

Chem. J. Chinese Universities ›› 2016, Vol. 37 ›› Issue (1): 108.doi: 10.7503/cjcu20150462
• Physical Chemistry • Previous Articles Next Articles
YANG Ming1, TANG Jia1, WANG Jingjing1, FAN Shuang1, ZHANG Huan1, TAO Liushi1, TAN Li2,*( )
)
Received:2015-06-12
Online:2016-01-10
Published:2015-12-20
Contact:
TAN Li
E-mail:tljw060606@163.com
CLC Number:
TrendMD:
YANG Ming, TANG Jia, WANG Jingjing, FAN Shuang, ZHANG Huan, TAO Liushi, TAN Li. Paal-Knorr Reaction Catalyzed by Fe-MIL-101 for Efficient Synthesis of Pyrroles†[J]. Chem. J. Chinese Universities, 2016, 37(1): 108.
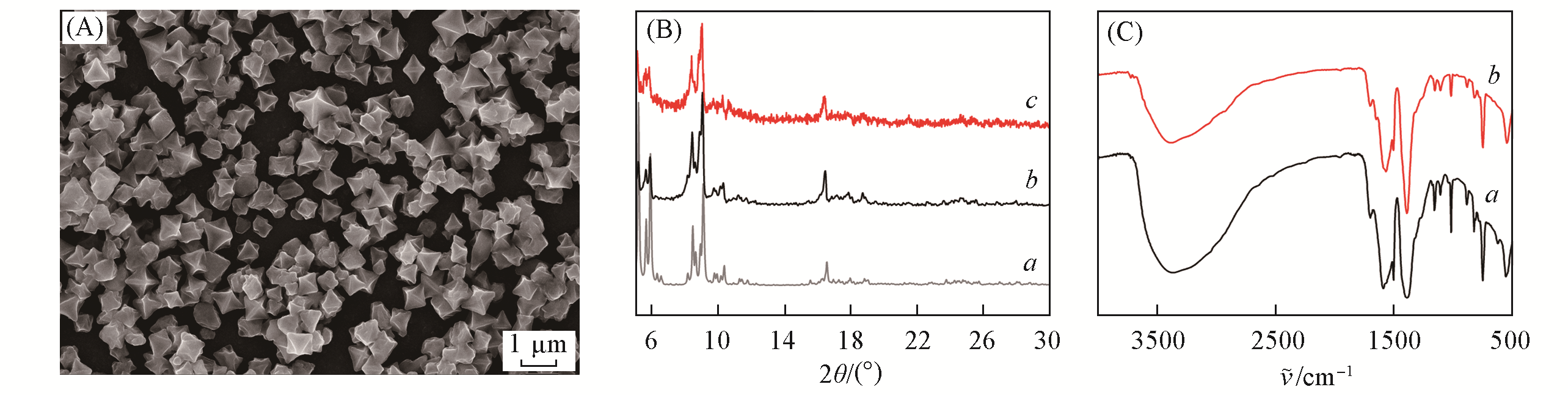
Fig.1 SEM image of Fe-MIL-101(A), PXRD patterns(B) of Cr-MIL-101-simulation(a), Fe-MIL-101(b), MIL-101(Fe)(c) and FTIR spectra(C) of Fe-MIL-101(a) and MIL-101(Fe)(b)
| Entry | Catalyst | Time/min | Conversion(%) | Selectivity(%) |
|---|---|---|---|---|
| 1 | 60 | 20.4 | 99.9 | |
| 2 | FeCl3·6H2O | 50 | 99.9 | 99.9 |
| 3 | Fe(NO3)3·9H2O | 50 | 99.9 | 99.9 |
| 4 | Fe2(SO4)3 | 50 | 98.8 | 99.9 |
| 5 | Fe(C5H5)2 | 50 | 13.3 | 99.9 |
| 6 | Fe-MIL-101 | 50 | 98.9 | 99.9 |
Table 1 Influence of Fe(Ⅲ)-catalysts on the Paal-Knorr reaction conversion*
| Entry | Catalyst | Time/min | Conversion(%) | Selectivity(%) |
|---|---|---|---|---|
| 1 | 60 | 20.4 | 99.9 | |
| 2 | FeCl3·6H2O | 50 | 99.9 | 99.9 |
| 3 | Fe(NO3)3·9H2O | 50 | 99.9 | 99.9 |
| 4 | Fe2(SO4)3 | 50 | 98.8 | 99.9 |
| 5 | Fe(C5H5)2 | 50 | 13.3 | 99.9 |
| 6 | Fe-MIL-101 | 50 | 98.9 | 99.9 |
| Entry | Solvent | Time/min | Conversion(%) | Selectivity(%) |
|---|---|---|---|---|
| 1 | | 50 | 2.4 | 99.9 |
| 2 | C2H5OHb | 50 | 9.1 | 99.9 |
| 3 | CH3CNb | 50 | 12.5 | 99.9 |
| 4 | CHC | 50 | 17.7 | 99.9 |
| 5 | C4H8Ob | 50 | 31.0 | 99.9 |
| 6 | C2H4C | 50 | 8.7 | 99.9 |
| 7 | H2Ob | 50 | 98.9 | 99.9 |
| 8 | C2H4C | 50 | 64.1 | 99.9 |
| 9 | C2H4C | 180 | 99.9 | 99.9 |
Table 2 Influence of different solvents on the Paal-Knorr reaction conversiona
| Entry | Solvent | Time/min | Conversion(%) | Selectivity(%) |
|---|---|---|---|---|
| 1 | | 50 | 2.4 | 99.9 |
| 2 | C2H5OHb | 50 | 9.1 | 99.9 |
| 3 | CH3CNb | 50 | 12.5 | 99.9 |
| 4 | CHC | 50 | 17.7 | 99.9 |
| 5 | C4H8Ob | 50 | 31.0 | 99.9 |
| 6 | C2H4C | 50 | 8.7 | 99.9 |
| 7 | H2Ob | 50 | 98.9 | 99.9 |
| 8 | C2H4C | 50 | 64.1 | 99.9 |
| 9 | C2H4C | 180 | 99.9 | 99.9 |
| Entry | Temperature/℃ | Time | Conversion(%) | Selectivity(%) |
