

Chem. J. Chinese Universities ›› 2014, Vol. 35 ›› Issue (2): 237.doi: 10.7503/cjcu20130960
• Analytical Chemistry • Previous Articles Next Articles
PAN Liying, WANG Chengjian, YUAN Jiangbei, ZHANG Ying, HUANG Linjuan, WANG Zhongfu*( )
)
Received:2013-09-29
Online:2014-02-10
Published:2013-12-11
Contact:
WANG Zhongfu
E-mail:wangzhf@nwu.edu.cn
Supported by:CLC Number:
TrendMD:
PAN Liying, WANG Chengjian, YUAN Jiangbei, ZHANG Ying, HUANG Linjuan, WANG Zhongfu. Qualitation and Quantitation for Comparative Analysis of N-glycans from Human Hepatocellular Carcinoma HepG2 and Normal Liver Cells L02 by Electrospray Ionization Mass Spectrometry†[J]. Chem. J. Chinese Universities, 2014, 35(2): 237.
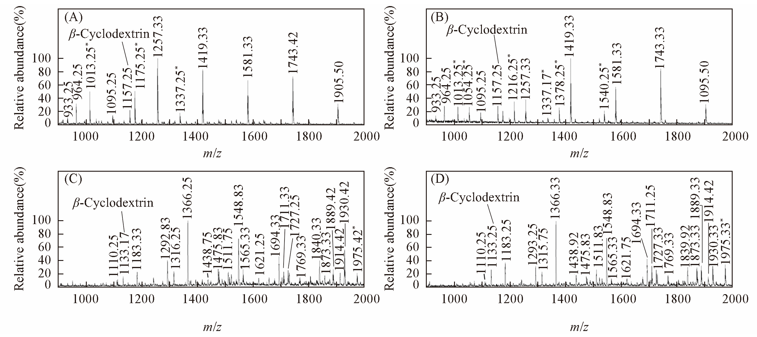
Fig.1 ESI-MS profiles of N-glycans drived HepG2(A, C) and L02 cells(B, D) under positive(A, B) and negative ion modes(C, D) (A, B) Eluted with 15% ACN;(C, D) eluted with 25% ACN. * Impurities.
| Glycan type | m/z | Monosaccharide composition | Peak intensity ratio of N-glycan to β-cyclodextrin (mean±SDa) | CV(%) | Relative ratio of N-glycans (HepG2 to L02) | Proposed structure | ||
|---|---|---|---|---|---|---|---|---|
| HepG2 (n=3) | L02 (n=3) | HepG2 (n=3) | L02 (n=3) | |||||
| High-Mannose | 933.25b | H3N2 | 0.526±0.023 | 0.512±0.038 | 4.42 | 7.55 | 1.028 | |
| 1095.33b | H4N2 | 0.673±0.059 | 0.680±0.036 | 8.81 | 5.33 | 0.990 | ||
| 1257.33b | H5N2 | 4.694±0.262 | 1.349±0.020 | 5.60 | 1.49 | 3.478 | ||
| 1419.33b | H6N2 | 4.011±0.241 | 3.755±0.233 | 6.01 | 6.21 | 1.068 | ||
| 1581.33b | H7N2 | 3.151±0.241 | 2.108±0.113 | 7.66 | 5.39 | 1.495 | ||
| 1743.33b | H8N2 | 3.547±0.278 | 3.127±0.136 | 7.86 | 4.37 | 1.134 | ||
| 1905.50b | H9N2 | 1.539±0.150 | 1.122±0.059 | 9.75 | 5.26 | 1.371 | ||
| 964.17c | 1.403±0.053 | 1.037±0.119 | 3.79 | 11.47 | 1.352 | |||
| Complex/hybrid | 1110.25(-)d | H5N4A2 | 0.611±0.070 | 0.333±0.013 | 11.51 | 4.11 | 1.834 | |
| (sialylated) | 1293.25(-)d | H6N5A2 | 2.597±0.102 | 1.108±0.181 | 3.94 | 16.38 | 2.344 | |
| 1315.75(-)e | 1.445±0.118 | 0.838±0.080 | 8.22 | 9.56 | 1.724 | |||
| 1438.75(-)d | H6N5A3 | 0.724±0.103 | 0.711±0.043 | 14.23 | 6.05 | 1.019 | ||
