

高等学校化学学报 ›› 2018, Vol. 39 ›› Issue (10): 2143.doi: 10.7503/cjcu20180224
收稿日期:2018-03-22
出版日期:2018-09-29
发布日期:2018-09-29
作者简介:联系人简介: 贲 腾, 男, 博士, 教授, 博士生导师, 主要从事有机多孔材料的制备与应用及手性高分子的制备与性能研究. E-mail:
基金资助:Received:2018-03-22
Online:2018-09-29
Published:2018-09-29
Contact:
BEN Teng
E-mail:tben@jlu.edu.cn
Supported by:摘要:
以天然海藻为原料, 通过自活化制备了碳材料SarCW-900-HF及LamCW-900-HF, 并对其进行表征和储甲烷能力测试. 其比表面积分别为812和671 m2/g, 孔体积分别为0.45和0.44 cm3/g, 孔分布的峰值均在1.1 nm, 在室温及3.5 MPa压强下储甲烷的能力分别为272 mg/g(质量比21.3%)和246 mg/g(质量比19.4%), 经20 MPa压强压片后吸附甲烷体积比达到274和449 cm3(STP)/cm3, 超过美国能源部提出的吸附剂的指标[263 cm3(STP)/cm3].
中图分类号:
TrendMD:
王昀, 贲腾. 海藻基微孔碳材料的储甲烷性能. 高等学校化学学报, 2018, 39(10): 2143.
WANG Yun,BEN Teng. Methane Storage Performace of Self-activated Seaweed-based Carbon Materials†. Chem. J. Chinese Universities, 2018, 39(10): 2143.
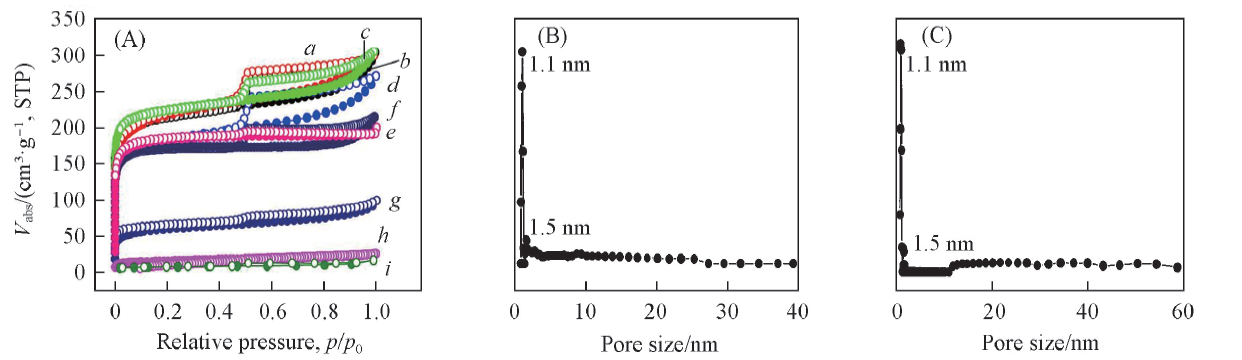
Fig.1 N2 sorption isotherms of different carbonized biomasses(A) and pore size distribution of SarCW-900-HF(B) and LamCW-900-HF(C)a. SarCW-900-HF; b. SarCW-850-HF; c. LamCW-900-HF; d. SarCW-950-HF; e. carbonized Walnut peel;f. LamCW-850-HF; g. LamCW-950-HF; h. carbonized passiflora edulia sims peel; i. Bentonite.

