

Chem. J. Chinese Universities ›› 2019, Vol. 40 ›› Issue (11): 2286.doi: 10.7503/cjcu20190464
• Analytical Chemistry • Previous Articles Next Articles
YU Zhaochuan1,MA Wenhui11,*( ),WU Tao1,WEN Jing1,ZHANG Yong2,WANG Liyan1,CHU Hongtao1
),WU Tao1,WEN Jing1,ZHANG Yong2,WANG Liyan1,CHU Hongtao1
Received:2019-08-26
Online:2019-11-10
Published:2019-10-18
Contact:
MA Wenhui1
E-mail:mwh972@163.com
Supported by:CLC Number:
TrendMD:
YU Zhaochuan, MA Wenhui, WU Tao, WEN Jing, ZHANG Yong, WANG Liyan, CHU Hongtao. Preparation of B, N, S co-Doped Graphene Quantum Dots for Fluorescence Detection of Fe 3+ and H2P
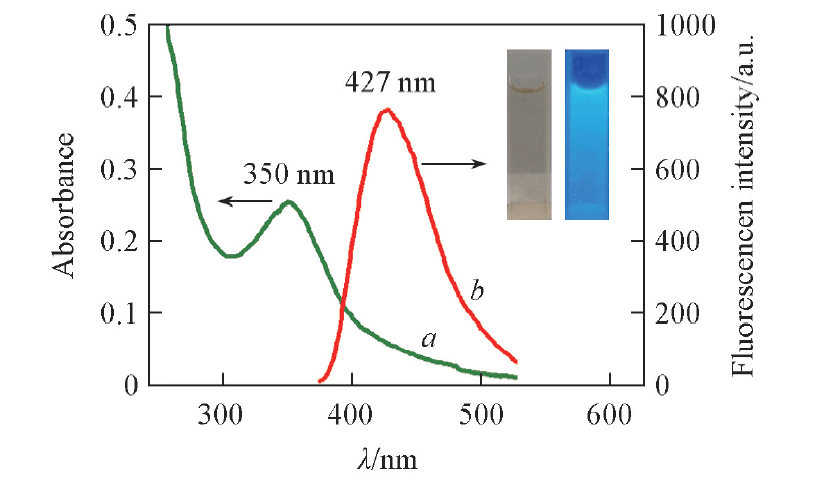
Fig.3 Ultraviolet absorption(a) and fluorescence(b)(λex=350 nm) spectra of BNS-GQDs Inset: the photographs of BNS-GQDs under visible(left) and UV(right, 365 nm) lights
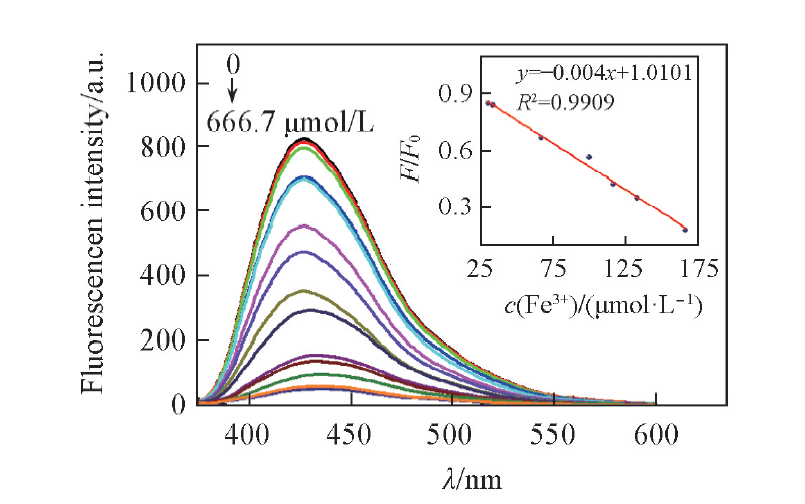
Fig.4 Fluorescence spectra of BNS-GQDs in the presence of different concentrations of Fe3+(0—666.7 μmol/L) in pure water Inset: variation of F/F0 as a function of the concentration of Fe3+.
