

Chem. J. Chinese Universities ›› 2016, Vol. 37 ›› Issue (12): 2275.doi: 10.7503/cjcu20160275
• Physical Chemistry • Previous Articles Next Articles
MAHMUTJAN Jelil1,2, ABLAT Mamat3,*( ), ALIMJAN Abaydulla1, MAMAT Tursun1,2
), ALIMJAN Abaydulla1, MAMAT Tursun1,2
Received:2016-04-25
Online:2016-12-10
Published:2016-11-15
Contact:
ABLAT Mamat
E-mail:ablet238@aliyun.com
Supported by:CLC Number:
TrendMD:
MAHMUTJAN Jelil, ABLAT Mamat, ALIMJAN Abaydulla, MAMAT Tursun. Investigation of Topology-distinct Stable Structures for Hydrogen Fluoride Clusters (HF)n(n=2—8) by the Methods of Graph Theoretical Enumerations and Quantum Chemical Calculations†[J]. Chem. J. Chinese Universities, 2016, 37(12): 2275.
| Project | No. of vertice | ||||||
|---|---|---|---|---|---|---|---|
| 2 | 3 | 4 | 5 | 6 | 7 | 8 | |
| Graph | 1 | 2 | 7 | 21 | 112 | 697 | 6386 |
| Digraph | 1 | 5 | 27 | 247 | 3272 | 55005 | 1104449 |
| Restrictive digraph | 2 | 2 | 9 | 51 | 320 | 2443 | |
| Local minima | 1 | 2 | 2 | 4 | 8 | 14 | |
Table 1 Numbers of graphs, digraphs, and restrictive digraphs of HF clusters(HF)n(n=2—8) and the number of stable structures of HF clusters(HF)n(n=2—7) under the calculation method of MP2/6-31G**(d,p)
| Project | No. of vertice | ||||||
|---|---|---|---|---|---|---|---|
| 2 | 3 | 4 | 5 | 6 | 7 | 8 | |
| Graph | 1 | 2 | 7 | 21 | 112 | 697 | 6386 |
| Digraph | 1 | 5 | 27 | 247 | 3272 | 55005 | 1104449 |
| Restrictive digraph | 2 | 2 | 9 | 51 | 320 | 2443 | |
| Local minima | 1 | 2 | 2 | 4 | 8 | 14 | |
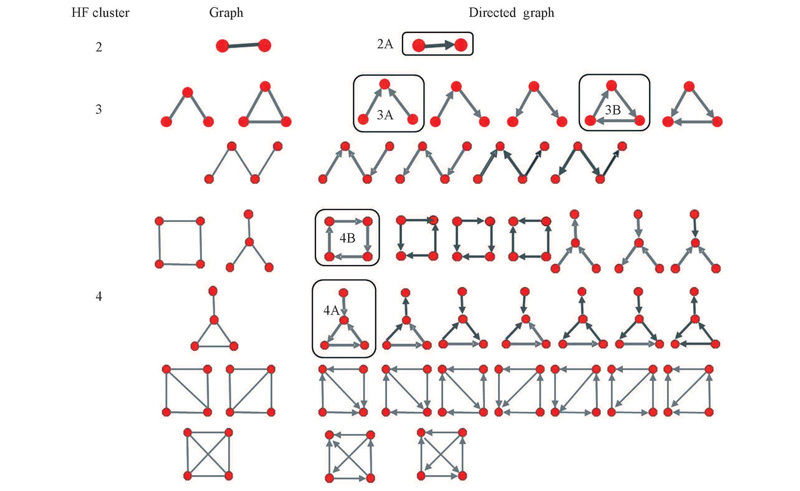
Fig.3 All the enumerated possible topology-distinct graphs and digraphs with n vertices for(HF)n(n=2—4) The designated patterns(2A, 3A, 3B, 4A and 4B) correspond to the stable structures.
