

高等学校化学学报 ›› 2021, Vol. 42 ›› Issue (11): 3367.doi: 10.7503/cjcu20210460
收稿日期:2021-07-01
出版日期:2021-11-10
发布日期:2021-09-10
通讯作者:
王雪强
E-mail:wangxq@hnu.edu.cn
基金资助:Received:2021-07-01
Online:2021-11-10
Published:2021-09-10
Contact:
WANG Xueqiang
E-mail:wangxq@hnu.edu.cn
Supported by:摘要:
核酸适体被称为“化学抗体”, 具有与抗体类似或更加优异的特异性和亲和力, 可以精准地靶向靶蛋白, 与靶蛋白特异性结合. 此外, 核酸适体还具有获取简单、 合成简便、 易于进行化学修饰、 不易变性、 靶标范围广、 免疫原性低及细胞内化快等优点, 已被广泛应用于众多研究领域. 在癌症治疗领域, 核酸适体作为一种优异的靶向识别工具和药物递送载体, 可实现抗肿瘤药物的精准递送. 将核酸适体与药物分子偶联, 可通过核酸适体的靶向作用使药物分子随核酸适体共同进入靶细胞, 实现药物分子在靶细胞内的富集, 进而促进靶细胞的死亡. 近年来, 核酸适体偶联药物已成为癌症靶向治疗的前沿新兴领域, 希望通过该领域的深入研究为癌症靶向治疗领域提供新思路. 本文综合评述了以生物偶联技术构建的核酸适体偶联药物及其应用研究.
中图分类号:
TrendMD:
赵卓, 王雪强. 核酸适体偶联药物的生物偶联构建技术与应用. 高等学校化学学报, 2021, 42(11): 3367.
ZHAO Zhuo, WANG Xueqiang. Investigations upon the Bioconjugation-based Construction Technologies and Applications of Aptamer-drug Conjugates. Chem. J. Chinese Universities, 2021, 42(11): 3367.
| Property | Aptamer | Antibody |
|---|---|---|
| Recognition pattern | Surface recognition Combining van der Waals forces, hydrogen bonds, and electrostatic interactions Reversal of activity by complementary antidote oligonucleotides | Bind pocket(key lock model) Three?dimensional interaction. recognizing epitopes loca? ted on the target antigen |
| Affinity | Strong affinity Multivalent aptamer with enhanced affinity and additional features | Strong affinity Related to the number of the same epitope on the target antigen |
| Specificity | High specificity Identifying single point mutations and conformational isomers | High specificity Different antibodies may bind to different epitopes of the same antigen |
| Method of obtaining | In vitro SELEX(2—15 rounds of selection) Available in about 2—8 weeks | Living biological system About 6 months or longer |
| Production and cost | Chemical solid phase synthesis Controllable and complete extracorporeal procedures 2 days for milligrams; 2 weeks for grams No or low pollution risk Low cost; high cost of specially modified long RNA(>60nt) | Production in animals Potential contamination in cell or animal production process 5—20 grams for 3 months From mammalian cells: high cost From genetically modified plants or animals: low cost |
| Physical and thermal stability | Very stable, long shelf life High temperature resistance(even up to 95 °C) and denatu? ration and renaturation cycles Long?term storage and transportation | Unstable and short shelf life Easily affected by temperature(even at room temperature or 37 °C), irreversible Refrigerated storage and transportation |
Table 1 Comparison of important parameters between aptamer and antibody
| Property | Aptamer | Antibody |
|---|---|---|
| Recognition pattern | Surface recognition Combining van der Waals forces, hydrogen bonds, and electrostatic interactions Reversal of activity by complementary antidote oligonucleotides | Bind pocket(key lock model) Three?dimensional interaction. recognizing epitopes loca? ted on the target antigen |
| Affinity | Strong affinity Multivalent aptamer with enhanced affinity and additional features | Strong affinity Related to the number of the same epitope on the target antigen |
| Specificity | High specificity Identifying single point mutations and conformational isomers | High specificity Different antibodies may bind to different epitopes of the same antigen |
| Method of obtaining | In vitro SELEX(2—15 rounds of selection) Available in about 2—8 weeks | Living biological system About 6 months or longer |
| Production and cost | Chemical solid phase synthesis Controllable and complete extracorporeal procedures 2 days for milligrams; 2 weeks for grams No or low pollution risk Low cost; high cost of specially modified long RNA(>60nt) | Production in animals Potential contamination in cell or animal production process 5—20 grams for 3 months From mammalian cells: high cost From genetically modified plants or animals: low cost |
| Physical and thermal stability | Very stable, long shelf life High temperature resistance(even up to 95 °C) and denatu? ration and renaturation cycles Long?term storage and transportation | Unstable and short shelf life Easily affected by temperature(even at room temperature or 37 °C), irreversible Refrigerated storage and transportation |
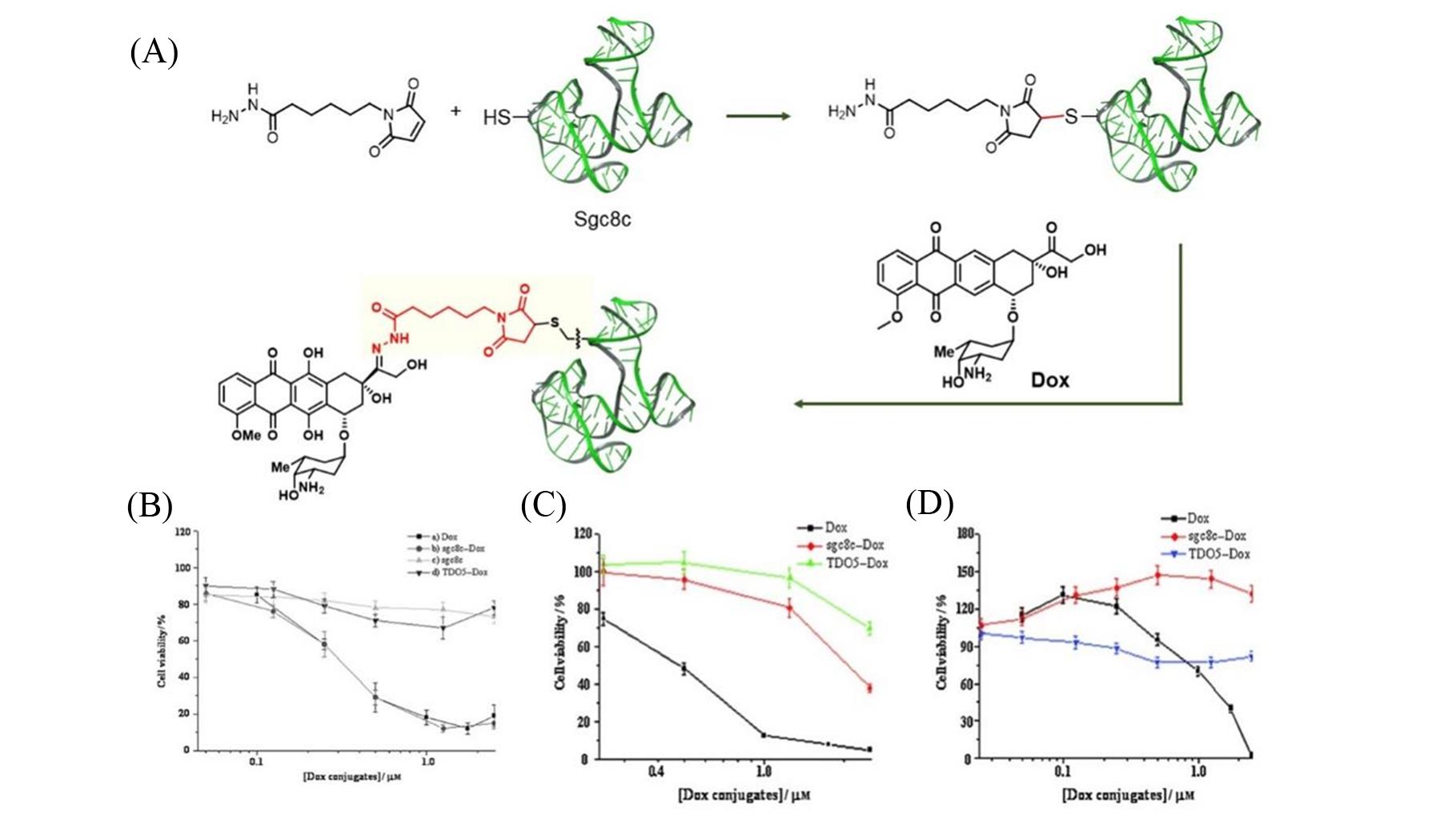
Fig.3 Research on the construction of conjugated drugs based on sulfhydryl modified nucleic acid aptamers and targeted drug delivery in vitro[68](A) The construction route of Sgc8c?Dox conjugate; cytotoxicity assays of CCRF?CEM cells(B), NB?4 cells(C) and Ramos cells(D). Copyright 2009, WILEY?VCH Verlag GmbH & Co. KGaA, Weinheim.
