

Chem. J. Chinese Universities ›› 2018, Vol. 39 ›› Issue (8): 1734.doi: 10.7503/cjcu20170768
• Physical Chemistry • Previous Articles Next Articles
YANG Meng, ZHANG Tengshuo, ZHENG Xuming, XUE Jiadan*( )
)
Received:2017-11-27
Online:2018-08-10
Published:2018-06-26
Contact:
XUE Jiadan
E-mail:jenniexue@zstu.edu.cn
Supported by:CLC Number:
TrendMD:
YANG Meng, ZHANG Tengshuo, ZHENG Xuming, XUE Jiadan. Study on the Decay Dynamic of Excited State and Photodissociation Channel for 2-Nitronaphthalene†[J]. Chem. J. Chinese Universities, 2018, 39(8): 1734.
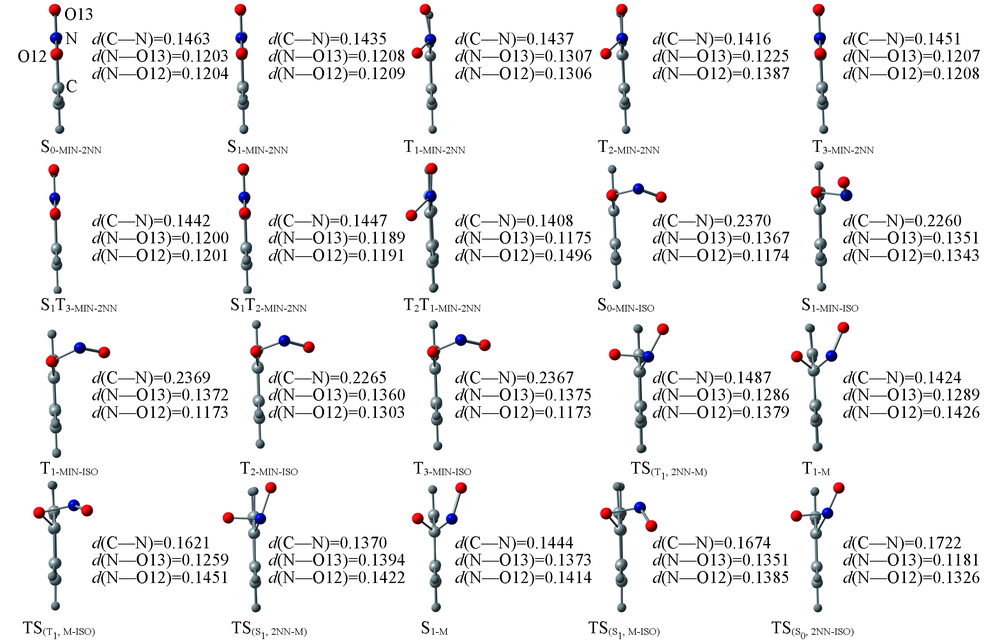
Fig.1 Front view of the optimized structures for stationary, transition states and curve-crossing points of 2NN and ISOSelected bonds distances are in nm.
| System | Species | Character | ΔE/(kJ·mol-1) | System | Species | Character | ΔE/(kJ·mol-1) |
|---|---|---|---|---|---|---|---|
| 2NN | S0-MIN-2NN | 0 | T | 270.7 | |||
| S1-FC-2NN | ππ* | 411.7 | T | 390.4 | |||
| T1-FC-2NN | ππ* | 347.3 | T1-MIN-M | 370.3 | |||
| T2-FC-2NN | πNOπ* | 375.7 | T | 392.0 | |||
| T3-FC-2NN | ππ* | 439.7 | T | 484.5 | |||
| S1-MIN-2NN | ππ* | 369.0 | S1-MIN-M | 448.1 | |||
| T3-MIN-2NN | ππ* | 410.5 | T | 468.2 | |||
| T2-MIN-2NN | nNOπ*+ππ* | 315.9 | ISO | 40.2 | |||
| T1-MIN-2NN | πNOπ* | 290.8 | S1-FC-ISO | ππ* | 470.7 | ||
| S1 | ππ*/ππ* | 405.1(389.5/420.9)a | T1-FC-ISO | ππ* | 370.7 | ||
| S1 | ππ*/ππ* | 396.6(397.9/395.4)a | S1-MIN-ISO | ππ* | 449.8 | ||
| T2 | πNOπ*/πNOπ* | 326.4(329.7/323.0)a | T1-MIN-ISO | ππ* | 306.3 |
Table 1 CASPT2(10,10)/6-31G(d)//CASPT2(10,10)/Aug-cc-PVDZ calculated excitation energies of the low-lying electronic states and curve-crossings of 2NN and ISO
| System | Species | Character | ΔE/(kJ·mol-1) | System | Species | Character | ΔE/(kJ·mol-1) |
|---|---|---|---|---|---|---|---|
| 2NN | S0-MIN-2NN | 0 | T | 270.7 | |||
| S1-FC-2NN | ππ* | 411.7 | T | 390.4 | |||
| T1-FC-2NN | ππ* | 347.3 | T1-MIN-M | 370.3 | |||
| T2-FC-2NN | πNOπ* | 375.7 | T | 392.0 | |||
| T3-FC-2NN | ππ* | 439.7 | T | 484.5 | |||
| S1-MIN-2NN | ππ* | 369.0 | S1-MIN-M | 448.1 | |||
| T3-MIN-2NN | ππ* | 410.5 | T | 468.2 | |||