Fig.2 SEM images of Sar(A—E), Lam(F—J), SarCW-900-HF(K, L), LamCW-900-HF(O, P), TEM images of SarCW-900-HF(M, N) and LamCW-900-HF(Q, R)Insets of (N) and (R) are the SAED of the sample.
| Material | Surface area/ (m2·g-1) | Pore volume/ (cm3·g-1) | Condition | CH4 storage capacity (mass ratio,%) | Excess CH4 uptake/ (cm3·cm-3) | Heat of adsorption/ (kJ·mol-1) | Ref. | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| p/MPa | T/K | |||||||||||||||||
| Porous | COF-102 | 3620 | 1.55 | 3.5 | 298 | 15.8 | 120 | 8.6 | [ | |||||||||
| polymers | COF-103 | 3530 | 1.54 | 3.5 | 298 | 14.9 | 152 | 9.5 | [ | |||||||||
| HCP-1 | 1904 | 0.54 | 1.5 | 298 | 6.4 | [ | ||||||||||||
| HCP-4 | 1366 | 0.55 | 3.6 | 298 | 8.1 | [ | ||||||||||||
| PIM-1 | 850 | 0.349 | 1.5 | 293 | 3.8 | [ | ||||||||||||
| PPN-3 | 2840 | 1.7 | 3.5 | 295 | 12.2 | 15.2 | [ | |||||||||||
| PPN-4 | 6461 | 3.04 | 5.5 | 295 | 21.5 | [ | ||||||||||||
| MOFs | Cu2(sbtc)[PCN-11] | 1931 | 0.91 | 2.5 | 298 | 14.0 | 171 | 14.6 | [ | |||||||||
| Ni(dhtp)[NiMOF-74,CPO-27-Ni] | 1027 | 0.54 | 3.5 | 298 | 11.9 | 206 | 21.5—22 | [ | ||||||||||
| Zn4O(bdc)3[MOF-5] | 1870 | 3.6 | 13.5 | 110 | 12.2 | [ | ||||||||||||
| LIFM-82 | 1624 | 0.71 | 3.5 | 298 | 15.2 | 196 | 17.5 | [ | ||||||||||
| MOF-905 | 3490 | 1.34 | 3.5 | 298 | 18.8 | 145 | 11.7 | [ | ||||||||||
| HKUST-1 | 1850 | 0.78 | 3.5 | 298 | 18.4 | 227 | 17.0 | [ | ||||||||||
| NU-125 | 3120 | 1.29 | 3.5 | 298 | 16.5 | 182 | 15.1 | [ | ||||||||||
| PCN-14 | 2000 | 0.829 | 3.5 | 298 | 17.4 | 202 | 17.6 | [ | ||||||||||
| UTSA-76 | 2820 | 1.06 | 3.5 | 298 | 15.4 | 211 | 15.4 | [ | ||||||||||
| UTSA-20 | 1620 | 3.5 | 298 | 18.1 | 184 | [ | ||||||||||||
| NOTT-102 | 3342 | 1.268 | 3.5 | 298 | 16.5 | 181 | 16.0 | [ | ||||||||||
| NOTT-103 | 2958 | 1.157 | 3.5 | 300 | 15.6 | 193 | 15.9 | [ | ||||||||||