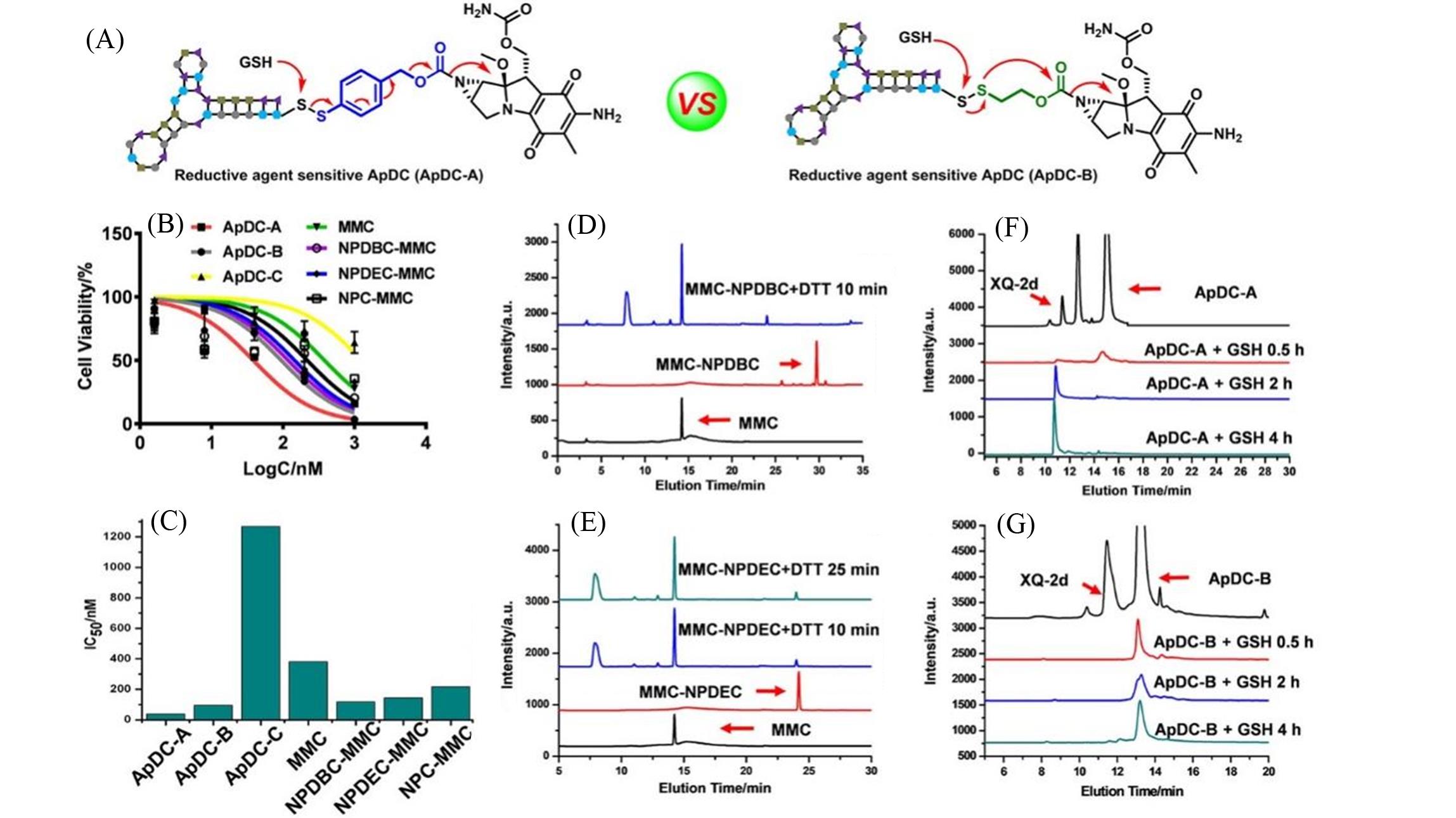
Fig.4 The effect of different linkers used for aptamer?MMC conjugates on the cytotoxicity of MMC to PL45 cells[74](A) The structure of reductive agent sensitive ApDC; (B) the toxicity test of ApDC with different linkers on PL45; (C) the IC50 value of ApDC; release rate of MMC from MMC?linkers NPDBC(D) and NPDEC(E) under the reduction of DTT characterized by HPLC; release rate of aptamer from ApDC?A(F) and ApDC?B(G) in the presence of GSH.Copyright 2020, American Chemical Society.
