

高等学校化学学报 ›› 2016, Vol. 37 ›› Issue (4): 736.doi: 10.7503/cjcu20150681
收稿日期:2015-08-28
出版日期:2016-04-10
发布日期:2016-03-16
基金资助:
WANG Wenliang, GENG Jing, LI Lufei, CHANG Jianmin*( )
)
Received:2015-08-28
Online:2016-04-10
Published:2016-03-16
Contact:
CHANG Jianmin
E-mail:cjianmin168@126.com
摘要:
以具备丰富中孔和大孔结构的快速热解炭(FPC) 为载体, 采用共浸渍法制备了不同Cu/Zn摩尔比的CuxZny/FPC负载型催化剂. 采用X射线衍射仪(XRD)、 高分辨场发射扫描电子显微镜(FE-SEM)及电子能谱仪(EDX) 对催化剂进行了表征, 采用热重分析仪(TG)和热解气质联用仪(Py-GC/MS) 评价了催化剂对碱木质素热裂解生成单酚类化合物的催化性能. 结果表明, 催化剂活性组分CuO和ZnO晶相结构均一, 很好地嵌入到FPC中孔和大孔结构中, 未发生聚集状态或生成CuZn合金; 随着Cu或Zn金属负载量的增大, 相应的Cu或Zn金属氧化物衍射峰强度逐渐增强, 平均晶粒尺寸逐渐增大. 热重分析结果表明, 催化剂降低了碱木质素热裂解残炭率和反应活化能, 提高了热裂解反应效率. 热解气质联用分析表明, CuxZny/FPC催化剂大幅度简化了碱木质素热裂解单酚类化合物种类(从23种减少到了10种), Cu0.67Zn0.33/FPC对单酚类化合物表现出最大的选择性(52.99%), 与Cu/FPC相比选择性增加49.7%.
中图分类号:
TrendMD:
王文亮, 耿晶, 李露霏, 常建民. 快速热解炭负载Cu-Zn对碱木质素热裂解生成单酚类化合物的催化性能. 高等学校化学学报, 2016, 37(4): 736.
WANG Wenliang, GENG Jing, LI Lufei, CHANG Jianmin. Catalytic Properties of Fast Pyrolysis Char Loaded with Cu-Zn on Alkali Lignin Pyrolysis for Monophenols†. Chem. J. Chinese Universities, 2016, 37(4): 736.
| Sample | Ultimate analysisa(mass fraction, %) | Proximate analysisc(mass fraction, %) | BET surface aread/ (m2·g-1) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| C | H | Ob | N | S | A | V | FCb | ||
| Alkali lignin | 62.40 | 6.14 | 29.43 | 0.26 | 1.77 | 6.21 | 66.43 | 27.36 | |
| FPC | 9.04 | 19.39 | 71.57 | 307.56±12.33 | |||||
Table 1 Basic analyses of alkali lignin and FPC
| Sample | Ultimate analysisa(mass fraction, %) | Proximate analysisc(mass fraction, %) | BET surface aread/ (m2·g-1) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| C | H | Ob | N | S | A | V | FCb | ||
| Alkali lignin | 62.40 | 6.14 | 29.43 | 0.26 | 1.77 | 6.21 | 66.43 | 27.36 | |
| FPC | 9.04 | 19.39 | 71.57 | 307.56±12.33 | |||||
| Catalyst | D(CuO)/nm | D(ZnO)/nm | Catalyst | D(CuO)/nm | D(ZnO)/nm |
|---|---|---|---|---|---|
| Cu/FPC | 27.9 | Cu0.33Zn0.67/FPC | 18.8 | 38.3 | |
| Cu0.67Zn0.33/FPC | 26.0 | 32.1 | Zn/FPC | 43.0 | |
