

Chem. J. Chinese Universities ›› 2018, Vol. 39 ›› Issue (8): 1676.doi: 10.7503/cjcu20180157
• Organic Chemistry • Previous Articles Next Articles
HUANG Chibao*( ), PAN Qi, CHEN Huashi, LIANG Xing, LÜ Guoling
), PAN Qi, CHEN Huashi, LIANG Xing, LÜ Guoling
Received:2018-02-28
Online:2018-08-10
Published:2018-06-20
Contact:
HUANG Chibao
E-mail:huangchibao@163.com
Supported by:CLC Number:
TrendMD:
HUANG Chibao, PAN Qi, CHEN Huashi, LIANG Xing, LÜ Guoling. Triphenylamine-based Dicyanostilbene Type Two-photon Fluorescence Solvatochromic Probe†[J]. Chem. J. Chinese Universities, 2018, 39(8): 1676.
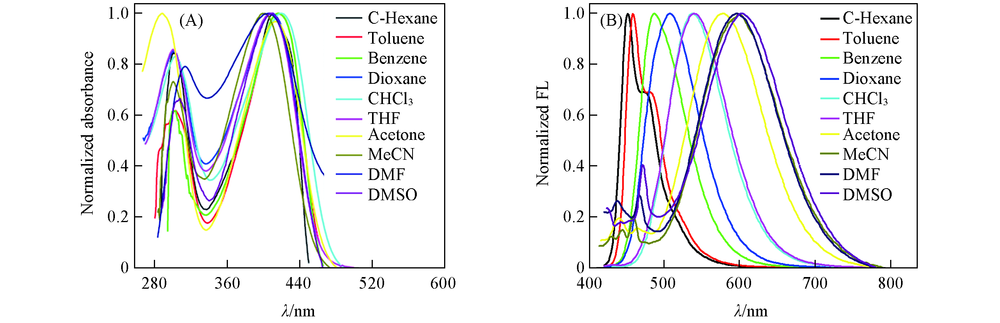
Fig.1 Normalized absorption(A) and one-photon emission spectra(B) of SP1 in various solventsc(SP1)(absorption)=10 μmol/L, c(SP1)(emission)=1 μmol/L, λex(OP)=412 nm.
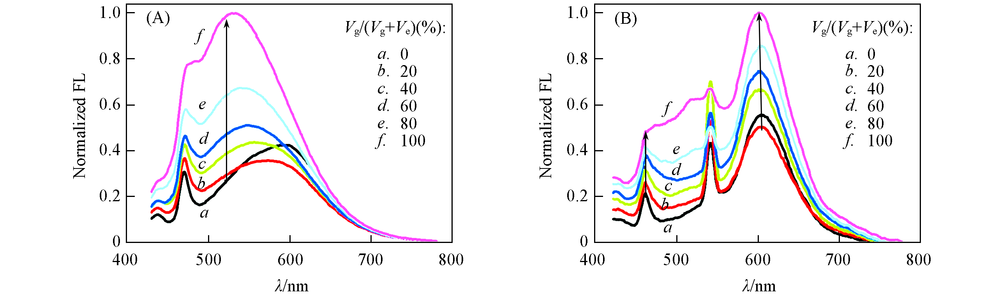
Fig.3 Normalized one-(A) and two-photon(B) emission spectra of SP1 in glycerol-ethanolOne-photon λex=412 nm; two-photon λex=810 nm; c(P2)=1 μmol/L; g: glycerol, e: ethanol.
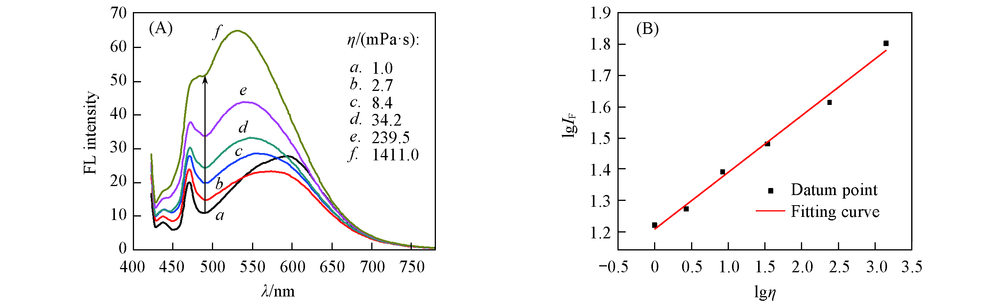
Fig.4 One-photon emission spectra(A) and linear fitting curve(B) in a double-logarithmic scale of SP1 in various viscosity(glycerol-ethanol solution)c(SP1)=1 μmol/L, λex(OP)=412 nm, 20 ℃.