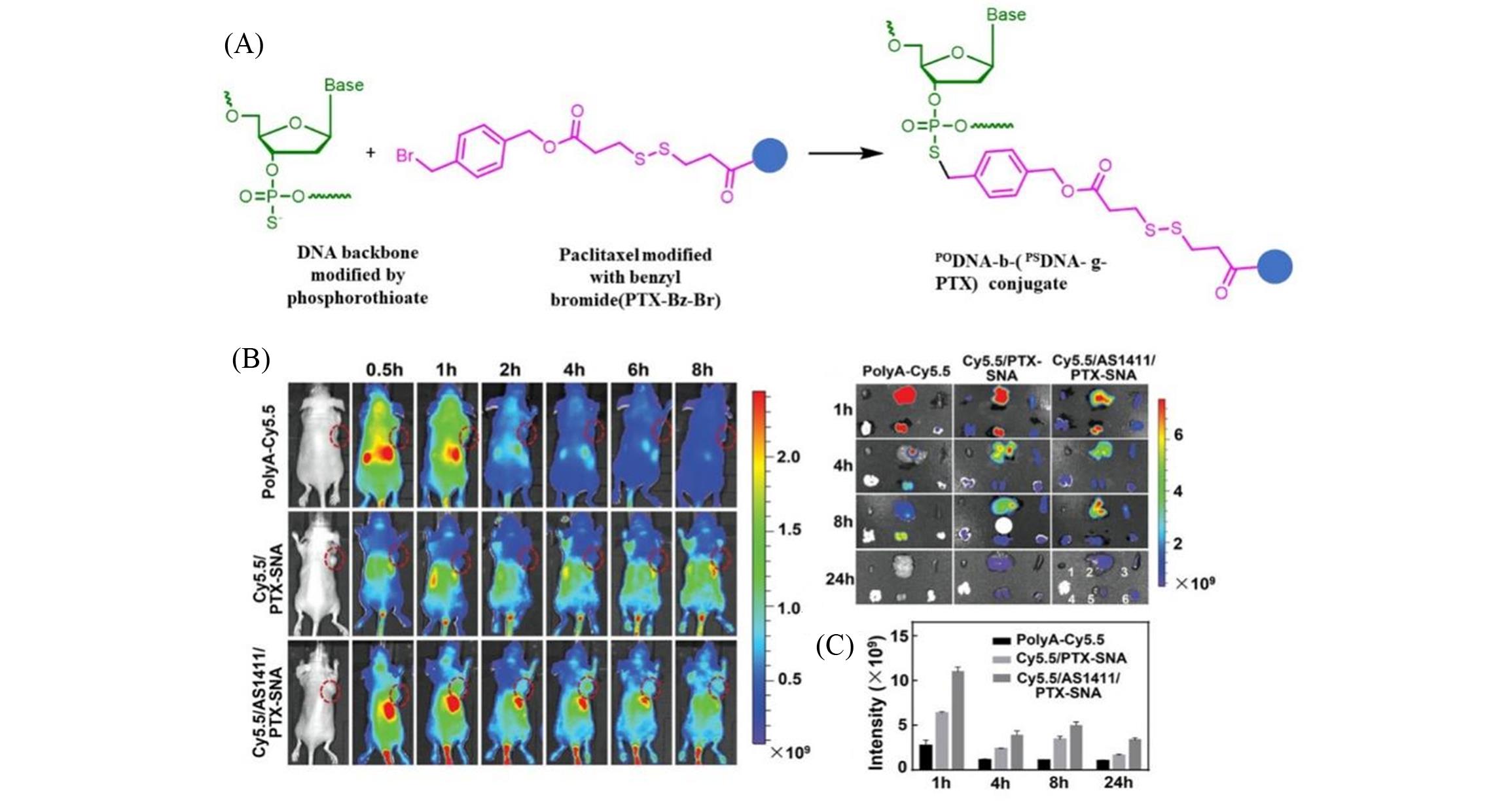
Fig.5 Thio?aptamer conjugated drugs, tetrahedral nano?drug research[76](A) The structure of the drug molecule; (B) in vivo tumor targeting and biodistribution of Cy5.5/PTX?SNA and Cy5.5/AS1411/PTX?SNA; (C) semi?quantitative analysis of fluorescent signals in tumors at different time points.Copyright 2019, WILEY?VCH Verlag GmbH & Co. KGaA, Weinheim.
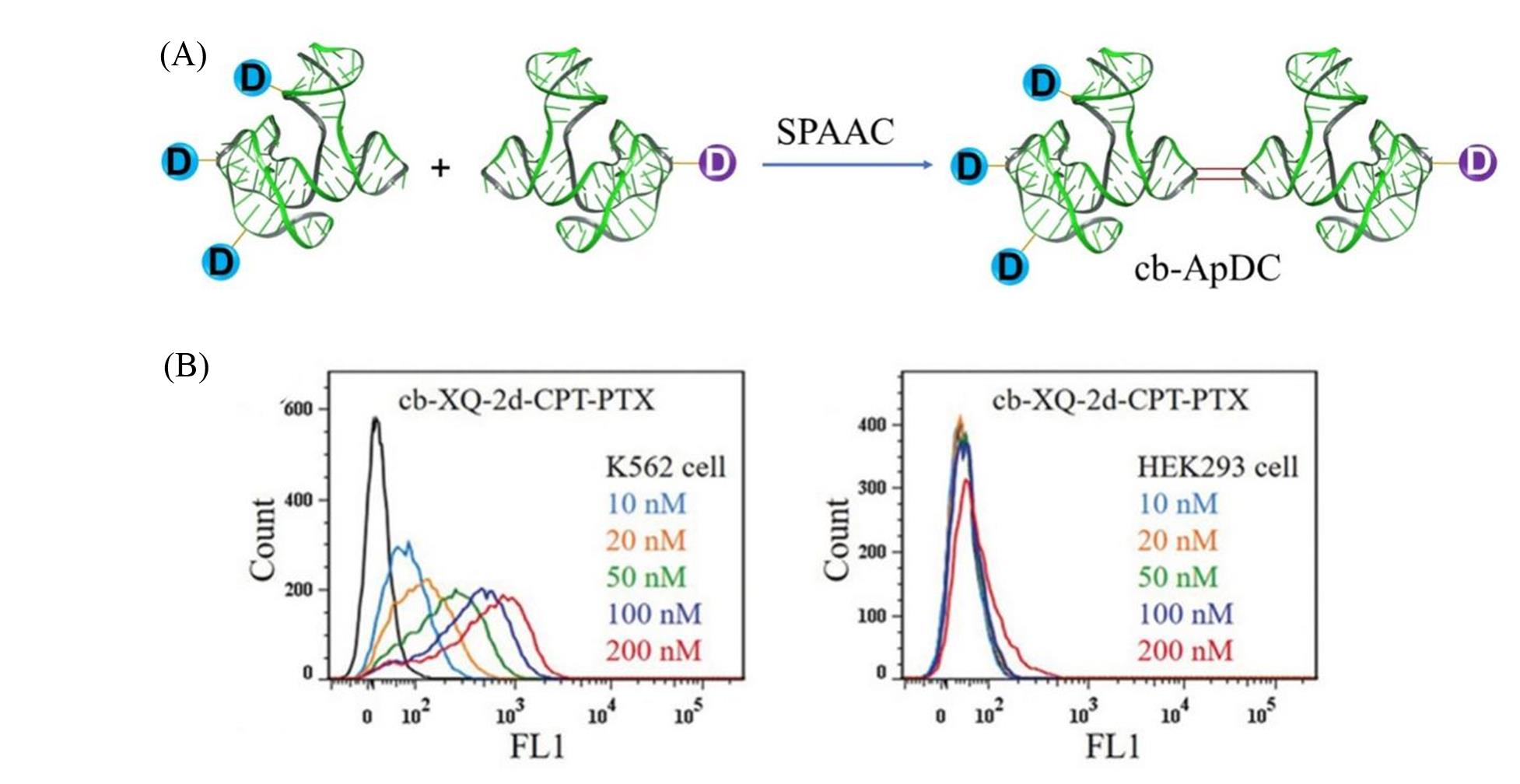
Fig.6 Cell experimental research on conjugated drugs of cyclic bivalent nucleic acid aptamer(cb?ApDC)[78](A) Construction of circular bivalent nucleic acid aptamer conjugated drugs; (B) the binding affinity of FAM?modified cb?XQ?2d?CPT?PTX for K562 cancer cells and HEK293 cells. Copyright 2019, Wiley?VCH Verlag GmbH & Co. KGaA, Weinheim.
