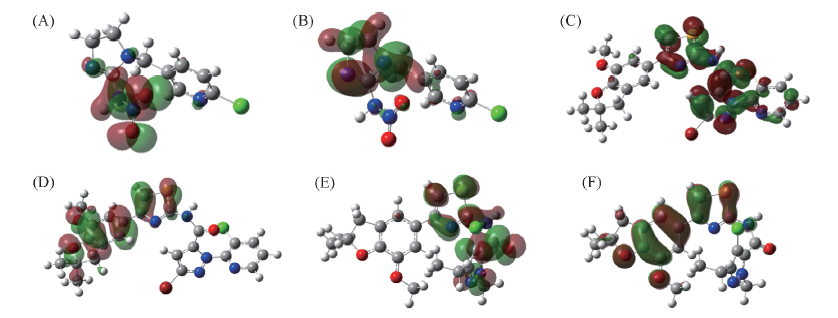
Fig.1 LUMO(A, C, E) and LUMO(B, D, F) maps for imidacloprid(A, B), B3(C, D) and B1(E, F) The green parts represent positive molecular orbital, and the red parts represent negative molecular orbital.
| DFT | Δ | ΔELUMO/Hartree | ΔEHOMO/Hartree | ΔEb/Hartree | Mortality rate(%) |
|---|---|---|---|---|---|
| Imidacloprid | -4613.41817 | -0.06741 | -0.23952 | 0.17211 | 100 |
| B1 | -2117.50553782 | -0.06094 | -0.19678 | 0.13584 | 18.21 |
| B4 | -4859.53075761 | -0.06146 | -0.20170 | 0.14024 | 100 |
| B3 | -4820.20321025 | -0.06211 | -0.20260 | 0.14049 | 100 |
| DFT | Δ | ΔELUMO/Hartree | ΔEHOMO/Hartree | ΔEb/Hartree | Mortality rate(%) |
|---|---|---|---|---|---|
| Imidacloprid | -4613.41817 | -0.06741 | -0.23952 | 0.17211 | 100 |
| B1 | -2117.50553782 | -0.06094 | -0.19678 | 0.13584 | 18.21 |
| B4 | -4859.53075761 | -0.06146 | -0.20170 | 0.14024 | 100 |
| B3 | -4820.20321025 | -0.06211 | -0.20260 | 0.14049 | 100 |
| [1] | Liu T., Tian H K.,. TongY. L., Yang C. L.,Ni Y. P Modern Agrochemicals, 2009,8(6), 5— 13 |
| ( 刘婷,田辉凯,童益利,杨春龙,倪珏萍.现代农药, 2009, 8(6), 5— 13) | |
| [2] | Kees K. L., Caggiano T., Steiner K., Fitzgerald J. J., Kates M. J., Christos T. E., Kulishoff J. M., Moore R. D., Mccaleb M. L., J. Med. Chem., 1995, 38( 4), 617— 628 |
| [3] | Zhou Q., Zhen D. D., Shi Y. J., Yao W., Qian H. W., Ding Y., Wei Z. H., Shen A. B., Feng X., Shi J., Dai H., Chin. J. Org. Chem., 2018, 38, 3318— 3325 |
| ( 周钱, 郑丹丹, 石玉军, 姚炜, 钱宏炜, 丁颖, 魏中昊, 沈爱宝, 冯霞, 石健, 戴红 . 有机化学, 2018, 38, 3318— 3325) | |
| [4] | Suresh L., Sagar V. K. P., Poornachandra Y., Ganesh K. C., Chandramouli G., Bioorg. Med. Chem. Lett., 2017, 27( 6), 1451— 1457 |
| [5] | Ma H. J., Li Y. H., Zhao Q. F., Zhang T., Xie R. L., Mei X. D., Ning J., J. Agric. Food Chem., 2010, 58, 4356— 4360 |
| [6] | Zhang Y. B., World Pesticides, ,2003, 25(6) 43— 45 |
| ( 张一宾.世界农药, 2003, 25(6), 43— 45) | |
| [7] | Bai Y. L., Agrochemicals Today, 2018, 10(11), 36— 40 |
| ( 柏亚罗.今日农药, 2018, 10(11), 36— 40) | |
| [8] | Yang G. Q., Huang Q., Chen L., Yang H. B., Yu H. B., Li B., World Pesticides, 2012, 34 6), 19— 21 |
| ( 杨桂秋,黄琦,陈霖,杨辉斌,于海波,李斌.世界农药, 2012, 34(6), 19— 21) | |
| [9] | Nagahori H., Yoshino H., Tomigahara Y., Isobe N., Kaneko H., Nakatsuka I., J. Agric. Food. Chem., 2000, 48( 11), 5760— 5770 |
| [10] | Zhang Q. K., World Pesticides, 2009, 31 3), 53— 53 |
| ( 张庆宽.世界农药, 2009, 31(3), 53— 53) | |
| [11] | Liu A. C., Yu C. H., Zhang S. K., Feng J. L., He X. L., Modern Agrochemicals, 2016, 15 1), 16— 18 |