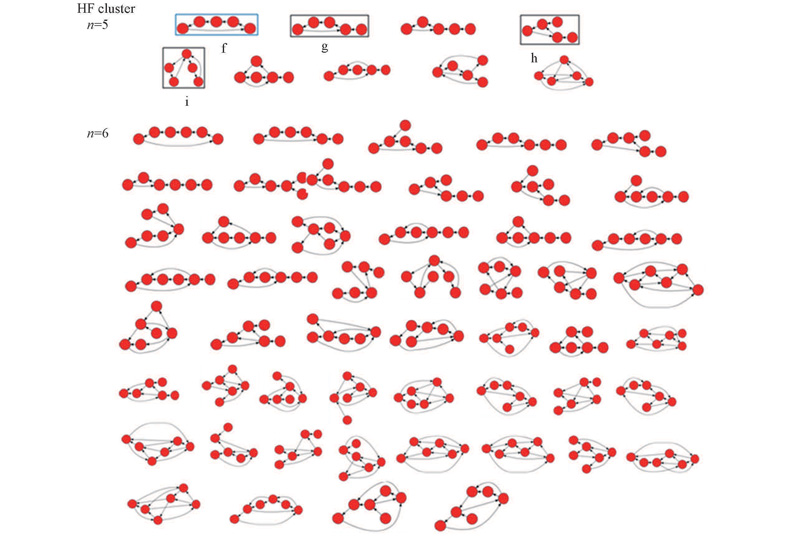
Fig.4 Enumerated restrictive digraphs generated from H-B matrices by Python program andGraphViz 2.37 program package corresponding to HF pentamer and hexamerThe designated patterns(f, g, h and i) correspond to the stable structures.
| Local minima | E/eV | ΔE/eV | ΔE/kJ |
|---|---|---|---|
| 7A | -19083.58598 | 0 | 0 |
| 7G | -19083.07658 | 0.50864 | 49.16 |
| 7F | -19083.04035 | 0.54672 | 52.68 |
| 7N | -19082.62860 | 0.95744 | 92.42 |
| 7H | -19082.71929 | 0.86768 | 83.64 |
| 7J | -19082.98659 | 0.59840 | 57.86 |
| 7D | -19083.34520 | 0.24208 | 23.22 |
| 7K | -19082.98847 | 0.59840 | 57.66 |
| 7L | -19082.93640 | 0.65008 | 20.89 |
| 7B | -19083.36094 | 0.22576 | 21.71 |
| 7E | -19083.09200 | 0.49504 | 47.70 |
| 7C | -19083.38822 | 0.19856 | 19.08 |
| 7M | -19082.85767 | 0.72896 | 70.29 |
| 7I | -19082.92199 | 0.66368 | 64.10 |
Table 2 14 local minima and corresponding total energy and relative total energy of the (HF)7 clusters[MP2/6-31G**(d,p)]
| Local minima | E/eV | ΔE/eV | ΔE/kJ |
|---|---|---|---|
| 7A | -19083.58598 | 0 | 0 |
| 7G | -19083.07658 | 0.50864 | 49.16 |
| 7F | -19083.04035 | 0.54672 | 52.68 |
| 7N | -19082.62860 | 0.95744 | 92.42 |
| 7H | -19082.71929 | 0.86768 | 83.64 |
| 7J | -19082.98659 | 0.59840 | 57.86 |
| 7D | -19083.34520 | 0.24208 | 23.22 |
| 7K | -19082.98847 | 0.59840 | 57.66 |
| 7L | -19082.93640 | 0.65008 | 20.89 |
| 7B | -19083.36094 | 0.22576 | 21.71 |
| 7E | -19083.09200 | 0.49504 | 47.70 |
| 7C | -19083.38822 | 0.19856 | 19.08 |
| 7M | -19082.85767 | 0.72896 | 70.29 |
| 7I | -19082.92199 | 0.66368 | 64.10 |
| [1] | Aakerðy C. B., Seddon K. R., Chem. Soc. Rew., 1993, 22(6), 397—407 |
| [2] | Scheiner S., Molecular Interactions from van der Waals to Strong Bound Complexes, John Wiley & Sons Lid., Chichester, 1997, 25—97 |
| [3] | Redington R. L., J. Phys. Chem., 1982, 86, 552—560 |
| [4] | Curtis L. A., Blander M., Chem. Rev., 1988, 88, 827—841 |
| [5] | Wang R. W., Hildeb J. H., Morell W. E., J. Am. Chem. Soc., 1943, 65, 182—187 |
| [6] | Briegleb G., Strohmeier W., Z. Elektrochem., 1953, 57, 668—674 |
| [7] | Frank U., Meyer F., Z. Elektrochem., 1959, 63, 571—578 |
| [8] | Barnes A. J., Hallan H. E., Jones D., Proc. R. Soc. London, Ser. A, 1973, 335, 97—104 |
| [9] | Smith D. F., J. Chem. Phys., 1958, 28, 1040—1047 |
| [10] | Yulia N., Struct. Chem., 2012, 23, 1253—1266 |
| [11] | Almeida T. S., Cabral B. J. C., J. Chem. Phys., 2010, 132, 094307 |