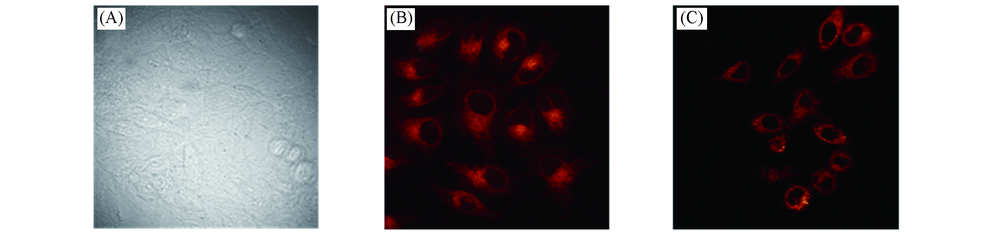
Fig.6 Bright-field two-photon excitation fluorescence image(A) and two-photon microscopy image of 1 μmol/L SP1-labeled(B) and 4 μmol/L X1-labeled(C) mouse fibroblast collected at 550—650 nmThe two-photon excitation fluorescence(TPEF) images were collected upon excitation at 800 nm with a femtosecond pulse.Cells shown were representative images from replicate experiments(n=5).
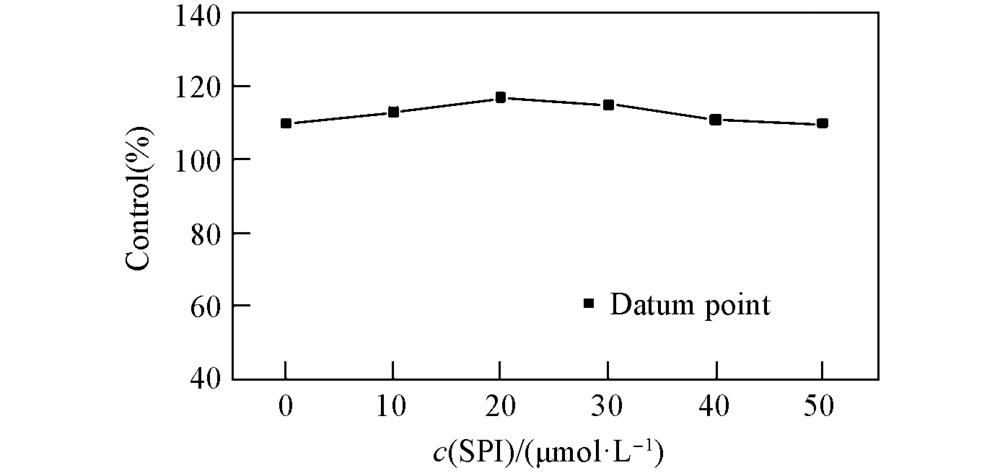
Fig.7 Response curve of fibroblast to SP1 dose from 0 to 50 μmol/L for 24 hData presented as percentage of control(n=3)±SD,SD: standard deviation, P<0.01.
