

Chem. J. Chinese Universities ›› 2017, Vol. 38 ›› Issue (1): 165.doi: 10.7503/cjcu20160190
• Polymer Chemistry • Previous Articles
WANG Huijie1,2, YU Haiyang2, ZHANG Dawei2, TANG Zhaohui2, CAO Qi1,*( )
)
Received:2016-03-28
Online:2017-01-10
Published:2016-12-08
Contact:
CAO Qi
E-mail:wjcaoqi@163.com
Supported by:CLC Number:
TrendMD:
WANG Huijie, YU Haiyang, ZHANG Dawei, TANG Zhaohui, CAO Qi. Synthesis and Characterization of Poly(E,K) and Poly(E,R)†[J]. Chem. J. Chinese Universities, 2017, 38(1): 165.
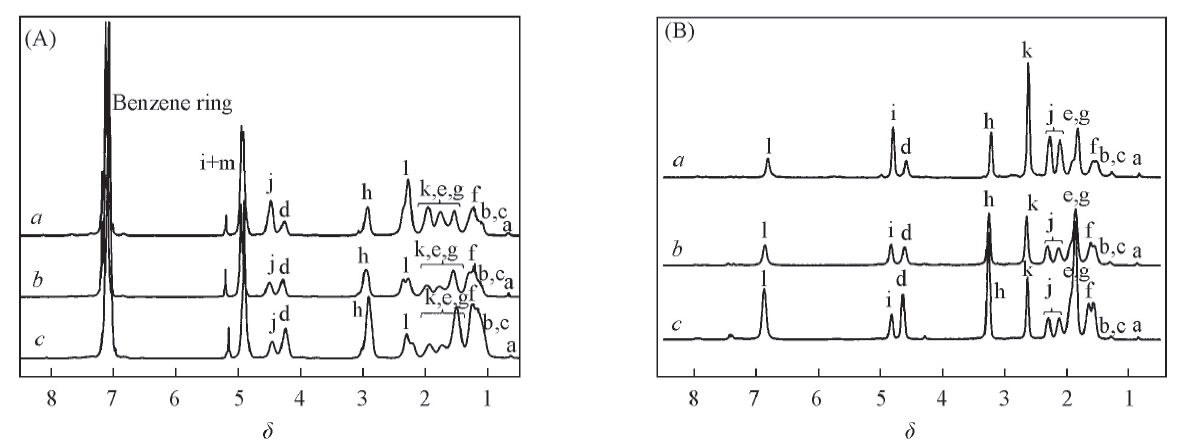
Fig.1 1H NMR spectra of Poly[E(Bn),K(Cbz)](A) and Poly(E,K)(B) in TFA-d (A) a. Poly[E(Bn),K(Cbz)]1; b. Poly[E(Bn),K(Cbz)]2; c. Poly[E(Bn),K(Cbz)]3. (B) a. Poly(E,K)1; b. Poly(E,K)2; c. Poly(E,K)3.
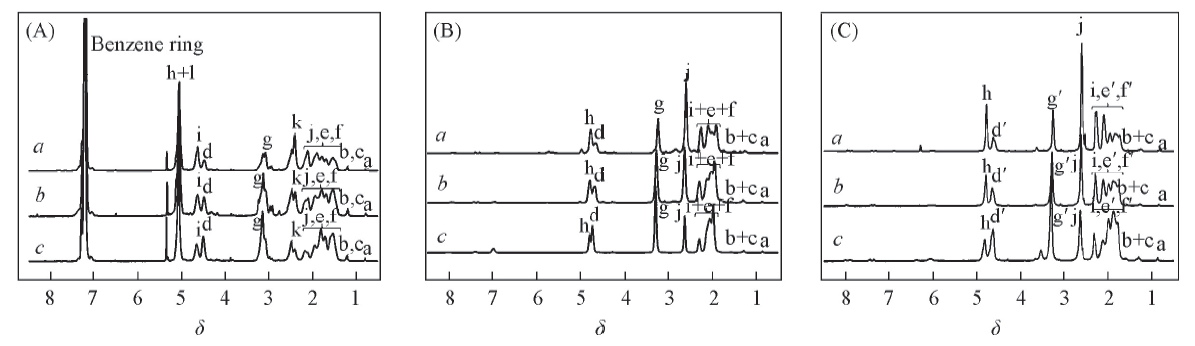
Fig.3 1H NMR spectra of Poly[E(Bn),O(Cbz)](A), Poly(E,O)(B) and Poly(E,R)(C) in TFA-d (A) a. Poly[E(Bn),O(Cbz)]1; b. Poly[E(Bn),O(Cbz)]2; c. Poly[E(Bn),O(Cbz)]3. (B) a. Poly(E,O)1; b. Poly(E,O)2; c. Poly(E,O)3. (C) a. Poly(E,R)1; b. Poly(E,R)2; c. Poly(E,R)3.
