

高等学校化学学报 ›› 2018, Vol. 39 ›› Issue (9): 1993.doi: 10.7503/cjcu20180169
收稿日期:2018-03-04
出版日期:2018-09-07
发布日期:2018-06-11
作者简介:联系人简介: 罗永春, 男, 博士, 教授, 主要从事储氢材料和电化学应用研究. E-mail:
基金资助:
ZHAO Lei1, LUO Yongchun1,2,*( ), DENG Anqiang1, JIANG Wanting1
), DENG Anqiang1, JIANG Wanting1
Received:2018-03-04
Online:2018-09-07
Published:2018-06-11
Contact:
LUO Yongchun
E-mail:luoyc@lut.cn
Supported by:摘要:
采用真空电弧熔炼和热处理(950 ℃×10 h)方法制备了新型无镁超点阵结构A2B7型La1-xYxNi3.25Mn0.15Al0.1(x=0, 0.25, 0.50, 0.67, 0.75, 0.85, 1.00)退火合金, 研究了A端稀土Y元素对退火合金微观组织结构、 储氢行为及电化学性能的影响. 结果表明, 退火合金微观组织的主相均由Ce2Ni7型结构组成, 随稀土Y含量x增大, Ce2Ni7型主相丰度呈先增加后减小的规律, 同时Ce2Ni7型主相的晶胞体积V逐渐减小. 气体储氢时, x=0~0.25合金无压力-组成-温度(PCT)曲线平台且易形成氢致非晶化; 当x≥0.50时, 合金能有效抑制储氢时的氢致非晶化倾向且具有明显的吸/放氢平台特征, 吸氢平台压范围为0.026~0.097 MPa, 最大储氢量为1.418%~1.48%(质量分数), 储氢性能得到极大改善. 电化学测试结果表明, x=0.50~0.85的合金具有较高的电化学放电容量(350.4~381 mA·h/g), 经100次充放电循环后容量保持率S100=52%~85%, 其中稀土Y含量x=0.67~0.75时的合金具有良好的储氢性能及较好的综合电化学性能. 合金电极的高倍率放电性能HRD900=64.5%~85.7%, 氢原子在合金体相中的扩散是电极反应动力学过程的控制步骤.
中图分类号:
TrendMD:
赵磊, 罗永春, 邓安强, 姜婉婷. 无镁超点阵结构A2B7型La1-xYxNi3.25Mn0.15Al0.1合金的储氢和电化学性能. 高等学校化学学报, 2018, 39(9): 1993.
ZHAO Lei,LUO Yongchun,DENG Anqiang,JIANG Wanting. Hydrogen Storage and Electrochemical Properties of the Mg-free A2B7-type La1-xYxNi3.25Mn0.15Al0.1 Alloys with Superlattice Structure†. Chem. J. Chinese Universities, 2018, 39(9): 1993.

Fig.1 Back scattered SEM images of the annealed La1-xYxNi3.25Mn0.15Al0.1(x=0—1) alloys (A) x=0; (B) x=0.25; (C) x=0.50; (D) x=0.67; (E) x=0.75; (F) x=0.85; (G) x=1.00.