| 1475.83(-)d | H7N6A2 | 1.779±0.176 | 0.657±0.041 | 9.94 | 6.25 | 2.705 | ||
| 1565.33(-)f | H4N3A1 | 1.559±0.109 | 0.564±0.055 | 7.03 | 9.82 | 2.763 | ||
| 1621.33(-)d | H7N6A3 | 0.905±0.058 | 0.547±0.049 | 6.48 | 9.11 | 1.653 | ||
| 1727.25(-)f | H5N3A1 | 1.533±0.125 | 1.109±0.102 | 8.18 | 9.26 | 1.382 | ||
| 1769.33(-)f | H4N4A1 | 0.849±0.083 | 0.731±0.087 | 9.80 | 12.00 | 1.162 | ||
| 1889.33(-)f | H6N3A1 | 1.624±0.097 | 1.378±0.126 | 6.02 | 9.15 | 1.179 | ||
| 1930.33(-)f | H5N4A1 | 3.355±0.296 | 1.294±0.073 | 8.83 | 5.70 | 2.592 | ||
| Complex/hybrid | 1183.33(-)d | H5N4A2F1 | 1.390±0.042 | 1.414±0.115 | 3.07 | 8.15 | 0.984 | |
| (fucosylated and | 1366.33(-)d | H6N5A2F1 | 6.244±0.478 | 3.798±0.036 | 7.66 | 0.95 | 1.644 | |
| sialylated) | 1511.75(-)d | H6N5A3F1 | 1.349±0.231 | 1.013±0.071 | 17.12 | 7.09 | 1.331 | |
| 1548.83(-)d | H7N6A2F1 | 4.285±0.453 | 2.937±0.141 | 10.58 | 4.80 | 1.459 | ||
| 1694.33(-)d | H7N6A3F1 | 3.071±0.205 | 1.721±0.093 | 6.68 | 5.44 | 1.785 | ||
| 1711.33(-)f | H4N3A1F1 | 1.301±0.194 | 1.264±0.110 | 14.93 | 8.73 | 1.029 | ||
| 1840.42(-)d | H7N6A4F1 | 2.575±0.170 | 1.202±0.077 | 6.61 | 6.48 | 2.142 | ||
| 1873.33(-)f | H5N3A1F1 | 0.551±0.049 | 1.289±0.127 | 9.03 | 9.89 | 0.427 | ||
| 1914.42(-)f | H4N4A1F1 | 1.265±0.174 | 0.635±0.024 | 13.78 | 3.78 | 1.990 | ||
Table 1 Compositions and proposed structures and the repeatability of β-cyclodextrin as inter standard of N-glycans from HepG2 and L02 cells
| Glycan type | m/z | Monosaccharide composition | Peak intensity ratio of N-glycan to β-cyclodextrin (mean±SDa) | CV(%) | Relative ratio of N-glycans (HepG2 to L02) | Proposed structure | ||
|---|---|---|---|---|---|---|---|---|
| HepG2 (n=3) | L02 (n=3) | HepG2 (n=3) | L02 (n=3) | |||||
| High-Mannose | 933.25b | H3N2 | 0.526±0.023 | 0.512±0.038 | 4.42 | 7.55 | 1.028 | |
| 1095.33b | H4N2 | 0.673±0.059 | 0.680±0.036 | 8.81 | 5.33 | 0.990 | ||
| 1257.33b | H5N2 | 4.694±0.262 | 1.349±0.020 | 5.60 | 1.49 | 3.478 | ||
| 1419.33b | H6N2 | 4.011±0.241 | 3.755±0.233 | 6.01 | 6.21 | 1.068 | ||
| 1581.33b | H7N2 | 3.151±0.241 | 2.108±0.113 | 7.66 | 5.39 | 1.495 | ||