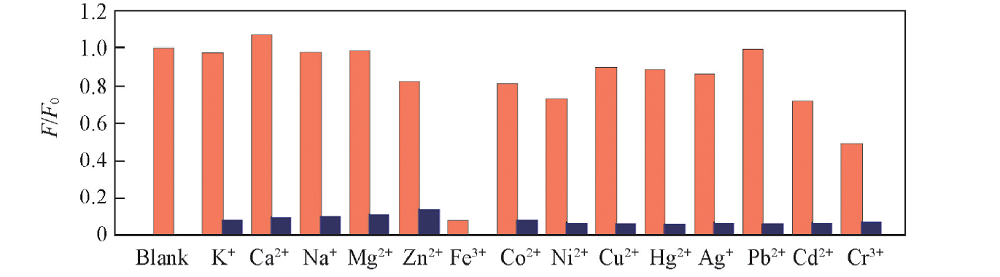
Fig.5 Selectivity of BNS-GQDs towards Fe3+ over the other competing metal ions Represent the fluorescence response of BNS-GQDs to different metal ions(333 μmol/L) in the absence of Fe3+, and represent the fluorescence response of BNS-GQDs to different metal ions(1000 μmol/L) in the presence of Fe3+(333 μmol/L). λex=350 nm, λem=427 nm.
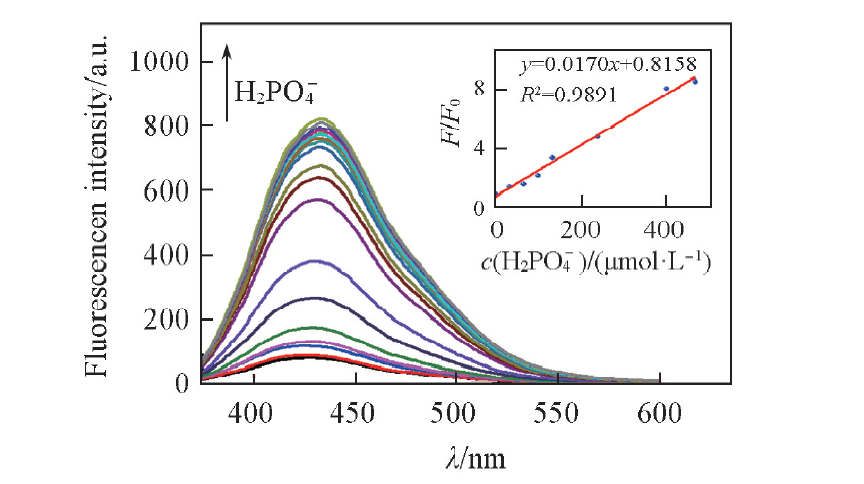
Fig.6 Fluorescence spectra of BNS-GQDs+Fe3+ in the presence of different concentrations of H2PO4-(0—1833.3 μmol/L) in pure water Inset: variation of F/F0 as a function of the concentration of H2P O 4 - .
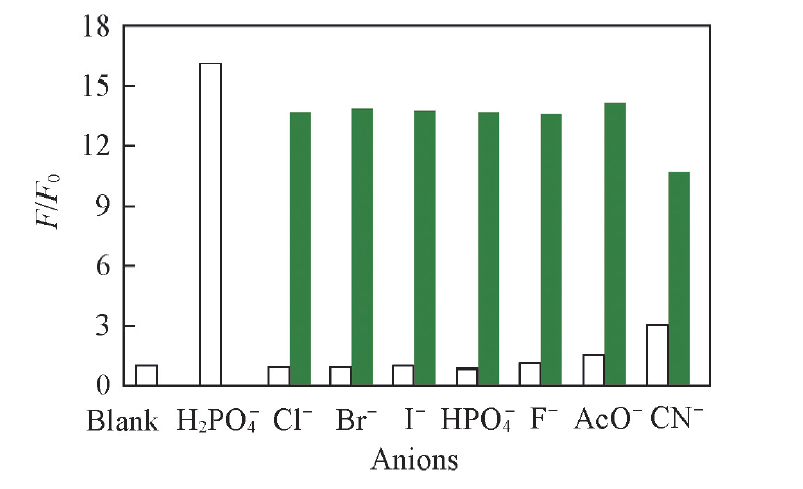
Fig.7 Selectivity of BNS-GQDs+Fe3+ towards H2PO4- over the other competing anions Represent the fluorescence response of BNS-GQDs+Fe3+ to different anions(1166.7 μmol/L) in the absence of H2P , and represent the fluorescence response of BNS-GQDs+Fe3+ to different anions(3000 μmol/L) in the presence of H2P (1166.7 μmol/L).