| Cu0.50Zn0.50/FPC | 25.8 | 33.3 |
Table 2 Average crystal sizes of CuO and ZnO in catalysts
| Catalyst | D(CuO)/nm | D(ZnO)/nm | Catalyst | D(CuO)/nm | D(ZnO)/nm |
|---|---|---|---|---|---|
| Cu/FPC | 27.9 | Cu0.33Zn0.67/FPC | 18.8 | 38.3 | |
| Cu0.67Zn0.33/FPC | 26.0 | 32.1 | Zn/FPC | 43.0 | |
| Cu0.50Zn0.50/FPC | 25.8 | 33.3 |
| Sample | Ts/℃ | Tmax/℃ | w(%) | E/(kJ·mol-1) | A/min-1 | R2 |
|---|---|---|---|---|---|---|
| Lignin | 268—463 | 326 | 53.7 | 35.4 | 86.7 | 0.977 |
| 670—779 | 161.3 | 8.5×108 | 0.958 | |||
| Cu | 267—463 | 343 | 48.4 | 29.2 | 19.8 | 0.988 |
| 669—765 | 134.4 | 2.7×107 | 0.964 | |||
| Cu0.67Zn0.33 | 267—463 | 349 | 49.4 | 29.9 | 21.5 | 0.985 |
| 670—765 | 135.0 | 2.3×107 | 0.952 | |||
| Cu0.50Zn0.50 | 268—463 | 345 | 47.5 | 27.9 | 14.5 | 0.984 |
| 669—764 | 111.7 | 8.8×105 | 0.927 | |||
| Cu0.33Zn0.67 | 268—462 | 350 | 48.4 | 29.5 | 19.7 | 0.985 |
| 670—765 | 113.7 | 1.2×106 | 0.940 | |||
| Zn | 268—462 | 344 | 47.3 | 29.4 | 19.2 | 0.983 |
| 670—765 | 104.0 | 3.2×105 | 0.943 |
Table 3 Kinetic parameters of catalytic pyrolysis of alkali lignin*
| Sample | Ts/℃ | Tmax/℃ | w(%) | E/(kJ·mol-1) | A/min-1 | R2 |
|---|---|---|---|---|---|---|
| Lignin | 268—463 | 326 | 53.7 | 35.4 | 86.7 | 0.977 |
| 670—779 | 161.3 | 8.5×108 | 0.958 | |||
| Cu | 267—463 | 343 | 48.4 | 29.2 | 19.8 | 0.988 |
| 669—765 | 134.4 | 2.7×107 | 0.964 | |||
| Cu0.67Zn0.33 | 267—463 | 349 | 49.4 | 29.9 | 21.5 | 0.985 |
| 670—765 | 135.0 | 2.3×107 | 0.952 | |||
| Cu0.50Zn0.50 | 268—463 | 345 | 47.5 | 27.9 | 14.5 | 0.984 |
| 669—764 | 111.7 | 8.8×105 | 0.927 | |||
| Cu0.33Zn0.67 | 268—462 | 350 | 48.4 | 29.5 | 19.7 | 0.985 |
| 670—765 | 113.7 | 1.2×106 | 0.940 | |||
| Zn | 268—462 | 344 | 47.3 | 29.4 | 19.2 | 0.983 |
| 670—765 | 104.0 | 3.2×105 | 0.943 |
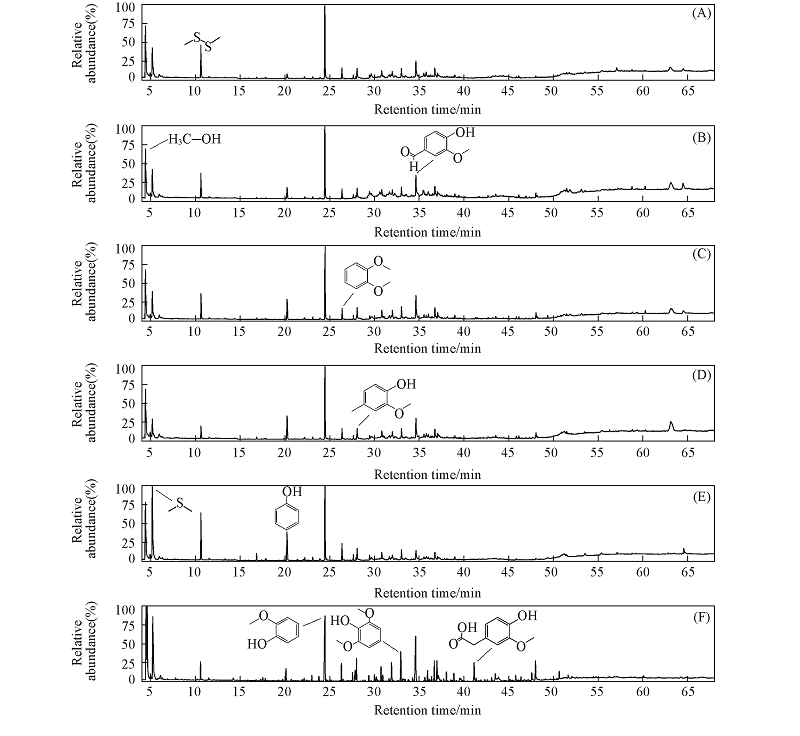
Fig.5 Total ion chromatograms of pyrolysis vapors of alkali lignin with catalysts^(A) Zn/FPC; (B) Cu0.33Zn0.67/FPC; (C) Cu0.50Zn0.50/FPC; (D) Cu0.67Zn0.33/FPC; (E) Cu/FPC; (F) Lignin.
