

高等学校化学学报 ›› 2018, Vol. 39 ›› Issue (9): 2080.doi: 10.7503/cjcu20180019
刘宜娜1, 杨荣杰1( ), 李定华1, 梁嘉香1, HU Weiguo2
), 李定华1, 梁嘉香1, HU Weiguo2
收稿日期:2018-01-08
出版日期:2018-09-07
发布日期:2018-08-03
作者简介:联系人简介: 杨荣杰, 男, 博士, 教授, 主要从事阻燃材料和高性能固体推进剂等方面的研究. E-mail:
基金资助:
LIU Yina1, YANG Rongjie1,*( ), LI Dinghua1, LIANG Jiaxiang1, HU Weiguo2
), LI Dinghua1, LIANG Jiaxiang1, HU Weiguo2
Received:2018-01-08
Online:2018-09-07
Published:2018-08-03
Contact:
YANG Rongjie
E-mail:yrj@bit.edu.cn
Supported by:摘要:
以液体定量31P核磁共振波谱(31P NMR)为主要表征手段, 通过建立水中加盐促溶的高聚合度聚磷酸铵(APP)水溶液的制备方法, 研究不同溶解温度、 测试温度及NMR参数等对APP聚合度的影响, 建立了31P NMR准确表征APP的聚合度及其链结构的方法. 研究结果表明, 于36 ℃在水中加盐溶解APP, 26 ℃下进行31P NMR测试, 既保证了APP溶液良好的溶解性, 又在水溶液制备和31P NMR测试过程中不引起APP降解, 测得的APP聚合度稳定、 准确. 利用液体31P NMR确定了不同温度下APP盐水溶液中端基磷和中间磷弛豫时间(T1), 发现T1与温度呈线性关系, 弛豫时间参数(D1)的设置影响APP聚合度结果, D1设置不满足磷所需的T1时, APP的聚合度结果偏大. 31P NMR表征APP链结构结果显示, APP的端基磷存在3种化学环境, 而溶液离子强度和温度会影响3种端基磷的化学位移; APP的中间磷中含有H+. 此外, 提出了中国化工行业标准HG/T 2770-2008中APP高聚合度测定存在的问题.
中图分类号:
TrendMD:
刘宜娜, 杨荣杰, 李定华, 梁嘉香, HU Weiguo. 31P核磁共振波谱表征高聚合度聚磷酸铵及其链结构. 高等学校化学学报, 2018, 39(9): 2080.
LIU Yina,YANG Rongjie,LI Dinghua,LIANG Jiaxiang,HU Weiguo. Characterization of High Polymerization Degree Ammonium Polyphosphate and Its Chain Structure by 31P NMR†. Chem. J. Chinese Universities, 2018, 39(9): 2080.
| Preparation conditon | Dissolving temperature/℃ | Testing temperature/℃ | Pfree | Pend | Pmid | n | |||
|---|---|---|---|---|---|---|---|---|---|
| Sfree | Ratio(%) | Send | Ratio(%) | Smid | Ratio(%) | ||||
| NaCl auxiliary | 26 | 26 | 6.69 | 0.64 | 2.62 | 0.25 | 1034.62 | 99.11 | 792 |
| dissolving | 36 | 26 | 6.77 | 0.61 | 2.46 | 0.22 | 1099.07 | 99.17 | 893 |
| 36 | 36 | 6.92 | 0.66 | 2.71 | 0.26 | 1040.72 | 99.08 | 770 | |
| 100 ℃ High temperature dissolving | 100 | 26 | 7.21 | 7.20 | 3.83 | 3.82 | 989.34 | 98.89 | 519 |
Table 1 Characteristic phosphorous and polymerization degree(n) of APP1# at different testing conditions
| Preparation conditon | Dissolving temperature/℃ | Testing temperature/℃ | Pfree | Pend | Pmid | n | |||
|---|---|---|---|---|---|---|---|---|---|