| MAF-38 | 2022 | 0.808 | 3.5 | 298 | 14.1 | 226 | 21.6 | [ | ||||||||||
| ZJU-35 | 2899 | 1.156 | 3.5 | 300 | 14.4 | 177 | [ | |||||||||||
| MIL-100(Cr) | 1900 | 1.1 | 3.5 | 11.7 | 144 | 19 | [ | |||||||||||
| Zeolites | CaX | 0.36 | 3.2 | 298 | 7.5 | 24.68 | [ | |||||||||||
| Porous | CMK-3 | 950 | 0.87 | 3.5 | 298 | 7.5 | 19.78 | [ | ||||||||||
| carbon | K-PAF-1-750 | 2926 | 3.5 | 298 | 17.1 | 17.9 | [ | |||||||||||
| material | Maxsorb A | 3100 | 4.0 | 298 | 16.4 | 152 | [ | |||||||||||
| KUA31752 | 3355 | 4.0 | 298 | 15.6 | 155 | [ | ||||||||||||
| PC | 1220 | 0.47 | 2.0 | 298 | 10.2 | 141 | [ | |||||||||||
| Saran A carbon 36X | 1650 | 3.2 | 298 | 11.5 | [ | |||||||||||||
| KF-1500 | 1500 | 3.2 | 298 | 7.6 | [ | |||||||||||||
| UlaC-950-HF | 1476 | 0.61 | 3.5 | 298 | 17 | 232 | 22.4 | [ | ||||||||||
Table 1 Summary of methane storage capacities of some porpus materials
| Material | Surface area/ (m2·g-1) | Pore volume/ (cm3·g-1) | Condition | CH4 storage capacity (mass ratio,%) | Excess CH4 uptake/ (cm3·cm-3) | Heat of adsorption/ (kJ·mol-1) | Ref. | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| p/MPa | T/K | |||||||||||||||||
| Porous | COF-102 | 3620 | 1.55 | 3.5 | 298 | 15.8 | 120 | 8.6 | [ | |||||||||
| polymers | COF-103 | 3530 | 1.54 | 3.5 | 298 | 14.9 | 152 | 9.5 | [ | |||||||||
| HCP-1 | 1904 | 0.54 | 1.5 | 298 | 6.4 | [ | ||||||||||||
| HCP-4 | 1366 | 0.55 | 3.6 | 298 | 8.1 | [ | ||||||||||||
| PIM-1 | 850 | 0.349 | 1.5 | 293 | 3.8 | [ | ||||||||||||
| PPN-3 | 2840 | 1.7 | 3.5 | 295 | 12.2 | 15.2 | [ | |||||||||||
| PPN-4 | 6461 | 3.04 | 5.5 | 295 | 21.5 | [ | ||||||||||||
| MOFs | Cu2(sbtc)[PCN-11] | 1931 | 0.91 | 2.5 | 298 | 14.0 | 171 | 14.6 | [ | |||||||||
| Ni(dhtp)[NiMOF-74,CPO-27-Ni] | 1027 | 0.54 | 3.5 | 298 | 11.9 | 206 | 21.5—22 | [ | ||||||||||
