

Chem. J. Chinese Universities ›› 2019, Vol. 40 ›› Issue (5): 1029.doi: 10.7503/cjcu20180668
• Physical Chemistry • Previous Articles Next Articles
DING Zhongxie1, LIANG Jinhua2, LIU Zhen1, SHEN Jiecan1, ZHANG Feng2, REN Xiaoqian1,*( ), JIANG Min3
), JIANG Min3
Received:2018-09-29
Online:2019-04-12
Published:2019-04-12
Contact:
REN Xiaoqian
E-mail:xqren@njtech.edu.cn
Supported by:CLC Number:
TrendMD:
DING Zhongxie,LIANG Jinhua,LIU Zhen,SHEN Jiecan,ZHANG Feng,REN Xiaoqian,JIANG Min. Functional Specific Heteropoly Acid Ionic Liquid Catalyzed Direct Esterification of Aqueous Succinate Solution†[J]. Chem. J. Chinese Universities, 2019, 40(5): 1029.
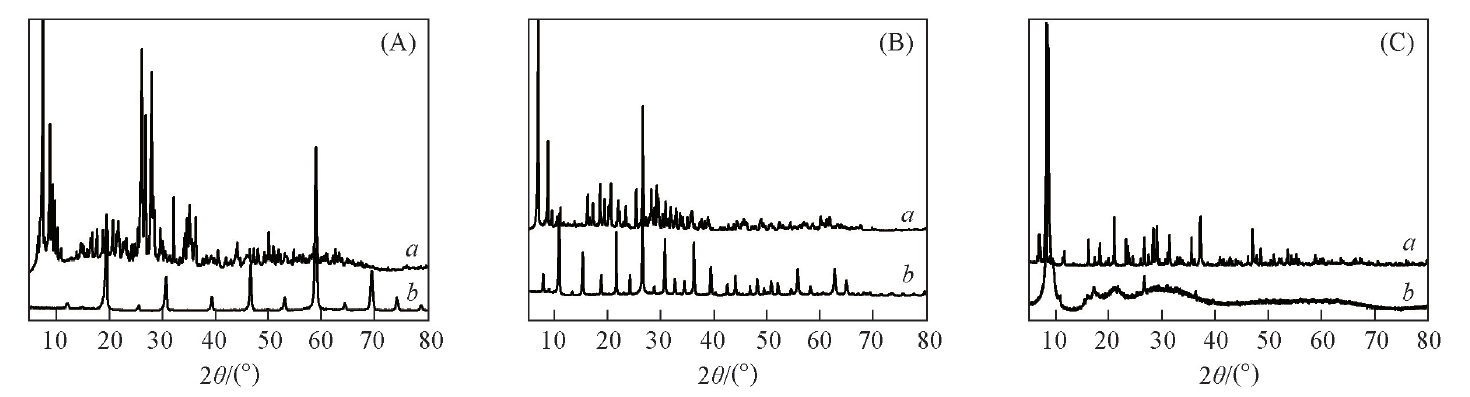
Fig.2 XRD patterns of [HMT-4PS][HPA](A) a. H3PMO12O40, b. [HMT-4PS]3[PMO]4; (B) a. H3PW12O40, b. [HMT-4PS]3[PW]4;(C) a. H4SiW12O40, b. [HMT-4PS][SiW].