| [12] | Bai X., Ming N., Brown R. E., J. Phys. Chem. A, 2011, 115, 10596—10599 |
| [13] | Sophy K. B., Kuo J. L., J. Chem. Phys., 2009, 131, 224307 |
| [14] | Swalina C., Wang Q., Chakraborty A., Hammes-Schiffer S., J. Phys. Chem. A, 2007, 111, 2206—2212 |
| [15] | Sun H., Watts R. O., Buck U., J. Chem. Phys., 1992, 96, 1810—1821 |
| [16] | Zhang C., Freeman D. L., Doll J. D., J. Chem. Phys., 1989, 91, 2489—2497 |
| [17] | Hosoya H., Gotoh M., Murakami M., Ikeda S., J. Chem., Inf. Comput. Sci., 1999, 39, 192—196 |
| [18] | Gutman I., Soldatovic' T., Vidovic D., Chem. Phys. Lett., 1998, 297, 428—432 |
| [19] | McDonald S., Ojamae L., Singer S. J., J. Phys. Chem. A, 1998, 102, 2824—2832 |
| [20] | Miyake T., Aida M., Chem. Phys. Lett., 2002, 363, 106—110 |
| [21] | Miyake T., Aida M., Internet Electron. J. Mol. Des., 2003, 2, 24—32 |
| [22] | Jieli M., Miyake T., Aida M., Bull. Chem. Soc. Jpn., 2007, 80(11), 2131—2136 |
| [23] | Jieli M., Aida M., J. Phys. Chem. A, 2009, 113, 1586—1594 |
| [24] | Jelil M., Abaydulla A., J. Chem. Phys., 2015, 143, 044301 |
| [25] | Alfred K., Int. J. Q. Chem., 2004, 38(S24), 129—140 |
| [26] | Curtis L. A., Blander M., Chem. Rev., 1988, 88, 827—841 |
| [27] | Sun H., Watts R. O., Buck U., J. Chem. Phys., 1992, 96, 1810—1821 |
| [28] | Zhang C., Freeman D. L., Doll J. D., J. Chem. Phys., 1989, 91, 2489—2497 |
| [29] | Quack M., Suhm M. A., J. Chem. Phys., 1991, 95, 28—59 |
| [30] | Klopper W., Quack M., Suhm M. A., J. Chem. Phys., 1998, 108, 10096—10115 |
| [31] | Grigorenko B. L., Nemukhin A. V., Apkarian V. A., J. Chem. Phys., 1998, 108, 4413—4425 |
| [32] | Ovchinnikov M., Apkarian V. A., J. Chem. Phys., 1999, 110, 9842—9852 |
| [33] | Guedes R. C., DoCouto P. C., Costa Cabral B. J., J. Chem. Phys., 2003, 118, 1272—1281 |
| [34] | Luis R., Rafael A., David G. A., Helena D. R., J. Chem. Phys., 2001, 114, 5552—5561 |
| [35] | Michael W. A., Philip T. G., Curottoa E., J. Chem. Phys., 2006, 124, 174305 |
| [36] | Howard B. J., Dyke T. R., Klemperer W., J. Chem. Phys., 1984, 81, 5417—5423 |
| [37] | Pine A. S., Howard B. J., J. Chem. Phys., 1986, 84, 590—596 |
| [38] | Yukhnevich G. V., Tarakanova E. G., Nemukhin A. V., Russian. Chem. Bull., 1997, 46, 414—422 |
| [39] | Arif B., GraphViz, Version 2.38, AT&TLabs, Inc., Austin, 2014 |
| [40] | ChemBio Office Ultra, Version 12.0, Cambridge Soft Corporation, Software Publishers Association Version 12.0, Cambridge Soft Corporation, Software Publishers Association, Washington DC., 2010 |
