

高等学校化学学报 ›› 2019, Vol. 40 ›› Issue (2): 279.doi: 10.7503/cjcu20180673
收稿日期:2018-10-08
出版日期:2019-02-10
发布日期:2018-11-26
作者简介:联系人简介: 刘海英, 女, 博士, 副教授, 主要从事理论计算与模拟化学方面的研究. E-mail:
基金资助:
CHENG Yingying, LIU Haiying*( ), TIAN Yigeng, LIU Zhongqi, LI Qingxin
), TIAN Yigeng, LIU Zhongqi, LI Qingxin
Received:2018-10-08
Online:2019-02-10
Published:2018-11-26
Contact:
LIU Haiying
E-mail:ss_liuhy@ujn.edu.cn
Supported by:摘要:
利用密度泛函理论并结合非平衡态格林函数方法, 研究了腺嘌呤A的碳2位氨基修饰对DNA导电性的影响. 结果表明, 形成的双氨基嘌呤D可以与胸腺嘧啶T通过3个氢键进行配对, 由于氨基修饰形成了新的氢键, 使配对碱基D和T之间的结合比AT更紧密. 修饰后体系的能隙和电离能大大降低, 紫外吸收光谱在一定程度上会发生红移, 并增加了一些电荷转移跃迁. 计算的沿氢键方向的横向电荷输运和沿DNA链方向的纵向电荷输运性质, 证明了氨基的取代修饰可以很好地提高DNA的电荷输运性质. 揭示了DNA导电性增强是由于修饰调整了碱基对DT的最高占据轨道(HOMO)能级, 使之较天然碱基对AT更靠近GC的HOMO, 从而降低了空穴在DNA中迁移的势垒.
中图分类号:
TrendMD:
程颖颖, 刘海英, 田宜耕, 刘仲奇, 李清新. 腺嘌呤的氨基修饰增强DNA导电性的理论研究. 高等学校化学学报, 2019, 40(2): 279.
CHENG Yingying,LIU Haiying,TIAN Yigeng,LIU Zhongqi,LI Qingxin. Theoretical Study on Enhancement Effect of Amino Modification of Adenine on Conductivity of DNA†. Chem. J. Chinese Universities, 2019, 40(2): 279.
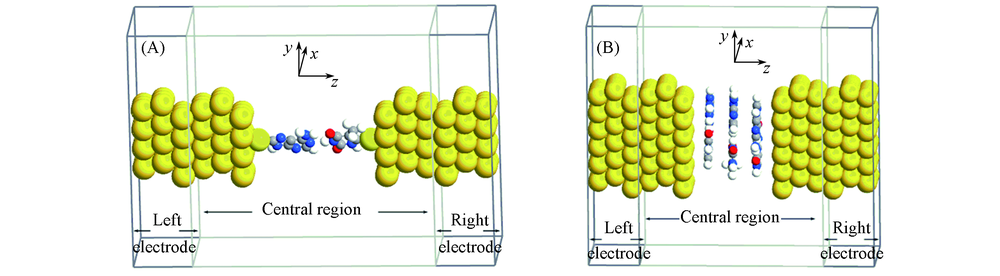
Fig.2 Schematic illustration of two-probe systems in charge transport calculation(A) The transverse charge transport model; (B) the longitudinal charge transport model.