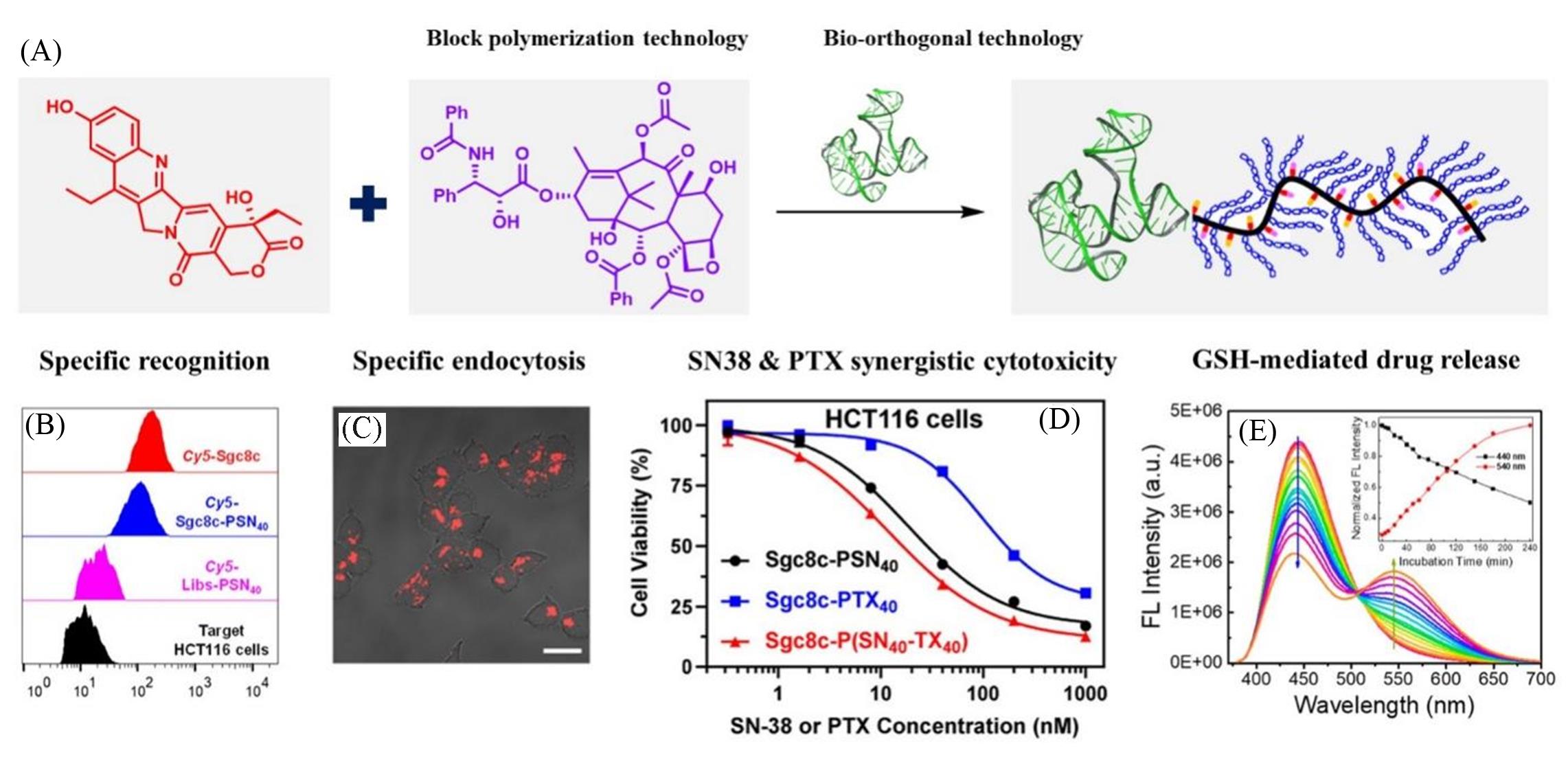
Fig.7 Targeted therapy research on the coupling of nucleic acid aptamers to polymerized drugs[79](A) The construction of Aptamer-Polyprodrug Conjugate(ApDC); (B) flow cytometry measurements of target HCT116 upon incubation with Cy5-Sgc8c, Cy5-Sgc8c-PSN40 and Cy5-Libs-PSN40; (C) representative confocal laser scanning microscopy(CLSM) images(scale bar: 20 μm), HCT116 upon incubation with Cy5-Sgc8c-PSN40 for 2 h at 37 °C.; (D) relative cell viabilities of Sgc8c-PSN40, Sgc8c-PTX40, and Sgc8c-P(SN40-TX40) against HCT116 cells, respectively; (E) time-dependent variations of fluorescence spectra(λex=365 nm) and normalized fluorescence intensity(inset, λem=440 and 540 nm) for Sgc8c-PSN40(1 μmol/L of DNA) upon incubation with GSH(10 mmol/L) in DPBS buffer(20 mmol/L, pH=7.4).Copy right 2020, American Chemical Society.
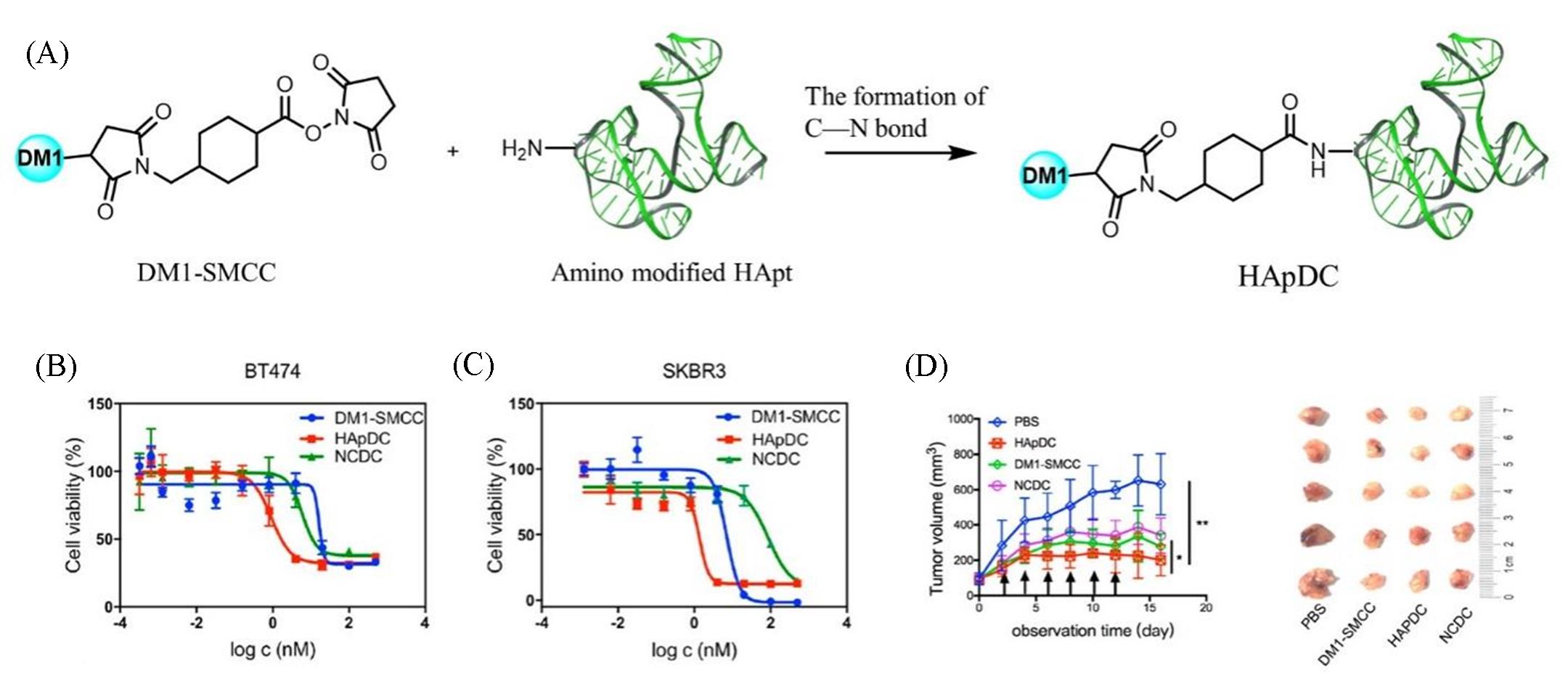
Fig.8 HER2 targeted cancer therapy study on nucleic acid aptamer maytansine conjugates[81](A) The construction of HER2 targeted DNA aptamer?DM1 conjugate(HApDC); cell viabilities of BT474(B) and SKBR3(C) after 72 h of treatment of HApDC, NCDC, and DM1?SMCC, evaluated via CCK8 assay; (D) analysis of tumor size every 2 days, along with the intravenous injection treatment. And tumors in each group were collected and viewed microscopically.Copyright 2020, American Chemical Society.