| Catalyst | ABET/(m2·g-1) | VT/(cm3·g-1) | AM/(m2·g-1) | VM/(cm3·g-1) |
|---|---|---|---|---|
| [HMT-4PS]3[PMO]4 | 33.334 | 0.026 | 12.887 | 0.006 |
| [HMT-4PS]3[PW]4 | 48.777 | 0.028 | 35.559 | 0.018 |
| [HMT-4PS][SiW] | 28.648 | 0.021 | 10.499 | 0.004 |
Table 1 Textural properties of the catalysts*
| Catalyst | ABET/(m2·g-1) | VT/(cm3·g-1) | AM/(m2·g-1) | VM/(cm3·g-1) |
|---|---|---|---|---|
| [HMT-4PS]3[PMO]4 | 33.334 | 0.026 | 12.887 | 0.006 |
| [HMT-4PS]3[PW]4 | 48.777 | 0.028 | 35.559 | 0.018 |
| [HMT-4PS][SiW] | 28.648 | 0.021 | 10.499 | 0.004 |
| Catalyst | Mass fraction(%, E/T*) | n(N)/n(S) | |||
|---|---|---|---|---|---|
| C | H | N | S | ||
| [HMT-4PS]3[PMO]4 | 6.95/7.06 | 1.10/1.18 | 1.92/1.83 | 3.99/4.18 | 1.10 |
| [HMT-4PS]3[PW]4 | 4.79/4.84 | 0.93/0.81 | 1.13/1.25 | 2.41/2.86 | 1.07 |
| [HMT-4PS][SiW] | 6.28/6.16 | 0.92/1.04 | 1.75/1.60 | 3.44/3.65 | 1.16 |
Table 2 Elemental analysis results of the catalysts
| Catalyst | Mass fraction(%, E/T*) | n(N)/n(S) | |||
|---|---|---|---|---|---|
| C | H | N | S | ||
| [HMT-4PS]3[PMO]4 | 6.95/7.06 | 1.10/1.18 | 1.92/1.83 | 3.99/4.18 | 1.10 |
| [HMT-4PS]3[PW]4 | 4.79/4.84 | 0.93/0.81 | 1.13/1.25 | 2.41/2.86 | 1.07 |
| [HMT-4PS][SiW] | 6.28/6.16 | 0.92/1.04 | 1.75/1.60 | 3.44/3.65 | 1.16 |
| Solvent | Solubility of catalyst | ||
|---|---|---|---|
| [HMT-4PS]3[PW]4 | [HMT-4PS]3[PMo]4 | [HMT-4PS][SiW] | |
| Water | * | * | * |
| Acetonitrile | * | * | * |
| Methanol | * | * | * |
| Ethanol | + | + | + |
| Isopropanol | + | + | + |
| Acetone | + | + | + |
| 1-Butanol | — | — | — |
| Ethyl acetate | — | — | — |
| Ethyl ether | — | — | — |
| Dichloromethane | — | — | — |
| 1,2-Dichloroethane | — | — | — |
| Benzene | — | — | — |
| Cyclohexane | — | — | — |
Table 3 Solubility of various catalysts
| Solvent | Solubility of catalyst | ||
|---|---|---|---|
| [HMT-4PS]3[PW]4 | [HMT-4PS]3[PMo]4 | [HMT-4PS][SiW] | |
| Water | * | * | * |
| Acetonitrile | * | * | * |
| Methanol | * | * | * |
| Ethanol | + | + | + |