| Base and base pair | VIP/eV | AIP/eV | Edef/eV | Eb/eV |
|---|---|---|---|---|
| A | 8.30 | 8.11 | 0.19 | - |
| D | 7.59 | 7.30 | 0.29 | - |
| G | 7.94 | 7.68 | 0.26 | - |
| AT | 7.87 | 7.70 | 0.17 | 0.46(0.52[ |
| DT | 7.16 | 6.90 | 0.26 | 0.55 |
| GC | 7.28 | 6.92(6.90[ | 0.36 | 1.00 |
Table 1 Comparison of the adiabatic ionization potentials(AIP), vertical ionization potentials(VIP), deformation energies(Edef) and binding energies(Eb)
| Base and base pair | VIP/eV | AIP/eV | Edef/eV | Eb/eV |
|---|---|---|---|---|
| A | 8.30 | 8.11 | 0.19 | - |
| D | 7.59 | 7.30 | 0.29 | - |
| G | 7.94 | 7.68 | 0.26 | - |
| AT | 7.87 | 7.70 | 0.17 | 0.46(0.52[ |
| DT | 7.16 | 6.90 | 0.26 | 0.55 |
| GC | 7.28 | 6.92(6.90[ | 0.36 | 1.00 |
| Base pair | EHOMO/eV | ELUMO/eV | Egap/eV | VIP/eV |
|---|---|---|---|---|
| GAG | -6.35 | -0.86 | 5.49 | 6.89 |
| GDG | -5.89 | -0.95 | 4.94 | 6.40 |
Table 2 Comparison of HOMO-LUMO gaps, VIP of three-layer stacked base pairs GAG and GDG, calculated by M06-2X/B3LYP*
| Base pair | EHOMO/eV | ELUMO/eV | Egap/eV | VIP/eV |
|---|---|---|---|---|
| GAG | -6.35 | -0.86 | 5.49 | 6.89 |
| GDG | -5.89 | -0.95 | 4.94 | 6.40 |
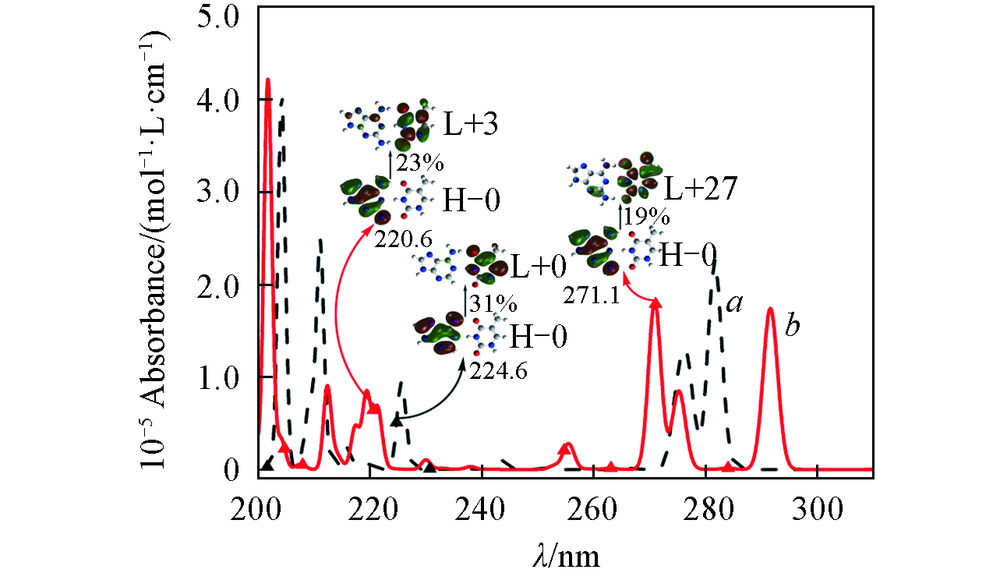
Fig.6 Absorption spectra of AT(a) and DT(b) in the ultraviolet region between 200 and 310 nm determined from the CIS resultsBlack and red solid triangles represent positions assigned to π→π* charge-transfer transition of AT and DT, respectively. H-0: HOMO, L+0: LUMO, L+3: LUMO+3, other markers are similar, and percentages represent proportions of transitions.
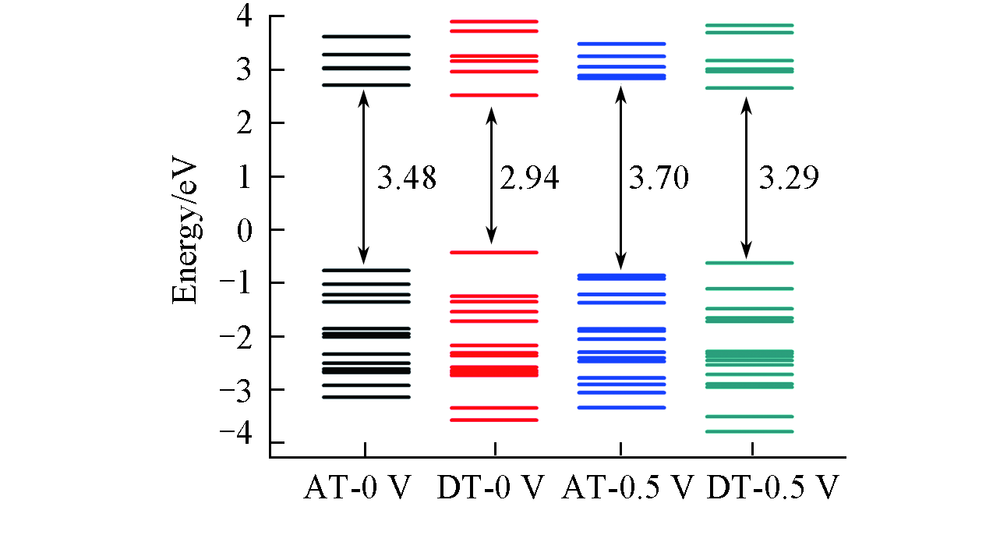
Fig.8 Energy levels of AT and DT junctions for the transverse transport model in the energy region of -4.0―4.0 eV under 0 and 0.5 V biasesThe average Fermi level is set as zero.