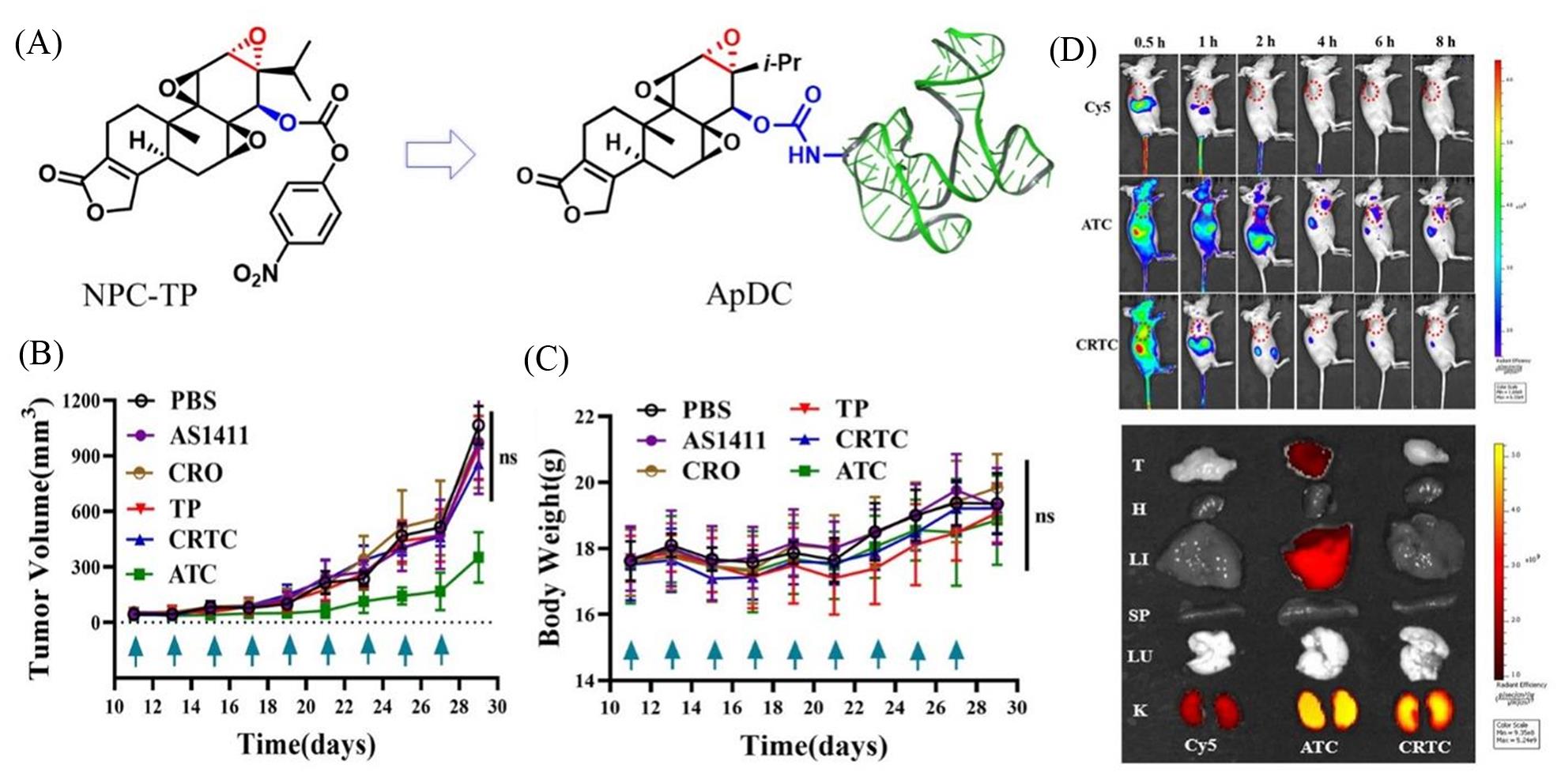
Fig.9 Research on the treatment of triple?negative breast cancer with the nucleic acid aptamer triptolide conjugated drugs[82](A) The construction of AS1411?triptolide conjugate(ATC); (B) analysis of tumor volume during 18 d of different treatments at a dosing frequency of every other day via intravenous injection(CRO, cytosine?rich oligonucleotide as the control sequence for AS1411; TP, triptolide; CRTC, CRO?triptolide); (C) analysis of body weight during 18 d of different treatments at a dosing frequency of every other day via intravenous injection; (D) in vivo distribution of Cy5, Cy5?modified ATC, and CRTC and distribution of Cy5, Cy5?modified ATC, and CRTC in the tumor(T) and major viscera(H, heart; LI, liver; SP, spleen; LU, lung; and K, kidney) at organ level 8 h post intravenous injection.Copyright 2020, American Chemical Society.
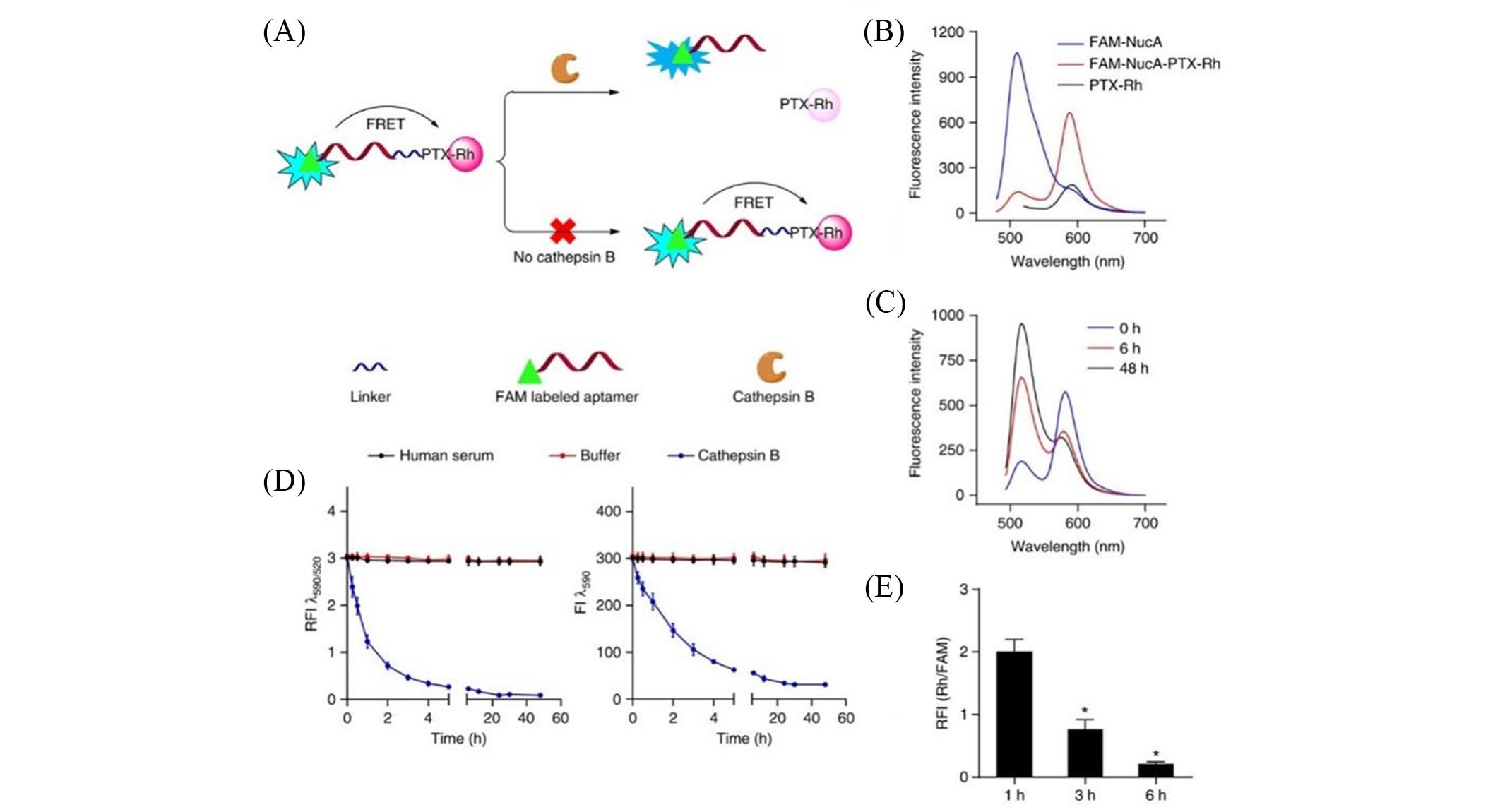
Fig.10 Study on aptamer paclitaxel conjugated drugs in ovarian cancer[83](A) Schematic diagram of the utilization of FRET in FAM?NucA?PTX?Rh for tracking the rupture of the cathepsin B?labile linker; (B) fluorescence emission spectra of FAM?NucA?PTX?Rh, FAM?NucA and PTX?Rh(λex=470 nm); (C) fluorescence emission spectra of FAM?NucA?PTX?Rh(λex=470 nm) in the presence of cathepsin B upon time; (D) the fluorescence analysis of FAM?NucA?PTX?Rh in human serum and buffer(pH=5.0, containing DTT) without or with cathepsin B upon time; (E) the intracellular release of PTX?Rh from FAM?NucA?PTX?Rh in SKOV3 cells monitored by flow cytometry. Copyright 2017, Springer Nature.