| ( 刘安昌,余彩虹,张树康,冯佳丽,贺晓露.现代农药, 2016, 15(1), 16— 18) | |
| [12] | Wang H. Y., Mao M. Z., Wang W ., Agrochemicals, 2018, 57 4), 255— 258 |
| ( 王海洋,毛明珍,王威.农药, 2018, 57(4), 255— 258) | |
| [13] | Shi Y. J., Zhou Q., Wang Y., Qian H. W., Ye L. Y., Feng X., Chen H., Li Y. T., Dai H., Wei Z. H., Wu J. M., Chin. J. Org. Chem., 2018, 38 9), 2450— 2457 |
| ( 石玉军,周钱,王杨,钱宏炜,叶林玉,冯霞,陈辉,李雅婷,戴红,魏中昊,吴锦明.有机化学, 2018, 38(9), 2450— 2457) | |
| [14] | Zhong L. K., Jiang T., Zhang F., Fu Q., Liu X. H., Xu T. M., Ding C. R., Chen J., Yuan J., Tan C. X., Chin. J. Org. Chem., 2019, 39, 2655— 2662 |
| ( 钟良坤, 江涛, 张帆, 付庆, 刘幸海, 许天民, 丁成荣, 陈杰, 袁静, 谭成侠 . 有机化学, 2019, 39, 2655— 2662) | |
| [15] | Hong Y., Dai H., Ye L. Y., Zhong S. L., Cao X. F., Shi Y. J., Li C. J., Shi J., Shi L., Chin. J. Org. Chem., 2017, 37 11), 3006— 3012 |
| ( 洪宇,戴红,叶林玉,仲苏林,曹雄飞,石玉军,李春建,石健,施磊.有机化学, 2017, 37(11), 3006— 3012) | |
| [16] | Yan Z. Z., Liu A. P., Huang M. Z., Liu M. H., Pei H., Huang L., Yi H. B., Liu W. D., Hu A. X ., Eur. J. Med. Chem. , 2018, 149, 170— 181 |
| [17] | Luo X. F., Hu A. X., Wang Y., Ye J., Wang X. G., Ou X. M., Chem. J. Chinese Universities, 2011, 32 12), 2800— 2805 |
| ( 罗先福,胡艾希,王宇,叶姣,王晓光,欧晓明.高等学校化学学报, 2011, 32(12), 2800— 2805) | |
| [18] | Hu A. X., Lin D., Dai M. C., Luo X. F., Ye J., Ou X. M., Application of 4-(Benzofuran-5-yl)-2-phenylaminothiazole as a Bactericide, CN103141486A, 2013-06-12 |
| ( 胡艾希, 林定, 戴明崇, 罗先福, 叶姣, 欧晓明 . 4-(苯并呋喃-5-基)-2-苯氨基噻唑作为杀菌剂的应用, CN103141486A, 2013 -06-12) | |
| [19] | Yang Z. H., Li B. B., Ye J., Hu A. X., Chem. J. Chinese Universities, 2016, 37 8), 1442— 1450 |
| ( 杨子辉,李贝贝,叶姣,胡艾希.高等学校化学学报, 2016, 37(8), 1442— 1450) | |
| [20] | Iwataki I., Iwasa T., Hatano R., Moyano E. L ., Insecticidal Aminothiazole Derivatives, US 6737382B1, 2004 -03-18 |
| [21] | Zhang H. Q., Gong D. W., Xu L. Z ., Agrochemicals, 2016, 55 3), 170— 173 |
| ( 张画轻,龚大伟,许良忠.农药, 2016, 55(3), 170— 173) | |
| [22] | Ye J., Sun X. X., Qiu S. Y., Hu A. X., Chin. J. Struc. Chem., 2014, 33( 3), 429— 433 |
| [23] | Shen F., Hu A. X., Luo X. F., Ye J., Ou X. M., Chin. J. Org. Chem., 2012, 32 2), 388— 392 |
| ( 沈芳,胡艾希,罗先福,叶姣,欧晓明.有机化学, 2012, 32(2), 388— 392) | |
| [24] | Li B B., . Synthesis and Biological Activity of 2-(4-aryloxyphenoxy) Propanamide Containing Furanophenol Ether. Hunan University,Changsha, 2015 |
| ( 李贝贝 . 含呋喃酚醚的2-(4-芳氧苯氧基)丙酰胺的合成与生物活性, 长沙: 湖南大学, 2015) | |
| [25] | Ding N ., Synthesis and Antitumor Activity of the N-[4-(t-Butyl)-5-benzylthiazole-2-yl] Acetamide Derivatives, Hunan University, Changsha, 2016 |
| ( 丁娜 . N-(4-叔丁基-5-苄基噻唑-2-基)乙酰胺衍生物的合成与抗肿瘤活性, 长沙: 湖南大学, 2016) | |