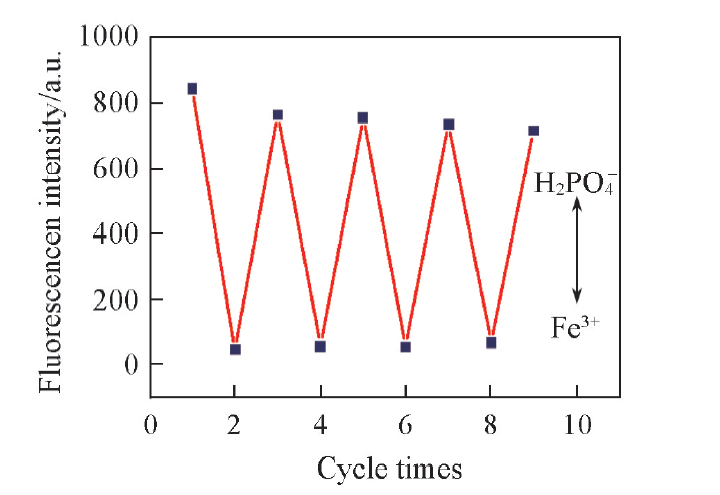
Fig.8 Change in the fluorescence intensity of BNS-GQDs system on alternate addition of Fe3+(333 μmol/L) and H2PO4-(1166.7 μmol/L) λex=350 nm, λem=427 nm.
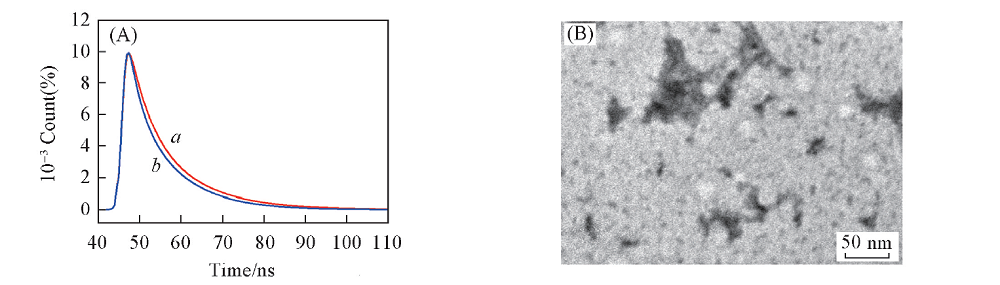
Fig.9 Fluorescence decay curves of BNS-GQDs before(a) and after(b) the addition of 333 μmol/L Fe3+(A) and TEM image of BNS-GQDs after the addition of 333 μmol/L Fe3+(B)

Fig.10 Images of Fe3+and H2PO4- in living Hela cells with BNS-GQDs The bright-field images(A—C), fluorescence images(D—F) and merged images(G—I) of the cells are treated with BNS-GQDs(A, D, G), BNS-GQDs+Fe3+(B, E, H), BNS-GQDs+Fe3++H2P O 4 - (C, F, I).
| [1] | Karimi M., Badiei A., Lashgari N., Ziarani M. G., J. Porous Mat., 2017,25(1), 137— 146 |
| [2] | Cheng H. Y., Li D. C., Cheng B. H., Jiang H ., Environ. Res., 2019,170, 443— 451 |
| [3] | Li Y. P., Hu Y., Jia Y., Jiang X. H., Cheng Z ., J., Anal. Lett., 2019,52(11), 1711— 1731 |
| [4] | Kim H., Kim K. B., Song E. J., Hwang I. H., Noh J. Y., Kim P. G., Jeong K. D., Kim C ., Inorg. Chem. Commun., 2013,36, 72— 76 |
| [5] | Mohapa S., Das R. K. X., Anal. Chim. Acta, 2019,1058, 146— 154 |
| [6] | Chen Z. H., Li Y. Y., Han J., Wang Y., Ren Y. P., Chen T., Tang X., Ni L., Chinese J. Anal. Chem., 2018,46(1), 20— 26 |
| ( 陈兆辉, 李媛媛, 韩娟, 王赟, 任彦鹏, 陈桐, 唐旭, 倪良 . 分析化学, 2018,46(1), 20— 26) | |
| [7] | Wang Y., Lao S., Ding W., Zhang Z., Liu S., Sensors Actuat. B: Chem., 2019,284, 186— 192 |
| [8] | Duong H. D., Jong I. R ., Sensors Actuat. B: Chem., 2018,274, 66— 77 |
| [9] | Gupta M., Lee H ., Dyes Pigments, 2019,167, 174— 180 |
| [10] | Ma W. H., Xia W., Xu Q., Han H. Y., Song B., Sun L. W., Liang C. H., Acta Chim. Sinica, 2012,70, 917— 920 |
| ( 马文辉, 夏薇, 徐群, 韩宏彦, 宋波, 梁春花 . 化学学报, 2012,70(7), 917— 920) | |
| [11] | Zhao B., Liu T., Fang Y., Wang L., Kan W., Deng Q., Song B., Sensors Actuat. B:Chem., 2017,246, 370— 379 |
| [12] | Singh G., Singh J., Singh J., Mangat S. S., J. Lumin., 2015,165, 123— 129. |
| [13] | Qi H.J., Teng M., Liu M., Liu S. X., Li J., Yu H. P., Teng C. B., Huang Z. H., Liu H., Shao Q., Umar A., Ding T., Gao Q., Guo Z., J. Colloid Interf. Sci., 2019,539, 332— 341 |
| [14] | Ge S. Y., He J. B., Ma C. X., Liu J. Y., Xi F. N., Dong X. P., Talanta, 2019,199, 581— 589 |
| [15] | Cao L., Wang X., Meziani M. J., Lu F., Wang H., Luo P. G., Luo Y. L., Harruff B. A., Veca L. M., Murray D., Xie S. Y., Sun Y. P., J. Am. Chem. Soc., 2007,129, 11318— 11319 |
| [16] | Zhou S.H., Li Q., Zhang T., Pang D. W., Tang H. W., Chem. J. Chinese Universities, 2019,40(8), 1593— 1599 |
| ( 周思慧, 李琼, 张婷, 庞代文, 唐宏武 . 高等学校化学学报, 2019,40(8), 1593— 1599) | |