| [1] | Su J., Huang S.S., He S., Zeng X. S., Chem. Res. Chinese Universities, 2016, 32(1), 20—27 |
| [2] | Bohne C., Ihmels H., Waidelich M., Yihwa C., J. Am. Chem. Soc., 2005, 127(49), 17158—17159 |
| [3] | Uchiyama S., Takehira K., Yoshihara T., Tobita S., Ohwada T., Organic Letters, 2006, 8(25), 5869—5872 |
| [4] | Sutharsan J., Lichlyter D., Wright N.E., Dakanali M., Haidekker M. A., Theodorakis E. A., Tetrahedron, 2010, 66, 2582—2588 |
| [5] | Droumaguet C.L., Mongin O., Werts M. H. V., Blanchard-Desce M., Chem. Commun., 2005, 41(22), 2802—2804 |
| [6] | Strehmel B., Sarker A.M., Detert H., ChemPhysChem, 2003, 4, 249—259 |
| [7] | Kim H.M., Jeong B. H., Hyon J. Y., An M. J., Seo M. S., Hong J. H., Lee K. J., Kim C. H., Joo T., Hong S. C., Cho B. R., J. Am. Chem. Soc., 2008, 130(13), 4246—4247 |
| [8] | Kim H.M., Choo H. J., Jung S. Y., Ko Y. G., Park W. H., Jeon S. J., Kim C. H., Joo T., Cho B. R., ChemBioChem, 2007, 8(5), 553—559 |
| [9] | Gers-Panther C.F., Fischer H., Nordmann J., Seiler T., Behnke T., Würth C., Frank W., Resch-Genger U., Müller T. J. J., J. Org. Chem., 2017, 82(1), 567—578 |
| [10] | Okada Y., Sugai M., Chiba K., J. Org. Chem., 2016, 81(22), 10922—10929 |
| [11] | Appelqvist H., Stranius K., Börjesson K., Nilsson K.P. R., Dyrager C., Bioconjugate Chem., 2017, 28(5), 1363—1370 |
| [12] | Gopikrishna P., Adil L.R., Iyer P. K., Mater. Chem. Front., 2017, 1, 2590—2598 |
| [13] | Mata G., Luedtke N.W., Org. Lett., 2013, 15(10), 2462—2465 |
| [14] | Xie X., Gutiérrez A., Trofimov V., Szilagyi I., Soldati T., Bakker E., Anal. Chem., 2015, 87(19), 9954—9959 |
| [15] | Liu H., Xu X., Shi Z., Liu K., Fang Y., Anal. Chem., 2016, 88(20), 10167—10175 |
| [16] | Yamashita H., Abe J., J. Phys. Chem. A, 2014, 118(8), 1430—1438 |
| [17] | Mukhopadhyay A., Mandal K.J., Moorthy J. N., Phys. Chem. Chem. Phys., 2018, doi: 10.1039/c7cp06766f |
| [18] | Kothavale S., Sekar N., Dyes and Pigments, 2017, 136, 116—130 |
| [19] | Warde U., Nagaiyan S., J. Photochem. Photobio. A: Chem., 2017, 337, 33—43 |
| [20] | Vyšniauskas A., López-Duarte I., Duchemin N., Vu T.T., Wu Y., Budynina E. M., Volkova Y. A., Cabrera E. P., Ramírez-Ornelas D. E., Kuimova M. K, Phys. Chem. Chem. Phys., 2017, 19, 25252—25259 |
| [21] | Ikejiri M., Mori K., Miyagi R., Konishi R., Chihara Y., Miyashita K., Org. Biomol. Chem., 2017, 15, 6948—6958 |
| [22] | Wang F., DeRosa C. A., Daly M. L., Song D., Sabat M., Fraser C. L., Mater. Chem. Front., 2017, 1, 1866—1874 |
| [23] | Zhang Y., Liang C., Jiang S., New J.Chem., 2017, 41, 8644—8649 |
| [24] | Suhina T., Amirjalayer S., Woutersen S., Bonn D., Brouwer A.M., Phys. Chem. Chem. Phys., 2017, 19, 19998—20007 |
| [25] | Kothavale S., Sekar N., RSC Adv., 2016, 6, 105387—105397 |
| [26] | Sasaki S., Drummen G.P. C., Konishi G. I., J. Mater. Chem. C, 2016, 4, 2731—2743 |
| [27] | Koenig M., Storti B., Bizzarri R., Guldi D.M., Brancato G., Bottari G., J. Mater. Chem. C, 2016, 4, 3018—3027 |
| [28] | Karpenko I.A., Niko Y., Yakubovskyi V. P., Gerasov A. O., Bonnet D., Kovtun Y. P., Klymchenko A. S., J. Mater. Chem. C, 2016, 4, 3002—3009 |
| [29] | Vu T.T., Méallet-Renault R., Clavier G., Trofimov B. A., Kuimova M. K., J. Mater. Chem. C, 2016, 4, 2828—2833 |