| 1743.33b | H8N2 | 3.547±0.278 | 3.127±0.136 | 7.86 | 4.37 | 1.134 | ||
| 1905.50b | H9N2 | 1.539±0.150 | 1.122±0.059 | 9.75 | 5.26 | 1.371 | ||
| 964.17c | 1.403±0.053 | 1.037±0.119 | 3.79 | 11.47 | 1.352 | |||
| Complex/hybrid | 1110.25(-)d | H5N4A2 | 0.611±0.070 | 0.333±0.013 | 11.51 | 4.11 | 1.834 | |
| (sialylated) | 1293.25(-)d | H6N5A2 | 2.597±0.102 | 1.108±0.181 | 3.94 | 16.38 | 2.344 | |
| 1315.75(-)e | 1.445±0.118 | 0.838±0.080 | 8.22 | 9.56 | 1.724 | |||
| 1438.75(-)d | H6N5A3 | 0.724±0.103 | 0.711±0.043 | 14.23 | 6.05 | 1.019 | ||
| 1475.83(-)d | H7N6A2 | 1.779±0.176 | 0.657±0.041 | 9.94 | 6.25 | 2.705 | ||
| 1565.33(-)f | H4N3A1 | 1.559±0.109 | 0.564±0.055 | 7.03 | 9.82 | 2.763 | ||
| 1621.33(-)d | H7N6A3 | 0.905±0.058 | 0.547±0.049 | 6.48 | 9.11 | 1.653 | ||
| 1727.25(-)f | H5N3A1 | 1.533±0.125 | 1.109±0.102 | 8.18 | 9.26 | 1.382 | ||
| 1769.33(-)f | H4N4A1 | 0.849±0.083 | 0.731±0.087 | 9.80 | 12.00 | 1.162 | ||
| 1889.33(-)f | H6N3A1 | 1.624±0.097 | 1.378±0.126 | 6.02 | 9.15 | 1.179 | ||
| 1930.33(-)f | H5N4A1 | 3.355±0.296 | 1.294±0.073 | 8.83 | 5.70 | 2.592 | ||
| Complex/hybrid | 1183.33(-)d | H5N4A2F1 | 1.390±0.042 | 1.414±0.115 | 3.07 | 8.15 | 0.984 | |
| (fucosylated and | 1366.33(-)d | H6N5A2F1 | 6.244±0.478 | 3.798±0.036 | 7.66 | 0.95 | 1.644 | |
| sialylated) | 1511.75(-)d | H6N5A3F1 | 1.349±0.231 | 1.013±0.071 | 17.12 | 7.09 | 1.331 | |
| 1548.83(-)d | H7N6A2F1 | 4.285±0.453 | 2.937±0.141 | 10.58 | 4.80 | 1.459 | ||
| 1694.33(-)d | H7N6A3F1 | 3.071±0.205 | 1.721±0.093 | 6.68 | 5.44 | 1.785 | ||
| 1711.33(-)f | H4N3A1F1 | 1.301±0.194 | 1.264±0.110 | 14.93 | 8.73 | 1.029 | ||
| 1840.42(-)d | H7N6A4F1 | 2.575±0.170 | 1.202±0.077 | 6.61 | 6.48 | 2.142 | ||
| 1873.33(-)f | H5N3A1F1 | 0.551±0.049 | 1.289±0.127 | 9.03 | 9.89 | 0.427 | ||
| 1914.42(-)f | H4N4A1F1 | 1.265±0.174 | 0.635±0.024 | 13.78 | 3.78 | 1.990 | ||
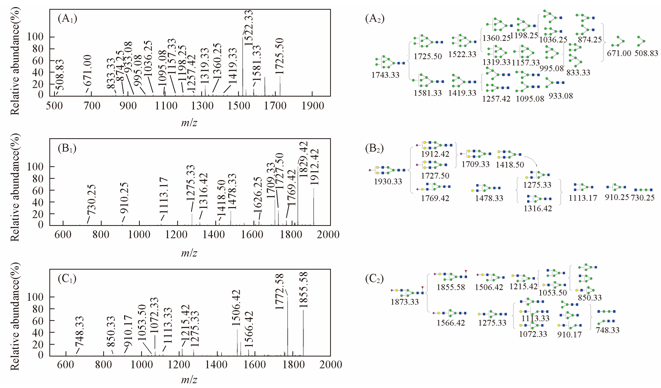
Fig.2 MS/MS spectra of N-glycans(A) MS/MS spectra of glycan with m/z 1743.33[M+Na]+ in the positive-ion mode; (B) MS/MS spectra of glycan with m/z 1930.33[M-H]- in the negative-ion mode; (C) MS/MS spectra of glycan with m/z 1873.33[M-H]- in the negative-ion mode.