| [17] | Peng J., Gao W., Gupta B. K., Liu Z., Romero-Aburto R., Ge L., Song L., Alemany L. B., Zhan X. B., Gao G. H., Vithayathil S. A., Kaipparettu B. A., Marti A. A., Hayashi T., Zhu. J. J., Ajayan P. M ., Nano Lett., 2012,12(2), 844— 849 |
| [18] | Kwok R.T. K., Leung C. W. T., Lam J. W. Y., Tang B. Z ., Chem. Soc. Rev., 2015,44(13), 4228— 4238 |
| [19] | Omer K. M., Tofiq D. I., Hassan A. Q., Microchim. Acta, 2018,185(10), 466— 453 |
| [20] | Su D. D., Li N., Liu Y., Wang M. K., Su X. G., Anal. Chim. Acta, 2019,1084, 78— 84 |
| [21] | Chen C., Zhao D., Hu T., Sun J., Yang X., Sensors Actuat.B:Chem., 2017,241, 779— 788 |
| [22] | Qu Z. B., hou X. G., Gu L., Lan R. M., Sun D. D., Yu D. J., Shi G. Y ., Chem. Commun., 2013,49(84), 9830— 9832 |
| [23] | He X. Y., Zhou L., Nesterenko E. P., Nesterenko P. N., Paull B., Omamogho J. O., Glennon J. D., Luong J. H. T ., Anal. Chem., 2012,84(5), 2351— 2357 |
| [24] | Wang Y.B., Chang Q., Hu S. L., Sensors Actuat. B: Chem., 2017,253, 928— 933 |
| [25] | Lakshmi V., Ravikanth M ., J. Mater. Chem. C, 2014,2(28), 5576— 5586 |
| [26] | Fan T.J., Zhang G. X., Jian L., Murtaza I., Meng H., Liu Y., Min Y.J ., Alloy. Compd., 2019,792, 844— 850 |
| [27] | Cheng C., Xing M., Wu Q., Mat. Sci. Eng.C-Mater., 2019,99, 611— 619 |
| [28] | Zhang Y. M., Chen X. P., Liang G. Y., Zhong K. P., Yao H., Wei, T. B., Lin Q., Can. J. Chem., 2018,96(4), 363— 370 |
| [29] | Wei T. B., Wu G. Y., Shi B. B., Lin Q., Yao H., Zhang Y. M., Chinese J. Chem., 2014,32(12), 1238— 1244 |
| [30] | Liu Y., Wang Q., Guo S., Jia P., Shui Y., Yao S., Huang C., Zhang M., Wang L., Sensors Actuat. B:Chem., 2018,275, 415— 421 |
| [31] | Kaur N., Sharma V., Tiwari P., Saini A. K., Mobin S. M., . Sensors Actuat. B: Chem., 2019,291, 275— 286 |
| [32] | Gao X. H., Du C., Zhuang Z. H., Che W., J. Mater. Chem. C, 2016,4(29), 6927— 694 |
| [1] | LI Anran, ZHAO Bing, KAN Wei, SONG Tianshu, KONG Xiangdong, BU Fanqiang, SUN Li, YIN Guangming, WANG Liyan. ON-OFF-ON Double Colorimetric and Fluorescent Probes Based on Phenanthro[9,10-d]imidazole Derivatives and Their Living Cells Imaging [J]. Chem. J. Chinese Universities, 2021, 42(8): 2403. |
| [2] | MU Yaxin,A Li,ZHUANG Qianfen,WANG Yong,NI Yongnian. Hydrothermal Synthesis of Ultrasmall Fluorescent Tungsten Disulfide Quantum Dots and Their Application in Cell imaging† [J]. Chem. J. Chinese Universities, 2019, 40(4): 693. |
| [3] | Yingying ZHANG,Yiwen HUANG,Bing ZHAO,Liyan WANG,Bo SONG. Synthesis of a Colorimetric Fluorescent Probe of Cr 3+ and Its Application in Cell Imaging † [J]. Chem. J. Chinese Universities, 2019, 40(12): 2486. |
| [4] | HUANG Chibao,PAN Qi,CHEN Huashi,LIANG Xing,LÜ Guoling. Dicyanostilbene-derived Two-photon Fluorescence Probe for Lead Ions† [J]. Chem. J. Chinese Universities, 2018, 39(5): 897. |