| [26] | Chen N C.. , Pesticide Bioassay Technology, Beijin Agricultural University Press, Beijing, 1991 |
| ( 陈年春 . 农药生物测定技术, 北京:北京农业大学出版社, 1991) | |
| [27] | Frisch M . J., Trucks G. W., Schlegel H. B.,Scuseria G. E., Robb M. A Cheeseman J. R., Scalmani G.,Barone V.,Mennucci B., Petersson G. A., Nakatsuji H.,Caricato M.,Li X., Hratchian H. P., Izmaylov A. F.,Bloino J.,Zheng G., Sonnenberg J. L., Hada M.,Ehara M.,Toyota K., Fukuda R., Hasegawa J.,Ishida M.,Nakajima T., Honda Y., Kitao O.,Nakai H.,Vreven T., Montgomery J. J. A., Peralta, J. E.,Ogliaro F., Bearpark M Heyd J. J., Brothers E.,Kudin K. N.,Staroverov V. N., Kobayashi R., Normand J.,Raghavachari K.,Rendell A., Burant J. C., Iyengar S. S.,Tomasi J.,Cossi M., Rega N., Millam N. J.,Klene M.,Knox J. E., Cross J. B., Bakken V.,Adamo C.,Jaramillo J., Gomperts R., Stratmann R. E.,Yazyev O.,Austin A. J., Cammi R., Pomelli C.,Ochterski J. W.,Martin R. L., Morokuma K., Zakrzewski V. G.,Voth G. A.,Salvador P., Dannenberg J. J., Dapprich S.,Daniels A. D.,Farkas O., Foresman J. B., Ortiz J. V.,Cioslowski JFox D. J., Gaussian 09. Gaussian, Inc,Wallingford CT, 2009 |
| [28] | Lu T., Chen F., J. Comput. Chem., 2012, 33, 580— 592 |
| [29] | Lu T., Chen F., J. Mol. Graph. Model, 2012, 38, 314— 323 |
| [1] | LI Kangming,CHEN Jia,YI Yangjie,YAN Zhongzhong,YE Jiao,LONG Chuyun,LIU Aiping,HU Aixi,LI Jianming. Design, Synthesis and Insecticidal Activity of 1-(4-Chlorophenyl)-2-cyclopropylpropan-1-one Oxime Ether † [J]. Chem. J. Chinese Universities, 2020, 41(5): 1026. |
| [2] | ZHAO Bangtun, TAO Jingjing, CHEN Xiaoji, FU Huimin, ZHU Weimin. Synthesis, Structure and Electrochemistry of Tetrathiafulvalene Vinylogues Bearing Thienyl and Pyridyl Groups† [J]. Chem. J. Chinese Universities, 2018, 39(7): 1449. |
| [3] | ZHAO Bangtun, MA Shuxiu, TAO Jingjing, ZHU Weimin. Synthesis, Structures and Electrochemical Properties of Pyridine-based Tetrathiafulvalene Derivatives† [J]. Chem. J. Chinese Universities, 2017, 38(2): 193. |
| [4] | LI Lin, LI Miao, CHAI Baoshan, YANG Jichun, SONG Yuquan, LIU Changling. Design, Synthesis and Biological Activity of Novel Substituted Diamides Derivatives Containing Thiophene Ring [J]. Chem. J. Chinese Universities, 2016, 37(9): 1649. |
| [5] | JIA Changqing, YANG Dongyan, CHE Chuanliang, MA Yongqiang, RUI Changhui, YAN Xiaojing, QIN Zhaohai. Synthesis, Structural Characterization, Insecticidal and Fungicidal Activity of (1H-1,2,4-Triazol-5-yl)carbamates† [J]. Chem. J. Chinese Universities, 2016, 37(5): 892. |