|---|---|---|---|---|
| 1 | 60 | 1 h | 1.91 | 99.9 |
| 2 | 60 | 24 h | 46.8 | 99.9 |
| 3 | 80 | 1 h | 37.1 | 99.9 |
| 4 | 80 | 3 h | 89.9 | 99.9 |
| 5 | 80 | 6 h | 99.5 | 99.9 |
| 6 | 100 | 50 min | 98.9 | 99.9 |
| 7 | 100 | 1 h | 99.6 | 99.9 |
Table 3 Influence of temperature and time on the Paal-Knorr reaction conversion*
| Entry | Temperature/℃ | Time | Conversion(%) | Selectivity(%) |
|---|---|---|---|---|
| 1 | 60 | 1 h | 1.91 | 99.9 |
| 2 | 60 | 24 h | 46.8 | 99.9 |
| 3 | 80 | 1 h | 37.1 | 99.9 |
| 4 | 80 | 3 h | 89.9 | 99.9 |
| 5 | 80 | 6 h | 99.5 | 99.9 |
| 6 | 100 | 50 min | 98.9 | 99.9 |
| 7 | 100 | 1 h | 99.6 | 99.9 |
| Entry | Amine | Product | Conversion(%) | Selectivity(%) | Entry | Amine | Product | Conversion(%) | Selectivity(%) |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 98.9 | 99.9 | 5 | 99.4 | 99.9 | ||||
| 2 | 99.9 | 99.9 | 6 | 99.7 | 99.9 | ||||
| 3 | 99.7 | 99.9 | 7 | 98.2 | 99.9 | ||||
| 4 | 99.0 | 99.9 | 8 | 99.4 | 99.9 | ||||
Table 4 Paal-Knorr reaction catalyzed by Fe-MIL-101 between tetrahydro-2,5-dimethoxyfuran and amines*
| Entry | Amine | Product | Conversion(%) | Selectivity(%) | Entry | Amine | Product | Conversion(%) | Selectivity(%) |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 98.9 | 99.9 | 5 | 99.4 | 99.9 | ||||
| 2 | 99.9 | 99.9 | 6 | 99.7 | 99.9 | ||||
| 3 | 99.7 | 99.9 | 7 | 98.2 | 99.9 | ||||
| 4 | 99.0 | 99.9 | 8 | 99.4 | 99.9 | ||||
| Entry | Amine | Product | Conversion(%) | Selectivity(%) | Entry | Amine | Product | Conversion(%) | Selectivity(%) |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 93.6 | 99.9 | 6 | 91.8 | 99.9 | ||||
| 2 | 53.2 | 99.9 | 7 | 67.8 | 99.9 | ||||
| 3 | 93.4 | 99.9 | 8 | 99.0 | 99.9 | ||||
| 4 | 99.0 | 99.9 | 9 | 99.0 | 99.9 | ||||
| 5 | 96.7 | 99.9 | |||||||
Table 5 Paal-Knorr reaction catalyzed by Fe-MIL-101 between acetonylacetone with amines*
| Entry | Amine | Product | Conversion(%) | Selectivity(%) | Entry | Amine | Product | Conversion(%) | Selectivity(%) |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 93.6 | 99.9 | 6 | 91.8 | 99.9 | ||||
| 2 | 53.2 | 99.9 | 7 | 67.8 | 99.9 | ||||
| 3 | 93.4 | 99.9 | 8 | 99.0 | 99.9 | ||||
| 4 | 99.0 | 99.9 | 9 | 99.0 | 99.9 | ||||
| 5 | 96.7 | 99.9 | |||||||
| [1] | Adhikari A., Radhakrishnan S., Vijayan M., J. Appl. Polym. Sci., 2012, 125, 1875—1881 |