| Sfree | Ratio(%) | Send | Ratio(%) | Smid | Ratio(%) | ||||
| NaCl auxiliary | 26 | 26 | 6.69 | 0.64 | 2.62 | 0.25 | 1034.62 | 99.11 | 792 |
| dissolving | 36 | 26 | 6.77 | 0.61 | 2.46 | 0.22 | 1099.07 | 99.17 | 893 |
| 36 | 36 | 6.92 | 0.66 | 2.71 | 0.26 | 1040.72 | 99.08 | 770 | |
| 100 ℃ High temperature dissolving | 100 | 26 | 7.21 | 7.20 | 3.83 | 3.82 | 989.34 | 98.89 | 519 |
| Preservation temperature/℃ | Polymerization degree, n | |||
|---|---|---|---|---|
| 2 h | 5 h | 8 h | 11 h | |
| 26 | 792 | 782 | 764 | 754 |
| 36 | 770 | 773 | 513 | 460 |
Table 2 Polymerization degree of APP1# solution(10 mg NaCl) experiencing preservation for different time
| Preservation temperature/℃ | Polymerization degree, n | |||
|---|---|---|---|---|
| 2 h | 5 h | 8 h | 11 h | |
| 26 | 792 | 782 | 764 | 754 |
| 36 | 770 | 773 | 513 | 460 |
| Testing number | n | Testing number | n |
|---|---|---|---|
| 1 | 893 | 6 | 838 |
| 2 | 883 | 7 | 857 |
| 3 | 878 | 8 | 865 |
| 4 | 866 | Average | 865 |
| 5 | 840 |
Table 3 Repeatability of polymerization degree(n) of APP1# in 31P NMR tests
| Testing number | n | Testing number | n |
|---|---|---|---|
| 1 | 893 | 6 | 838 |
| 2 | 883 | 7 | 857 |
| 3 | 878 | 8 | 865 |
| 4 | 866 | Average | 865 |
| 5 | 840 |
| APP sample | Sfree(%) | Send(%) | Smid(%) | n |
|---|---|---|---|---|
| 5125# | 0.52 | 0.17 | 98.64 | 1162 |
| 61231# | 0.10 | 0.06 | 95.74 | 3193 |
| 714# | 0.16 | 0.02 | 94.17 | 9419 |
| 715# | 0.08 | — | 102.6 | — |
Table 4 Characteristic phosphorus and polymerization degree of different APP samples with 10 mg NaCl auxiliary dissolving at 36 ℃
| APP sample | Sfree(%) | Send(%) | Smid(%) | n |
|---|---|---|---|---|
| 5125# | 0.52 | 0.17 | 98.64 | 1162 |
| 61231# | 0.10 | 0.06 | 95.74 | 3193 |
| 714# | 0.16 | 0.02 | 94.17 | 9419 |
| 715# | 0.08 | — | 102.6 | — |
| D1/s | Send | Smid | n |
|---|---|---|---|
| 1 | 1.68 | 957.85 | 1142 |
| 10 | 2.48 | 1058.56 | 855 |
| 20 | 2.54 | 1058.78 | 835 |
| 30 | 2.54 | 1058.77 | 835 |
Table 5 Characteristic phosphorus and polymerization degree of APP1# with 10 mg NaCl auxiliary dissolving at 36 ℃ at different relaxation time parameters