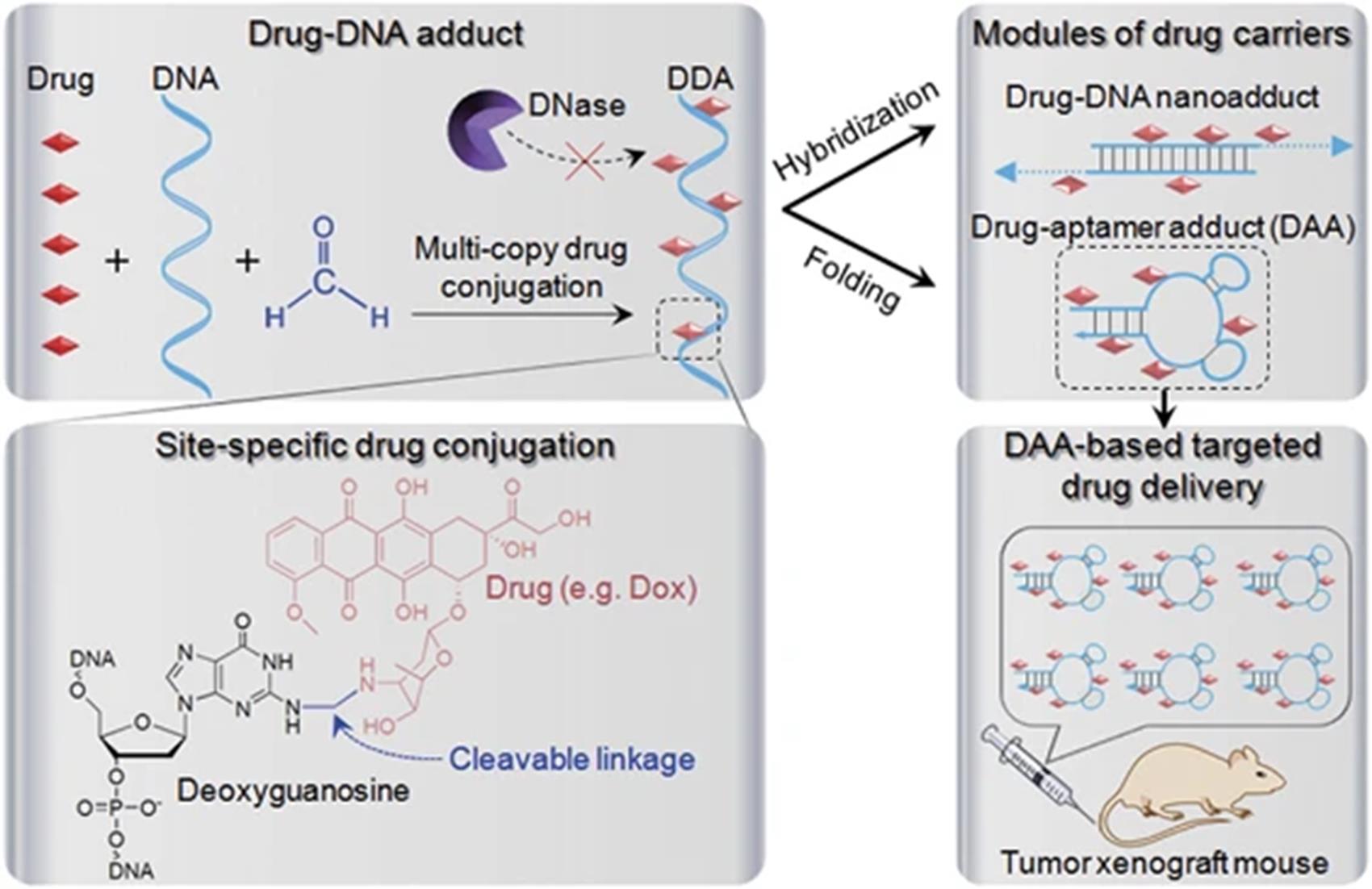
Fig.11 Research on aptamer Dox conjugated drugs[84]Multiple copies of drugs(e.g., doxorubicin) were specifically conjugated on deoxyguanosine of DNA, making it programmable to design DNA and drugs for drug?DNA adduct?based drug delivery.Copyright 2015, Springer Nature.