| T2-MIN-2NN | nNOπ*+ππ* | 315.9 | ISO | 40.2 | |||
| T1-MIN-2NN | πNOπ* | 290.8 | S1-FC-ISO | ππ* | 470.7 | ||
| S1 | ππ*/ππ* | 405.1(389.5/420.9)a | T1-FC-ISO | ππ* | 370.7 | ||
| S1 | ππ*/ππ* | 396.6(397.9/395.4)a | S1-MIN-ISO | ππ* | 449.8 | ||
| T2 | πNOπ*/πNOπ* | 326.4(329.7/323.0)a | T1-MIN-ISO | ππ* | 306.3 |
| 2NN | Electron density | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 41 (πH-4) | 42 (πNOH-3) | 43 (πH-2) | 44 (πH-1) | 45 (πH) | 45 (πH+nH) | 46 ( | 46 ( | 47 ( | 48 ( | 49 ( | 50 ( | |
| S1T3-MIN-2NN | 1.87 | 1.93 | 1.90 | 1.53 | 1.36 | — | 0.50 | — | 0.06 | 0.10 | 0.12 | 0.64 |
| 1.87 | 1.94 | 1.92 | 1.13 | 1.74 | — | 0.87 | — | 0.05 | 0.12 | 0.09 | 0.27 | |
| S1T2-MIN-2NN | 1.87 | 1.93 | 1.90 | 1.52 | 1.36 | — | 0.644 | — | 0.06 | 0.10 | 0.12 | 0.50 |
| 1.88 | 1.94 | 1.89 | 1.53 | 1.37 | — | 0.51 | — | 0.10 | 0.05 | 0.12 | 0.62 | |
| T2T1-MIN-2NN | 1.99 | 1.00 | 1.95 | 1.99 | — | 1.93 | — | 1.00 | 0.01 | 0.08 | 0.05 | 0.00 |
| 1.99 | 1.00 | 1.95 | 1.99 | 1.93 | — | — | 1.00 | 0.00 | 0.08 | 0.05 | 0.01 | |
Table 2 CASPT2(10,10) calculated one electron density associated the curve-crossing points of 2NN
| 2NN | Electron density | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 41 (πH-4) | 42 (πNOH-3) | 43 (πH-2) | 44 (πH-1) | 45 (πH) | 45 (πH+nH) | 46 ( | 46 ( | 47 ( | 48 ( | 49 ( | 50 ( | |
| S1T3-MIN-2NN | 1.87 | 1.93 | 1.90 | 1.53 | 1.36 | — | 0.50 | — | 0.06 | 0.10 | 0.12 | 0.64 |
| 1.87 | 1.94 | 1.92 | 1.13 | 1.74 | — | 0.87 | — | 0.05 | 0.12 | 0.09 | 0.27 | |
| S1T2-MIN-2NN | 1.87 | 1.93 | 1.90 | 1.52 | 1.36 | — | 0.644 | — | 0.06 | 0.10 | 0.12 | 0.50 |
| 1.88 | 1.94 | 1.89 | 1.53 | 1.37 | — | 0.51 | — | 0.10 | 0.05 | 0.12 | 0.62 | |
| T2T1-MIN-2NN | 1.99 | 1.00 | 1.95 | 1.99 | — | 1.93 | — | 1.00 | 0.01 | 0.08 | 0.05 | 0.00 |
| 1.99 | 1.00 | 1.95 | 1.99 | 1.93 | — | — | 1.00 | 0.00 | 0.08 | 0.05 | 0.01 | |
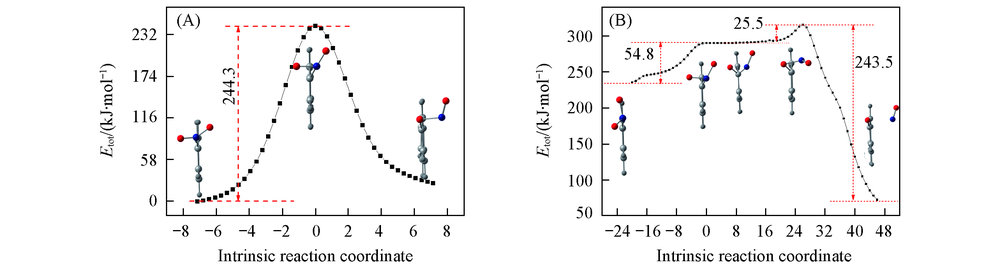
Fig.3 IRC scans of the transition states in S0 and T1 state(A) Reactant S0-MIN-2NN, transition state TS(S0,2NN?ISO), product S0?MIN?ISO; (B) transient intermediates, reactant T1-MIN-2NN, transition state TS(T1,2NN?M), TS(T1,M?ISO), product T1-MIN-ISO.