| No. | Compound | Relative content(area)(%) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lignin | Cu | Cu0.67Zn0.33 | Cu0.5Zn0.5 | Cu0.33Zn0.67 | Zn | ||||||||
| Phenols | 7.32 | 4.93 | 4.46 | 3.79 | 1.97 | 1.15 | |||||||
| 1 | Phenol | 1.53 | 4.30 | 3.89 | 3.16 | 1.97 | 1.15 | ||||||
| 2 | o-Cresol | 0.62 | 0.63 | 0.57 | 0.63 | ||||||||
| 3 | p-Cresol | 0.69 | |||||||||||
| 4 | 3-Ethoxyphenol | 1.88 | |||||||||||
| 5 | 4-Methylcatechol | 1.77 | |||||||||||
| 6 | 4-Hydroxybenzaldehyde | 0.58 | |||||||||||
| 7 | 5-tert-Butylpyrogallol | 0.25 | |||||||||||
| Guaiacols | 39.61 | 28.08 | 44.88 | 41.80 | 41.36 | 42.07 | |||||||
| 8 | Guaiacol | 13.74 | 17.82 | 25.26 | 21.98 | 24.77 | 25.84 | ||||||
| 9 | Isocreosol | 0.96 | 1.15 | 1.09 | 1.11 | 1.12 | 1.12 | ||||||
| 10 | Creosol | 2.83 | 3.06 | 3.32 | 3.21 | 3.03 | 3.25 | ||||||
| 11 | 3-Methoxycatechol | 0.55 | |||||||||||
| 12 | 4-Ethylguaiacol | 1.70 | 1.56 | 3.10 | 2.71 | 1.97 | 2.49 | ||||||
| 13 | 4-Acetyl-3-methylphenol | 2.51 | |||||||||||
| 14 | 4-Vinylguaiacol | 1.30 | 1.82 | 2.58 | 1.85 | 1.86 | |||||||
| 15 | 2-Methoxyquinol | 0.45 | |||||||||||
| 16 | Vanillin | 9.29 | 2.74 | 8.67 | 8.66 | 8.62 | 7.51 | ||||||
| 17 | (E)-Isoeugenol | 1.29 | |||||||||||
| 18 | Acetovanillone | 2.77 | 0.45 | 1.62 | 1.55 | ||||||||
| 19 | Guaiacylacetone | 1.04 | |||||||||||
| 20 | Homovanillic acid | 2.48 | |||||||||||
| Syringols | 4.93 | 2.39 | 3.65 | 3.80 | 3.14 | 3.48 | |||||||
| 21 | Syringol | 4.10 | 2.39 | 3.65 | 3.80 | 3.14 | 3.48 | ||||||
| 22 | Syringaldehyde | 0.39 | |||||||||||
| 23 | Syringone | 0.44 | |||||||||||
| Alcohols | 10.63 | 17.09 | 19.02 | 16.80 | 18.85 | 21.33 | |||||||
| 24 | Methanol | 8.75 | 17.09 | 18.53 | 16.54 | 18.85 | 21.33 | ||||||
| 25 | Dimethylhexanol | 0.49 | 0.26 | ||||||||||
| 26 | 1-Dodecanol | 0.52 | |||||||||||
| 27 | Pentadecanol | 0.44 | |||||||||||
| 28 | Nonadecanol | 0.92 | |||||||||||
| Ethers | 6.17 | 6.86 | 8.54 | 7.76 | 8.38 | 6.89 | |||||||
| 29 | Methylvanillin | 2.61 | 1.51 | 3.36 | 3.38 | 4.47 | 2.72 | ||||||
| 30 | 4-Vinylveratrole | 0.45 | |||||||||||
| 31 | Anisole | 0.35 | |||||||||||
| 32 | Veratrole | 1.92 | 3.51 | 3.05 | 2.92 | 2.97 | 3.35 | ||||||
| 33 | Homoveratrole | 0.58 | 0.98 | 0.90 | 0.83 | 0.94 | 0.82 | ||||||
| 34 | Triglycol monomethyl ether | 1.23 | 0.63 | ||||||||||
| 35 | 1,2,3-Trimethoxybenzene | 0.25 | 0.32 | ||||||||||