| 1 | Airley R., Cancer Chemotherapy. Wiley-Blackwell, 2009, ISBN 978⁃0⁃470⁃09254⁃5 |
| 2 | Elad S., Zadik Y., Hewson I., Hovan A., Correa M. E. P., Logan R., Elting L. S., Spijkervet F. K. L., Brennan M. T., Support. Care Cancer, 2010, 18(8), 993—1006 |
| 3 | Schwartz R. S., N. Engl. J. Med., 2004, 350(11), 1079—1080 |
| 4 | Imai K., Takaoka A., Nat. Rev. Cancer, 2006,6(9),714—727 |
| 5 | Schuler M., Wu Y. L., Hirsh V., O’Byrne K., Yamamoto N., Mok T., Popat S., Sequist L. V., Massey D., Zazulina V., Yang J. C. H., J. Thorac. Oncol., 2016, 11(3), 380—390 |
| 6 | Noronha V., Patil V. M., Joshi A., Menon N., Chougule A., Mahajan A., Janu A., Purandare N., Kumar R., More S., J. Clin. Oncol., 2020, 38,(2), 124—136 |
| 7 | Bachelot T., Romieu G., Campone M., Diéras V., Cropet C., Dalenc F., Jimenez M., Rhun E. L., Pierga J. Y., Gonçalves A., Lancet Oncol., 2013, 14, 64—71 |
| 8 | Rizvi N. F., Smith G. F., Bioorg. Med. Chem. Lett., 2017, 27(23), 5083—5088 |
| 9 | Mrinal M. Gounder M. M., Mahoney M. R., Tine B. A. T., Vinod Ravi V., Steven Attia S., Deshpande H. A., Abha A., Gupta A. A., Milhem M. M., N. Engl. J. Med., 2018, 379(25), 2417—2428 |
| 10 | Obradovic M., Mrhar A., Kos M., Clin. Thera., 2009, 31(12), 2940—2952 |
| 11 | Shan L. L., Liu M., Wu C., Zhao L., Li S. W., Xu L. S., Cao W. G., Gao G. Z., Gu Y. Q., Int. J. Nanomedicine., 2015, 10, 5571—5591 |
| 12 | Lazo J. S., McQueeney K. E., Burnett J. C., Wipf P., Sharlow E. R., Int. J. Biochem. Cell Biol., 2018, 96, 171—181 |
| 13 | Murugan R. N., Park J. E., Kim E. H., Shin S. Y., Cheong C., Lee K. S., Bang J. K., Mol. Cells, 2011, 32(3), 209—220 |
| 14 | Ohnmacht S. A., Neidle S., Bioorg. Med. Chem. Lett., 2014, 24(12), 2602—2612 |
| 15 | Lu Y. H., Gao X. Q., Wu M., Zhang⁃Negrerie D., Gao Q., Mini. Rev. Med. Chem., 2011, 11(7), 611—624 |
| 16 | Cazzamalli S., Ziffels B., Widmayer F., Murer P., Pellegrini G., Pretto F., Wulhfard S., Neri D., Clin. Cancer Res., 2018, 24(15), 3656—3667 |
| 17 | Wang Y., Cheetham A. G., Angacian G., Su H., Xie L. S., Cui H. G., Adv. Drug Deliv. Rev., 2017, 110, 112—126 |
| 18 | Barve A., Jain A., Liu H., Jin W., Cheng K., Nanomedicine: NBM, 2016, 12(8), 2373—2381 |
| 19 | Hoppenz P., Els⁃Heindl S., Beck⁃Sickinger A. G., Front. Chem., 2020, 8, 571 |
| 20 | Burns K. E., Hensley H., Robinson M. K., Thévenin D., Mol. Pharmaceutics, 2017, 14, 2, 415—422 |
| 21 | Kulhari H., Pooja D., Shrivastava S., Telukutala S. R., Barui A. K., Patra C. R., Vegi G. M. N., Adams D. J., Sistla R., Nanomedicine: NBM, 2015, 11(6), 1511—1520 |
| 22 | Dissanayake S., Denny W.A., Gamage S., Sarojini V., J. Control. Release, 2017, 250, 62—76 |
| 23 | Cox N., Kintzing J. R., Smith M., Grant G. A., Cochran J. R., Angew. Chem. Int. Ed., 2016, 55(34), 9894—9897 |
| 24 | Li S. T., Zhao H. L., Mao X. X., Fan Y. F., Liang X. J., Wang R. X., Xiao L. J., Wang J. P., Liu Q., Zhao G. Q., Pharm. Res., 2019, 36(12), 168 |
| 25 | Vrettos E. I., Mező G., Tzakos A. G., Beilstein J. Org. Chem., 2018,14, 930—954 |
| 26 | Saunders L. R., Bankovich A. J., Anderson W. C., Aujay M. A., Bheddah S., Black K., Desai R., Escarpe P. A., Hampl J., Laysang A., Sci. Transl. Med., 2015, 7(302), 302ra136 |
| 27 | Lambert J. M., Chari R. V. J., J. Med. Chem., 2014, 57(16), 6949—6964 |
| 28 | Cardillo T. M., Govindan S. V., Sharkey R. M., Trisal P., Goldenberg D. M., Clin. Cancer Res., 2011, 17(10), 3157—3169 |
| 29 | Shor B., Gerber H. P., Sapra P., Mol. Immunol., 2015, 67(2), 107—116 |
| 30 | Faltas B., Goldenberg D. M., Ocean A. J., Govindan S. V., Wilhelm F., Sharkey R. M., Hajdenberg J., Hodes G., Nanus D. M., Tagawa S. T., Clin. Genitourin. Cancer, 2016, 14(1), E75—E79 |
| 31 | Kamath A. V., Iyer S., Pharm. Res., 2015, 32(11), 3470—3479 |
| 32 | Chen W. H., Sun Z., Lu L. H., Angew. Chem. Int. Ed., 2021, 60(11), 5626—5643 |
| 33 | Thomas A., Teicher B. A., Hassan R. T., Lancet Oncol., 2016, 17(6), E254—E262 |
| 34 | Lambert J. M., Morris C. Q., Adv. Ther., 2017, 34(5), 1015—1035 |
| 35 | Hammood M., Craig A. W., Leyton J. V., Pharmaceuticals, 2021, 14(7), 674 |
| 36 | Kalim M., Chen J., Wang S. H., Lin C. Y., Ullah S., Liang K. Y., Ding Q., Chen S. Q., Zhan J. B., Drug Des. Dev. Ther., 2017, 11, 2265—2276 |
| 37 | Lv C., Yang C., Ding D., Sun Y., Wang R. W., Han D., Tan W. H., Anal. Chem., 2019, 91(21), 13818—13823 |
| 38 | Boswell C. A., Mundo E. E., Zhang C., Bumbaca D., Valle N. R., Kozak K. R., Fourie A., Chuh J., Koppada N., Saad O., Bioconjugate Chem., 2011, 22(10), 1994—2004 |
| 39 | de Goeij B. E. C. G., Satijn D., Freitag C. M., Wubbolts R., Bleeker W. K., Khasanov A., Zhu T., Chen G., Miao D., van Berkel P. H.C., Mol. Cancer Ther., 2015, 14(5), 1130—1140 |
| 40 | Beck A., Goetsch L., Dumontet C., Corvaïa N., Nat. Rev. Drug Discov., 2017, 16(5), 315—337 |
| 41 | Ellington A. D., Szostak J. W., Nature, 1990, 346(6287), 818—822 |
| 42 | Tuerk C., Gold L., Science, 1990, 249(4968), 505—510 |
| 43 | Dunn M. R., Jimenez R. M., Chaput J. C., Nat. Rev. Chem., 2017, 1(10), 0076 |
| 44 | Wen W., Huang D. D., Bao J. X., Zhang J. Z. H., Chem. J. Chinese Universities, 2021, 42(7), 2161—2169(温炜, 黄达锭, 鲍劲霄, 张增辉. 高等学校化学学报, 2021,42(7), 2161—2169) |
| 45 | Wang R. W., Zhu G. Z., Mei L., Xie Y., Ma H. B., Ye M., Qing F. L., Tan W. H., J. Am. Chem. Soc., 2014, 136(7), 2731—2734 |
| 46 | Cai S. D., Yan J. H., Xiong H. J., Liu Y. F., Peng D. M., Liu Z. B., Analyst, 2018, 143(22), 5317—5338 |
| 47 | Li L., Xu S. J., Yan H., Li X. W., Yazd H. S., Li X., Huang T., Cui C., Jiang J. H., Tan W. H., Angew. Chem. Int. Ed., 2021, 60(5), 2221—2231 |