| Isopropanol | + | + | + |
| Acetone | + | + | + |
| 1-Butanol | — | — | — |
| Ethyl acetate | — | — | — |
| Ethyl ether | — | — | — |
| Dichloromethane | — | — | — |
| 1,2-Dichloroethane | — | — | — |
| Benzene | — | — | — |
| Cyclohexane | — | — | — |
| No. | Catalyst | Conv.(%) | SD(%) | SM(%) | A/(mmol·g-1) | Reaction phenomenon |
|---|---|---|---|---|---|---|
| 1 | [HMT-4PS]3[PMO]4 | 74.8 | 89.8 | 10.2 | 1.03 | Emulsion |
| 2 | [HMT-4PS]3[PW]4 | 79.3 | 92.9 | 7.1 | 1.16 | Emulsion |
| 3 | [HMT-4PS][SiW] | 77.1 | 90.9 | 9.1 | 1.08 | Emulsion |
| 4 | [MIMPS]3PMo12O40 | 57.6 | 76.5 | 23.5 | 1.04 | Heterogeneous |
| 5 | [MIMPS]3PW12O40 | 62.2 | 80.4 | 19.6 | 0.66 | Heterogeneous |
| 6 | [MIMPS]4SiW12O40 | 56.3 | 78.2 | 21.8 | 0.87 | Heterogeneous |
| 7 | H2SO4 | 46.8 | 68.5 | 31.5 | — | Homogeneous |
| 8 | — | 13.6 | 53.9 | 46.1 | — | Homogeneous |
Table 4 Catalytic performance of various catalysts*
| No. | Catalyst | Conv.(%) | SD(%) | SM(%) | A/(mmol·g-1) | Reaction phenomenon |
|---|---|---|---|---|---|---|
| 1 | [HMT-4PS]3[PMO]4 | 74.8 | 89.8 | 10.2 | 1.03 | Emulsion |
| 2 | [HMT-4PS]3[PW]4 | 79.3 | 92.9 | 7.1 | 1.16 | Emulsion |
| 3 | [HMT-4PS][SiW] | 77.1 | 90.9 | 9.1 | 1.08 | Emulsion |
| 4 | [MIMPS]3PMo12O40 | 57.6 | 76.5 | 23.5 | 1.04 | Heterogeneous |
| 5 | [MIMPS]3PW12O40 | 62.2 | 80.4 | 19.6 | 0.66 | Heterogeneous |
| 6 | [MIMPS]4SiW12O40 | 56.3 | 78.2 | 21.8 | 0.87 | Heterogeneous |
| 7 | H2SO4 | 46.8 | 68.5 | 31.5 | — | Homogeneous |
| 8 | — | 13.6 | 53.9 | 46.1 | — | Homogeneous |
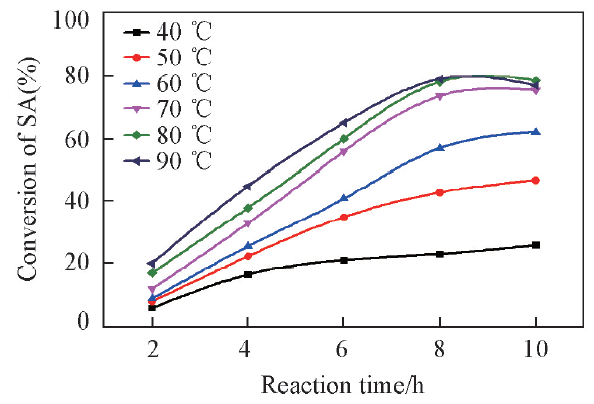
Fig.4 Effects of reaction temperatures on conversion of SAReaction conditions: succinic acid(mass fraction of 8% in aqueous solution), n(methanol)∶n(succinic acid)=30∶1, catalyst(mass fraction of 5% based on SA aqueous solution).
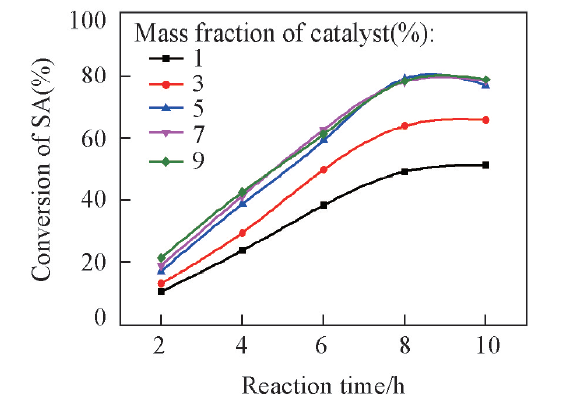
Fig.5 Effects of catalyst amounts on conversion of SAReaction conditions: succinic acid(mass fraction of 8% in aqueous solution), n(methanol)∶n(succinic acid)=30∶1, 80 ℃.