| 36 | 1,2,4-Trimethoxybenzene | 0.36 | 0.19 | ||||||||||
| No. | Compound | Relative content(area)(%) | |||||||||||
| Lignin | Cu | Cu0.67Zn0.33 | Cu0.5Zn0.5 | Cu0.33Zn0.67 | Zn | ||||||||
| Ketones | 1.77 | 0.59 | 2.54 | 2.29 | 2.28 | 2.30 | |||||||
| 37 | Acetone | 0.80 | 1.78 | 1.31 | 1.43 | 1.52 | |||||||
| 38 | Acetoveratrone | 0.97 | |||||||||||
| 39 | 2,4-Dimethoxyacetophenone | 0.59 | 0.76 | 0.98 | 0.85 | 0.78 | |||||||
| Esters | 0.36 | 1.90 | 1.50 | 3.60 | 1.96 | 1.24 | |||||||
| 40 | Allyl acetate | 1.48 | |||||||||||
| 41 | Butyrolactone | 0.36 | |||||||||||
| 42 | Aceteugenol | 0.42 | 0.78 | 0.98 | 0.78 | ||||||||
| 43 | Diisobutyl phthalate | 0.72 | 0.57 | 0.71 | 0.46 | ||||||||
| 44 | Dibutyl phthalate | 0.39 | |||||||||||
| 45 | Isopropyl Palmitate | 0.57 | |||||||||||
| 46 | Mono(2-ethylhexyl) phthalate | 1.09 | 1.25 | ||||||||||
| Hydrocarbons | 0.25 | 1.64 | 2.15 | 2.79 | 4.33 | 1.37 | |||||||
| 47 | o-Cymene | 0.25 | 0.42 | 0.28 | |||||||||
| 48 | 2,4-Dimethyl styrene | 0.29 | |||||||||||
| 49 | 1-Ethylidene-1H-indene | 0.42 | 0.68 | ||||||||||
| 50 | α-Cedrene | 0.51 | 2.15 | 1.83 | 4.33 | 1.37 | |||||||
| Carboxylic acids | 5.96 | 1.28 | 2.54 | 3.36 | 1.37 | ||||||||
| 51 | Acetic acid | 0.40 | 1.28 | 0.72 | 0.87 | ||||||||
| 52 | Myristic acid | 0.75 | 0.50 | ||||||||||
| 53 | Pentadecylic acid | 0.66 | |||||||||||
| 54 | Petroselic acid | 1.07 | |||||||||||
| 55 | Palmitic acid | 2.57 | |||||||||||
| 56 | Tridecylic acid | 1.82 | 1.99 | 1.37 | |||||||||
| 57 | Oleic Acid | 0.51 | |||||||||||
| Sulfur compounds | 23.00 | 35.24 | 10.72 | 14.01 | 16.36 | 20.17 | |||||||
| 58 | Methanethiol | 8.28 | |||||||||||
| 59 | Dimethyl sulfide | 12.76 | 24.31 | 6.79 | 8.68 | 10.13 | 11.68 | ||||||
| 60 | 2,3-Dithiabutane | 1.96 | 9.57 | 3.25 | 5.33 | 6.23 | 8.49 | ||||||
| 61 | 2,4-Dithiapentane | 1.36 | 0.68 | ||||||||||
Table 4 Main components of pyrolysis vapors of alkali lignin with catalysts
| No. | Compound | Relative content(area)(%) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lignin | Cu | Cu0.67Zn0.33 | Cu0.5Zn0.5 | Cu0.33Zn0.67 | Zn | ||||||||
| Phenols | 7.32 | 4.93 | 4.46 | 3.79 | 1.97 | 1.15 | |||||||
| 1 | Phenol | 1.53 | 4.30 | 3.89 | 3.16 | 1.97 | 1.15 | ||||||
| 2 | o-Cresol | 0.62 | 0.63 | 0.57 | 0.63 | ||||||||
| 3 | p-Cresol | 0.69 | |||||||||||
| 4 | 3-Ethoxyphenol | 1.88 | |||||||||||
| 5 | 4-Methylcatechol | 1.77 | |||||||||||