| Zn4O(bdc)3[MOF-5] | 1870 | 3.6 | 13.5 | 110 | 12.2 | [ | ||||||||||||
| LIFM-82 | 1624 | 0.71 | 3.5 | 298 | 15.2 | 196 | 17.5 | [ | ||||||||||
| MOF-905 | 3490 | 1.34 | 3.5 | 298 | 18.8 | 145 | 11.7 | [ | ||||||||||
| HKUST-1 | 1850 | 0.78 | 3.5 | 298 | 18.4 | 227 | 17.0 | [ | ||||||||||
| NU-125 | 3120 | 1.29 | 3.5 | 298 | 16.5 | 182 | 15.1 | [ | ||||||||||
| PCN-14 | 2000 | 0.829 | 3.5 | 298 | 17.4 | 202 | 17.6 | [ | ||||||||||
| UTSA-76 | 2820 | 1.06 | 3.5 | 298 | 15.4 | 211 | 15.4 | [ | ||||||||||
| UTSA-20 | 1620 | 3.5 | 298 | 18.1 | 184 | [ | ||||||||||||
| NOTT-102 | 3342 | 1.268 | 3.5 | 298 | 16.5 | 181 | 16.0 | [ | ||||||||||
| NOTT-103 | 2958 | 1.157 | 3.5 | 300 | 15.6 | 193 | 15.9 | [ | ||||||||||
| MAF-38 | 2022 | 0.808 | 3.5 | 298 | 14.1 | 226 | 21.6 | [ | ||||||||||
| ZJU-35 | 2899 | 1.156 | 3.5 | 300 | 14.4 | 177 | [ | |||||||||||
| MIL-100(Cr) | 1900 | 1.1 | 3.5 | 11.7 | 144 | 19 | [ | |||||||||||
| Zeolites | CaX | 0.36 | 3.2 | 298 | 7.5 | 24.68 | [ | |||||||||||
| Porous | CMK-3 | 950 | 0.87 | 3.5 | 298 | 7.5 | 19.78 | [ | ||||||||||
| carbon | K-PAF-1-750 | 2926 | 3.5 | 298 | 17.1 | 17.9 | [ | |||||||||||
| material | Maxsorb A | 3100 | 4.0 | 298 | 16.4 | 152 | [ | |||||||||||
| KUA31752 | 3355 | 4.0 | 298 | 15.6 | 155 | [ | ||||||||||||
| PC | 1220 | 0.47 | 2.0 | 298 | 10.2 | 141 | [ | |||||||||||
| Saran A carbon 36X | 1650 | 3.2 | 298 | 11.5 | [ | |||||||||||||
| KF-1500 | 1500 | 3.2 | 298 | 7.6 | [ | |||||||||||||
| UlaC-950-HF | 1476 | 0.61 | 3.5 | 298 | 17 | 232 | 22.4 | [ | ||||||||||
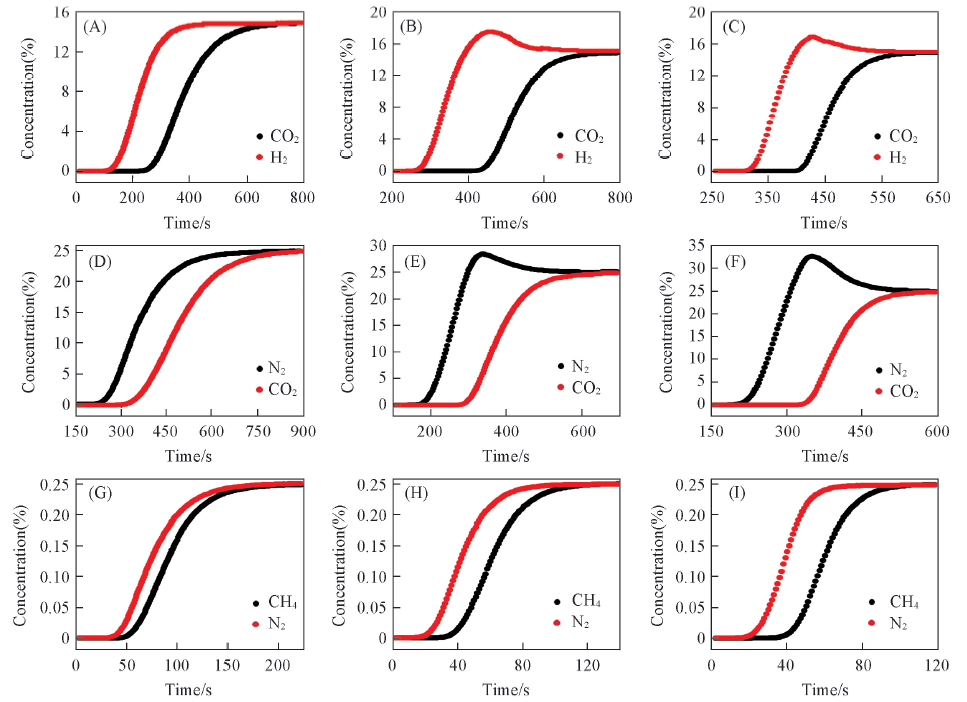
Fig.11 Breakthrough curves of LamCW-900-HF for CO2/H2/He(A—C), N2/CO2/He(D—F) and CH4/N2/He(G—I) mixture with ratio of 1:1:2 at 299 K(A), (D), (G): 100 kPa; (B), (E), (H): 200 kPa; (C), (F), (I): 300 kPa.