| [41] | Dennington, Keith T., Millam J., GaussView, Version 5, Semichem Inc., Shawnee Mission, KS, 2009 |
| [42] | Frisch M.J., Trucks G. W., Schlegel H. B., Scuseria G. E., Robb M. A., Cheeseman J. R., Scalmani G., Barone V., Mennucci B., Petersson G. A., Nakatsuji H., Caricato M., Li X., Hratchian H. P., Izmaylov A. F., Bloino J., Zheng G., Sonnenberg J. L., Hada M., Ehara M., Toyota K., Fukuda R., Hasegawa J., Ishida M., Nakajima T., Honda Y., Kitao O., Nakai H., Vreven T., Montgomery J. A., Peralta J. E., Ogliaro F., Bearpark M., Heyd J. J., Brothers E., Kudin K. N., Staroverov V. N., Kobayashi R., Normand J., Raghavachari K., Rendell A., Burant J. C., Iyengar S. S., Tomasi J., Cossi M., Rega N., Millam J. M., Klene M., Knox J. E., Cross J. B., Bakken V., Adamo C., Jaramillo J., Gomperts R., Stratmann R. E., Yazyev O., Austin O. A. J., Cammi R., Pomelli C., Ochterski J. W., Martin R. L., Morokuma K., Zakrzewski V. G., Voth G. A., Gaussian 09, Revision A.02, Gaussian Inc., Wallingford CT, 2009 |
| [43] | Abu-Awwad F. M., Chem. Phys. Lett., 2002, 360, 340—348 |
| [44] | Friedrich J., Perlt E., Roatsch M., Spickermann C., Kirchner B., J. Chem., Theory Comput., 2011, 7, 843—851 |
| [45] | Muller H., Franke R., Vogtner S., Jaquet R., Kutzelnig W., Theor. Chem. Acc., 1998, 100, 85—102 |
| [46] | Andrews L., Bondybev V. E., English J. H., J. Chem. Phys., 1984, 81, 3452—3457 |
| [1] | LI Kangming, LI Yansai, YI Yangjie, XU Leitao, YE Jiao, OU Xiaoming, LI Jianming, HU Aixi. Design, Synthesis and Biological Activity of 5-Pyrazole Carboxamides † [J]. Chem. J. Chinese Universities, 2020, 41(4): 716. |
| [2] | ZHAO Bangtun, TAO Jingjing, CHEN Xiaoji, FU Huimin, ZHU Weimin. Synthesis, Structure and Electrochemistry of Tetrathiafulvalene Vinylogues Bearing Thienyl and Pyridyl Groups† [J]. Chem. J. Chinese Universities, 2018, 39(7): 1449. |
| [3] | ZHAO Bangtun, MA Shuxiu, TAO Jingjing, ZHU Weimin. Synthesis, Structures and Electrochemical Properties of Pyridine-based Tetrathiafulvalene Derivatives† [J]. Chem. J. Chinese Universities, 2017, 38(2): 193. |
| [4] | CHEN Jiuju. Theoretical Studies on the of Ambipolar Charge Transport in Terazulene Single Crystal† [J]. Chem. J. Chinese Universities, 2016, 37(1): 121. |
| [5] | XIANG Junfeng, YI Pinggui, YU Xianyong, CHEN Jian, HAO Yanlei, REN Zhiyong. Excited-state Proton Transfer of 2-(2-Hydroxyphenyl)benzothiazole in the Confined Nanocavity† [J]. Chem. J. Chinese Universities, 2015, 36(4): 654. |