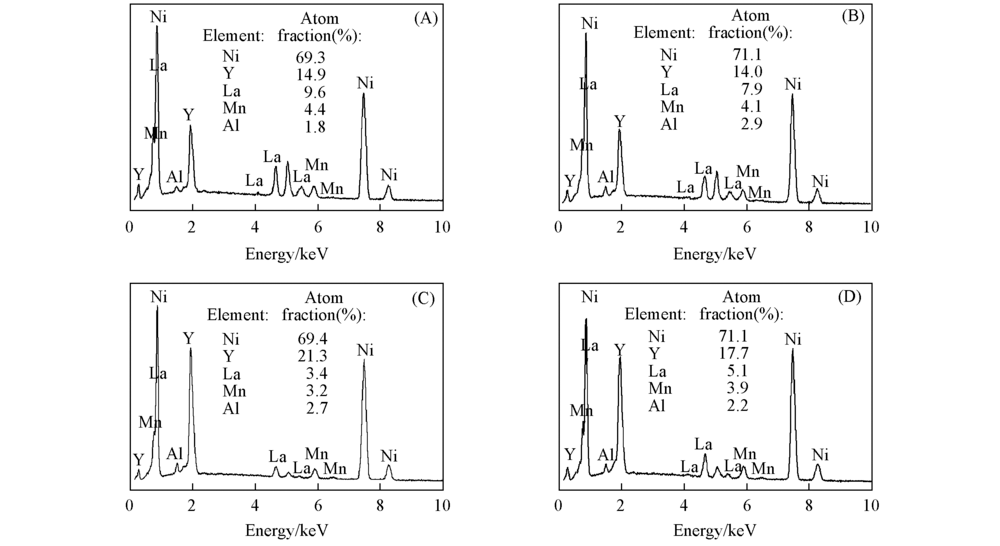
Fig.2 EDS results for different regions of the annealed La1-xYxNi3.25Mn0.15Al0.1 alloys(x=0.5, 0.85) in Fig.1 (A) Area 1 of x=0.50; (B) area 2 of x=0.50; (C) area 3 of x=0.85; (D) area 4 of x=0.85.
| x | Normal composition | Chemical compositions by ICP, w(%) | Stoichiometric B/A ratio | ||||
|---|---|---|---|---|---|---|---|
| La | Y | Ni | Mn | Al | |||
| 0 | LaNi3.25Mn0.15Al0.1 | 40.80 | 0 | 54.42 | 3.14 | 1.64 | 3.52 |
| 0.25 | La0.75Y0.25Ni3.25Mn0.15Al0.1 | 31.01 | 6.48 | 58.67 | 2.93 | 0.91 | 3.56 |
| 0.50 | La0.5Y0.5Ni3.25Mn0.15Al0.1 | 21.67 | 14.10 | 60.53 | 2.66 | 0.94 | 3.54 |
| 0.67 | La0.33Y0.67Ni3.25Mn0.15Al0.1 | 14.49 | 19.35 | 62.74 | 2.56 | 0.87 | 3.56 |
| 0.75 | La0.25Y0.75Ni3.25Mn0.15Al0.1 | 10.84 | 22.35 | 63.34 | 2.54 | 0.92 | 3.45 |
| 0.85 | La0.15Y0.85Ni3.25Mn0.15Al0.1 | 6.28 | 25.70 | 64.66 | 2.41 | 0.94 | 3.53 |
| 1.00 | YNi3.25Mn0.15Al0.1 | 0 | 31.51 | 65.14 | 2.71 | 0.93 | 3.44 |
Table 1 Chemical compositions of the annealed alloys La1-xYxNi3.25Mn0.15Al0.1 by ICP analysis
| x | Normal composition | Chemical compositions by ICP, w(%) | Stoichiometric B/A ratio | ||||
|---|---|---|---|---|---|---|---|
| La | Y | Ni | Mn | Al | |||
| 0 | LaNi3.25Mn0.15Al0.1 | 40.80 | 0 | 54.42 | 3.14 | 1.64 | 3.52 |
| 0.25 | La0.75Y0.25Ni3.25Mn0.15Al0.1 | 31.01 | 6.48 | 58.67 | 2.93 | 0.91 | 3.56 |
| 0.50 | La0.5Y0.5Ni3.25Mn0.15Al0.1 | 21.67 | 14.10 | 60.53 | 2.66 | 0.94 | 3.54 |
| 0.67 | La0.33Y0.67Ni3.25Mn0.15Al0.1 | 14.49 | 19.35 | 62.74 | 2.56 | 0.87 | 3.56 |
| 0.75 | La0.25Y0.75Ni3.25Mn0.15Al0.1 | 10.84 | 22.35 | 63.34 | 2.54 | 0.92 | 3.45 |
| 0.85 | La0.15Y0.85Ni3.25Mn0.15Al0.1 | 6.28 | 25.70 | 64.66 | 2.41 | 0.94 | 3.53 |
| 1.00 | YNi3.25Mn0.15Al0.1 | 0 | 31.51 | 65.14 | 2.71 | 0.93 | 3.44 |
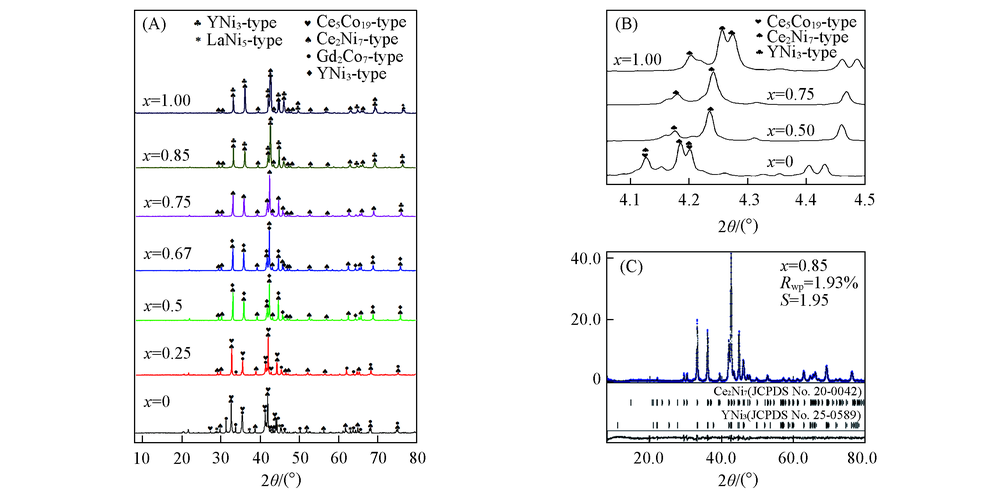
Fig.3 XRD patterns of the annealed La1-xYxNi3.25Mn0.15Al0.1(x=0—1.00) alloys(A, B) and Rietveld refinement XRD profile of the La0.15Y0.85Ni3.25Mn0.15Al0.1 alloy(C)