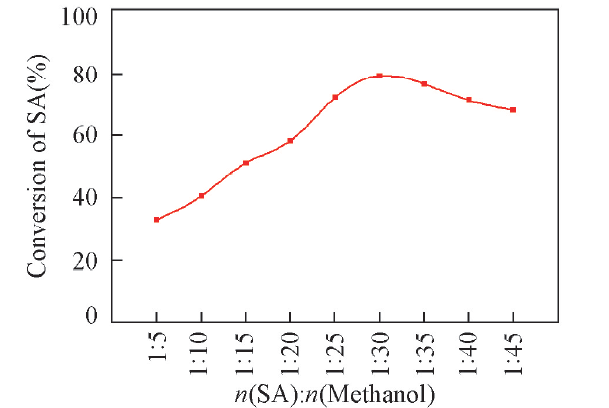
Fig.6 Effects of molar ratio of SA to methanol on conversion of SAReaction conditions: succinic acid(mass fraction of 8% in aqueous solution), 80 ℃, 8 h, catalyst(mass fraction of 5% based on SA aqueous solution).
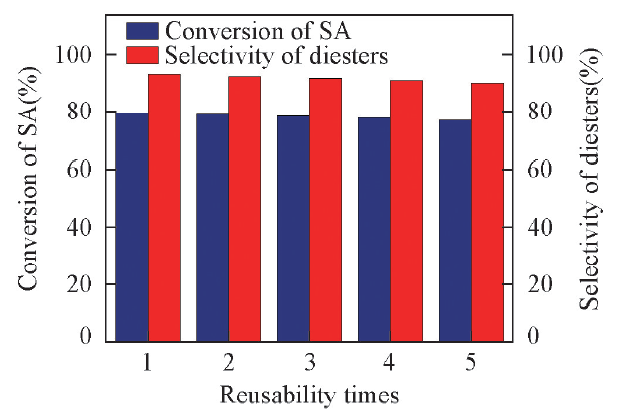
Fig.7 Reusability of catalyst [HMT-4PS]3[PW]4Reaction conditions: succinic acid(mass fraction of 8% in aqueous solution), 80 ℃, 8 h, n(methanol)∶n(succinic acid)=30∶1, catalyst(mass fraction of 5% based on SA aqueous solution).
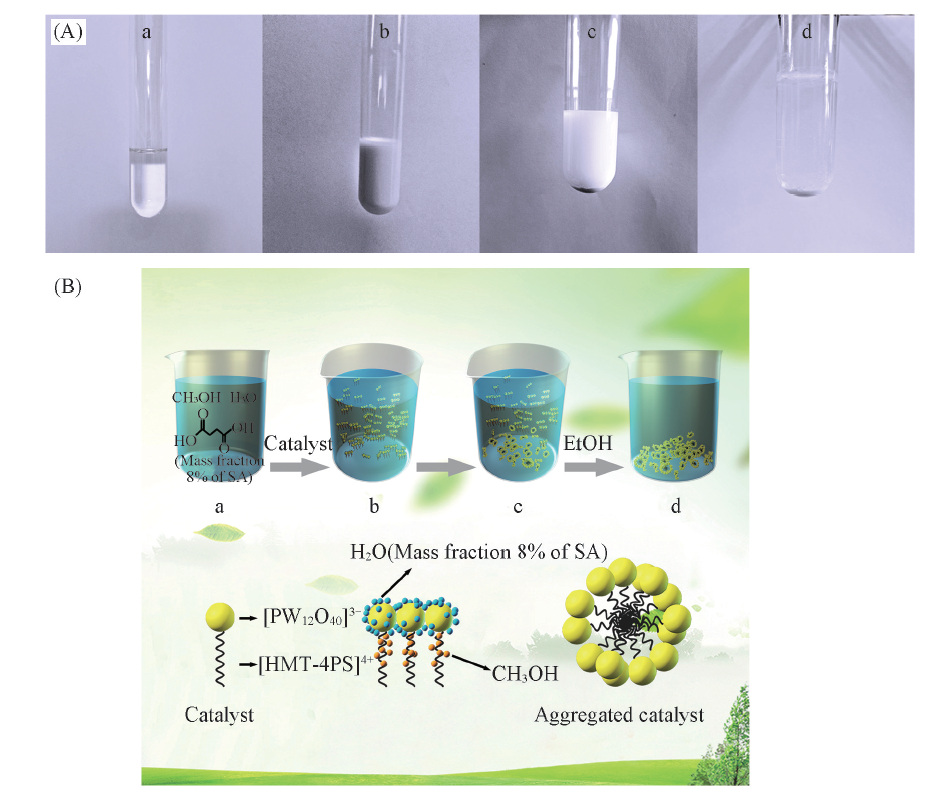
Fig.8 Reaction phenomena(A) and reaction process(B) Catalyst esterification and recovery of low concentration succinic acid solution: a. Before esterification; b. catalytic esterification of low concentration succinic acid solution in emulsion droplets; c. after esterification; d. catalyst recovery.