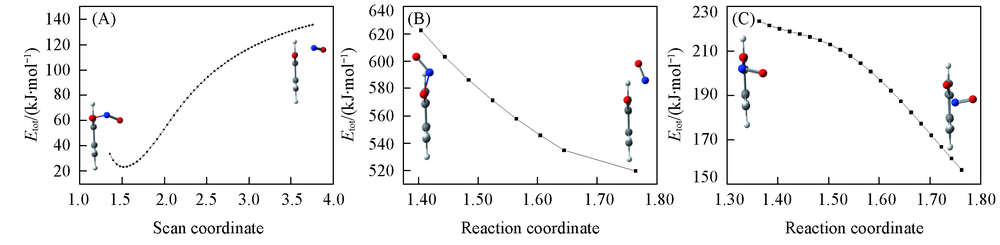
Fig.4 Potential energy scans connecting S0-MIN-ISO, S1-MIN-ISO and T1-MIN-ISO along the N—O reaction coordinate(A) S0-MIN-ISO; (B) S1-MIN-ISO; (C) T1-MIN-ISO.
| [1] | Arce R., Pino E.F., Valle C., Negrón-Encarnación I., Morel M., J. Phys. Chem. A, 2011, 115(2), 152—160 |
| [2] | Reynission J., Stiborová M., Martínek V., Environ. Mol. Mutagen., 2008, 49(8), 659—667 |
| [3] | Pande P., Malik C.K., Bose A., Jasti V. P., Basu A. K, Biochem., 2014, 53(32), 5323—5331 |
| [4] | Vogt R.A., Hahman S., Crespo-Hernández C. E, Practical Aspects Comput. Chem., 2009, 19(1), 217—240 |
| [5] | Feiberg A., Kamens R.M., Strommen M. R., Nielsen T, Polycyclic Aromat. Compd., 1999, 14(1—4), 151—160 |
| [6] | Phousongphouang P.T., Arey J., J. Photochem. Photobiol. A, 2003, 157(2/3), 301—309 |
| [7] | Yu H.J., Environ. Sci. Health. C, 2002, 20(2), 149—183 |
| [8] | Atkinson R., Aschmann S.M., Arey J., Zielinska B., Schuetzle D, Atmospheric Environment., 1989, 23(12), 2679—2690 |
| [9] | Healy R.M., Chen Y., Kourtchev I., Kalberer M., O’Shea D., Wenger J. C, Environ. Sci. Technol., 2012, 46(21), 11813—11820 |
| [10] | Brister M.M., Piñero-Santiago L. E., Morel M., Arce R., Crespo-Hernández C. E., J. Phys. Chem. Lett., 2016, 7, 5086—5092 |
| [11] | Brister M.M., Piñero-Santiago L. E., Morel M., Arce R., Crespo-Hernández C. E., J. Phys. Chem. A, 2017, 121(43), 8197—8206 |
| [12] | Vogt R.A., Crespo-Hernández C. E., J. Phys. Chem. A, 2013, 117, 14100—14108 |
| [13] | Morales-Cueto R., Esquivelzeta-Rabell M., Saucedo-Zugazagoitia J., Peon J., J. Phys. Chem. A, 2007, 111(4), 552—527 |
| [14] | Larsen M.A., Thogersen J., Stephansen A. B., Peon J., Solling T. I., Keiding S. R., J. Phys. Chem. A, 2016, 120(1), 28—35 |
| [15] | Fukuhara K., Kurihara M., Miyata N., J. Am. Chem. Soc., 2001, 123(36), 8662—8666 |
| [16] | Wolfbeis O.S., Posch W., Gübitz G., Tritthart P., Analy. Chim. Acta, 1983, 147, 405—410 |
| [17] | Rusakowicz B., Testa A.C., Spectrochimica Acta Part A: Molecular Spectroscopy, 1971, 27(6), 787—792 |
| [18] | García-Berríos Z.I., Arce R., J. Phys. Chem. A, 2012, 116(14), 3652—3664 |
| [19] | Arce R., Pion E.F., Valle C., Ágreda J., J. Phys. Chem. A, 2008, 112(41), 10294—10304 |
| [20] | Reichardt C., Vogt R.A., Crespo-Hernndez C. E., J. Chem. Phys., 2009, 131(22), 224518 |
| [21] | Giussani A., J. Chem. Theory Comput., 2014, 10(9), 3987—3995 |
| [22] | Vogt R.A., Reichardt C., Crespo-Hernández C. E., J. Phys. Chem. A, 2013, 117(30), 6580—6588 |
| [23] | Brown H.W., Pimentel G. C., J. Chem. Phys., 1958, 29(4), 883—888 |
| [24] | Chapman O.L., Heckert D. C., Reasoner J. W., Thackaberry S. P., J. Am. Chem. Soc., 1966, 88(23), 5550—5554 |