| x | Phase type | Space group | Lattice parameter | Phase abundance, w(%) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| a/nm | c/nm | c/a | V/nm3 | ||||||||
| 0 | Ce2Ni7-type | P63/mmc(194) | 0.5060 | 2.4586 | 4.859 | 0.545154 | 39.56 | ||||
| Gd2Co7-type | R | 0.5001 | 3.6326 | 7.264 | 0.786796 | 18.24 | |||||
| Ce5Co19-type | R | 0.4952 | 4.9093 | 9.914 | 1.042585 | 17.64 | |||||
| LaNi5-type | P6/mmm(191) | 0.5013 | 0.3987 | 0.795 | 0.086771 | 24.56 | |||||
| 0.25 | Ce2Ni7-type | P63/mmc(194) | 0.5049 | 2.4521 | 4.857 | 0.541352 | 48.26 | ||||
| Gd2Co7-type | R | 0.5000 | 3.6320 | 7.264 | 0.786351 | 10.38 | |||||
| Ce5Co9-type | R | 0.4948 | 4.8743 | 9.851 | 1.033480 | 41.36 | |||||
| 0.50 | Ce2Ni7-type | P63/mmc(194) | 0.5021 | 2.4393 | 4.858 | 0.532569 | 81.03 | ||||
| PuNi3-type | R | 0.5000 | 2.4350 | 4.870 | 0.527193 | 18.97 | |||||
| 0.67 | Ce2Ni7-type | P63/mmc(194) | 0.5015 | 2.4332 | 4.852 | 0.530880 | 86.61 | ||||
| PuNi3-type | R | 0.5000 | 2.4350 | 4.870 | 0.529966 | 13.39 | |||||
| 0.75 | Ce2Ni7-type | P63/mmc(194) | 0.5003 | 2.4303 | 4.858 | 0.526807 | 100.00 | ||||
| 0.85 | Ce2Ni7-type | P63/mmc(194) | 0.4977 | 2.4223 | 4.867 | 0.519630 | 62.54 | ||||
| YNi3-type | R | 0.4986 | 2.4223 | 4.948 | 0.521511 | 37.46 | |||||
| 1.00 | Ce2Ni7-type | P63/mmc(194) | 0.4965 | 2.4237 | 4.882 | 0.517426 | 56.72 | ||||
| YNi3-type | R | 0.4978 | 2.4361 | 4.894 | 0.522800 | 43.28 | |||||
Table 2 Characteristics of phase structures and lattice parameters of the annealed alloys La1-xYxNi3.25Mn0.15Al0.1
| x | Phase type | Space group | Lattice parameter | Phase abundance, w(%) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| a/nm | c/nm | c/a | V/nm3 | ||||||||
| 0 | Ce2Ni7-type | P63/mmc(194) | 0.5060 | 2.4586 | 4.859 | 0.545154 | 39.56 | ||||
| Gd2Co7-type | R | 0.5001 | 3.6326 | 7.264 | 0.786796 | 18.24 | |||||
| Ce5Co19-type | R | 0.4952 | 4.9093 | 9.914 | 1.042585 | 17.64 | |||||