| 6 | 4-Hydroxybenzaldehyde | 0.58 | |||||||||||
| 7 | 5-tert-Butylpyrogallol | 0.25 | |||||||||||
| Guaiacols | 39.61 | 28.08 | 44.88 | 41.80 | 41.36 | 42.07 | |||||||
| 8 | Guaiacol | 13.74 | 17.82 | 25.26 | 21.98 | 24.77 | 25.84 | ||||||
| 9 | Isocreosol | 0.96 | 1.15 | 1.09 | 1.11 | 1.12 | 1.12 | ||||||
| 10 | Creosol | 2.83 | 3.06 | 3.32 | 3.21 | 3.03 | 3.25 | ||||||
| 11 | 3-Methoxycatechol | 0.55 | |||||||||||
| 12 | 4-Ethylguaiacol | 1.70 | 1.56 | 3.10 | 2.71 | 1.97 | 2.49 | ||||||
| 13 | 4-Acetyl-3-methylphenol | 2.51 | |||||||||||
| 14 | 4-Vinylguaiacol | 1.30 | 1.82 | 2.58 | 1.85 | 1.86 | |||||||
| 15 | 2-Methoxyquinol | 0.45 | |||||||||||
| 16 | Vanillin | 9.29 | 2.74 | 8.67 | 8.66 | 8.62 | 7.51 | ||||||
| 17 | (E)-Isoeugenol | 1.29 | |||||||||||
| 18 | Acetovanillone | 2.77 | 0.45 | 1.62 | 1.55 | ||||||||
| 19 | Guaiacylacetone | 1.04 | |||||||||||
| 20 | Homovanillic acid | 2.48 | |||||||||||
| Syringols | 4.93 | 2.39 | 3.65 | 3.80 | 3.14 | 3.48 | |||||||
| 21 | Syringol | 4.10 | 2.39 | 3.65 | 3.80 | 3.14 | 3.48 | ||||||
| 22 | Syringaldehyde | 0.39 | |||||||||||
| 23 | Syringone | 0.44 | |||||||||||
| Alcohols | 10.63 | 17.09 | 19.02 | 16.80 | 18.85 | 21.33 | |||||||
| 24 | Methanol | 8.75 | 17.09 | 18.53 | 16.54 | 18.85 | 21.33 | ||||||
| 25 | Dimethylhexanol | 0.49 | 0.26 | ||||||||||
| 26 | 1-Dodecanol | 0.52 | |||||||||||
| 27 | Pentadecanol | 0.44 | |||||||||||
| 28 | Nonadecanol | 0.92 | |||||||||||
| Ethers | 6.17 | 6.86 | 8.54 | 7.76 | 8.38 | 6.89 | |||||||
| 29 | Methylvanillin | 2.61 | 1.51 | 3.36 | 3.38 | 4.47 | 2.72 | ||||||
| 30 | 4-Vinylveratrole | 0.45 | |||||||||||
| 31 | Anisole | 0.35 | |||||||||||
| 32 | Veratrole | 1.92 | 3.51 | 3.05 | 2.92 | 2.97 | 3.35 | ||||||
| 33 | Homoveratrole | 0.58 | 0.98 | 0.90 | 0.83 | 0.94 | 0.82 | ||||||
| 34 | Triglycol monomethyl ether | 1.23 | 0.63 | ||||||||||
| 35 | 1,2,3-Trimethoxybenzene | 0.25 | 0.32 | ||||||||||
| 36 | 1,2,4-Trimethoxybenzene | 0.36 | 0.19 | ||||||||||
| No. | Compound | Relative content(area)(%) | |||||||||||
| Lignin | Cu | Cu0.67Zn0.33 | Cu0.5Zn0.5 | Cu0.33Zn0.67 | Zn | ||||||||
| Ketones | 1.77 | 0.59 | 2.54 | 2.29 | 2.28 | 2.30 | |||||||
| 37 | Acetone | 0.80 | 1.78 | 1.31 | 1.43 | 1.52 | |||||||
| 38 | Acetoveratrone | 0.97 | |||||||||||
| 39 | 2,4-Dimethoxyacetophenone | 0.59 | 0.76 | 0.98 | 0.85 | 0.78 | |||||||
| Esters | 0.36 | 1.90 | 1.50 | 3.60 | 1.96 | 1.24 | |||||||