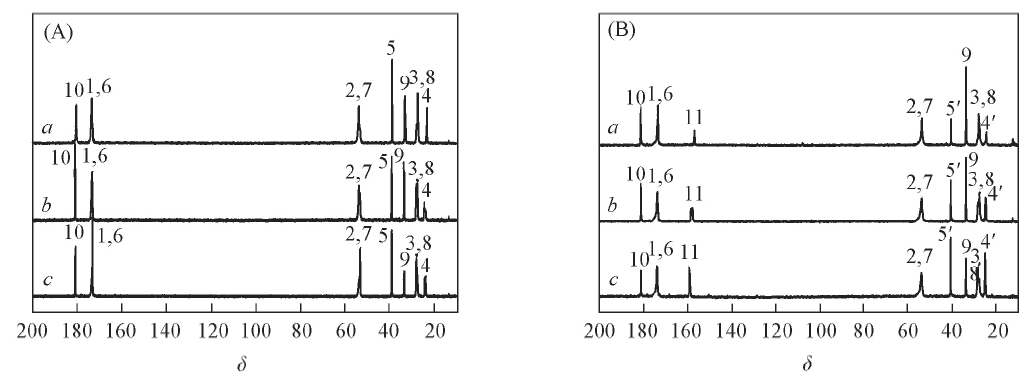
Fig.4 Quantitative 13C NMR spectra of Poly(E,O)(A) and Poly(E,R)(B) in D2O/NaOD (A) a. Poly(E,O)1; b. Poly(E,O)2; c. Poly(E,O)3. (B) a. Poly(E,R)1; b. Poly(E,R)2; c. Poly(E,R)3.
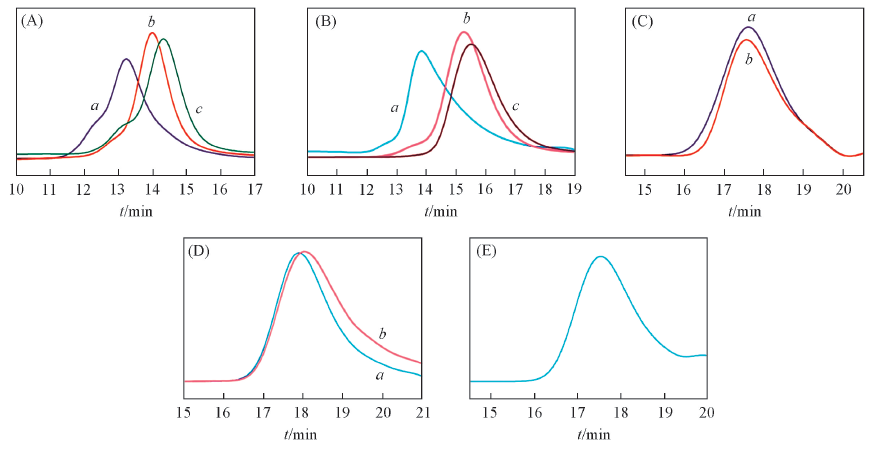
Fig.5 GPC curves of Poly[E(Bn),K(Cbz)](A), Poly[E(Bn),O(Cbz)](B), Poly(E,K)(C), Poly(E,O)(D) and Poly(E,R)(E) (A) a. Poly[E(Bn),K(Cbz)]1; b. Poly[E(Bn),K(Cbz)]2; c. Poly[E(Bn),K(Cbz)]3. (B) a. Poly[E(Bn),O(Cbz)]1; b. Poly[E(Bn),O(Cbz)]2; c. Poly[E(Bn),O(Cbz)]3. (C) a. Poly(E,K)1; b. Poly(E,K)2. (D) a. Poly(E,O)1; b. Poly(E,O)2. (E) Poly(E,R)1.