| LaNi5-type | P6/mmm(191) | 0.5013 | 0.3987 | 0.795 | 0.086771 | 24.56 | |||||
| 0.25 | Ce2Ni7-type | P63/mmc(194) | 0.5049 | 2.4521 | 4.857 | 0.541352 | 48.26 | ||||
| Gd2Co7-type | R | 0.5000 | 3.6320 | 7.264 | 0.786351 | 10.38 | |||||
| Ce5Co9-type | R | 0.4948 | 4.8743 | 9.851 | 1.033480 | 41.36 | |||||
| 0.50 | Ce2Ni7-type | P63/mmc(194) | 0.5021 | 2.4393 | 4.858 | 0.532569 | 81.03 | ||||
| PuNi3-type | R | 0.5000 | 2.4350 | 4.870 | 0.527193 | 18.97 | |||||
| 0.67 | Ce2Ni7-type | P63/mmc(194) | 0.5015 | 2.4332 | 4.852 | 0.530880 | 86.61 | ||||
| PuNi3-type | R | 0.5000 | 2.4350 | 4.870 | 0.529966 | 13.39 | |||||
| 0.75 | Ce2Ni7-type | P63/mmc(194) | 0.5003 | 2.4303 | 4.858 | 0.526807 | 100.00 | ||||
| 0.85 | Ce2Ni7-type | P63/mmc(194) | 0.4977 | 2.4223 | 4.867 | 0.519630 | 62.54 | ||||
| YNi3-type | R | 0.4986 | 2.4223 | 4.948 | 0.521511 | 37.46 | |||||
| 1.00 | Ce2Ni7-type | P63/mmc(194) | 0.4965 | 2.4237 | 4.882 | 0.517426 | 56.72 | ||||
| YNi3-type | R | 0.4978 | 2.4361 | 4.894 | 0.522800 | 43.28 | |||||
| x | Hydriding capacity at 8 MPa, w(%) | Plateau pressure/MPa | Hydrogen capacity at 8 MPa, w(%) | Hydrogen capacity at 0.1 MPa, w(%) | △H 0—/ (kJ·mol-1 H2) | △S 0—/(J· K-1·mol-1 H2) | Hf | Sf | ||
|---|---|---|---|---|---|---|---|---|---|---|
| The 1st cycle | The 3rd cycle | Abs. | Des. | |||||||
| 0 | 1.522 | 1.271 | 0.173 | 0.052 | 1.024 | 0.603 | -38.26 | 96.29 | 0.52 | 3.66 |
| 0.25 | 1.485 | 1.119 | 0.218 | 0.037 | 0.983 | 0.573 | -35.80 | 108.10 | 0.77 | 4.52 |
| 0.50 | 1.530 | 1.389 | 0.026 | 0.009 | 1.418 | 1.122 | -34.25 | 77.22 | 0.46 | 1.10 |
| 0.67 | 1.442 | 1.444 | 0.054 | 0.031 | 1.425 | 1.128 | -33.75 | 91.59 | 0.24 | 1.66 |
| 0.75 | 1.489 | 1.434 | 0.044 | 0.033 | 1.435 | 1.216 | -34.19 | 78.11 | 0.12 | 0.88 |
| 0.85 | 1.528 | 1.513 | 0.094 | 0.046 | 1.490 | 0.804 | -33.10 | 80.12 | 0.32 | 1.26 |
| 1.00 | 1.465 | 1.444 | 0.097 | 0.057 | 1.432 | 0.749 | -32.91 | 79.21 | 0.22 | 1.59 |