| D1/s | Send | Smid | n |
|---|---|---|---|
| 1 | 1.68 | 957.85 | 1142 |
| 10 | 2.48 | 1058.56 | 855 |
| 20 | 2.54 | 1058.78 | 835 |
| 30 | 2.54 | 1058.77 | 835 |
| Testing temperature/℃ | T1/s | ||
|---|---|---|---|
| Pfree | Pend | Pmid | |
| 26 | 8.32 | 2.99 | 0.52 |
| 31 | 8.52 | 4.54 | 0.55 |
| 36 | 8.89 | 5.60 | 0.59 |
| 41 | 10.70 | 6.81 | 0.63 |
| 46 | 10.68 | 7.99 | 0.66 |
| 51 | 10.21 | 8.97 | 0.69 |
| 56 | 11.20 | 10.10 | 0.73 |
Table 6 Relaxation time(T1) of phosphorus at different testing temperatures in 31P NMR for APP1# with 10 mg NaCl auxiliary dissolving at 36 ℃
| Testing temperature/℃ | T1/s | ||
|---|---|---|---|
| Pfree | Pend | Pmid | |
| 26 | 8.32 | 2.99 | 0.52 |
| 31 | 8.52 | 4.54 | 0.55 |
| 36 | 8.89 | 5.60 | 0.59 |
| 41 | 10.70 | 6.81 | 0.63 |
| 46 | 10.68 | 7.99 | 0.66 |
| 51 | 10.21 | 8.97 | 0.69 |
| 56 | 11.20 | 10.10 | 0.73 |
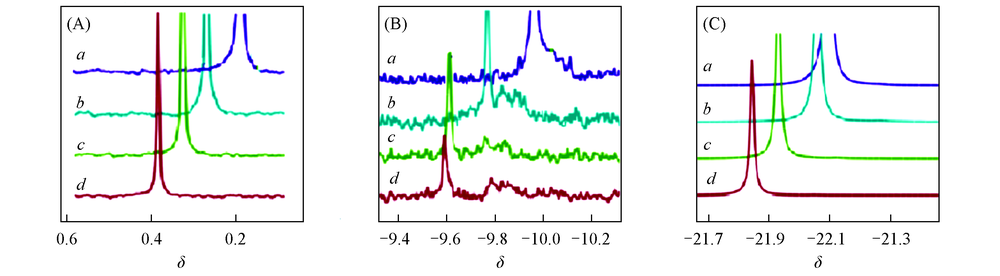
Fig.4 Phosphorous resonance peaks of APP1# with 10 mg NaCl auxiliary dissolving at 36 ℃ at different testing temperatures in 31P NMR (A) Pfree; (B) Pend; (C) Pmid. Temperature/℃: a. 26; b. 36; c. 46; d. 56.
| Acetic acid content(%) | Pfree ratio(%) | Pend ratio(%) | Short chain Pmid ratio(%) | Long chain Pmid ratio(%) | ||
|---|---|---|---|---|---|---|
| Peak 1 | Peak 2 | Peak 3 | ||||
| 0 | 1.18 | 0.14 | 0.14 | 0.16 | 0.21 | 98.2 |
| 30 | 1.40 | 0.13 | 0.22 | 0.20 | 0.21 | 97.8 |
| 50 | 1.41 | 0.12 | 0.32 | 0.37 | 0.27 | 97.5 |
Table 7 Characteristic phosphorus in APP1# solution with acetic acid added
| Acetic acid content(%) | Pfree ratio(%) | Pend ratio(%) | Short chain Pmid ratio(%) | Long chain Pmid ratio(%) | ||
|---|---|---|---|---|---|---|
| Peak 1 | Peak 2 | Peak 3 | ||||
| 0 | 1.18 | 0.14 | 0.14 | 0.16 | 0.21 | 98.2 |
| 30 | 1.40 | 0.13 | 0.22 | 0.20 | 0.21 | 97.8 |
| 50 | 1.41 | 0.12 | 0.32 | 0.37 | 0.27 | 97.5 |
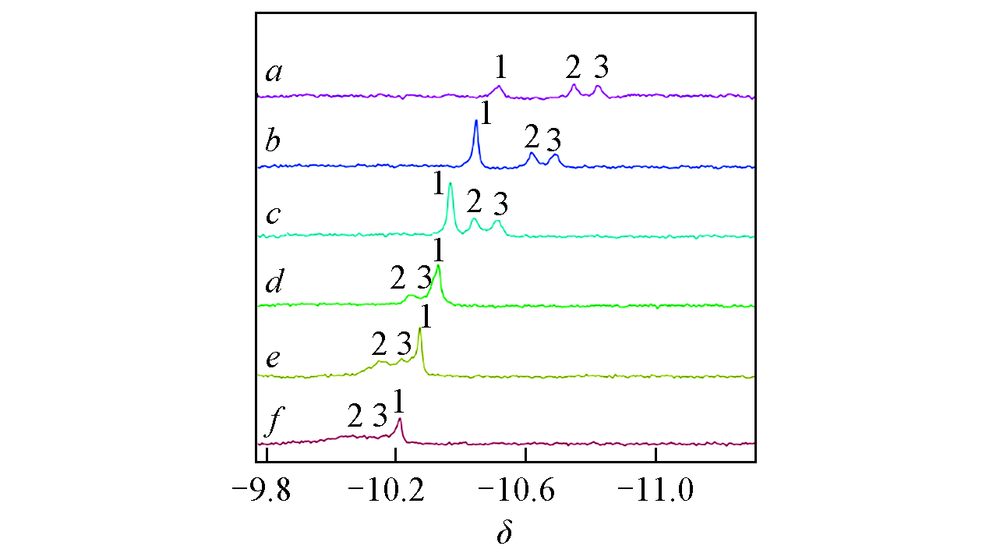
Fig.9 Pend characteristics of of APP1# solutions with different NaCl concentrationsNaCl concentration/(mg·mL-1): a. 0(100 ℃); b. 5; c. 10; d. 15; e. 20; f. 25.