| [30] | Jung H.S., Verwilst P., Kim W. Y., Kim J. S., Chem. Soc. Rev., 2016, 45, 1242—1256 |
| [31] | Klymchenko A.S., Acc. Chem. Res., 2017, 50(2), 366—375 |
| [32] | Fares M., Li Y., Liu Y., Miao K., Gao Z., Zhai Y., Zhang X., Bioconjugate Chem., 2018, 29(1), 215—224 |
| [33] | Ibarra-Rodríguez M., Muñoz-Flores B.M., Dias H. V. R., Sánchez M., Gomez-Treviño A., Santillan R., Farfán N., Jiménez-Pérez V. M., J. Org. Chem., 2017, 82(5), 2375—2385 |
| [34] | Liu G.H., QiuX. P., Siddiq M., Bo S. Q., Ji X. L., Chem. Res. Chinese Universities, 2013, 29(5), 1022—1028 |
| [35] | Huang C.B., Liang X., Zeng Q. H., Chen H. S., Zeng B. P., Yi D. S., Chen X. Y., Chem. J. Chinese Universities, 2015, 36(4), 646—653 |
| (黄池宝, 梁兴, 曾启华, 陈华仕, 曾伯平, 易道生, 陈晓远. 高等学校化学学报, 2015, 36(4), 646—653) | |
| [36] | Huang C.B., Chen H. S., Zeng B. P., Chen X. Y., Chinese Journal of Analytical Chemistry, 2015, 43(4), 507—511 |
| (黄池宝, 陈华仕, 曾伯平, 陈晓远. 分析化学, 2015, 43(4), 507—511) | |
| [37] | Huang C.B., Pan Q., Chen X. Y., Zhao G. L., Chen H. S., Liang X., Lü G. L., Chem. J. Chinese Universities, 2017, 38(10), 646—653 |
| (黄池宝, 潘淇, 陈晓远, 赵光练, 陈华仕, 梁兴, 吕国岭. 高等学校化学学报, 2017, 38(10), 646—653) | |
| [38] | Huang C.B., Pan Q., Chen H. S., Liang X., Lv G. L., Chem. J. Chinese Universities, 2018, 39(5), 646—653 |
| (黄池宝, 潘淇, 陈华仕, 梁兴, 吕国岭. 高等学校化学学报, 2018, 39(5), 646—653) | |
| [39] | Huang H., He Q., Lin H., Bai F., Sun Z., Li Q., Polym. Adv. Technol., 2004, 15(1—2), 84—88 |
| [40] | Huang C.B., Fan J. L., Peng X. J., Lin Z. Y., Guo B. P., Ren A. X., Cui J. Q., Sun S. G., J. Photochem. Photobio. A: Chem., 2008, 199(2/3), 144—149 |
| [41] | Baker Ⅲ T.N., Jr. Doherty W. P., Kelley W. S., Newmeyer W., Jr. Rogers J. E., Spalding R. E., Walter R. I., J. Org. Chem., 1965, 30(11), 3714—3718 |
| [42] | Bent D.V., Sohulte-Frohlinde D., J. Phys. Chem., 1974, 78(4), 446—450 |
| [43] | Schanze K.S., Shin D. M., Whitten D. G., J. Am. Chem. Soc., 1985, 107(2), 507—509 |
| [44] | Shin D.M., Whitten D. G., J. Phys. Chem., 1988, 92(10), 2945—2956 |
| [45] | Lewis F.D., Sinks L. E., Weigel W., Sajimon M. C., Crompton E. M., J. Phys. Chem. A, 2005, 109(11), 2443—2451 |
| [46] | Jager W.F., Volkers A. A., Neckers D. C., Macromolecules, 1995, 28(24), 8153—8158 |
| [47] | Loutfy R.O., Law K. Y., J. Phys. Chem., 1980, 84(21), 2803—2808 |
| [48] | Förster T., Hoffmann G., Z. Phys. Chem., 1971, 75(1/2), 63—75 |
| [49] | Sutharsan J., Lichlyter D., Wright N.E., Dakanali M., Haidekker M. A., Theodorakis E. A., Tetrahedron, 2010, 66(14), 2582—2588 |
| [50] | Huang C.B., Ren A. X., Li H. B., Yang N. F., Chem. J. Chinese Universities, 2010, 31(11), 2222—2227 |
| (黄池宝, 任安祥, 李海渤, 阳年发. 高等学校化学学报, 2010, 31(11), 2222—2227) | |
| [51] | Huang C.B., Ren A. X., Acta Chimica Sinica, 2007, 65(23), 2765—2770 |
| (黄池宝, 任安祥. 化学学报, 2007, 65(23), 2765—2770) | |
| [52] | Huang C.B., Zhang D. H., Zeng B. P., Liu Q. B., Chen H. S., Kang S., Chen X. Y., Chem. J. Chinese Universities, 2018, 37(4), 638—642 |
| (黄池宝, 张道海, 曾伯平, 刘其斌, 陈华仕, 康帅, 陈晓远. 高等学校化学学报, 2016, 37(4), 638—642) | |
| [53] | Xie X.J., Gutiérrez A., Trofimov V., Szilagyi I., Soldati T., Bakker E, Anal. Chem., 2015, 87, 9954—9959 |
| [54] | Fotakis G., Timbrell J.A., Toxicology Letters, 2006, 160, 171—177 |