Table 3 Hydriding absorption and hydrogen storage thermodynamic properties of the La1-xYxNi3.25Mn0.15Al0.1 alloys
| x | Hydriding capacity at 8 MPa, w(%) | Plateau pressure/MPa | Hydrogen capacity at 8 MPa, w(%) | Hydrogen capacity at 0.1 MPa, w(%) | △H 0—/ (kJ·mol-1 H2) | △S 0—/(J· K-1·mol-1 H2) | Hf | Sf | ||
|---|---|---|---|---|---|---|---|---|---|---|
| The 1st cycle | The 3rd cycle | Abs. | Des. | |||||||
| 0 | 1.522 | 1.271 | 0.173 | 0.052 | 1.024 | 0.603 | -38.26 | 96.29 | 0.52 | 3.66 |
| 0.25 | 1.485 | 1.119 | 0.218 | 0.037 | 0.983 | 0.573 | -35.80 | 108.10 | 0.77 | 4.52 |
| 0.50 | 1.530 | 1.389 | 0.026 | 0.009 | 1.418 | 1.122 | -34.25 | 77.22 | 0.46 | 1.10 |
| 0.67 | 1.442 | 1.444 | 0.054 | 0.031 | 1.425 | 1.128 | -33.75 | 91.59 | 0.24 | 1.66 |
| 0.75 | 1.489 | 1.434 | 0.044 | 0.033 | 1.435 | 1.216 | -34.19 | 78.11 | 0.12 | 0.88 |
| 0.85 | 1.528 | 1.513 | 0.094 | 0.046 | 1.490 | 0.804 | -33.10 | 80.12 | 0.32 | 1.26 |
| 1.00 | 1.465 | 1.444 | 0.097 | 0.057 | 1.432 | 0.749 | -32.91 | 79.21 | 0.22 | 1.59 |
| x | Na | Cmax(mA·h/g) | S100(%) | HRD900(%) | I0/ (mA·g-1) | 1010D0/ (cm2·s-1) | Ecorr/V | icorr/ (mA·cm-2) | |
|---|---|---|---|---|---|---|---|---|---|
| 60 mA/g | 300 mA/g | ||||||||
| 0 | 2 | 211.3 | 152.3 | 92.1 | 64.5 | 321.8 | 0.91 | -0.916 | 6.69 |
| 0.25 | 2 | 184.2 | 137.6 | 95.5 | 74.4 | 267.9 | 1.01 | -0.919 | 6.57 |
| 0.50 | 1 | 376.1 | 340.6 | 75.6 | 75.6 | 249.0 | 1.32 | -0.915 | 6.74 |
| 0.67 | 2 | 376.3 | 335.3 | 85.1 | 83.4 | 248.3 | 1.71 | -0.912 | 5.81 |
| 0.75 | 1 | 381.6 | 347.2 | 80.3 | 85.7 | 274.3 | 2.56 | -0.912 | 6.29 |
| 0.85 | 2 | 350.4 | 297.5 | 52.6 | 75.4 | 235.4 | 1.58 | -0.927 | 8.75 |
| 1.00 | 1 | 307.2 | 164.1 | 22.1 | 68.1 | 51.9 | 0.94 | -0.935 | 8.74 |
Table 4 Electrochemical properties of La1-xYxNi3.25Mn0.15Al0.1 alloy electrodes
| x | Na | Cmax(mA·h/g) | S100(%) | HRD900(%) | I0/ (mA·g-1) | 1010D0/ (cm2·s-1) | Ecorr/V | icorr/ (mA·cm-2) | |
|---|---|---|---|---|---|---|---|---|---|