| [6] | MAHMUTJAN Jelil, ABLAT Mamat, ALIMJAN Abaydulla, MAMAT Tursun. Investigation of Topology-distinct Stable Structures for Hydrogen Fluoride Clusters (HF)n(n=2—8) by the Methods of Graph Theoretical Enumerations and Quantum Chemical Calculations† [J]. Chem. J. Chinese Universities, 2016, 37(12): 2275. |
| [7] | CHEN Jiuju. Theoretical Studies on the of Ambipolar Charge Transport in Terazulene Single Crystal† [J]. Chem. J. Chinese Universities, 2016, 37(1): 121. |
| [8] | XIANG Junfeng, YI Pinggui, YU Xianyong, CHEN Jian, HAO Yanlei, REN Zhiyong. Excited-state Proton Transfer of 2-(2-Hydroxyphenyl)benzothiazole in the Confined Nanocavity† [J]. Chem. J. Chinese Universities, 2015, 36(4): 654. |
| [9] | YANG Dongyan, WANG Lei, JIA Changqing, LI Changsheng, MA Yongqiang, RUI Changhui, XU Yanjun, QIN Zhaohai. Syntheses and Insecticidal Activities of Nitro Arylideneamino Guanidine Derivatives† [J]. Chem. J. Chinese Universities, 2014, 35(8): 1703. |
| [10] | GAO Chenggui, LONG Zhengwen, TAN Xingfeng, LONG Bo, ZHANG Weijun, LONG Chaoyun, QIN Shuijie. Theoretical Investigation on the Reaction Between HRnCCH and X(X=H2O, NH3) in Gas Phase† [J]. Chem. J. Chinese Universities, 2014, 35(2): 344. |
| [11] | ZHANG Xiu-Lan, WANG Bao-Lei, MAO Ming-Zhen, XIONG Li-Xa, YU Shu-Jing, LI Zheng-Ming. Synthesis and Insecticidal Activity of 5-Chloro-N-[4-chloro- 2-(substitutedcarbamoyl)-6-methylphenyl]-1-aryl-3- (trifluoromethyl)-1H-pyrazole-4-carboxamide [J]. Chem. J. Chinese Universities, 2013, 34(1): 96. |
| [12] | YAN Tao, LIU Peng-Fei, ZHANG Ji-Feng, YU Shu-Jing, XIONG Li-Xia, LI Zheng-Ming. Design, Synthesis and Biological Activities of Novel Ortho-dicarboxyamides [J]. Chem. J. Chinese Universities, 2012, 33(08): 1745. |
| [13] | LIU Peng-Fei, ZHOU Sha, XIONG Li-Xia, YU Shu-Jing, ZHANG Xiao, LI Zheng-Ming. Design, Synthesis and Biological Activities of New Ryanodine Receptor Pesticides Based on Ugi Reaction [J]. Chem. J. Chinese Universities, 2012, 33(04): 738. |
| [14] | QIHE Ri-Ma, ZHAO Xiao-Guang, ZHANG Yun-Hong, YUAN Hui, XU Guang-Tong. Investigation of Selectivity over HDS Catalysts by in situ IR Spectra of Adsorbed CO and Molecular Simulation Calculation [J]. Chem. J. Chinese Universities, 2012, 33(02): 383. |
| [15] | YAN Tao, YU Guan-Ping, XIONG Li-Xia, YU Shu-Jing, WANG Su-Hua, LI Zheng-Ming*. Design, Synthesis and Biological Activities of Novel Anthranilic Isophthaloyl Amide [J]. Chem. J. Chinese Universities, 2011, 32(8): 1750. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||