| Copolymer | Feed molar ratio | Resultant molar ratio | 10-3 | PDIh | Conversion ratio(%) |
|---|---|---|---|---|---|
| Poly[E(Bn),K(Cbz)]1 | 2:1a | 2.2:1.0c | 20.4 | 1.30 | 86 |
| Poly[E(Bn),K(Cbz)]2 | 1:1a | 1.1:1.0c | 16.6 | 1.25 | 87 |
| Poly[E(Bn),K(Cbz)]3 | 1:2a | 1.0:1.8c | 12.9 | 1.26 | 81 |
| Poly(E,K)1 | 2:1a | 2.2:1.0d | 5.5 | 1.33 | 95 |
| Poly(E,K)2 | 1:1a | 1.0:1.0d | 6.0 | 1.38 | 96 |
| Poly(E,K)3 | 1:2a | 1.0:1.8d | 93 | ||
| Poly[E(Bn),O(Cbz)]1 | 2:1b | 2.2:1.0e | 16.2 | 1.37 | 84 |
| Poly[E(Bn),O(Cbz)]2 | 1:1b | 1.2:1.0e | 11.9 | 1.28 | 83 |
| Poly[E(Bn),O(Cbz)]3 | 1:2b | 1.0:1.9e | 10.5 | 1.29 | 87 |
| Poly(E,O)1 | 2:1b | 2.0:1.0f | 7.9 | 1.41 | 96 |
| Poly(E,O)2 | 1:1b | 1.0:1.0f | 6.1 | 1.45 | 94 |
| Poly(E,O)3 | 1:2b | 1.0:2.0f | 93 | ||
| Poly(E,R)1 | 2:1b | 2.2:1.0g | 15.9 | 1.43 | 95 |
| Poly(E,R)2 | 1:1b | 1.2:1.0g | 93 | ||
| Poly(E,R)3 | 1:2b | 1.0:1.9g | 92 |
Table 1 Characterization of copolymers
| Copolymer | Feed molar ratio | Resultant molar ratio | 10-3 | PDIh | Conversion ratio(%) |
|---|---|---|---|---|---|
| Poly[E(Bn),K(Cbz)]1 | 2:1a | 2.2:1.0c | 20.4 | 1.30 | 86 |
| Poly[E(Bn),K(Cbz)]2 | 1:1a | 1.1:1.0c | 16.6 | 1.25 | 87 |
| Poly[E(Bn),K(Cbz)]3 | 1:2a | 1.0:1.8c | 12.9 | 1.26 | 81 |
| Poly(E,K)1 | 2:1a | 2.2:1.0d | 5.5 | 1.33 | 95 |
| Poly(E,K)2 | 1:1a | 1.0:1.0d | 6.0 | 1.38 | 96 |
| Poly(E,K)3 | 1:2a | 1.0:1.8d | 93 | ||
| Poly[E(Bn),O(Cbz)]1 | 2:1b | 2.2:1.0e | 16.2 | 1.37 | 84 |
| Poly[E(Bn),O(Cbz)]2 | 1:1b | 1.2:1.0e | 11.9 | 1.28 | 83 |
| Poly[E(Bn),O(Cbz)]3 | 1:2b | 1.0:1.9e | 10.5 | 1.29 | 87 |
| Poly(E,O)1 | 2:1b | 2.0:1.0f | 7.9 | 1.41 | 96 |
| Poly(E,O)2 | 1:1b | 1.0:1.0f | 6.1 | 1.45 | 94 |
| Poly(E,O)3 | 1:2b | 1.0:2.0f | 93 | ||
| Poly(E,R)1 | 2:1b | 2.2:1.0g | 15.9 | 1.43 | 95 |
| Poly(E,R)2 | 1:1b | 1.2:1.0g | 93 | ||
| Poly(E,R)3 | 1:2b | 1.0:1.9g | 92 |
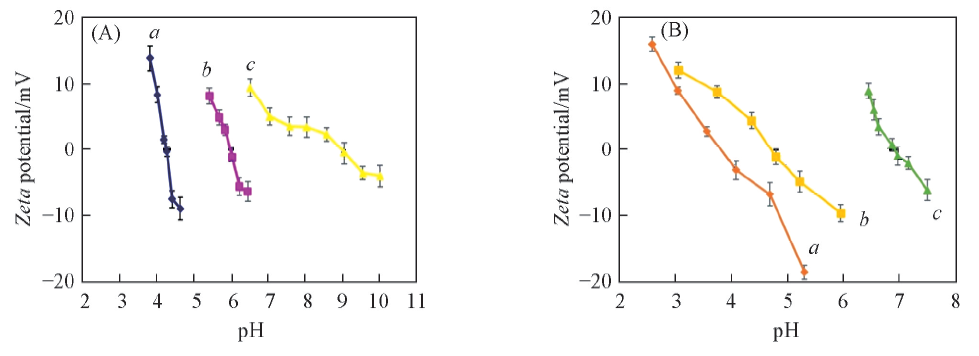
Fig.7 Zeta potentials of Poly(E,K)(A) and Poly(E,R)(B) at different pH(A) a. Poly(E,K)1; b. Poly(E,K)2; c. Poly(E,K)3. (B) a. Poly(E,R)1; b. Poly(E,R)2; c. Poly(E,R)3.
