

Chem. J. Chinese Universities ›› 2014, Vol. 35 ›› Issue (2): 314.doi: 10.7503/cjcu20130613
• Organic Chemistry • Previous Articles Next Articles
YANG Gang1, ZHU Liangliang2, LÜ Xia2, WU Dachang1, XIA Yangliu2, MA Xiaochi1, XIN Yi1, HOU Jie1,*( )
)
Received:2013-07-04
Online:2014-02-10
Published:2014-01-02
Contact:
HOU Jie
E-mail:houjie@nankai.edu.cn
Supported by:TrendMD:
YANG Gang, ZHU Liangliang, LÜ Xia, WU Dachang, XIA Yangliu, MA Xiaochi, XIN Yi, HOU Jie. Highly Efficient Preparation of Bisphenol A Glucuronide†[J]. Chem. J. Chinese Universities, 2014, 35(2): 314.
| No. | Substrate concentrationa/(mmol·L-1) | RLM concentrationb/(mg·mL-1) | Incubation timec/h |
|---|---|---|---|
| 1 | 0.5 | 0.2 | 1 |
| 2 | 1.0 | 0.4 | 2 |
| 3 | 1.5 | 0.6 | 4 |
Table 1 Factors and levels in orthogonal array design for reaction optimization
| No. | Substrate concentrationa/(mmol·L-1) | RLM concentrationb/(mg·mL-1) | Incubation timec/h |
|---|---|---|---|
| 1 | 0.5 | 0.2 | 1 |
| 2 | 1.0 | 0.4 | 2 |
| 3 | 1.5 | 0.6 | 4 |
| No. | A | B | C | D(Blank) | Experiment scheme |
|---|---|---|---|---|---|
| 1 | 1 | 1 | 1 | 1 | A1B1C1 |
| 2 | 1 | 2 | 2 | 2 | A1B2C2 |
| 3 | 1 | 3 | 3 | 3 | A1B3C3 |
| 4 | 2 | 1 | 2 | 3 | A2B1C2 |
| 5 | 2 | 2 | 3 | 1 | A2B2C3 |
| 6 | 2 | 3 | 1 | 2 | A2B3C1 |
| 7 | 3 | 1 | 3 | 2 | A3B1C3 |
| 8 | 3 | 2 | 1 | 3 | A3B2C1 |
| 9 | 3 | 3 | 2 | 1 | A3B3C2 |
Table 2 Orthogonal assay for reaction optimization
| No. | A | B | C | D(Blank) | Experiment scheme |
|---|---|---|---|---|---|
| 1 | 1 | 1 | 1 | 1 | A1B1C1 |
| 2 | 1 | 2 | 2 | 2 | A1B2C2 |
| 3 | 1 | 3 | 3 | 3 | A1B3C3 |
| 4 | 2 | 1 | 2 | 3 | A2B1C2 |
| 5 | 2 | 2 | 3 | 1 | A2B2C3 |
| 6 | 2 | 3 | 1 | 2 | A2B3C1 |
| 7 | 3 | 1 | 3 | 2 | A3B1C3 |
| 8 | 3 | 2 | 1 | 3 | A3B2C1 |
| 9 | 3 | 3 | 2 | 1 | A3B3C2 |
| No. | A | B | C | D(Blank) | Conversion(%) |
|---|---|---|---|---|---|
| 1 | 1 | 1 | 1 | 1 | 92.7 |
| 2 | 1 | 2 | 2 | 2 | 94.4 |
| 3 | 1 | 3 | 3 | 3 | 91.8 |
| 4 | 2 | 1 | 2 | 3 | 98.9 |
| 5 | 2 | 2 | 3 | 1 | 96.2 |
| 6 | 2 | 3 | 1 | 2 | 85.1 |
| 7 | 3 | 1 | 3 | 2 | 84.5 |
| 8 | 3 | 2 | 1 | 3 | 72.2 |
| 9 | 3 | 3 | 2 | 1 | 89.3 |
Table 3 Orthogonal assay results for reaction optimization
| No. | A | B | C | D(Blank) | Conversion(%) |
|---|---|---|---|---|---|