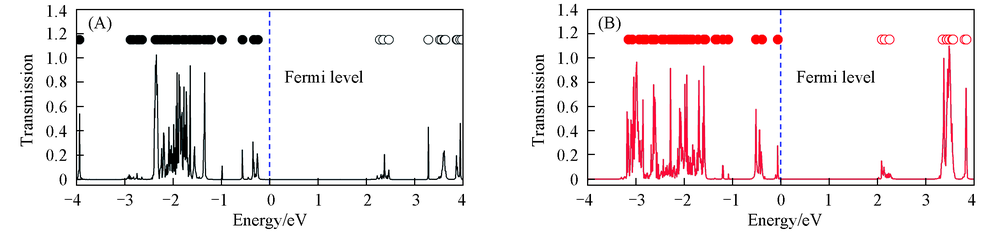
Fig.10 Transmission spectra of 3-layer stacked GAG(A) and GDG(B) junctions at zero bias for the longitudinal electronic transport modelThe dotted dots on the top of each picture represent the molecular projected self-consistent Hamiltonian(MPSH) eigenvalue positions. The solid dots represent the occupied molecular orbital energies and the hollow dots correspond to the unoccupied molecular orbital energies.
| [1] | Guo C. L., Wang K., Zerah-Harush E., Hamill J., Wang B., Dubi Y., Xu B. Q., Nat. Chem.,2016, 8(5), 484-490 |
| [2] | Xiang D., Wang X. L., Jia C. C., Lee T., Guo X. F., Chem. Rev.,2016, 116(7), 4318-4440 |
| [3] | Wang L., Li Z., Shen X. Q., Ma N., Chem. J. Chinese Universities,2018, 39(1), 32-40 |
| (王莉, 李智, 沈晓琴, 马楠. 高等学校化学学报, 2018, 39(1), 32-40) | |
| [4] | Behnia S., Fathizadeh S., Akhshani A., J. Phys. Chem. C,2016, 120(5), 2973-2983 |
| [5] | Shipman S. L., Nivala J., Macklis J. D., Church G. M., Nature,2017, 547(7663), 345-349 |
| [6] | Lin M. H., Wen Y. L., Li L. Y., Pei H., Liu G., Song H. Y., Zuo X. L., Fan C. H., Huang Q., Anal. Chem.,2014, 86(5), 2285-2288 |
| [7] | Li M., Kong H. F., Guo Z. H., Chem. J. Chinese Universities,2016, 37(7), 1269-1275 |
| (李敏, 孔慧芳, 郭志慧. 高等学校化学学报, 2016, 37(7), 1269-1275) | |
| [8] | Kasumov A. Y., Kociak M., Guéron S., Reulet B., Volkov V. T., Klinov D. V., Bouchiat H., Science,2001, 291(5502), 280-282 |
| [9] | Porath D., Bezryadin A., de Vries S., Dekker C., Nature,2000, 403(6770), 635-638 |
| [10] | De Pablo P. J., Moreno-Herrero F., Colchero J., Gómez-Herrero J., Herrero P., Baró A. M., Ordejón P., Soler J. M., Artacho E., Phys. Rev. Lett.,2000, 85(23), 4992-4995 |
| [11] | Steinbrecher T., Koslowski T., Case D. A., J. Phys. Chem. B,2008, 112(51), 16935-16944 |
| [12] | Kawai K., Kodera H., Osakada Y., Majima T., Nat. Chem., 2009, 1(2), 156-159 |
| [13] | Nakatani K., Dohno C., Saito I., J. Am. Chem. Soc.,2000, 122(24), 5893-5894 |
| [14] | Lee A. H. F., Kool E. T., J. Am. Chem. Soc.,2006, 128(28), 9219-9230 |
| [15] | Hernández A. R., Kool E. T., Org. Lett., 2011, 13(4), 676-679 |
| [16] | Liu H. Y., Li G. Q., Zhao P., Chen G., Bu Y. X., Int. J. Quantum Chem., 2014, 114(14), 911-919 |
| [17] | Brancolini G., Felice R. D., J. Phys. Chem. B,2008, 112(45), 14281-14290 |
| [18] | Liu H. Y., Li G. Q., Ai H. Q., Li J. L., Bu Y. X., J. Phys. Chem. C,2011, 115(45), 22547-22556 |
| [19] | Kawai K., Kodera H., Majima T., J. Am. Chem. Soc., 2010, 132(2), 627-630 |