| [1] | Kantor F. S., Ojeda A., Benacerraf B., Journal of Experimental Medicine,1963, 117(1), 55—69 |
| [2] | Benacerraf B., Ojeda A., Maurer P. H., Journal of Experimental Medicine,1963, 118(6), 945—952 |
| [3] | Nair L. S., Laurencin C. T., Progress in Polymer Science,2007, 32(8/9), 762—798 |
| [4] | Li M. Q., Tang Z. H., Sun H., Ding J. X., Song W. T., Chen X. S., Polymer Chemistry,2013, 4, 1199—1207 |
| [5] | Cheng Y. L., He C. L., Xiao C. S., Ding J. X., Zhuang X. L., Huang Y. B., Chen X. S., Biomacromolecules,2012, 13(7), 2053—2059 |
| [6] | Osada K., Kataoka K., Peptide Hybrid Polymers,2006, 202, 113—153 |
| [7] | Yu M. Y., Deming T. J., Macromolecules,1998, 31(15), 4739—4745 |
| [8] | Leuchs H., Berichte Der Deutschen Chemischen Gesellschaft, 1906, 39, 857—861 |
| [9] | Curtius T., Sieber W., Berithte Der Dewtschen Chemischen Gesellschaft,1921, 54, 1430—1437 |
| [10] | Bae Y., Kataoka K., Advanced Drug Delivery Reviews,2009, 61, 768—784 |
| [11] | Miyata K., Christie R. J., Kataoka K., Reactive and Functional Polymers,2011, 71(3), 227—234 |
| [12] | Wang W., Ding J. X., Xiao C. S., Tang Z. H., Li D., Chen J., Zhuang X. L., Chen X. S., Biomacromolecules,2011, 12(7), 2466—2474 |
| [13] | Ding J. X., Shi F. H., Xiao C. S., Lin L., Chen L., He C. L., Zhuang X. L., Chen X. S., Polymer Chemistry,2011, 2(12), 2857—2864 |
| [14] | Kataoka K., Harada A., Nagasaki Y., Advanced Drug Delivery Reviews,2001, 47(1), 113—131 |
| [15] | Asayama S., Ogawa A., Kawakami H., Nagaoka S., Molecular Pharmaceutics,2007, 5(1), 162—164 |
| [16] | Milhaud P. G., Compagnon B., Bienvenue A., Philippotet J. R., Bioconjugate Chem., 1992, 3(5), 402—407 |
| [17] | Gillies E. R., Fréchet J. M. J., Bioconjugate Chem., 2005, 16(2), 361—368 |
| [18] | Daly W. H., Poché D., Tetrahedron Letters,1988, 29, 5859—5862 |
| [19] | Deming T. J., J. Polym. Sci. Pol. Chem., 2000, 38, 3011—3018 |
| [20] | Cabral H., Matsumoto Y., Mizuno K., Chen Q., Murakami M., Kimura M., Terada Y., Kano M. R., Miyazono K., Uesaka M., Nishiyama N., Kataoka K., Nature Nanotechnology,2011, 6(12), 815—823 |
| [21] | Tang L., Yang X., Yin Q., Cai K., Wang H., Chaudhury I., Yao C., Zhou Q., Kwon M., Hartman J. A., Dobrucki I. T., Dobrucki L. W., Borst L. B., Lezmig S., Helferich W. G., Ferguson A. L., Fan T. M., Cheng J., Proc. Natl. Acad . Sci. USA,2014, 111(43), 15344—15349 |
| [1] | ZHANG Wanbin, WANG Yanmeng, WANG Shaowu, TONG Xin, HAN Xiaoqian, ZHANG Ce, ZHANG Guanghua, ZHU Xiuzhong. Synthesis of Poly(allyl glycidyl ether) Bearing Alkyl Functional Side Groups and Its Plasticizing and Antistatic Effects for PVC [J]. Chem. J. Chinese Universities, 2021, 42(9): 2961. |
| [2] | MA Yukun, JIN Hui, REN Chuanli, LI Zhibo. Ring-opening Polymerization of Cyclic Esters Using Recyclable Polystyrene Supported Urea-Base Binary Catalyst [J]. Chem. J. Chinese Universities, 2021, 42(9): 2968. |