| 1 | 1 | 1 | 1 | 1 | 92.7 |
| 2 | 1 | 2 | 2 | 2 | 94.4 |
| 3 | 1 | 3 | 3 | 3 | 91.8 |
| 4 | 2 | 1 | 2 | 3 | 98.9 |
| 5 | 2 | 2 | 3 | 1 | 96.2 |
| 6 | 2 | 3 | 1 | 2 | 85.1 |
| 7 | 3 | 1 | 3 | 2 | 84.5 |
| 8 | 3 | 2 | 1 | 3 | 72.2 |
| 9 | 3 | 3 | 2 | 1 | 89.3 |
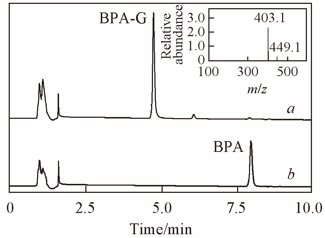
Fig.1 UFLC-MS of SPE purification with RLM(a) and without RLM(b) BPA was incubated in appropriate incubation buffer system at 37 ℃ for 6 h. Inset the mass spectrum of BPA-G.
| No. | SPE recovery rate*(%) | BPA-G yield(%) |
|---|---|---|
| 1 | 95.4 | 94.4 |
| 2 | 98.5 | 97.4 |
| 3 | 97.2 | 96.1 |
Table 4 Recovery rates of the sample
| No. | SPE recovery rate*(%) | BPA-G yield(%) |
|---|---|---|
| 1 | 95.4 | 94.4 |
| 2 | 98.5 | 97.4 |
| 3 | 97.2 | 96.1 |
| Position | BPA | BPA-G | ||
|---|---|---|---|---|
| δH(Number of protons) | δC | δH (Number of protons) | δC | |
| 1 | 40.8 | 40.6 | ||
| 2, 2' | 1.53(6H) | 30.7 | 1.52—1.3(6H) | 30.2 |
| A1, B1 | 140.9 | 140.5, 141.3 | ||
| A2, A6 | 6.65—6.98(8H) | 127.1 | 6.68—7(8H) | 126.9 |
| B2, B6 | ||||
| A3, A5 | 114.4 | 114.5 | ||
| B3, B5 | 116.1 | |||
| A4, B4 | 154.7 | 154.8, 155.5 | ||
| G1 | 5.02(1H, 7.8, d) | 101.9 | ||
| G2 | 3.47(1H, m) | 73.4 | ||
| G3 | 3.31(1H, m) | 76.3 | ||
| G4 | 3.46(1H, m) | 71.8 | ||
| G5 | 4.01(1H, m) | 76.9 | ||
| G6 | 171.7 | |||
Table 5 1H NMR and 13C NMR data for BPA and BPA-G*
| Position | BPA | BPA-G | ||
|---|---|---|---|---|
| δH(Number of protons) | δC | δH (Number of protons) | δC | |
| 1 | 40.8 | 40.6 | ||
| 2, 2' | 1.53(6H) | 30.7 | 1.52—1.3(6H) | 30.2 |
| A1, B1 | 140.9 | 140.5, 141.3 | ||
| A2, A6 | 6.65—6.98(8H) | 127.1 | 6.68—7(8H) | 126.9 |
| B2, B6 | ||||
| A3, A5 | 114.4 | 114.5 | ||
| B3, B5 | 116.1 | |||
| A4, B4 | 154.7 | 154.8, 155.5 | ||
| G1 | 5.02(1H, 7.8, d) | 101.9 | ||
| G2 | 3.47(1H, m) | 73.4 | ||
| G3 | 3.31(1H, m) | 76.3 | ||
| G4 | 3.46(1H, m) | 71.8 | ||
| G5 | 4.01(1H, m) | 76.9 | ||
| G6 | 171.7 | |||
| [1] | Wu J., Wei W., Yang N. Y., Shen X. Y., Tsuji I., Yamamura T., Li J., Li X. M., Chem. Res. Chinese Universities, 2013, 29(3), 512—518 |
| [2] | Hu P., Xiong Z. H., Liu J. M., Chem. J. Chinese Universities, 2012, 33(9), 2111—2116 |
| (胡品, 熊振湖, 刘建明.高等学校化学学报,2012, 33(9), 2111—2116) | |