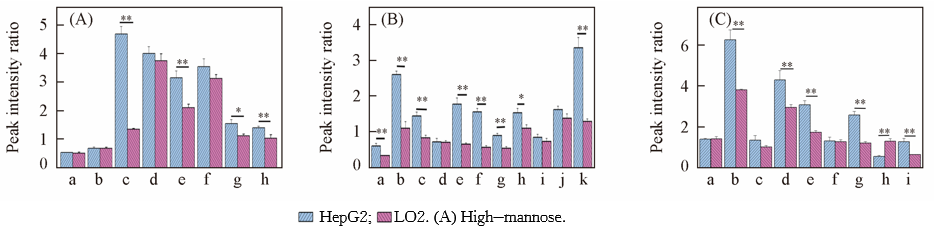
Fig.3 Peak intensity ratio of N-glycans to β-cyclodextrin from HepG2 and L02a. H3N2; b. H4N2; c. H5N2; d. H6N2; e. H7N2; f. H8N2; g. H9N2([M+Na]+); h. H9N2([M+2Na]2+). (B) Complex/hybrid(sialylated). a. H5N4A2; b. H6N5A2; c. H6N5A2; d. H6N5A3; e. H7N6A2; f. H4N3A1; g. H7N6A3; h. H5N3A1; i. H4N4A1; j. H6N3A1; k. H5N4A1. (C) Complex/hybrid(fucosylated and sialylated). a. H5N4A2F1; b. H6N5A2F1; c. H6N5A3F1; d. H7N6A2F1; e. H7N6A3F1; f. H4N3A1F1; g. H7N6A4F1; h. H5N3A1F1; i. H4N4A1F1. ** p<0.01; * p<0.05; H: hexose; N: N-acetyl hexosamine; F: fucose; A: sialic acaid.
| [1] | Apweiler R., Hermjakob H., Sharon N., Biochimica et Biophysica Acta(BBA)-General Subjects, 1999, 1473(1), 4—8 |
| [2] | Furukawa K., Kobata A., Current Opinion in Biotechnology, 1992, 3(5), 554—559 |
| [3] | Ohtsubo K., Marth J. D., Cell, 2006, 126(5), 855—867 |
| [4] | Parodi A. J., Annual Review of Biochemistry, 2000, 69(1), 69—93 |
| [5] | Helenius A., Aebi M., Science, 2001, 291(5512), 2364—2369 |
| [6] | Isailovic D., Kurulugama R. T., Plasencia M. D., Stokes S. T., Kyselova Z. K., Goldman R., Mechref Y., Novotny M. V., Clemmer D. E., J. Proteome Res., 2008, 7(3), 1109—1117 |
| [7] | Bai C., Wu G., Zhao L., Science China Chemistry, 2010, 53(4), 807—811 |
| [8] | Skolnick A. A., The Journal of the American Medical Association, 1996, 276(18), 1458—1459 |
| [9] | Marrero J. A., Clinics in Liver Disease, 2005, 9(2), 235—251 |
| [10] | Gupta S., Bent S., Kohlwes J., Annals of Internal Medicine, 2003, 139(1), 46—50 |
| [11] | Goldman R., Ressom H. W., Varghese R. S., Goldman L., Bascug G., Loffredo C. A., Abdel-Hamid M., Gouda I., Ezzat S., Kyselova Z., Mechref Y., Novotny M. V., Clinical Cancer Research, 2009, 15(5), 1808—1813 |
| [12] | Xu N. S., Yang H. M., Cui M., Song F. R., Liu Z. Q., Liu S. Y., Chem. J. Chinese Universities, 2012, 33(11), 2430—2434 |
| (徐牛生, 杨洪梅, 崔勐, 宋凤瑞, 刘志强, 刘淑莹.高等学校化学学报,2012, 33(11), 2430—2434) | |
| [13] | Orvisky E., Drake S. K., Martin B. M., Abdel-Hamid M., Ressom H. W., Varghese R. S., An Y., Saha D., Hortin G. L., Loffredo C. A., Goldman R., Proteomics, 2006, 6(9), 2895—2902 |
| [14] | Ressom H. W., Varghese R. S., Goldman L., An Y., Loffredo C. A., Abdel-Hamid M., Kyselova Z., Mechref Y., Novotny M., Drake S. K., Goldman R., J. Proteome Res., 2008, 7(2), 603—610 |
| [15] | Shimizu Y., Nakata., Kuroda Y., Tsutsumi F., Kojima N., Mizuochi T., Carbohydrate Research, 2001, 332, 381—388 |
| [16] | Packer N. H., Lawson M. A., Jardine D. R., Redmond J. W., Glycoconjugate Journal, 1998, 15(8), 737—747 |