| 60 mA/g | 300 mA/g | ||||||||
| 0 | 2 | 211.3 | 152.3 | 92.1 | 64.5 | 321.8 | 0.91 | -0.916 | 6.69 |
| 0.25 | 2 | 184.2 | 137.6 | 95.5 | 74.4 | 267.9 | 1.01 | -0.919 | 6.57 |
| 0.50 | 1 | 376.1 | 340.6 | 75.6 | 75.6 | 249.0 | 1.32 | -0.915 | 6.74 |
| 0.67 | 2 | 376.3 | 335.3 | 85.1 | 83.4 | 248.3 | 1.71 | -0.912 | 5.81 |
| 0.75 | 1 | 381.6 | 347.2 | 80.3 | 85.7 | 274.3 | 2.56 | -0.912 | 6.29 |
| 0.85 | 2 | 350.4 | 297.5 | 52.6 | 75.4 | 235.4 | 1.58 | -0.927 | 8.75 |
| 1.00 | 1 | 307.2 | 164.1 | 22.1 | 68.1 | 51.9 | 0.94 | -0.935 | 8.74 |
| [1] | Cuevas F., Joubert J. M., Latroche M., Percheron-Guégan A., Appl. Phys. A, 2001, 72(2), 225—238 |
| [2] | Ouyang L. Z., Huang J. L., Wang H., Liu J. W., Zhu M., Mater. Chem. Phys., 2017, 200, 164—178 |
| [3] | Young K. H., Ng K., Bendersky L., Batteries, 2016, 2(1), 2—15 |
| [4] | Gao Z. J., Kang L., Luo Y. C., Chem. J. Chinese Universities, 2012, 33(9), 2035—2042 |
| (高志杰, 康龙, 罗永春. 高等学校化学学报, 2012, 33(9), 2035—2042) | |
| [5] | Kohno T., Yoshida H., Kawashima F., Inaba T., Sakai I., Yamamoto M., Kanda M., J. Alloys Compd., 2000, 311, L5—L7 |
| [6] | Yasuoka S., Magari Y., Murata T., Tanaka T., Ishida J., Nakamura H., Nohma T., Kihara M., Baba Y., Teraoka H., J. Power Sources, 2006, 156(2), 662—666 |
| [7] | Liu J. J., Han S. M., Li Y., Zhang L., Zhao Y. M., Yang S. Q., Liu B. Z., Int. J. Hydrogen Energy, 2016, 41(44), 20261—20275 |
| [8] | Denys R. V., Riabov A. B., Yartys V. A., Sato M., Delaplane R. G., J. Solid State Chem., 2008, 181(4), 812—821 |
| [9] | Crivello J. C., Zhang J., Latroche M., J. Phys. Chem. C, 2011, 115(51), 25470—25478 |
| [10] | Gal L., Charbonnier V., Zhang J., Goubault L., Bernard P., Latroche M., Int. J. Hydrogen Energy, 2015, 40(47), 17017—17020 |
| [11] | Iwase K., Mori K., Terashita N., Tashiro S., Suzuki T., J. Solid State Chem., 2016, 247, 142—146 |
| [12] | Subramanian P. R., Smith J. F., Metall. Trans. B, 1985, 16(3), 577—584 |