| [3] | Roy A., Bauer S., Lawrence B. P., PloS One, 2012, 7(6), e38448-1—e38448-12 |
| [4] | Deng M. X., Wu D. S., Zhan L., Journal of Environment and Health, 2011, 18(3), 134—136 |
| (邓茂先, 吴德生, 詹立.环境与健康杂志,2011, 18(3), 134—136) | |
| [5] | Ayyanan A., Laribi O., Schuepbach-Mallepell S., Schrick C., Gutierrez M., Tanos T., Lefebvre G., Rougemont J., Yalcin-Ozuysal O., Brisken C. P., Molecular Endocrinol., 2011, 25(11), 1915—1923 |
| [6] | Golub M. S., Wu K. L., Kaufman F. L., Li L. H., Moran-Messen F., Zeise L., Alexeeff G. V., Donald J. M., Birth Defects Res. B: Development Reproductive Toxicology, 2010, 89(6), 441—466 |
| [7] | Inoue H., Tsuruta A., Kudo S., Ishii T., Fukushima Y., Iwano H., Yokota H., Kato S., Drug Metab Dispos., 2004, 33(1), 55—59 |
| [8] | Genius S. J., Beesoon S. B., Birkholz D., Lobo R. A., J. Environ. Public Health, 2012, 2012, 185731-1—185731-10 |
| [9] | Inoue H., Yokota H., Makino T., Yuasa A., Kato S., Drug Metab. Dispos., 2001, 29(8), 1084—1087 |
| [10] | Arakawa C., Fujimaki K., Yoshinaga J., Imai H., Serizawa S., Shiraishi H., Environ. Health Prev. Med., 2004, 9, 22—26 |
| [11] | Silver M. K., O’Neill M. S., Sowers M. R., Park S. K., PloS One, 2011, 6(10), e26868-1—e26868-9 |
| [12] | Zeng J., Kuang H., Hu C., Shi X., Yan M., Xu L., Wang L., Xu C., Xu G., Environ. Sci. Technol., 2013, 47(13), 7457—7465 |
| [13] | Court M. H., Zhang X., Ding X., Yee K. K., Hesse L. M., Finel M., Xenobiotica, 2012, 42(3), 266—277 |
| [14] | Harbourt D. E., Fallon J. K., Ito S., Baba T., Ritter J. K., Glish G. L., Smith P. C., Anal. Chem., 2012, 84(1), 98—105 |
| [15] | Pallmann T., Jonas U., Wagner M., Thevis M., Kaeferstein H., Rothschild M. A., Bender K., Eur. J. Pharm. Sci., 2010, 39(4), 233—240 |
| [16] | Liang S. C., Ge G. B., Liu H. X., Zhang Y. Y., Zhang J. W., Yin L., Li W., Fang Z. Z., Wang L. M., Yang L., Drug Metab. Dispos., 2010, 38(6), 973—980 |
| [17] | Zhu L. L., Ge G. B., Zhang H. B., Liu H. X., He G. Y., Liang S. C., Zhang Y. Y., Fang Z. Z., Dong P. P., Finel M., Drug Metab. Dispos., 2012, 40(3), 529—538 |
| [18] | Zhu L., Ge G. B., Liu Y., Guo Z., Peng C., Zhang F., Cao Y., Wu J., Fang Z., Liang X., Chem. Res. Toxicol., 2012, 25(12), 2663—2669 |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||