| [2] | Andersen R. J., Faulkner D. J., Cun-heng H., van Duyne G. D., Clardy J., J. Am. Chem. Soc., 1985, 107(19), 5492—5495 |
| [3] | Fürstner A., Angew. Chem. Int. Ed., 2003, 42, 308—311 |
| [4] | Dieter R. K., Yu H., Org. Lett., 2000, 2, 2283—2286 |
| [5] | Hantzsch A., Dtsch Ber., Chem. Ges., 1890, 23, 1474—1476 |
| [6] | Ferreira V. F., de Souza M. C. B. V., Cunha A. C., Pereira L. O. R., Ferreira M. L. G., Org. Prep. Proced. Int., 2001, 33, 411—454 |
| [7] | Berree F., Marchand E., Morel G., Tetrahedron Lett., 1992, 33, 6155—6158 |
| [8] | Lee C. F., Yang I. M., Hwu T. Y., Feng A. S., Tseng J. C., Luh T. Y., J. Am. Chem. Soc., 2000, 122, 4992—4993 |
| [9] | Chiu P. K., Sammes M. P., Tetrahedron,1990, 46, 3439—3456 |
| [10] | Wang B., Gu Y., Luo C., Yang T., Yang L., Suo J., Tetrahedron Lett., 2004, 45, 3417—3419 |
| [11] | Chen J. X., Wu H. T., Zheng Z. G., Jin C., Zhang X. X., Su W. K., Tetrahedron Lett., 2006, 47, 5383—5387 |
| [12] | Luo H. T., Kang Y. R., Li Q., Yang L. M., Heteroat. Chem., 2008, 19, 144—148 |
| [13] | Azizi N., Davoudpour A., Eskandari F., Batebi E., Monatsh. Chem., 2013, 144, 405—409 |
| [14] | Monnier F., Taillefer M., Angew. Chem. Int. Ed., 2009, 48, 6954—6971 |
| [15] | Deng H. J., Fang Y. J., Chen G. W., Liu M. C., Wu H. Y., Chen J. X., Appl. Organomet. Chem., 2012, 26, 164—167 |
| [16] | Jafari A. A., Amini S., Tamaddon F., J. Appl. Polym. Sci., 2012, 125, 1339—1345 |
| [17] | Polshettiwar V., Varma R. S., Tetrahedron,2010, 66, 1091—1097 |
| [18] | Polshettiwar V., Baruwati B., Varma R.S.,Chem. Commun., 2009, 1837—1839 |
| [19] | Sreekumar R., Raghavakaimal P., Synth. Commun., 1998, 28(9), 1661—1665 |
| [20] | Song G. Y., Wang B., Wang G., Kang Y. R., Yang T., Yang L. M., Synth. Commun., 2005, 35, 1051—1057 |
| [21] | Xie S. M., Zhang Z. J., Yuan L. M., Chem. J. Chinese Universities,2014, 35(8), 1652—1657 |
| (谢生明, 张泽俊, 袁黎明. 高等学校化学学报, 2014, 35(8), 1652—1657>) | |
| [22] | Song L. F., Zhang J., Sun L. X., Xu F., Li F., Zhang H. Z., Si X. L., Jiao C. L., Li Z. B., Liu S., Liu Y. L., Zhou H. Y., Sun D., Du Y., Cao Z., Gabelica Z., Energy Environ. Sci., 2012, 5, 7508—7520 |
| [23] | Xu L., Chen C., Wang R., Luo J. H., Liu Y. L., Zhang N., Chem. J. Chinese Universities,2013, 34(8), 1907—1912 |
| (徐力, 陈超, 王瑞, 罗家还, 刘云凌, 张宁. 高等学校化学学报, 2013, 34(8), 1907—1912) | |
| [24] | Furukawa H., Cordova K. E., O’Keeffe M., Yaghi O. M., Science,2013, 341, 1230444-1—12 |