| [13] | Nagasaki S., Hirabayashi M., Binary Alloy Phase-Diagrams, Translated by Liu A. S., Metallurgical Industry Press, Beijing, 2004, 119—220 |
| (刘安生[译]. 二元合金状态图集, 北京: 冶金工业出版社, 2004, 119—220) | |
| [14] | Shrivastava D., Sanyal S. P., Indian J. Phys., 2016, 9(2), 183—196 |
| [15] | Baddour-Hadjean R., Meyer L., Pereira-Ramos J. P., Latroche M., Percheron-Guegan A., Electrochim. Acta, 2001, 46(15), 2385—2393 |
| [16] | Berezovets V. V., Denys R. V., Ryabov O. B., Zavaliil I. Y., Mater Sci., 2007, 43(4), 499—507 |
| [17] | Charbonnier V., Zhang J. X., Monnier J., Goubault L., Bernard P., Magén C., Serin V., Latroche M., J. Phys. Chem. C, 2015, 119(22), 12218—12225 |
| [18] | Charbonnier V., Monnier J., Zhang J. X., Paul-Boncour V., Joiret S., Puga B., Goubault L., Bernard P., Latroche M., J. Power Sources, 2016, 326, 146—155 |
| [19] | Yan H. Z., Xiong W., Wang L., Li B. Q., Li J., Zhao X., Int. J. Hydrogen Energy, 2016, 42(4), 2257—2264 |
| [20] | Xiong W., Yan H. Z., Wang L., Zhao X., Li J., Li B Q., Wang Y., Int. J. Hydrogen Energy, 2017, 42(22), 15319—15327 |
| [21] | Wang H., Luo Y. C., Deng A. Q., Zhao L., Jiang W. T., J. Inorg. Mater., 2018, 33(4), 434—440 |
| (王浩, 罗永春, 邓安强, 赵磊, 姜婉婷. 无机材料学报, 2018, 33(4), 434—440) | |
| [22] | Young R. A., Cryst. Res. Technol., 1993, 210(8), 252—254 |
| [23] | Zhang L., Han S. M., Li Y., Yang S. Q., Zhao X., Liu J. J., J. Electrochem. Soc., 2014, 161(12), A1844—A1850 |
| [24] | Aoki K., Masumoto T., J. Alloys Compd., 1995, 231(1/2), 20—28 |
| [25] | Zhang J., Zhou G. Y., Chen G. R., Latroche M., Percheron-Guégan A., Sun D. L., Acta Mater., 2008, 56(19), 5388—5394 |
| [26] | Zhang Q. A., Chen Z. L., Li Y. T., Fang F., Sun D. L., Ouyang L. Z., Zhu M., J. Phys. Chem. C, 2015, 119(9), 4719—4727 |
| [27] | Senohl H., Yonei T., Takeshita H., Nobuhiko T., Takeichi N., Tanaka H., Kuriyama N., Mater. Trans., 2005, 46(2), 152—154 |
| [28] | Li S., Lan C., Hong H., Deng X. X., Chen W., Chen D. M., Yang K., J. Xi’an Jiaotong University, 2008, 42(3), 359—363 |