| [25] | Roos B.O., Taylor P. R., Sigbahn P. E. M, Chem. Phys., 1980, 48(2), 157—173 |
| [26] | Roos B.O., Advances in Chemical Physics: Ab Initio Methods in Quantum Chemistry Part 2, 1987, 69, 399—445 |
| [27] | Andersson K., Malmqvist P.Å., Roos B. O., J. Chem. Phys., 1992, 96(2), 1218—1226 |
| [28] | Werner H.J., Mol. Phys., 1996, 89(2), 645—661 |
| [29] | Celani P., Werner H.J., J. Chem. Phys., 2000, 112(13), 5546—5557 |
| [30] | Becke A.D., J. Chem. Phys., 1993, 98(2), 1372—1377 |
| [31] | Barone V., Cossi M., Tomasi J., J. Chem. Phys., 1997, 107(8), 3210—3221 |
| [32] | Lee C., Yang W., Parr R.G., Phys. Rev. B: Condens. Mater. Phys., 1988, 37(2), 785—789 |
| [33] | Frisch M.J., Trucks G. W., Schlegel H. B., Scuseria G. E., Robb M. A., Cheeseman J. R., Scalmani G., Barone V., Mennucci B., Petersson G. A., Nakatsuji H., Caricato M., Li X., Hratchian H. P., Izmaylov A. F., Bloino J., Zheng G., Sonnenberg J. L., Hada M., Ehara M., Toyota K., Fukuda R., Hasegawa J., Ishida M., Nakajima T., Honda Y., Kitao O., Nakai H., Vreven T., Montgomery J. A. Jr., Peralta J. E., Ogliaro F., Bearpark M., Heyd J. J., Brothers E., Kudin K. N., Staroverov V. N., Kobayashi R., Normand J., Raghavachari K., Rendell A., Burant J. C., Iyengar S. S., Tomasi J., Cossi M., Rega N., Millam J. M., Klene M., Knox J. E., Cross J. B., Bakken V., Adamo C., Jaramillo J., Gomperts R., Stratmann R. E., Yazyev O., Austin A. J., Cammi R., Pomelli C., Ochterski J. W., Martin R. L., Morokuma K., Zakrzewski V. G., Voth G. A., Salvador P., Dannenberg J. J., Dapprich S., Daniels A. D., Farkas Ö., Foresman J. B., Ortiz J. V., Cioslowski J., Fox D. J., Gaussian 09, Revision A.01, Gaussian Inc., Wallingford CT, 2009 |
| [34] | Werner H.J., Knowles P. J., Knizia G., Manby F. R., Schütz M, WIREs Comput. Mol. Sci., 2012, 2(2), 242—253 |
| [35] | James A.D., J. Am. Chem. Soc., 2009, 131(33), 2416—2416 |
| [1] | LIU Suyu, DING Fei, LI Qian, FAN Chunhai, FENG Jing. Azobenzene-integrated DNA Nanomachine [J]. Chem. J. Chinese Universities, 2022, 43(8): 20220122. |
| [2] | GAO Jian, FENG Yiyu, FANG Wenyu, WANG Hui, GE Jing, FENG Wei. Alkane Grafted Phase Change Azobenzene Materials Based on Low Temperature Heat Release [J]. Chem. J. Chinese Universities, 2022, 43(8): 20220146. |
| [3] | ZHOU Min, SHI Yingying, LI Shuqi, ZHANG Kailin, CUI Yongliang, ZHANG Sen, ZHANG Xianyi, KONG Xianglei. 193 nm UV Photodissociation Mass Spectrometry for Ubiquitin Ions with Different Charge States [J]. Chem. J. Chinese Universities, 2021, 42(8): 2436. |