| [20] | Brancolini G., Migliore A., Corni S., Fuentes-Cabrera M., Luque F. J., Di Felice R., ACS Nano,2013, 7(10), 9396-9406 |
| [21] | Becke A. D., J. Chem. Phys., 1993, 98(7), 5648-5652 |
| [22] | Lee C., Yang W., Parr R. G., Phys. Rev. B: Condens. Matter,1988, 37(2), 785-789 |
| [23] | Frisch M. J., Pople J. A., Binkley J. S., J. Chem. Phys.,1984, 80(7), 3265-3269 |
| [24] | Frisch M.J., Trucks G. W., Schlegel H. B., Scuseria G. E., Robb M. A., Cheeseman J. R., Scalmani G., Barone V., Mennucci B., Petersson G. A., Nakatsuji H., Caricato M., Li X., Hratchian H. P., Izmaylov A. F., Bloino J., Zheng G., Sonnenberg J. L., Hada M., Ehara M., Toyota K., Fukuda R., Hasegawa J., Ishida M., Nakajima T., Honda Y., Kitao O., Nakai H., Vreven T., Montgomery J. A., Peralta J. E., Ogliaro F., Bearpark M., Heyd J. J., Brothers E., Kudin K. N., Staroverov V. N., Kobayashi R., Normand J., Raghavachari K., Rendell A., Burant J. C., Iyengar S. S., Tomasi J., Cossi M., Rega N., Millam J. M., Klene M., Knox J. E., Cross J. B., Bakken V., Adamo C., Jaramillo J., Gomperts R., Stratmann R. E., Yazyev O., Austin A. J., Cammi R., Pomelli C., Ochterski J. W., Martin R. L., Morokuma K., Zakrzewski V. G., Voth G. A., Salvador P., Dannenberg J. J., Dapprich S., Daniels A. D., Farkas O., Foresman J. B., Ortiz J. V., Cioslowski J., Fox D. J., Gaussian 09, Revision A.02, Gaussian Inc., Wallingford CT, 2009 |
| [25] | Rappé A. K., Bernstein E. R., J. Phys. Chem. A,2000, 104(26), 6117-6128 |
| [26] | Boys S. F., Bernardi F., Mol. Phys.,1970, 19(4), 553-566 |
| [27] | Wang M., Wang J., Bu Y. X., Chem. J. Chinese Universities,2015, 36(11), 2271-2282 |
| (王梅, 王军, 步宇翔. 高等学校化学学报, 2015, 36(11), 2271-2282) | |
| [28] | Liu C., Wang Y., Zhao D., Gong L. D., Yang Z. Z., J. Mol. Graph. Model.,2014, 47, 62-76 |
| [29] | Yang B., Rodgers M. T., J. Am. Chem. Soc.,2014, 136(1), 282-290 |
| [30] | Jissy A. K., Datta A., J. Phys. Chem. Lett.,2014, 5(1), 154-166 |
| [31] | Foresman J. B., Head-Gordon M., Pople J. A., Frisch M. J., J. Phys. Chem.,1992, 96(1), 135-149 |
| [32] | Dreuw A., Head-Gordon M., Chem. Rev.,2005, 105(11), 4009-4037 |
| [33] | Shukla M. K., Leszczynski J., J. Phys. Chem. B,2005, 109(36), 17333-17339 |
| [34] | Zhang L., Bu Y., J. Phys. Chem. B,2008, 112(34), 10723-10731 |
| [35] | Broo A., Holmén A., J. Phys. Chem. A,1997, 101(19), 3589-3600 |
| [36] | Holmén A., Broo A., Albinsson B., Nordén B., J. Am. Chem. Soc.,1997, 119(50), 12240-12250 |