| [25] | Song F.J., Zhang T., Wang C., Lin W. B., Proc. R. Soc. A., 2012, 468,2035—2052 |
| [26] | Zhang Y., Yang X. G., Wang Q. Y., Yao J., Hu J., Wang G. Y., Chem. J. Chinese Universities,2014, 35(3), 613—618 |
| ( 张毅, 杨先贵, 王庆印, 姚洁, 胡静, 王公应. 高等学校化学学报, 2014, 35(3), 613—618) | |
| [27] | O’Keeffe M., Yaghi O. M., Chem. Rev., 2012, 112, 675—702 |
| [28] | Lee J.Y., Farha O. K., Roberts J., Scheidt K. A., Nguyen S. B. T., Hupp J. T., Chem. Soc. Rev., 2009, 38, 1450—1459 |
| [29] | Tang J., Dong W. J., Wang G., Yao Y. Z., Cai L., Liu Y., Zhao X., Xu J. Q., Tan L., RSC Adv., 2014, 4, 42977—42982 |
| [30] | Tang J., Yang M., Yang M., Dong W. J., Wang G., New J. Chem., 2015, 39, 4919—4923 |
| [31] | Phan N. T.S., Nguyen T.T., Luu Q. H., Nguyen L. T. L., J. Mol. Catal. A: Chem., 2012, 363/364, 178—185 |
| [32] | Dhakshinamoorthy A., Alvaro M., Garcia H., Adv. Synth. Catal., 2010, 352, 3022—3030 |
| [33] | Kathryn M. L., Pashow T., Rocca J. D., Xie Z. G., Tran S., Lin W. B., J. Am. Chem. Soc., 2009, 131, 14261—14263 |
| [34] | Azizi N., Khajeh-Amiri A., Ghafuri H., Bolourtchian M., Saidi M. R., Synlett., 2009, 14, 2245—2248 |
| [35] | Wang F. H., Zhu X. D., Miao H., J. Anhui Poly. Univ., 2014, 29(2), 26—30 |
| (王芬华, 朱贤东, 苗慧. 安徽工程大学学报, 2014, 29(2), 26—30) |
| [1] | GUO Biao, ZHAO Chencan, LIU Xinxin, YU Zhou, ZHOU Lijing, YUAN Hongming, ZHAO Zhen. Effects of Surface Hydrothermal Carbon Layer on the Photocatalytic Activity of Magnetic NiFe2O4 Octahedron [J]. Chem. J. Chinese Universities, 2022, 43(11): 20220472. |
| [2] | DONG Yanhong, LU Xinhuan, YANG Lu, SUN Fanqi, DUAN Jingui, GUO Haotian, ZHANG Qinjun, ZHOU Dan, XIA Qinghua. Preparation of Bifunctional Metal-organic Framework Materials and Application in Catalytic Olefins Epoxidation [J]. Chem. J. Chinese Universities, 2022, 43(11): 20220458. |
| [3] | WANG Zumin, MENG Cheng, YU Ranbo. Doping Regulation in Transition Metal Phosphides for Hydrogen Evolution Catalysts [J]. Chem. J. Chinese Universities, 2022, 43(11): 20220544. |
| [4] | HE Jianyun, JIANG Yunbo, ZHANG Aimin, TANG Zhenyan, LI Hongpeng. Preparation and application of a novel porphyrin-based porous organic polymer COP-180 supported palladium catalyst [J]. Chem. J. Chinese Universities, 0, (): 20220535. |
| [5] | XIA Wenwen, YU Hongjing, WANG Shiye, YAO Li, LI Xiangyuan. Combustion Mechanism Construction Based On Minimized Reaction Network: Combustion of Aromatic hydrocarbon [J]. Chem. J. Chinese Universities, 0, (): 20220616. |