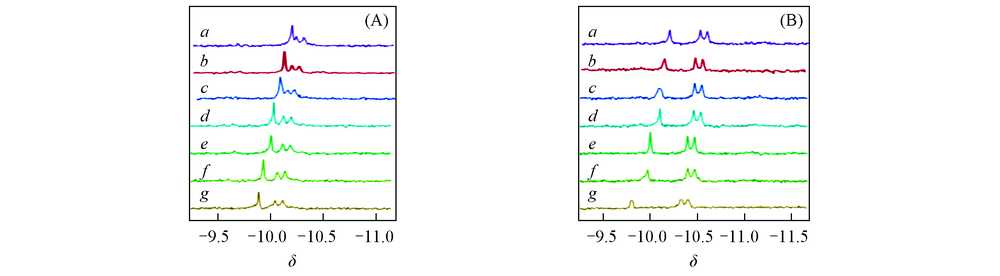
Fig.10 Pend characteristics of APP1# solutions at different testing temperatures (A) NaCl auxiliary dissolving at 36 ℃; (B) high temperature dissolving at 100 ℃. Testing temperature/℃: a. 26; b. 31; c. 36; d. 41; e. 46; f. 51; g. 56.
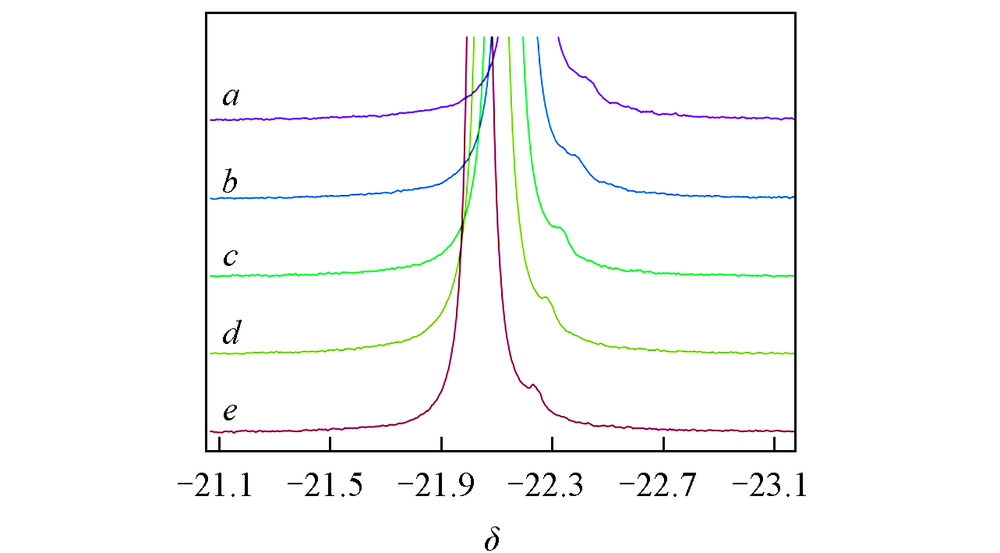
Fig.11 Resonance peaks at different testing temperature of middle phosphorus Pmid in APP1# solution with 10 mg NaCl auxiliary dissolving at 36 ℃Testing temperature/℃: a. 26; b. 31; c. 36; d. 41; e. 46.