| [1] | ZHAO Baodong, LIU Yajing, PAN Yongfei, LIU Weixiao, GAO Fulei, WANG Yinglei. Synthesis and Properties of Energetic Plasticizer 2,2-Dinitropropyl Trifluoropropanoate [J]. Chem. J. Chinese Universities, 2021, 42(9): 2815. |
| [2] | JIANG Lei, YAN Shengdi, LIN Yu, WU Guozhang. Studies on Viscosity and Chemical Structure Changes in Polycarbonate Melts under Nitrogen Protection [J]. Chem. J. Chinese Universities, 2021, 42(3): 884. |
| [3] | HUANG Chibao,PAN Qi,CHEN Huashi,LIANG Xing,LÜ Guoling. Dicyanostilbene-derived Two-photon Fluorescence Environment-sensitive Probe† [J]. Chem. J. Chinese Universities, 2019, 40(6): 1128. |
| [4] | LI Haocheng, GAO Yuan, XIN Yingchun, ZONG Hua, LIU Huiying, ZHANG Lei, ZHANG Lu. Interfacial Dilational Rheology of Polyether Demulsifiers [J]. Chem. J. Chinese Universities, 2019, 40(4): 809. |
| [5] | MA Haizhu,HAN Wentao,TAO Fei. Effect of Side Groups on Conformational Characteristics of Containing Phosphor, Fluoride, Oxygen and Silane Polymers† [J]. Chem. J. Chinese Universities, 2018, 39(10): 2304. |
| [6] | LI Juanli, PENG Zenghui, LI Jian, HU Minggang, AN Zhongwei, ZHANG Lu. Synthesis and Properties of Novel Isothiocyanatotolane Liquid Crystals with Terminal Difluorovinyl Substitute† [J]. Chem. J. Chinese Universities, 2017, 38(10): 1788. |
| [7] | LI Yunxi, YUE Xigui, JIANG Zhenhua. Synthesis and Performance of Poly(ether ether ketone)s with Low Melt Viscosity† [J]. Chem. J. Chinese Universities, 2015, 36(11): 2160. |
| [8] | LI Dan, FANG Wen-Jun, LIU Li, WU Qian, ZHANG Hong-Yan, SUN Xiao-Ri. Volumetric and Viscous Properties for Binary Mixtures of N-Methylpiperazine with Ethyl Acetate or Butyl Acetate from 298.15 K to 313.15 K [J]. Chem. J. Chinese Universities, 2013, 34(8): 1924. |
| [9] | WANG Hui-Xia, YAO Lin, DING Bin, LUO Jian-Hui, ZHOU Ge, JIANG Bo. Molecular Simulation Study on Hydrophobically Modified Polyacrylamide Solutions [J]. Chem. J. Chinese Universities, 2013, 34(5): 1295. |
| [10] | LIU Fei, WU Tong, HU Ming-Ming, PENG Xiao-Jun, FAN Jiang-Li. A Novel Carbazole-based Cyanine as a Fluorescent Probe for Viscosity Detection [J]. Chem. J. Chinese Universities, 2012, 33(10): 2239. |
| [11] | CHEN Kun, LI Hai-Ping, HOU Wan-Guo. Zero-shear Viscosity and Critical Concentrations of Aqueous Solution of Partially Hydrolyzed Polyacrylamide [J]. Chem. J. Chinese Universities, 2012, 33(06): 1301. |
| [12] | CUI Xiao-Hong, ZHANG Lei, ZHAO Rong-Hua, LUO Lan, ZHANG Lu*, ZHAO Sui, YU Jia-Yong. Interfacial Interactions Among Aromatic Side Chained N-Acyltaurates Studied by Interfacial Tension Relaxation Measurements [J]. Chem. J. Chinese Universities, 2011, 32(7): 1556. |
| [13] | YIN Hong-Gen, HOU Feng-Wen, LI Yi*, CHENG Rong-Shi. Adsorption of Bovine Serum Albumin on the Surface of Medical Polyurethane Elastomer [J]. Chem. J. Chinese Universities, 2011, 32(11): 2691. |
| [14] | CAO Xu-Long*, ZHANG Lei, CHENG Jian-Bo. Effect of Gemini surfactant on the Interfacial Dilational Properties of Hydrophobically Modified Partly Hydrolyzed Polyacrylamide [J]. Chem. J. Chinese Universities, 2010, 31(5): 998. |
| [15] | . Effect of Interactions Among Flooding Chemicals on Interfacial Properties of Adsorption Film [J]. Chem. J. Chinese Universities, 2010, 31(12): 2445. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||