| [6] | LI Huaike, YUE Guichu, XIE Haiyun, LIU Jing, GAO Songwei, HOU Lanlan, LI Shuai, MIAO Beibei, WANG Nü, BAI Jie, CUI Zhimin, ZHAO Yong. Application of Electrospun Hollow Nanofibers in Catalysis [J]. Chem. J. Chinese Universities, 0, (): 20220625. |
| [7] | KUANG Huayi, CHEN Chen. Synthesis methods and electrocatalytic performance of noble-metal nanoframes catalysts [J]. Chem. J. Chinese Universities, 0, (): 20220586. |
| [8] | . Ni-La/SiO2 Catalysts Prepared by Dielectric Barrier Discharge Plasma Applying in the Dry Reforming of Methane [J]. Chem. J. Chinese Universities, 0, (): 20220503. |
| [9] | ZHU Jipeng, LIU Runhui, SONG Gonghua. Application of Bisoxazoline Grafted Amino Acid Polymer as Chiral Catalytic Center in Asymmetric Henry Reaction [J]. Chem. J. Chinese Universities, 0, (): 20220569. |
| [10] | CHENG Yuanyuan, XI Biying. Theoretical Study on the Fragmentation Mechanism of CH3SSCH3 Radical Cation Initiated by OH Radical [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220271. |
| [11] | LI Xueyu, WANG Zhao, CHEN Ya, LI Keke, LI Jianquan, JIN Shunjing, CHEN Lihua, SU Baolian. Enhanced Catalytic Performance of Supported Nano-gold by the Localized Surface Plasmon Resonance for Selective Hydrogenation of Butadiene [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220174. |
| [12] | SUN Jinshi, CHEN Peng, JING Liping, SUN Fuxing, LIU Jia. Synthesis of Hierarchical Porous Aromatic Frameworks for Immobilization of Thiourea Catalyst [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220171. |
| [13] | SONG Jiaxin, CUI Jing, FAN Xiaoqiang, KONG Lian, XIAO Xia, XIE Zean, ZHAO Zhen. Preparation of mesoporous silica supported highly dispersed vanadium catalyst and their catalytic performance for selective oxidation of ethane [J]. Chem. J. Chinese Universities, 0, (): 20220532. |
| [14] | TANG Quanjun, LIU Yingxin, MENG Rongwei, ZHANG Ruotian, LING Guowei, ZHANG Chen. Application of Single-atom Catalysis in Marine Energy [J]. Chem. J. Chinese Universities, 2022, 43(9): 20220324. |
| [15] | LIN Zhi, PENG Zhiming, HE Weiqing, SHEN Shaohua. Single-atom and Cluster Photocatalysis: Competition and Cooperation [J]. Chem. J. Chinese Universities, 2022, 43(9): 20220312. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||