| [1] | Camino G., Luda M. P., Fire Retardancy of Polymers: the Use of Intumescence, The Royal Society of Chemistry, Cambrige, 1988, 48—73 |
| [2] | Bbargava H. N., Varma B. K., Agrawal P. K., Colloid and Polymer Science, 1978, 256, 452—458 |
| [3] | Dong Y., Wang G. J., Su Q., Surface Coatings Technology, 2014, 246, 71—76 |
| [4] | Griffith E. J., J. Am. Chem. Soc., 1957, 79, 509—513 |
| [5] | Liu G. S., Duan X. Z., Advanced Materials Research, 2011, 832—835 |
| [6] | Yang R.J., Yi D. Q., Ammonium Polyphosphate, Science Press, Beijing, 2015 |
| (杨荣杰, 仪德启. 聚磷酸铵, 北京:科学出版社, 2015) | |
| [7] | Greenfield S., Oxford M.C., Analytical Chemistry of the Condensed Phosphates, Pergamon Press, New York, 1975, 18—32 |
| [8] | Van Wazer J. R., Callis C. F., Shoolery J. N., J. Am. Chem. Soc., 1955, 77(18), 4945—4946 |
| [9] | Van Wazer J. R., Phosphorous and Its Compounds, Interscience, New Yourk, 1958, 419—477 |
| [10] | Van Wazer J. R., J. Am. Chem. Soc., 1950, 72, 906—908 |
| [11] | Callis C. F., van Wazer J. R., Shoolery J. N., J. Am. Chem. Soc., 1957, 78, 2719—2726 |
| [12] | Callis C. F., van Wazer J. R., Arvan P. G., Chemical Reviews, 1954, 54, 777—796 |
| [13] | Macdonald J. C., Mazurek M., Journal of Magnetic Rreonance, 1987, 72, 48—60 |
| [14] | Glonek T., Costello J. R., Myers T. C., van Wazer J. R., The Journal of Physical Chemistry, 1975, 79(12), 1214—1218 |
| [15] | Wang Q. C., Yang Y. J., Inorganic Chemicals Industry, 2005, 32(12), 53—56 |
| (王清才, 杨荣杰. 无机盐工业, 2005, 32(12), 53—56) | |
| [16] | Greenwood N. N., Earnshaw A., Chemistry of the Elements, 2nd Ed., Butterworth-Heinermann, Oxford and Boston, 1997, 473—531 |
| [17] | Waerstad K. R., Mcclellan G., Food Chemistry, 1976, 24(2), 412—415 |
| [18] | HG/T 2770-2008, Chemical Industry Standard of the People's Republic of China, Chemical Industry Press, Beijing, 2008 |
| (HG/T 2770-2008, 中华人民共和国行业标准, 北京: 化学工业出版社, 2008) | |
| [19] | Wang Q. C., Studies on Characterization of the Molecular Weight and Crystal Structure of Ammonium Polyphosphate, Beijing Institute of Technology, Beijing, 2006 |