| [17] | de Leoz M. L. A., An H. J., Kronewitter S., Kim J., Beecroft S., Vinall R., Lebrilla C., Disease Markers, 2008, 25(4), 243—258 |
| [18] | Hua S., Williams C. C., Dimapasoc L. M., Ro G. S., Ozcan S., Miyamoto S., Lebrilla C. B., An H. J., Leiserowitz G. S., J. Chromatogr. A, 2013, 1279, 58—67 |
| [29] | Kudo T., Nakagawa H., Takahashi M., Hamaguchi J., Kamiyama N., Yokoo H., Nakanishi K., Nakagawa T., Kamiyama T., Deguchi K., Nishimura S. I., Todo S., Molecular Cancer, 2007, 6(1), 32 |
| [20] | Amano M., Yamaguchi M., Takegawa Y., Yamashita T., Terashima M., Furukawa J., Miura Y., Shinohara Y., Iwasaki N., Minami A., Nishimura S. I., Molecular & Cellular Proteomics, 2010, 9(3), 523—537 |
| [21] | Wang C., Fan W., Zhang P., Wang Z., Huang L., Proteomics, 2011, 11(21), 4229—4242 |
| [22] | Nakagawa T., Moriwaki K., Terao N., Nakagawa T., Miyamoto Y., Kamada Y., Miyoshi E., J. Proteome Res., 2012, 11(5), 2798—2806 |
| [23] | Dennis J. W., Granovsky M., Warren C. E., Biochimica et Biophysica Acta(BBA)-General Subjects, 1999, 1473(1): 21—34 |
| [24] | Taylor M.E., Drickamer K.; Translated by Ma Y. J., Introduction to Glycobiology, Chemical Industry Press, Beijing, 2006, 193—195 |
| (Taylor M.E., Drickamer K., 马毓甲[译]. 糖生物学导论, 北京: 化学工业出版社, 2006, 193—195) |
| [1] | DENG Jiewei, YANG Yunyun, LIN Li, LUAN Tiangang. Rapid Classification of Daphniamagna and Daphnia pulex by Surface-coated Probe Nanoelectrospray Ionization Mass Spectrometry Lipidomics [J]. Chem. J. Chinese Universities, 2020, 41(9): 2011. |
| [2] | LI Xiaoqian, ZHANG Hua, LU Haijian, LIU Chang, LIU Qinglong, MA Xiayu, FANG Yuanping, LIANG Dapeng. Mechanism of Photocatalytic Degradation of Rhodamine B by TiO2 Nanowire Array with Internal Extraction Electrospray Ionization Mass Spectrometry [J]. Chem. J. Chinese Universities, 2020, 41(9): 2003. |
| [3] | WANG Xiaoqun, HUANG Guangming. Investigation of Lipid Distributions by Trifluoroacetic Acid-enhanced Desorption Electrospray Ionization Mass Spectrometry Imaging [J]. Chem. J. Chinese Universities, 2020, 41(12): 2673. |
| [4] | LI Cheng,WANG Chengjian,JIN Wanjun,HAN Jianli,YANG Meifang,GAO Xi,HUANG Linjuan,WANG Zhongfu. Mass Spectrometric Analysis of N-Glycans of Glycoprotein Separated by SDS-PAGE Gel from Ginkgo Seed† [J]. Chem. J. Chinese Universities, 2019, 40(1): 69. |
| [5] | WU Fangling,CHU Yanqiu,CHEN Xin,WEI Wanghui,DING Chuanfan. Critical Factors Affecting Noncovalent Interaction Between Pentapeptides Explored by Electrospray Ionization Mass Spectrometry† [J]. Chem. J. Chinese Universities, 2018, 39(9): 1927. |