| [5] | HAN Ruixia,LÜ Jitao,ZHANG Shuzhen. Molecular Probe for the Determination of Hydroxyl Radicals in Heterogeneous Systems: Coumarin† [J]. Chem. J. Chinese Universities, 2018, 39(12): 2658. |
| [6] | YANG Mei,LIU Qing,TANG Qing,WANG Chenghui,YANG Meixiang,SUN Tao,HUANG Ying,TAO Zhu. Water-soluble Supramolecular Fluorescent Probe for Sensing Carbendazim and Its Application in Living Cell Imaging† [J]. Chem. J. Chinese Universities, 2018, 39(12): 2665. |
| [7] | WANG Fengyan, YAN Ni, WEI Junji, XIA Huiyun, SONG Lifang, SONG Jiale, GAO Lining, YAN Luke. Synthesis, Characterization and Cell Imaging of Water Soluble Polythiophene Derivative† [J]. Chem. J. Chinese Universities, 2018, 39(11): 2594. |
| [8] | XU Wanzhen, QIU Chunxiao, HUANG Weihong, LIU Hong, YANG Wenming. Computer Simulation Design, Preparation and Application of Fluorescence Sensors Based on Quantum Dots for Selective Detection of 4-Nitrophenol in River Water† [J]. Chem. J. Chinese Universities, 2017, 38(7): 1155. |
| [9] | HUANG Chibao, PAN Qi, CHEN Xiaoyuan, ZHAO Guanglian, CHEN Huashi, LIANG Xing, LÜ Guoling. Dicyanostilbene-derived Two-photon Fluorescence Probe for Mercury Ions† [J]. Chem. J. Chinese Universities, 2017, 38(10): 1751. |
| [10] | WANG Caiping, LI Youfen, ZHANG Yidong. Preparation of Glass Ceramics Containing CaF2 by One-step Method and Analysis of the Fluorescent Probe of Eu3+ Ion† [J]. Chem. J. Chinese Universities, 2016, 37(4): 607. |
| [11] | SONG Chunxia, YANG Xiaohai, WANG Kemin, WANG Qing, LIU Jianbo, HUANG Jin, LI Wenshan, HUANG Haihua, LIU Wei. Application of Polymers in Fluorescence Analysis† [J]. Chem. J. Chinese Universities, 2016, 37(2): 201. |
| [12] | MI Xiaolong, JIAO Xiaojie, LIU Chang, HE Song, ZENG Xianshun. Rhodamine-based Cell Permeable Fluoresecent Turn-on Probes for Cupric Ion† [J]. Chem. J. Chinese Universities, 2016, 37(10): 1784. |
| [13] | HUANG Chibao, LIANG Xing, ZENG Qihua, CHEN Huashi, ZENG Boping, YI Daosheng, CHEN Xiaoyuan. Dicyanostilbene-derived Two-photon Fluorescence Probe for Free Zinc Ions in Live Cells and Living Tissues† [J]. Chem. J. Chinese Universities, 2015, 36(4): 646. |
| [14] | XU Hui, DAI Yanna, SHAN Hongyan, FEI Qiang, HUAN Yanfu, LI Guanghua, FENG Guodong. Rhodamine Derivative ABDO/Er3+ Composite Fluorescent Probe in Response to Temperature† [J]. Chem. J. Chinese Universities, 2014, 35(4): 736. |
| [15] | CHEN Yi, ZHANG Yan, ZENG Xi, MU Lan, LI Jun, SUN Qiang, ZHANG Jian-Xin, WEI Gang. Synthesis and Probe Properties of Rhodamine-triazine Derivatives [J]. Chem. J. Chinese Universities, 2013, 34(7): 1598. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||