| 40 | Allyl acetate | 1.48 | |||||||||||
| 41 | Butyrolactone | 0.36 | |||||||||||
| 42 | Aceteugenol | 0.42 | 0.78 | 0.98 | 0.78 | ||||||||
| 43 | Diisobutyl phthalate | 0.72 | 0.57 | 0.71 | 0.46 | ||||||||
| 44 | Dibutyl phthalate | 0.39 | |||||||||||
| 45 | Isopropyl Palmitate | 0.57 | |||||||||||
| 46 | Mono(2-ethylhexyl) phthalate | 1.09 | 1.25 | ||||||||||
| Hydrocarbons | 0.25 | 1.64 | 2.15 | 2.79 | 4.33 | 1.37 | |||||||
| 47 | o-Cymene | 0.25 | 0.42 | 0.28 | |||||||||
| 48 | 2,4-Dimethyl styrene | 0.29 | |||||||||||
| 49 | 1-Ethylidene-1H-indene | 0.42 | 0.68 | ||||||||||
| 50 | α-Cedrene | 0.51 | 2.15 | 1.83 | 4.33 | 1.37 | |||||||
| Carboxylic acids | 5.96 | 1.28 | 2.54 | 3.36 | 1.37 | ||||||||
| 51 | Acetic acid | 0.40 | 1.28 | 0.72 | 0.87 | ||||||||
| 52 | Myristic acid | 0.75 | 0.50 | ||||||||||
| 53 | Pentadecylic acid | 0.66 | |||||||||||
| 54 | Petroselic acid | 1.07 | |||||||||||
| 55 | Palmitic acid | 2.57 | |||||||||||
| 56 | Tridecylic acid | 1.82 | 1.99 | 1.37 | |||||||||
| 57 | Oleic Acid | 0.51 | |||||||||||
| Sulfur compounds | 23.00 | 35.24 | 10.72 | 14.01 | 16.36 | 20.17 | |||||||
| 58 | Methanethiol | 8.28 | |||||||||||
| 59 | Dimethyl sulfide | 12.76 | 24.31 | 6.79 | 8.68 | 10.13 | 11.68 | ||||||
| 60 | 2,3-Dithiabutane | 1.96 | 9.57 | 3.25 | 5.33 | 6.23 | 8.49 | ||||||
| 61 | 2,4-Dithiapentane | 1.36 | 0.68 | ||||||||||
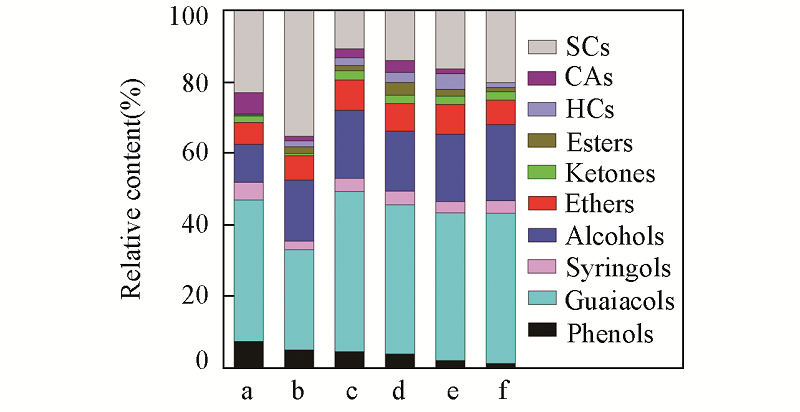
Fig.6 Component distribution of pyrolysis vapors of alkali lignin with catalysts^a. Lignin; b. Cu; c. Cu0.67Zn0.33; d. Cu0.50Zn0.50; e. Cu0.33Zn0.67; f. Zn.
| [1] | Zakzeski J., Bruijnincx P. C. A., Jongerius A. L., Weckhuysen B. M., Chem. Rev., 2010, 110(6), 3552—3599 |
| [2] | Jiang T.D., Lignin, Chemical Industry Press, Beijing, 2009, 19—45 |
| (蒋挺大. 木质素, 北京:化学工业出版社, 2009, 19—45) | |
| [3] | Ouyang X. P., Tan Y. D., Qiu X. Q., J. Fuel Chem. Technol., 2014, 42(6), 677—682 |
| (欧阳新平, 谭友丹, 邱学青. 燃料化学学报, 2014, 42(6), 677—682) | |
| [4] | Yang Z. X., Kumar A., Huhnke R. L., Renew. Sust. Energ. Rev., 2015, 50, 859—870 |
| [5] | Jiang X. X., Ellis N., Zhong Z. P., Chinese J. Chem. Eng., 2010, 18(6), 1018—1022 |
| [6] | Fan D. B., Chang J. M., Gou J. S., Xia B. H., Ren X. Y., J. Adhesion, 2010, 86(8), 834—843 |
| [7] | Kim J. S., Bioresource Technol., 2015, 178, 90—98 |
| [8] | Amen-Chen C., Pakdel H., Roy C., Bioresource Technol., 2001, 79, 277—299 |