| 48 | Nan M. N., Bi Y., Xue H. L., Long H. T., Xue S. L., Pu L. M., Prusky D., Food Chem., 2021, 337, 127761 |
| 49 | Gao L., Wang H. X., Deng Z. B., Xiang W. W., Shi H. F., Xie B., Shi H. X., New J. Chem., 2020, 44(6), 2571—2577 |
| 50 | Duan C. J., Jiao J., Zheng J., Li D. Y., Ning L. M., Xiang Y., Li G. X., Anal. Chem., 2020, 92(22), 15162—15168 |
| 51 | Fu B. S., Park Y., Kim K. T., Chen K., Zou G. R., Wei Q., Peng S., Chen Y. Q., Kim B. H., Zhou X., Chem. Commun., 2018, 54(81), 11487—11490 |
| 52 | Wu Z., Tang L. J., Zhang X. B., Jiang J. H., Tan W. H., ACS Nano, 2011, 5(10), 7696—7699 |
| 53 | Xiong H. J., Yan J. H., Cai S. D., He Q. Y., Peng D. M., Liu Z. B., Liu Y. F., Int. J. Biol. Macromol., 2019, 132, 190—202 |
| 54 | Dong Q., Li Z. Q., Peng T. H., Chen Z., Tan W. H., Chem. J. Chinese Universities, 2020, 41(12), 2648—2657(董倩, 李兆倩, 彭天欢, 陈卓, 谭蔚泓. 高等学校化学学报, 2020,41(12), 2648—2657) |
| 55 | Shangguan D., Li Y., Tang Z. W., Cao Z. H. C., Chen H. W., Mallikaratchy P., Sefah K., Yang C. Y. J., Tan W. H., Proc. Natl. Acad. Sci. USA, 2006, 103(32), 11838—11843 |
| 56 | Tan W. H., Donovan M. J., Jiang J. H., Chem. Rev., 2013, 113(4), 2842—2862 |
| 57 | Zhou J. H., Rossi J., Nat. Rev. Drug Discov., 2017, 16(3), 181—202 |
| 58 | Ni X., Castanares M., Mukherjee A., Lupold S.E., Curr. Med. Chem., 2011, 18(27), 4206—4214 |
| 59 | Moreno A., Pitoc G. A., Ganson N. J., Layzer J. M., Hershfield M. S., Tarantal A. F., Sullenger B. A., Cell Chem. Biol., 2019, 26(5), 634—644 |
| 60 | Querques G., Bux A. V., Martinelli D., Iaculli C., Noci N. D., Acta Ophthalmol., 2009, 87(6), 623—630 |
| 61 | Gold L., Janjic N., Jarvis T., Schneider D., Walker J. J., Wilcox S. K. Zichi D., Cold Spring Harb. Perspect. Biol., 2012, 4(3), a003582 |
| 62 | Zhou W. H., Ding J. S., Liu J. W., Theranostics, 2017, 7(4), 1010—1025 |
| 63 | McConnell E. M., Cozma I., Mou Q. B., Brennan J. D., Lu Y., Li Y. F., Chem. Soc. Rev., 2021,50(16), 8954—8994 |
| 64 | Yan J. H., Xiong H. J., Cai S. D., Wen N. C., He Q. Y., Liu Y. F., Peng D. M., Liu Z. B., Talanta, 2019, 200, 124—144 |
| 65 | Meng H. M., Fu T., Zhang X. B., Tan W. H., Natl. Sci. Rev., 2015, 2(1), 71—84 |
| 66 | Nimjee S. M., White R. R., Becker R. C., Sullenger B. A., Annu. Rev. Pharmacol. Toxicol., 2017,57, 61—79 |
| 67 | Liu J., Wu X. H., Lu J., Huang G. X., Dang L., Zhang H. R., Zhong C. X., Zhang Z. K., Li D. J., Li F. F., Nat. Aging, 2021, 1(4), 368—384 |
| 68 | Huang Y. F., Shangguan D. H., Liu H. P., Phillips J. A., Zhang X. L., Chen Y., Tan W. H., ChemBioChem, 2009, 10(5), 862—868 |
| 69 | Chen K., Liu B., Yu B., Zhong W., Lu Y., Zhang J. N., Liao J., Liu J., Pu Y., Qiu L. P., Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol., 2017, 9(3), e1438 |
| 70 | Liu M. T., Ma W. J., Li Q. S., Zhao D., Shao X. R., Huang Q., Hao L. Y., Lin Y. F., Cell Proliferat., 2019, 52(1), e12511 |
| 71 | Leitner M., Poturnayova A., Lamprecht C., Weich S., Snejdarkova M., Karpisova I., Hianik T., Ebner A., Anal. Bioanal. Chem., 2017, 409(11), 2767—2776 |
| 72 | Poturnayová A., Buríková M., Bízik J., Hianik T., ChemPhysChem, 2019, 20(4), 545—554 |
| 73 | Luo Y. L., Shiao Y. S., Huang Y. F., ACS Nano, 2011, 5(10), 7796—7804 |
| 74 | Yang Q. X., Deng Z. Y., Wang D., He J. X., Zhang D. L., Tan Y., Peng T. H., Wang X. Q., Tan W. H., J. Am. Chem. Soc., 2020, 142(5), 2532—2540 |
| 75 | Li Y., Wang X. L., Gao L., Hu P., Jiang L. C., Ren T. Y., Fu R., Yang D. W., Jiang X. H., J. Mater. Sci., 2018, 53(20), 14138—14148 |
| 76 | Guo Y. Y., Zhang J., Ding F., Pan G. F., Li J., Feng J., Zhu X. Y., Zhang C., Adv. Mater., 2019, 31(16), 1807533 |
| 77 | Zhang J., Guo Y. Y., Ding F., Pan G. F., Zhu X. Y., Zhang C., Angew. Chem. Int. Ed., 2019, 58(39), 13794—13798 |
| 78 | Zhou F., Wang P., Peng Y. B., Zhang P. G., Huang Q., Sun W. D., He N. Y., Fu T., Zhao Z. L., Fang X. H., Tan W. H., Angew. Chem. Int. Ed., 2019, 58(34), 11661—11665 |
| 79 | Deng Z. Y., Yang Q. X., Peng Y. B., He J. X., Xu S. J., Wang D., Peng T. H., Wang R. W., Wang X. Q., Tan W. H., Bioconjugate Chem., 2020, 31(1), 37—42 |
| 80 | Ríos⁃Luci C., García⁃Alonso S., Díaz⁃Rodríguez E., Nadal⁃Serrano M., Arribas J., Ocaña A., Pandiella A., Cancer Res.,2017, 77(17), 4639—4651 |
| 81 | Tan Y., Peng Y. B., Ai L. L., Li Y. Y., Qu Y. X., Wang D., Su Y. Y., Deng T. G., Fu T., Zhao Z. L., Bioconjugate Chem., 2020, 31(7), 1766—1774 |
| 82 | He J. X., Peng T. H., Peng Y. B., Ai L. L., Deng Z. Y., Wang X. Q., Tan W. H., J. Am. Chem.Soc., 2020, 142(6), 2699—2703 |
| 83 | Li F. F., Lu J., Liu J., Liang C., Wang M. L., Wang L. Y., Li D. F., Yao H. Z., Zhang Q. L., Wen J., Nat. Commun., 2017, 8, 1390 |
| 84 | Zhu G. Z., Cansiz S., You M. X., Qiu L. P., Han D., Zhang L. Q., Mei L., Fu T., Chen Z., Tan W. H., NPG Asia Mater., 2015, 7, e169 |
| [1] | 柯梦婷, 袁江培, 张恒, 方煜. 多孔配位聚合物靶向亚细胞器用于生物成像和诊疗[J]. 高等学校化学学报, 2021, 42(11): 3295. |
| [2] | 董倩, 李兆倩, 彭天欢, 陈卓, 谭蔚泓. 核酸适体在癌症诊疗中的研究进展[J]. 高等学校化学学报, 2020, 41(12): 2648. |
| [3] | 丁吉勇, 沈洪辰, 刘夫锋. p53突变体Y220C小分子稳定剂的虚拟筛选[J]. 高等学校化学学报, 2016, 37(4): 706. |
| [4] | 付翠翠, 梁丽佳, 齐国华, 徐抒平, 徐蔚青. SERS生物传感技术及其应用进展[J]. 高等学校化学学报, 2015, 36(11): 2134. |
| [5] | 王海, 张超, 张琳华, 刘兰霞, 郑义, 朱敦皖. 载紫杉醇星型M-PLA-TPGS纳米颗粒的合成及其用于前列腺癌治疗药物载体的研究[J]. 高等学校化学学报, 2014, 35(10): 2239. |
| [6] | 梅素容, 蔡凌霜, 姚庆红, 邢钧, 许国旺, 吴采樱. 毛细管电泳-柱末安培检测癌症病人尿中8-羟基脱氧鸟苷[J]. 高等学校化学学报, 2003, 24(11): 1987. |
| [7] | 朱尔一, 王小如, 邓志威, 杨芃原, 黄本立. 多变量判别分析用于癌症诊断研究[J]. 高等学校化学学报, 1993, 14(5): 621. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