| [1] | Ouyang P.K., Bio-based Polymer Material, Chemical Industry Press, Beijing, 2012, 122—136 |
| (欧阳平凯. 生物基高分子材料, 北京: 化学工业出版社, 2012, 122—136) | |
| [2] | Bechthold I., Bretz K., Kabasci S., Kopitzky R., Springer A., Chem. Eng. Technol., 2008, 31(5), 647—654 |
| [3] | Cheng K. K., Zhao X. B., Zeng J., Zhang J. A., Biofuels. Bioprod. Biorefin., 2012, 6(3), 302—318 |
| [4] | Muzumdar A. V., Sawant S. B., Pangarkar V. G., Org. Process Res. Dev., 2004, 8(4), 685—688 |
| [5] | Kondrashova M. N., Ljubimova T. B., Maevskij E. I., Method for Preparing Succinic Acid, RU 2237056,2004-09-27 |
| [6] | Glassner D. A., Datta R., Process for the Production and Purification of Succinic Acid, US 5143834,1992-09-01 |
| [7] | Guan G. F., Xu C., Yao H. Q., Environ. Prot. Chem. Ind., 2004, 24(4), 240—243 |
| (管国锋, 徐晨, 姚虎卿. 化工环保, 2004, 24(4), 240—243) | |
| [8] | Ye X. J., Gong L., Wang H. L., Wang X. J., Xu H. Z., Xue D. H., Chem. Bioeng., 2014, 31(4), 32—36 |
| (叶小金, 宫莉, 王红蕾, 王晓俊, 徐洪章, 薛冬桦. 化学与生物工程, 2014, 31(4), 32—36) | |
| [9] | Li Q., Wang D., Wu Y., Li W., Zhang Y., Xing J., Su Z., Sep. Purif. Technol., 2010, 72(3), 294—300 |
| [10] | Sun X., Wang Q., Zhao W., Ma H., Sakata K., Sep. Purif. Technol., 2006, 49(1), 43—48 |
| [11] | Zhao W., Sun X., Wang Q., Ma H., Teng Y., Biomass Bioenergy,2009, 33(1), 21—25 |
| [12] | Budarin V., Luque R., Macquarrie D. J., Clark J. H., Chem. Eur. J., 2010, 13(24), 6914—6919 |
| [13] | Delhomme C., Goh S. L. M., Kühn F. E., Weuster-Botz D. J., J. Mol. Catal. B: Enzym., 2012, 80, 39—47 |
| [14] | Leng Y., Wang J., Zhu D., Ren X., Ge H., Shen L., Angew. Chem. Int. Ed.,2010, 48(1), 168—171 |
| [15] | Cole A. C., Jensen J. L., Ntai I., Tran K. L. T., Weaver K. J., Forbes D. C., Davis J. H., J. Am. Chem. Soc., 2002, 124(21), 5962—5963 |
| [16] | Liu S., Zhou H., Yu S., Xie C., Liu F., Song Z., Chem. Eng. J.,2012, 174(1), 396—399 |
| [17] | Gai B. B., He H., Zhao Y. H., Mao Z., Fu H., Chem. Res. Chinese Universities,2016, 32(4), 527—529 |
| [18] | Ma Q., Tong J. H., Su L. D., Wang W. H., Ma W. M., Bo L. L., Acta Phys. Chim. Sin., 2016, 32(12), 2961—2967 |
| (马青, 童金辉, 宿玲弟, 王文慧, 马文梅, 薄丽丽. 物理化学学报, 2016, 32(12), 2961—2967) | |
| [19] | Ilgen F., Ott D., Kralisch D., Reil C., Palmberger A., König B., Green Chem., 2009, 11(12), 1948—1954 |
| [20] | Cheng M., Tian S., Guan H., Wang S., Wang X., Jiang Z., Appl. Catal. B,2011, 107(1), 104—109 |