| [4] | YING Fuming, JI Chenru, SU Peifeng, WU Wei. λ-DFCAS: A Hybrid Density Functional Complete Active Space Self Consistent Field Method [J]. Chem. J. Chinese Universities, 2021, 42(7): 2218. |
| [5] | YANG Yiying, ZHU Rongxiu, ZHANG Dongju, LIU Chengbu. Theoretical Study on Gold-catalyzed Cyclization of Alkynyl Benzodioxin to 8-Hydroxy-isocoumarin [J]. Chem. J. Chinese Universities, 2021, 42(7): 2299. |
| [6] | LIN Dan, CHENG Min, DU Yikui, ZHU Qihe. Photofragment Translational Spectroscopy of CF3I at 238 nm:Vibrational Distribution of CF3 Fragments andCurve Crossing Probability† [J]. Chem. J. Chinese Universities, 2018, 39(8): 1713. |
| [7] | FANG Sheng, WANG Meiyan, LIU Jingjing, LIU Jingyao. Reaction Mechanism of Nickel Complex Catalyzed Isomerization of N-Allylamides† [J]. Chem. J. Chinese Universities, 2018, 39(7): 1475. |
| [8] | LI Chuansong, DU Yanyan, XUE Xuzhi, XIANG Xianzheng, LI Jiusheng, REN Tianhui. Preparation and Performance of Pt/IZM-2 Catalyst on the Hydroisomerization of n-Dodecane† [J]. Chem. J. Chinese Universities, 2018, 39(4): 729. |
| [9] | ZHANG Yujian, ZHAI Xuli, FU Kaimei, JIANG Tao. Synthesis of Nano ZSM-23 Zeolites with Low L/D Value Morphology and Their Hydroisomerization Performance† [J]. Chem. J. Chinese Universities, 2017, 38(2): 231. |
| [10] | CAO Dan, LI Yuanying, SU Qingqing, WANG Bin, LIU Fengyi, WANG Wenliang. CASSCF and MS-CASPT2 Studies on an Electron-tunable,1,2-Dicyanoethylene-based Optical Molecular Switch† [J]. Chem. J. Chinese Universities, 2016, 37(6): 1128. |
| [11] | OUYANG Bing, XUE Jiadan, ZHENG Xuming. Excited State Dynamics of γ-Crotonolactone: Resonance Raman Spectroscopy and Complete Active Space Self-consistent Field(CASSCF) Study† [J]. Chem. J. Chinese Universities, 2015, 36(10): 1995. |
| [12] | SUO Yanhua, LI Xiumin, CHEN Gang, CUI Yingxue, SUN Yujia, WANG Yingjun. Ni/SAPO-11 Promoted by Rare Earth Element Ce for Hydroisomerization of n-Heptane† [J]. Chem. J. Chinese Universities, 2014, 35(6): 1252. |
| [13] | SONG Hua, SHI Yang, SONG Hua-Lin. Preparation of Pt-S2O82-/ZrO2-Al2O3 Catalyst by Microemulsion Method and Its Performance for Isomerization [J]. Chem. J. Chinese Universities, 2012, 33(09): 2061. |
| [14] | ZHANG Ding-Lin, FU Hai-Yan, ZHAO Xian-Ying, ZHAO Hua-Wen, CHEN Hua, LIU Yi-Min, LI Xian-Jun. HRhCO(TPPTS)3/HAP Hydroformylate 1-Hexene High Selectivity and Special Isomerization Performance [J]. Chem. J. Chinese Universities, 2012, 33(08): 1835. |
| [15] | CI Cheng-Gang, DUAN Xue-Mei, LIU Jing-Yao*, SUN Chia-Chung. Theoretical Studies on Photodissociation Mechanism of Glycoladehyde [J]. Chem. J. Chinese Universities, 2011, 32(7): 1588. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||