| [1] | |
| [2] | Piñero E.R., Leroux F., Béguin F., Adv. Mater., 2006, 14(18), 1877—1882 |
| [3] | Laine J., Calafat A., Labady M., Carbon, 1989, 27(2), 191—195 |
| [4] | Laine J., Calafat A., Carbon, 1991, 29(7), 949—953 |
| [5] | Ahmadpour A., Do D.D., Carbon, 1997, 35(12), 1723—1732 |
| [6] | Pandolfo A.G., Amoli M. A., Killingley J. S., Carbon, 1994, 32(5), 1015—1019 |
| [7] | Fuertes A.B., Ferrero G. A., Sevilla M., J. Mater. Chem.A, 2014, 2(35), 14439—14448 |
| [8] | Deng J., Xiong T.Y., Xu F., Li M. M., Han C. L., Gong Y. T., Wang H. Y., Wang Y., Green Chem., 2015, 7, 4053—4060 |
| [9] | Wang Z., Tan Y.T., Yang Y. L., Zhao X. N., Liu Y., Niu L. Y., Tichnell B., Kong L. B., Kang L., Liu Z., Ran F., J. Power Sources, 2018, 378, 499—510 |
| [10] | Laine J., Simoni S., Calles R., Chem. Eng. Commun., 1991, 99(1), 15—23 |
| [11] | Bergius F., Z. Angew. Phys. Chem., 1913, 20, 260—260 |
| [12] | Sevilla M., Antonio J., Antonio M.A., Fuertes B., Biomass Bioenerg., 2011, 35(7), 3152—3159 |
| [13] | Biswal M., Banerjee A., Deo M., Ogale S., Energ. Environ. Sci., 2013, 6(4), 1249—1259 |
| [14] | Lu W.J., Huang S. Z., Miao L., Liu M. X., Zhu D. Z., Li L. C., Duan H., Gan Z. H., Chinese Chem. Lett., 2017, 28(6), 1324—1329 |
| [15] | Zhong Y., Xia X.H., Deng S. J., Zhan J. Y., Fang R. Y., Xia Y., Wang X. L., Zhang Q., Tu J. P., Adv. Energy Mater., 2018, 8(1), 1701110 |
| [16] | Caturla F., Molina-Sabio M., Rodríguez-Reinoso F., Carbon, 1991, 29(7), 999—1007 |
| [17] | Gong Y., Li D., Luo C., Fu Q., Pan C., Green Chem., 2017, 19(17), 4132—4140 |
| [18] | Paskaleva E.E., Lin X. D., Duus K., Mc Sharry J. J., Veille J. C. L., Thornber C., Liu Y. Z., Lee D. Y. W., Canki M., Virol. J., 2008, 5(1), 8 |
| [19] | Zhu Y., Murali S., Stoller M.D., Ganesh K. J., Cai W., Ferreira P. J., Pirkle A., Wallace R. M., Cychosz K. A., Thommes M., Su D., Stach E. A., Ruoff R. S., Science, 2011, 332(6037), 1537—1541 |
| [20] | Konstas K., Osl T., Yang Y.X., Batten M., Burke N., Hill A. J., Hill M. R., J. Mater. Chem., 2012, 22(33), 16698—16708 |
| [21] | Wood C.D., Tan B., Trewin A., Su F. B., Rosseinsky M. J., Bradshaw D., Sun Y., Zhou L., Cooper A. I., Adv. Mater., 2008, 20(10), 1916—1921 |
| [22] | Larsen G.S., Lin P., Siperstein F. R., Colina C. M., Adsorption, 2011, 17(1), 21—26 |
| [23] | Makal T.A., Li J. R., Lu W. G., Zhou H. C., Chem. Soc. Rev., 2012, 41(23), 7761—7779 |
| [24] | Yuan D.Q., Lu W. G., Zhao D., Zhou H. C., Adv. Mater., 2011, 23(32), 3723—3725 |
| [25] | Zhou W., Chem. Rec., 2010, 10(3), 200—204 |
| [26] | Chen C.X., Wei Z. W., Jiang J. J., Zheng S. P., Wang H. P., Qiu Q. F., Cao C. C., Fenske D., Su C. Y., J. Am. Chem. Soc., 2017, 139(17), 6034—6037 |
| [27] | Jiang J., Furukawa H., Zhang Y.B., Yaghi O. M., J. A.Chem. Soc., 2016, 138(32), 10244—10251 |