| [21] | Duan X., Sun G., Sun Z., Li J., Wang S., Wang X., Li S., Jiang Z., Catal. Commun., 2013, 42(4), 125—128 |
| [22] | An Y., Lu L., Li C. M., Cheng S. F., Gao G. H., Chinese J. Catal., 2009, V30(12), 1222—1226 |
| (安莹, 陆亮, 李才猛, 程时富, 高国华. 催化学报,2009, V30(12), 1222—1226) | |
| [23] | Ramesh S., Lu S. C., J. Mater. Res., 2011, 26(23), 2945—2951 |
| [24] | Nemanashi M., Noh J. H., Meijboom R. J., Mater. Sci., 2018, 53(18), 12663—12678 |
| [25] | Park C., Lee J., Kim C., Chem. Commun., 2011, 47(44), 12042—12056 |
| [26] | Tomalia D. A., Naylor A. M., Iii W. A. G., Angew. Chem. Int. Ed.,2010, 29(2), 138—175 |
| [27] | Zhang F., Liang J. H., Hu X., Wang J. G., Chen L. L., Ren X. Q., Jiang M., J. Chem. Eng., 2015, 66(7), 2488—2497 |
| (张锋, 梁金花, 胡曦, 王俊格, 陈刘龙, 任晓乾, 姜岷. 化工学报, 2015, 66(7), 2488—2497) | |
| [28] | Kapoor M. P., Kuroda H., Yanagi M., Nanbu H., Juneja L. R., TopCatal., 2009, 52(6/7), 634—642 |
| [29] | Duan X. X., Zhong S., Li X. Y., Wang X. H., Wang S. T., Li S. W., Energy Technol., 2015, 3(8), 871—877 |
| [30] | Li H., Qiao Y., Hua L., Hou Z., Feng B., Pan Z., Hu Y., Wang X., Zhao X., Yu Y., Chem. Cat. Chem,2010, 2(9), 1165—1170 |
| [31] | Li C., Jiang Z. X., Gao J. B., Yang Y. X., Wang S. J., Tian F. P., Sun F. X., Sun X. P., Ying P. L., Han C. R., Chem. Eur. J.,2010, 10(9), 2277—2280 |
| [1] | CUI Wei, ZHAO Deyin, BAI Wenxuan, ZHANG Xiaodong, YU Jiang. CO2 Absorption in Composite of Aprotic Solvent and Iron-based Ionic Liquid [J]. Chem. J. Chinese Universities, 2022, 43(8): 20220120. |
| [2] | ZHOU Zixuan, YANG Haiyan, SUN Yuhan, GAO Peng. Recent Progress in Heterogeneous Catalysts for the Hydrogenation of Carbon Dioxide to Methanol [J]. Chem. J. Chinese Universities, 2022, 43(7): 20220235. |
| [3] | PENG Kuilin, LI Guilin, JIANG Chongyang, ZENG Shaojuan, ZHANG Xiangping. Research Progress for the Role of Electrolytes in the CO2 Electrochemical Reduction [J]. Chem. J. Chinese Universities, 2022, 43(7): 20220238. |
| [4] | JI Shuangqi, JIN Zhao, GUAN Wenna, PAN Xiangyu, GUAN Tong. Preparation and Chromatographic Performance of Mixed-mode Silica Stationary Phase Modified by Double Cationic Ionic Liquid and Octadecyl Group [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220008. |
| [5] | LI Jiafu, ZHANG Kai, WANG Ning, SUN Qiming. Research Progress of Zeolite-encaged Single-atom Metal Catalysts [J]. Chem. J. Chinese Universities, 2022, 43(5): 20220032. |