| (王清才. 聚磷酸铵(APP)分子链表征及结晶结构研究, 北京: 北京理工大学, 2006) | |
| [20] | Deng Y. P., Characterization on Polymerization Degree of Long Chain Ammonium Polyphosphate, Beijing Institute of Technology, Beijing, 2015 |
| (邓银萍. 长链聚磷酸铵的聚合度表征研究, 北京: 北京理工大学, 2015) | |
| [21] | Van Wazer J. R., Callis C. F., Shoolery J. N., Jones R. C., J. Am. Chem. Soc., 1956, 78(22), 5715—5726 |
| [22] | Gao H.B., Zhang Z. F., Nuclear Magnetic Resonance Principle and Experimental Method, Wuhan University Press,Wuhan, 2008 |
| (高汉宾, 张振芳. 核磁共振原理与实验方法, 武汉: 武汉大学出版社, 2008) | |
| [23] | Crutchfield M. M., Callis C. V., Irani R. R., Roth G. C., Inorganic Chemistry, 1962, 1(4), 813—817 |
| [1] | 刘宜娜, 杨荣杰, HUWeiguo, 李定华. 高聚合度聚磷酸铵中特征磷的31P核磁共振分析[J]. 高等学校化学学报, 2020, 41(12): 2832. |
| [2] | 宋丽, 林家祥, 黄定海. 步进扫描差示扫描量热法研究不同链结构的聚乙烯类聚烯烃热力学特性[J]. 高等学校化学学报, 2019, 40(8): 1740. |
| [3] | 王娜, 杨菲, 张静, 方庆红. 卡拉胶包覆APP微球阻燃水性环氧树脂[J]. 高等学校化学学报, 2019, 40(2): 385. |
| [4] | 李霈, 付海, 赵欧, 来方, 陈仕梅, 梅贵友, 赵伟, 班大明. 聚磷酸酯阻燃剂复配聚磷酸铵对环氧树脂阻燃性能的影响[J]. 高等学校化学学报, 2017, 38(2): 294. |
| [5] | 彭艺芳, 王承健, 王晶晶, 李玲梅, 晋万军, 强珊, 师红丹, 张英, 黄琳娟, 王仲孚. 花生致敏糖蛋白Ara h1糖链决定簇的质谱分析[J]. 高等学校化学学报, 2016, 37(9): 1622. |
| [6] | 刘建超, 许苗军, 李斌. 聚磷酸铵的疏水改性及聚丙烯阻燃性能[J]. 高等学校化学学报, 2015, 36(6): 1228. |
| [7] | 周丽娟, 张秀芹, 李荣波, 苏允兰, 赵莹, 王笃金, 徐端夫. 等规聚丙烯在γ射线辐照下的链结构和结晶行为[J]. 高等学校化学学报, 2012, 33(04): 850. |
| [8] | 陈文彦 刘心纯 刘够生 于建国. 网状结构结晶V型聚磷酸铵的合成与表征[J]. 高等学校化学学报, 2010, 31(12): 2494. |
| [9] | 王美玲, 臧洪俊, 蔡白雪, 程博闻. 纤维素在离子液体[AMMor]Cl/[AMIM]Cl混合溶剂中的溶解性能[J]. 高等学校化学学报, 2009, 30(7): 1469. |
| [10] | 郭立颖, 史铁钧, 李忠, 段衍鹏, 王于刚. 新型功能化离子液体[HeEIM]Cl的合成及其对棉纤维的溶解性能[J]. 高等学校化学学报, 2008, 29(9): 1901. |
| [11] | 林梅钦, 董朝霞, 李明远, 吴肇亮 . 低浓度HPAM/AlCit交联体系的27Al NMR研究[J]. 高等学校化学学报, 2007, 28(8): 1573. |
| [12] | 李东华, 徐晓明, 周文婷, 韩国彬 . 季铵盐型Gemini表面活性剂的胶束化动力学研究[J]. 高等学校化学学报, 2006, 27(10): 1927. |
| [13] | 孔旭新, 徐昆, 王丕新, 陈群. 淀粉-丙烯酸钠接枝共聚物的固体高分辨核磁共振研究[J]. 高等学校化学学报, 2005, 26(9): 1735. |
| [14] | 高红昌, 毛诗珍, 袁汉珍, 杜有如. 三氟乙酸溶液中尼龙6链凝聚缠结的NMR弛豫研究[J]. 高等学校化学学报, 2004, 25(8): 1555. |
| [15] | 周雪琴, 许晶, 赵晓东, 张志慧, 姚康德, 刘东志. 氮酮对鼠角质层角蛋白作用的光谱证据[J]. 高等学校化学学报, 2004, 25(7): 1273. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||