| [9] | Jung K. A., Woo S. H., Lim S. R., Park J. M., Chem. Eng. J., 2015, 259, 107—116 |
| [10] | Yan L., Pang H., Huang Y. B., Fu Y., Acta Chim. Sinica, 2014, 72(9), 1005—1011 |
| (严龙, 庞欢, 黄耀兵, 傅尧. 化学学报, 2014, 72(9), 1005—1011) | |
| [11] | Wang W. L., Ren X. Y., Li L. F., Chang J. M., Cai L. P., Geng J., Fuel Process. Technol., 2015, 134, 345—351 |
| [12] | Wang W. L., Ren X. Y., Chang J. M., Cai L. P., Shi S. Q., Fuel Process. Technol., 2015, 138, 605—611 |
| [13] | Peng C. N., Zhang G. Y., Yue J. R., Xu G. W., Fuel Process. Technol., 2014, 124, 212—221 |
| [14] | Singh S. K., Ekhe J. D., Catal. Sci. Technol., 2015, 5(4), 2117—2124 |
| [15] | Shen D. K., Zhao J., Xiao R., Gu S., J. Anal. Appl. Pyrol., 2015, 111, 47—54 |
| [16] | Deepa A. K., Dhepe P. L., ACS Catal., 2015, 5(1), 365—379 |
| [17] | Adhikari S., Srinivasan V., Fasina O., Energy Fuels, 2014, 28(7), 4532—4538 |
| [18] | Bu Q., Lei H. W., Wang L., Wei Y., Zhu L., Zhang X. S., Liu Y. P., Yadavalli G., Tang J. M., Bioresource Technol., 2014, 162, 142—147 |
| [19] | Li Z. X., Na W., Wang H., Gao W. G., Chem. J. Chinese Universities, 2014, 35(12), 2616—2623 |
| (李志雄, 纳薇, 王华, 高文桂. 高等学校化学学报, 2014, 35(12), 2616—2623) | |
| [20] | Xi J. Y., Lü G. X., Wang Z. F., Acta Phys-Chim. Sin., 2001, 17(7), 655—658 |
| (席靖宇, 吕功煊, 王志飞. 物理化学学报, 2001, 17(7), 655—658) | |
| [21] | Wang D., Yang G. H., Ma Q. X., Yoneyama Y., Tan Y. S., Han Y. Z., Tsubaki N., Fuel, 2013, 109, 54—60 |
| [22] | Wang W. L., Yu Y. X., Chang J. M., Bai T. T., J. Fuel Chem. Technol., 2013, 41(11), 1310—1315 |
| (王文亮, 虞宇翔, 常建民, 白甜甜. 燃料化学学报, 2013, 41(11), 1310—1315) | |
| [23] | Chang J. M., Wang W. L., Ren X. Y., Li L. F., Si H., Yu Y. X., Preparation and Application of the Catalysts Used for Biomass Fast Pyrolysis Process, CP 103691429B, 2015-06-10 |
| (常建民, 王文亮, 任学勇, 李露霏, 司慧, 虞宇翔. 用于生物质快速热裂解液化的催化剂及其制备方法和应用, CP 103691429B, 2015-06-10) | |
| [24] | Wang Y. E., Cao S., Wu W. H., Wu M., Tang Y. W., Lu T. H., Chem. J. Chinese Universities, 2014, 35(11), 2455—2459 |
| (王彦恩, 曹爽, 武伟红, 吴敏, 唐亚文, 陆天虹. 高等学校化学学报, 2014, 35(11), 2455—2459) | |
| [25] | Lu X. C., Jiang J. C., Sun K., Xie X.P., B. Korean Chem. Soc., 2014, 35(1), 103—110 |
| [26] | Wang J., Zhang M. X., Chen M. Q., Min F. F., Zhang S. P., Ren Z. W., Yan Y. J., Thermochim. Acta, 2006, 444(1), 110—114 |
| [27] | Collard F. X., Blin J., Bensakhria A., Valette J., J. Anal. Appl. Pyrol., 2012, 95, 213—226 |