| [6] | CHANG Sihui, CHEN Tao, ZHAO Liming, QIU Yongjun. Thermal Degradation Mechanism of Bio-based Polybutylactam Plasticized by Ionic Liquids [J]. Chem. J. Chinese Universities, 2022, 43(11): 20220353. |
| [7] | LI Haibo, XIAO Changfa, JIANG Long, HUANG Yun, DAN Yi. Copolymerization of Methyl Acrylate and 1-Octene Catalyzed by the Loaded Aluminum Chloride on MCM-41 Molecular Sieve [J]. Chem. J. Chinese Universities, 2021, 42(9): 2974. |
| [8] | WAN Ren, SONG Fan, PENG Changjun, LIU Honglai. Group Contribution Method for Infinite Dilution Molar Conductivity of Unconventional Ions in Water [J]. Chem. J. Chinese Universities, 2021, 42(12): 3672. |
| [9] | WANG Man, WANG Xin, ZHOU Jing, GAO Guohua. Efficient Synthesis of Dimethyl Carbonate via Transesterification of Methanol and Ethylene Carbonate Catalyzed by Poly(ionic liquid)s [J]. Chem. J. Chinese Universities, 2021, 42(12): 3701. |
| [10] | ZHOU Molin, JIANG Xin, YI Ting, YANG Xiangguang, ZHANG Yibo. Improvement of Interface Stability Between Sulfide Solid Electrolyte Li10GeP2S12 and Lithium Metal [J]. Chem. J. Chinese Universities, 2020, 41(8): 1810. |
| [11] | CHENG Shifu,HU Hao,CHEN Bihua,WU Haihong,GAO Guohua,HE Mingyuan. Preparation and Electrochemical Performance of Porous Carbons Prepared from Binary Ionic Liquids † [J]. Chem. J. Chinese Universities, 2020, 41(5): 1048. |
| [12] | LIU Hengshuo,YU Zhiquan,SUN Zhichao,WANG Yao,LIU Yingya,WANG Anjie. Copper Salt Anchored on a Covalent Organic Framework as Heterogeneous Catalyst for Chan-Lam Coupling Reaction † [J]. Chem. J. Chinese Universities, 2020, 41(5): 1091. |
| [13] | GAO Chong,YU Fengli,XIE Congxia,YU Shitao. Baeyer-Villiger Oxidation of Cyclic Ketones Catalyzed by Amino Alcohol Heteropoly Acid Ionic Liquid † [J]. Chem. J. Chinese Universities, 2020, 41(5): 1101. |
| [14] | GAO Naiwei, MA Qiang, HE Yonglin, WANG Yapei. Green Electronic Devices Based on Ionic Liquids † [J]. Chem. J. Chinese Universities, 2020, 41(5): 901. |
| [15] | PIAO Huilan,MA Pinyi,QIN Zucheng,JIANG Yanxiao,SUN Ying,WANG Xinghua,SONG Daqian. Determination of Triazine Herbicides from Fruit Juice Samples Using Effervescence Assisted Microextraction Method Based on Acidic Ionic Liquid Packed Syringe [J]. Chem. J. Chinese Universities, 2020, 41(2): 228. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||