| [37] | Gorelsky S.I., SWizard Program, Revision 4.4, Centre for Catalysis Research and Innovation, University of Ottawa, Ottawa, 2010( |
| [38] | Atomistix Tool Kit Version 2015.1, Quantum Wise A/S, De nmark, 2015( Version 2015.1, Quantum Wise A/S, De nmark, 2015() |
| [39] | Brandbyge M., Mozos J. L., Ordejón P., Taylor J., Stokbro K., Phys. Rev. B,2002, 65, 165401 |
| [40] | Staykov A., Tsuji Y., Yoshizawa K., J. Phys. Chem. C,2011, 115(8), 3481-3490 |
| [41] | Smeu M., Wolkow R. A., Guo H., J. Am. Chem. Soc.,2009, 131(31), 11019-11026 |
| [42] | Jiang L. M., Qiu W., Al-Dirini F., Hossain F. M., Evans R., Skafidas E., Appl. Phys., 2016, 120(2), 025501 |
| [43] | Liu H. M., Wang N., Zhao J. W., Guo Y., Yin X., Boey F. Y. C., Zhang H., Chem. Phys. Chem.,2008, 9(10), 1416-1424 |
| [44] | Cohen R., Stokbro K., Martin J. M. L., Ratner M. A., J. Phys. Chem. C,2007, 111(40), 14893-14902 |
| [45] | Tada T., Kondo M., Yoshizawa K., Chem. Phys. Chem.,2003, 4(11), 1256-1260 |
| [46] | Lukas M., Kelly R. E. A., Kantorovich L. N., Otero R., Xu W., Laegsgaard E., Stensgaard I., Besenbacher F., J. Chem. Phys.,2009, 130(2), 024705 |
| [47] | Perdew J. P., Burke K., Ernzerhof M., Phys. Rev. Lett.,1996, 77(18), 3865-3868 |
| [48] | Troullier N., Martins J. L., Phys. Rev. B,1991, 43(3), 1993-2006 |
| [49] | Soler J. M., Artacho E., Gale J. D., Garcia A., Junquera J., Ordejon P., Sanchez-Portal D., Physics,2002, 14(11), 2745-2779 |
| [50] | Monkhorst H. J., Pack J. D., Phys. Rev. B,1976, 13(12), 5188-5192 |
| [51] | Grimme S., Antony J., Ehrlich S., Krieg H., J. Chem. Phys.,2010, 132(15), 154104 |
| [52] | Piccirilli J. A., Krauch T., Moroney S. E., Benner S. A., Nature,1990, 343(6253), 33-37 |
| [53] | Okamoto A., Maeda Y., Tsukamoto T., Ishikawa Y., Kurita N., Comp. Mater. Sci.,2012, 53(1), 416-424 |
| [54] | Nakano S., Sugimoto N., Molecules,2014, 19(8), 11613-11627 |
| [55] | Gervasio F. L., Boero M., Parrinello M., Angew. Chem. Int. Ed.,2006, 45(34), 5606-5609 |
| [56] | Reynisson J., Steenken S., Phys. Chem. Chem. Phys.,2002, 4, 5353-5358 |
| [57] | Zhao Y., Truhlar D. G., Acc. Chem. Res.,2008, 41(2), 157-167 |
| [58] | Zhao Y., Truhlar D. G., Theor. Chem. Acc.,2008, 120(1—3), 215-241 |
| [1] | 李敏, 孔慧芳, 郭志慧. 基于铜离子与DNA相互作用的铜离子检测[J]. 高等学校化学学报, 2016, 37(7): 1269. |
| [2] | 王梅, 王军, 步宇翔. 与蛋白质调控DNA空穴迁移相关的具有负离解能特征的亚稳态氢键[J]. 高等学校化学学报, 2015, 36(11): 2271. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||