| [28] | Ye J. W., Jin C. D., Song P. A., Wu Q., Chem. Ind. For. Prod., 2010, 30)6), 40—44 |
| (叶结旺, 金春德, 宋平安, 吴强. 林产化学与工业, 2010, 30(6), 40—44) | |
| [29] | Wang X. Y., Wan X. J., Wu F. Y., Appl. Chem. Ind., 2010, 39(3), 377—379 |
| (王新运, 万新军, 吴凤义. 应用化工, 2010, 39(3), 377—379) | |
| [30] | Coats A. W., Redfern J. P., Nature, 1964, 201, 68—69 |
| [31] | Liu J. Y., Wu S. B., Guo Y. L., Chem. Ind. For. Prod., 2008, 28(4), 65—70 |
| (刘江燕, 武书彬, 郭伊丽. 林产化学与工业, 2008, 28(4), 65—70) | |
| [32] | Li Z.Z., Chemistry of Plant Fiber Resources, China Light Industry Press, Beijing, 2012, 203—204 |
| (李忠正. 植物纤维化学, 北京: 中国轻工业出版社, 2012, 203—204) | |
| [33] | Cheng H., Yu J., Yao M. Q., Xu G. W., CIESC Journal, 2013, 64(5), 1757—1765 |
| (程辉, 余剑, 姚梅琴, 许光文. 化工学报, 2013, 64(5), 1757—1765) | |
| [34] | Geng J., Wang W. L., Ren X. Y., Chang J. M., Chin. Sciencepaper, 2015, 10(12), 1450—1454 |
| (耿晶, 王文亮, 任学勇, 常建民. 中国科技论文, 2015, 10(12), 1450—1454) |
| [1] | 董妍红, 鲁新环, 杨璐, 孙凡棋, 段金贵, 郭昊天, 张钦峻, 周丹, 夏清华. 双功能金属有机骨架材料的制备及催化烯烃环氧化性能[J]. 高等学校化学学报, 2022, 43(11): 20220458. |
| [2] | 钟声广, 夏文生, 张庆红, 万惠霖. 电中性团簇MCu2Ox(M=Cu2+, Ce4+, Zr4+)上甲烷和二氧化碳直接合成乙酸的理论研究[J]. 高等学校化学学报, 2021, 42(9): 2878. |
| [3] | 赵鹏,张晋腾,林艳红. Mg-ZnO复合物的紫外光催化效率及协同作用研究[J]. 高等学校化学学报, 2020, 41(3): 538. |
| [4] | 杨秀荣,张驰,高红旭,赵凤起,牛诗尧,郭兆琦,马海霞. NO与NO2在ZnO表面吸附的密度泛函理论研究[J]. 高等学校化学学报, 2019, 40(10): 2121. |
| [5] | 王芙香, 陈子玉, 杨玮婷, 刘丽娟, 任国建, 刘艳凤, 潘勤鹤. ZnO@ZIF-8核壳微球的制备及对U(Ⅵ)的吸附性能[J]. 高等学校化学学报, 2019, 40(1): 24. |
| [6] | 黄雨婷, 英祖萍, 郑继兴, 庄驷耕, 刘璐, 冯威. 分级多孔结构ZnO的秸秆生物模板法制备及光催化性能[J]. 高等学校化学学报, 2018, 39(9): 2031. |
| [7] | 张岩, 张盛明, 方桂珍. Ce掺杂固体酸活化的碱木质素的抗氧化性能[J]. 高等学校化学学报, 2018, 39(6): 1255. |
| [8] | 周列, 吴青云, 徐莹, 王晨光, 马隆龙, 李文志, 陈佩丽. Ni/SiO2-Al2O3固体酸催化剂催化氢解碱木质素制备芳香类化合物[J]. 高等学校化学学报, 2018, 39(4): 735. |
| [9] | 薛佼, 王润伟, 张宗弢, 裘式纶. 新型Zn2+掺杂C/Nb2O5纳米催化剂的制备及光催化性能[J]. 高等学校化学学报, 2018, 39(2): 319. |
| [10] | 张金, 史天彩, 罗力文, 刘佳, 刘荣, 刘乐, 梁明, 马养民. 纳米氧化铜催化一锅法合成β-咔啉类化合物[J]. 高等学校化学学报, 2018, 39(11): 2411. |
| [11] | 符方宝, 王欢, 钟锐生, 邱学青, 杨东杰. 木质素/氧化锌复合颗粒的制备及在水性聚氨酯中的应用[J]. 高等学校化学学报, 2018, 39(10): 2335. |
| [12] | 胡海霞, 胡世荣, 董培辉, 唐元军, 洪可俊, 王松华. 新型氧化锌纳米材料在电化学检测对硝基苯酚中的应用[J]. 高等学校化学学报, 2017, 38(7): 1171. |
| [13] | 孙连志, 赵圣哲, 高志伶, 程志强. Ag/ZnO纳米纤维的制备及光催化性能[J]. 高等学校化学学报, 2017, 38(6): 907. |
| [14] | 李圆圆, 杨东杰, 邱学青. pH响应木质素基胶体球的制备和表征[J]. 高等学校化学学报, 2017, 38(5): 880. |
| [15] | 陈宇, 王磊, 王波. 胶带表面优异防覆冰性能[J]. 高等学校化学学报, 2017, 38(4): 631. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||