| [6] | GAO Chenggui, LONG Zhengwen, TAN Xingfeng, LONG Bo, ZHANG Weijun, LONG Chaoyun, QIN Shuijie. Theoretical Investigation on the Reaction Between HRnCCH and X(X=H2O, NH3) in Gas Phase† [J]. Chem. J. Chinese Universities, 2014, 35(2): 344. |
| [7] | QIHE Ri-Ma, ZHAO Xiao-Guang, ZHANG Yun-Hong, YUAN Hui, XU Guang-Tong. Investigation of Selectivity over HDS Catalysts by in situ IR Spectra of Adsorbed CO and Molecular Simulation Calculation [J]. Chem. J. Chinese Universities, 2012, 33(02): 383. |
| [8] | FENG Jin-Ling, ZHANG Jian-Guo*, WANG Kun, ZHANG Tong-Lai*. Crystal Structure and Theoretical Studies of 3,6-Dihydrazino-1,2,4,5-tetrazine [J]. Chem. J. Chinese Universities, 2011, 32(7): 1519. |
| [9] | BAI Feng-Ying, LI Xiao-Tian*, ZHU Guang-Shan, XING Yong-Heng , ZENG Xiao-Qing,... Synthesis, Structure and Molecular Configuration of a Series of Ionic Salt Compounds of Pyrophosphate Ester of Thiamine [J]. Chem. J. Chinese Universities, 2009, 30(10): 1919. |
| [10] | LI Xiao-Li, DUAN Ran, ZHANG Ying-Hui, RUAN Wen-Juan, ZHU Zhi-Ang. Studies on Molecular Recognition of Chiral Asymmetry Salen Ni to Guests Imidazole and Diamine Derivatives [J]. Chem. J. Chinese Universities, 2006, 27(9): 1689. |
| [11] | DU Jin-Tang, LI Yan-Mei, CHEN Zhong-Zhou, TAN Bo, JIANG Yang, ZHONG Ru-Gang, ZHAO Yu-Fen . Theoretical Studies on N-Phosphorylized Amino Acids Properties by Quantum Chemical Calculation [J]. Chem. J. Chinese Universities, 2005, 26(3): 540. |
| [12] | YANG Guo-Bo, ZHOU Yin-Hua, TIAN Wen-Jing, ZHOU Xin, PAN Qing-Jiang, REN Ai-Min. Quantum Chemical Study on the Energy Level Structure of Thiophene Oligomers [J]. Chem. J. Chinese Universities, 2004, 25(6): 1104. |
| [13] | QIU Yong-Qing, YU Hai-Ying, SU Zhong-Min, ZHAO Liang, WANG Rong-Shun, ZHU Yu-Lan, . Theoretical Studies on Electronic Spectrum and Third-order Nonlinear Optical Properties of Centrosymmetric Pyrazine Derivatives [J]. Chem. J. Chinese Universities, 2004, 25(12): 2335. |
| [14] | RUAN Wen-Juan, ZHANG Yu-Ling, ZHAO Xiao-Jing, JIANG Dong-Qing, ZHU Zhi-Ang . Studies on Chiral Molecular Recognition of Salen Zn with Amino Acid Esters [J]. Chem. J. Chinese Universities, 2003, 24(9): 1657. |
| [15] | SUN Yan-Bo, LI Ze-Sheng, HUANG Xu-Ri, SUN Chia-Chung . Quantum-chemical Investigation on Structure of B2H5 Cation and Radical [J]. Chem. J. Chinese Universities, 2002, 23(8): 1542. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||