| [28] | Peng Y., Krungleviciute V., Eryazici I., Hupp J.T., Farha O. K., Yildirim T., J. Am. Chem. Soc., 2013, 135(32), 11887—11894 |
| [29] | Wilmer C.E., Farha O. K., Yildirim T., Eryazici I., Krungleviciute V., Sarjeant A. A., Snurr R. Q., Hupp J. T., Energ. Environ. Sci., 2013, 6(4), 1158—1163 |
| [30] | Li B., Wen H.M., Wang H., Wu H., Yildirim T., Zhou W., Chen B. L., Energ. Environ. Sci., 2015, 8(8), 2504—2511 |
| [31] | Li B., Wen H.M., Wang H., Wu H., Tyagi M., Yildirim T., Zhou W., Chen B., J. Am. Chem. Soc., 2014, 136(17), 6207—6210 |
| [32] | Guo Z.Y., Wu H., Srinivas G., Zhou Y. M., Xiang S. C., Chen Z. X., Yang Y. T., Zhou W., O’Keeffe M., Chen B. L., Angew. Chem. Int. Ed., 2011, 50(14), 3178—3181 |
| [33] | He Y., Zhou W., Yildirim T., Chen B., Energ. Environ. Sci., 2013, 6(9), 2735—2744 |
| [34] | Lin J.M., He C. T., Liu Y., Liao P. Q., Zhou D. D., Zhang J. P., Chen X. M., Angew. Chem. Int. Ed., 2016, 55(15), 4674—4678 |
| [35] | Kong G.Q., Han Z. D., He Y. B., Ou S., Zhou W., Yildirim T., Krishna R., Zou C., Chen B. L., Wu C. D., Chem-Eur. J., 2013, 19(44), 14886—14894 |
| [36] | Llewellyn P.L., Bourrelly S., Serre C., Vimont A., Daturi M., Hamon L., de Weireld G., Chang J. S., Hong D. Y., Hwang Y. K., Jhung S. H., Ferey G., Langmuir, 2008, 24(14), 7245—7250 |
| [37] | Zhang S.Y., Talu O., Hayhurst D. T., J. Chem. Phys., 1991, 95(4), 1722—1726 |
| [38] | Zhou H.S., Zhu S. M., Honma I., Seki K., Chem. Phys. Lett., 2004, 396(4), 252—255 |
| [39] | Li Y.Q., Ben T., Zhang B. Y., Fu Y., Qiu S. L., Sci. Rep-UK, 2013, 3(33), 2420 |
| [40] | Castelló D.L., Amorós D. C., Solano A. L., Energ.Fuel, 2002, 16(5), 1321—1328 |
| [41] | Menon V.C., Komarneni S., J. Porous Mater., 1998, 5(1), 43—58 |
| [42] | Wang Y., Ben T., Qiu S.L., Chem. J. Chinese Universities, 2016, 37(5), 801—809) |
| (王昀, 贲腾, 裘式纶. 高等学校化学学报, 2016, 37(5), 801—809) | |
| [43] | Lu J.L., Plant Nutrition, China Agricultural University Press, Beijing, 2003, 61—72 |
| (陆景陵. 植物营养学, 北京: 中国农业大学出版社, 2003, 61—72) | |
| [44] | Mendoza-Cortés J.L., Han S. S., Furukawa H., Yaghi O. M., Goddard III W. A., J. Phys. Chem.A, 2010, 114(40), 10824—10833 |
| [45] | Bojan M.J., Slooten R. V., Steele W., Sep. Sci. Technol., 1992, 27(14), 1837—1856 |
| [46] | Chen X.S., Mcenaney B., Mays T. J., Alcaniz-Monge J., Cazorla-Amoros D., Linares-Solano A., Carbon, 1997, 35(9), 1251—1258 |
| [47] | Lv Y.K., Gan L. H., Liu M. X., Xiong W., Xu Z. J., Zhu D. Z., Wright D. S., J. Power Sources, 2012, 209(209), 152—157 |
| [48] | Bhatia S.K., Myers A. L., Langmuir, 2006, 22(4), 1688—1700 |
| [1] | 王昀, 贲腾. 海藻基含杂原子碳材料的制备及电化学性质[J]. 高等学校化学学报, 2018, 39(12): 2627. |
| [2] | 仲娜,王小如,杨黄浩,曹煊,王庚 . 高效液相色谱与电感耦合等离子体质谱联用技术用于胁迫富硒海带硒形态研究[J]. 高等学校化学学报, 2008, 29(1): 77. |
| [3] | 田媛, 宋文波, 姜艳霞, 刘颖, 陈旭, 许宏鼎. 微分脉冲阳极溶出伏安法测定碘离子[J]. 高等学校化学学报, 1999, 20(1): 28. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