| (李慎, 兰程, 宏辉, 邓小霞, 陈伟, 陈德敏, 杨柯. 西安交通大学学报, 2008, 42(3), 359—363) | |
| [29] | Luo G., Hu X. C., Li S. L., Chen W., Han X. B., Chen J. P., Chen D. M., Yang K., Rare Metal Mat. Eng., 2012, 41(10), 1693—1699 |
| [30] | Liu J. J., Li Y., Han D., Yang S. Q., Chen X. C., Zhang L. Han. S. M., J. Power Sources, 2015, 300, 77—86 |
| [31] | Enomoto M., Ohata Y., Uchida H., J. Alloys Compd., 2013, 580(Suppl. 1), S3—S5 |
| [1] | 侯从聪, 王惠颖, 李婷婷, 张志明, 常春蕊, 安立宝. N-CNTs/NiCo-LDH复合材料的制备及电化学性能[J]. 高等学校化学学报, 2022, 43(10): 20220351. |
| [2] | 鲍俊全, 郑仕兵, 苑旭明, 史金强, 孙田将, 梁静. 有机盐PTO(KPD)2作为高性能锂离子电池正极材料的研究[J]. 高等学校化学学报, 2021, 42(9): 2911. |
| [3] | 项厚政, 谢鸿翔, 李文超, 刘晓磊, 冒爱琴, 俞海云. 尖晶石型高熵氧化物的制备和电化学性能[J]. 高等学校化学学报, 2020, 41(8): 1801. |
| [4] | 白延群,王存国,李雪,樊文琪,宋鹏豪,顾圆春,刘法谦,刘光烨. 孔道有序排列高容量S@C复合电极材料的制备及电化学性能[J]. 高等学校化学学报, 2020, 41(6): 1306. |
| [5] | 邓安强,罗永春,夏元华,彭思慧,马伟旗,赵旭东,杨洋,侯晓东. 退火处理对新型无镁Y0.7La0.3Ni3.25Al0.1Mn0.15储氢合金结构和电化学性能的影响[J]. 高等学校化学学报, 2020, 41(1): 145. |
| [6] | 陈耀燕, 赵昕, 王哲, 董杰, 张清华. 制备条件对MXene形貌、 结构与电化学性能的影响[J]. 高等学校化学学报, 2019, 40(6): 1249. |
| [7] | 钱明, 徐志民, 刘炜炜, 韩冰. 吡咯-(3,4-乙烯二氧噻吩)共聚物有序纳米阵列薄膜的制备及表征[J]. 高等学校化学学报, 2019, 40(3): 612. |
| [8] | 王坤, 黄梦怡, 张小颂, 黄俊杰, 邓祥, 刘长路. LiNi1/3Co1/3Mn1/3O2@C复合材料的制备及电化学性能[J]. 高等学校化学学报, 2018, 39(1): 141. |
| [9] | 郑卓, 吴振国, 向伟, 滑纬博, 郭孝东. Na+掺杂锂离子电池正极材料LiNi0.6 Co0.2 Mn0.2 O2的制备及电化学性能[J]. 高等学校化学学报, 2017, 38(8): 1458. |
| [10] | 潘超, 谷海腾, 宗飞旭, 高婧怡. 静电纺氧化锰复合碳纳米纤维柔性膜的电化学性能[J]. 高等学校化学学报, 2017, 38(6): 953. |
| [11] | 李兴盛, 赫文秀, 张永强, 于慧颖, 李子庆. β-Ni(OH)2纳米线的相转化法制备及电化学性能[J]. 高等学校化学学报, 2017, 38(2): 261. |
| [12] | 李方方, 王洪宾, 王润伟, 裘式纶, 张宗弢. NiCo2O4@C纳米复合材料的水热合成及电化学储锂性能[J]. 高等学校化学学报, 2017, 38(11): 1913. |
| [13] | 张勃, 何珺, 华正伸, 王新, 彭会芬. S |
| [14] | 孙洪丹, 贺诗词, 夏丙波, 方国清, 刘伟伟, 仇光超, 张茜, 金子信悟, 郑军伟, 李德成. LiNi0.5-xAl2xMn1.5-xO4的制备、结构及电化学特征[J]. 高等学校化学学报, 2013, 34(5): 1059. |
| [15] | 柯川, 蔡芳共, 杨峰, 程翠华, 赵勇. CuS/TiO2纳米管异质结阵列的制备及光电性能[J]. 高等学校化学学报, 2013, 34(2): 423. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||