| [3] | XU Mengyi, HUANG Xuewen, LI Xiaojie, WEI Wei, LIU Xiaoya. Fabrication of Biosensor Based on “Beads-on-a-String” Shaped Composite Nano-assembly Modified Screen Printed Electrode [J]. Chem. J. Chinese Universities, 2021, 42(6): 1768. |
| [4] | LI Rongye, NI Yunxia, LIU Dandan, LI Zhi, CHENG Yuxin, XIA Mingxin, FU Xiaohui. Synthesis and Characterization of Thermoresponsive Polypeptide/polypeptoid Block Copolymers [J]. Chem. J. Chinese Universities, 2021, 42(3): 850. |
| [5] | LI Chen, LI Yuesheng. Living Ring-opening Polymerization of O-Carboxyanhydrides Catalyzed by Pyridine Derivatives [J]. Chem. J. Chinese Universities, 2021, 42(10): 3203. |
| [6] | MA Qian,WU Xiaohui,YU Lin,DING Jiandong. Design and Synthesis of Novel Iodinated Polycarbonates with Inherent X-Ray Opacity † [J]. Chem. J. Chinese Universities, 2019, 40(10): 2233. |
| [7] | MENG Jiafeng, NI Xufeng, ZHENG Hao, SHEN Zhiquan. Copolymerization of Norbornene and 1-Octene Catalyzed by Bis(phenoxy-imine) Titanium Complex† [J]. Chem. J. Chinese Universities, 2018, 39(8): 1853. |
| [8] | LI Juan, DU Fanfan, FENG Rui, HU Qian, JIE Suyun, LI Bogeng. Synthesis of Cyclic and Linear Block Copolyesters via Ring-opening Copolymerization of ε-Caprolatone and L-Lactide Catalyzed by Zinc Complex† [J]. Chem. J. Chinese Universities, 2018, 39(6): 1297. |
| [9] | WANG Luyuan, LIU Shuxue, LI Huimin, HUANG Yaodong. Preparation and Properties of the Two-component Hydrogels Based on Pyrazine Dicarboxylic Acid and Melamine† [J]. Chem. J. Chinese Universities, 2017, 38(5): 806. |
| [10] | LIU Yajie, ZHANG Peng, DU Jianwei, WANG Youxiang. pH-Responsive PEGylated Gene Delivery System† [J]. Chem. J. Chinese Universities, 2016, 37(5): 1003. |
| [11] | LIU Ruiqing, LIANG Shuang, JIANG Cun, XU Zushun, XU Haibo. Synthesis and Proporties of Temperature and pH-sensitive and Gadolinium Contained Polymeric Magnetic Resonance Imaging Contrast Agent† [J]. Chem. J. Chinese Universities, 2016, 37(1): 155. |
| [12] | WANG Lei, YANG Li, GAO Chengyong. Synthesis, Characterization of Methoxyl-bridged Aluminium-lithium Heterobimetallic Complex and Catalysis for the Ring-opening Polymerization of ε-Caprolactone† [J]. Chem. J. Chinese Universities, 2015, 36(4): 794. |
| [13] | QU Zhi, LI Xiang, PANG Xuan, DUAN Ranlong, GAO Bo, CHEN Xuesi. Aluminum Schiff Base Catalyst for Ring-opening Polymerization of ε-Caprolactone† [J]. Chem. J. Chinese Universities, 2014, 35(4): 869. |
| [14] | GUO Kai, SUN Liang, WU Meng-Chun, WANG Shao-Dan, WANG Li-Yan*. Effects of Inclusion Complexation and Degree of Protonation on Cloud Point of Poly(N-acryloyl-N′-propylpiperazine) [J]. Chem. J. Chinese Universities, 2011, 32(9): 2223. |
| [15] | LIANG Zhen-Hua, NI Xu-Feng*, SHEN Zhi-Quan. Controlled Ring\|opening Polymerization of ε-Caprolactone Initiated by in situ Formed Yttrium Tribenzyloxide Complex [J]. Chem. J. Chinese Universities, 2011, 32(8): 1881. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||