| [6] | KE Mufang, HAN Jing, ZHU Tenggao, LIU Wenjie, ZHANG Hua, KOU Wei, LIANG Dapeng. Direct and Rapid Analysis of Avocado Using Internal Extractive Electrospray Ionization Mass Spectrometry† [J]. Chem. J. Chinese Universities, 2017, 38(5): 738. |
| [7] | HUANG Yandong, WU Ruofei, CHU Yanqiu, DING Chuanfan. Effect of Side Chain of α-Amino Acids and Esters on the Stability Constants for β-Cyclodextrin Complexes† [J]. Chem. J. Chinese Universities, 2017, 38(5): 743. |
| [8] | PENG Yifang, WANG Chengjian, WANG Jingjing, LI Lingmei, JIN Wanjun, QIANG Shan, SHI Hongdan, ZHANG Ying, HUANG Linjuan, WANG Zhongfu. Analysis of Antigenic Determinant Glycans of Peanut Allergy Glycoprotein Ara h1 by Mass Spectrometry† [J]. Chem. J. Chinese Universities, 2016, 37(9): 1622. |
| [9] | LI Penghui, DENG Lingli, LUO Jiao, LI Wei, NING Jing, DING Jianhua, WU Xiaoping. EESI-MS Detection and Statistical Analysis of Multi-batch of Exhaled Breath Metabolomics Data of Liver Failure Patients [J]. Chem. J. Chinese Universities, 2016, 37(4): 626. |
| [10] | WU Ruofei, CHU Yanqiu, XU Chongsheng, LIU Zhipan, DING Chuanfan. Fragmentation Reaction of Complexes of Alkali Metal Cations with Tripeptides in Gas Phase† [J]. Chem. J. Chinese Universities, 2016, 37(12): 2150. |
| [11] | CHEN Huanhuan, ZHAO Xia, LUAN Xiaohong, YU Guangli. Application of Electrospray Tandem Mass Spectrometry in Sequence Analysis of Oligosaccharides† [J]. Chem. J. Chinese Universities, 2015, 36(1): 1. |
| [12] | DAI Yulin, YU Shanshan, ZHANG Ying, HAO Ying, ZHONG Wei, YUE Hao, LIU Shuying. Studies on the Isoflavone in Extract of the Flower of Pueraria Lobata by RRLC-Q-TOF MS/MS† [J]. Chem. J. Chinese Universities, 2014, 35(7): 1396. |
| [13] | FANG Feifei, DU Shangguang, DAI Ximo, GUO Xiali, CHEN Huanwen, LUO Liping. Rapid Analysis Alkaloids in Lotus Seeds by Extractive Electrospray Ionization Mass Spectrometry† [J]. Chem. J. Chinese Universities, 2014, 35(4): 730. |
| [14] | XU Niu-Sheng, YANG Hong-Mei, CUI Meng, SONG Feng-Rui, LIU Zhi-Qiang, LIU Shu-Ying. Investigation of Interaction of Fangchinoline with G-quadruplex DNA by Electrospray Ionization Mass Spectrometry [J]. Chem. J. Chinese Universities, 2012, 33(11): 2430. |
| [15] | LIU Zhen-Shuang, GAO Shang, ZHAO Wai-Ou, QIN Yu-Jiao, WANG Bing, GUO Xin-Hua. Recognition of Flavonoids Towards to DNA G-quadruplexes and Duplexes by ESI-MS [J]. Chem. J. Chinese Universities, 2012, 33(08): 1681. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||