

Chem. J. Chinese Universities ›› 2015, Vol. 36 ›› Issue (8): 1596.doi: 10.7503/cjcu20150105
• Physical Chemistry • Previous Articles Next Articles
LI Ming, LI Youji*( ), XU Peng, LIN Xiao, HAN Wenxuan
), XU Peng, LIN Xiao, HAN Wenxuan
Received:2015-01-29
Online:2015-08-10
Published:2015-07-17
Contact:
LI Youji
E-mail:bcclyj@163.com
Supported by:CLC Number:
TrendMD:
LI Ming, LI Youji, XU Peng, LIN Xiao, HAN Wenxuan. Preparation and Photoelectrocatalytic Performance of Mesoporous Titanium Dioxide/Conductive Carbon Felt Electrode†[J]. Chem. J. Chinese Universities, 2015, 36(8): 1596.
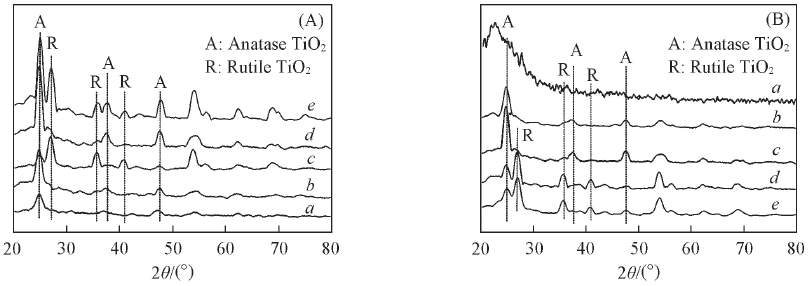
Fig.2 XRD patterns of MPT/CCF and MPT calcined at different temperatures(A) and different load times(B) (A) a. MPT; b. MPT-200; c. MPT-300; d. 2-MPT/CCF-500; e. 2-MPT/CCF-600. (B) a. CCF; b. 1-MPT/CCF-500; c. 2-MPT/CCF-500; d. 3-MPT/CCF-500; e. 4-MPT/CCF-500.
| Sample | Calcination temperature/℃ | |||
|---|---|---|---|---|
| 400 | 500 | 600 | 700 | |
| MPT | 16.9 | 19.1 | 20.9 | 27.7 |
| 1-MPT/CCF | 12.5 | 13.8 | 18.7 | 22.4 |
| 2-MPT/CCF | 9.9 | 18.5 | 20.2 | 27.1 |
| 3-MPT/CCF | 12.7 | 22.3 | 16.1 | 27.4 |
| 4-MPT/CCF | 10.4 | 19.4 | 20.5 | 22.2 |
Table 1 Grain size parameters(nm) of different samples
| Sample | Calcination temperature/℃ | |||
|---|---|---|---|---|
| 400 | 500 | 600 | 700 | |
| MPT | 16.9 | 19.1 | 20.9 | 27.7 |
| 1-MPT/CCF | 12.5 | 13.8 | 18.7 | 22.4 |
| 2-MPT/CCF | 9.9 | 18.5 | 20.2 | 27.1 |
| 3-MPT/CCF | 12.7 | 22.3 | 16.1 | 27.4 |
| 4-MPT/CCF | 10.4 | 19.4 | 20.5 | 22.2 |
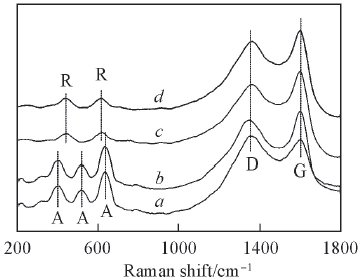
Fig.3 Raman spectra of MPT/CCFA: Anatase; R: rutile; D and G: D-band and G-band of CCF. a. 2-MPT/CCF-400; b. 2-MPT/CCF-500; c. 2-MPT/CCF-600; d. 2-MPT/CCF-700.

Fig.4 SEM(A—E) and TEM(F) images of CCF(A) and MPT/CCF(B—F)(B) 1-MPT/CCF-500; (C, F) 2-MPT/CCF-500; (D) 3-MPT/CCF-500; (E) 4-MPT/CCF-500. Inset of (F) is HRTEM image of 2-MPT/CCF-500.
| Sample | TiO2 contenta(%) | Average pore sizec/nm | O1s contentd(%) | |||
|---|---|---|---|---|---|---|
| Ti—O | C—O | O—H | ||||
| MPT | 100 | 103.377 | 16.628 | 86.3 | 13.6 | 0.1 |
| 2-MPT/CCF-400 | 9.73 | 139.137 | 18.153 | 83.7 | 16.1 | 0.2 |
| 2-MPT/CCF-500 | 9.25 | 144.015 | 19.360 | 77.9 | 10.0 | 12.1 |
| 2-MPT/CCF-600 | 9.14 | 119.120 | 24.843 | 73.7 | 19.8 | 6.5 |
| 2-MPT/CCF-700 | 9.03 | 72.618 | 15.656 | 78.4 | 18.7 | 2.9 |
| 1-MPT/CCF-500 | 7.67 | 39.530 | 19.134 | 77.5 | 22.4 | 0.1 |
| 3-MPT/CCF-500 | 11.26 | 41.173 | 19.107 | 68.9 | 26.5 | 4.6 |
| 4-MPT/CCF-500 | 12.89 | 66.709 | 19.152 | 78.1 | 20.0 | 1.9 |
| CCF | 0 | 58.786 | 15.430 | |||
Table 2 Structural parameters of the samples
| Sample | TiO2 contenta(%) | Average pore sizec/nm | O1s contentd(%) | |||
|---|---|---|---|---|---|---|
| Ti—O | C—O | O—H | ||||
| MPT | 100 | 103.377 | 16.628 | 86.3 | 13.6 | 0.1 |
| 2-MPT/CCF-400 | 9.73 | 139.137 | 18.153 | 83.7 | 16.1 | 0.2 |
| 2-MPT/CCF-500 | 9.25 | 144.015 | 19.360 | 77.9 | 10.0 | 12.1 |
| 2-MPT/CCF-600 | 9.14 | 119.120 | 24.843 | 73.7 | 19.8 | 6.5 |
| 2-MPT/CCF-700 | 9.03 | 72.618 | 15.656 | 78.4 | 18.7 | 2.9 |
| 1-MPT/CCF-500 | 7.67 | 39.530 | 19.134 | 77.5 | 22.4 | 0.1 |
| 3-MPT/CCF-500 | 11.26 | 41.173 | 19.107 | 68.9 | 26.5 | 4.6 |
| 4-MPT/CCF-500 | 12.89 | 66.709 | 19.152 | 78.1 | 20.0 | 1.9 |
| CCF | 0 | 58.786 | 15.430 | |||
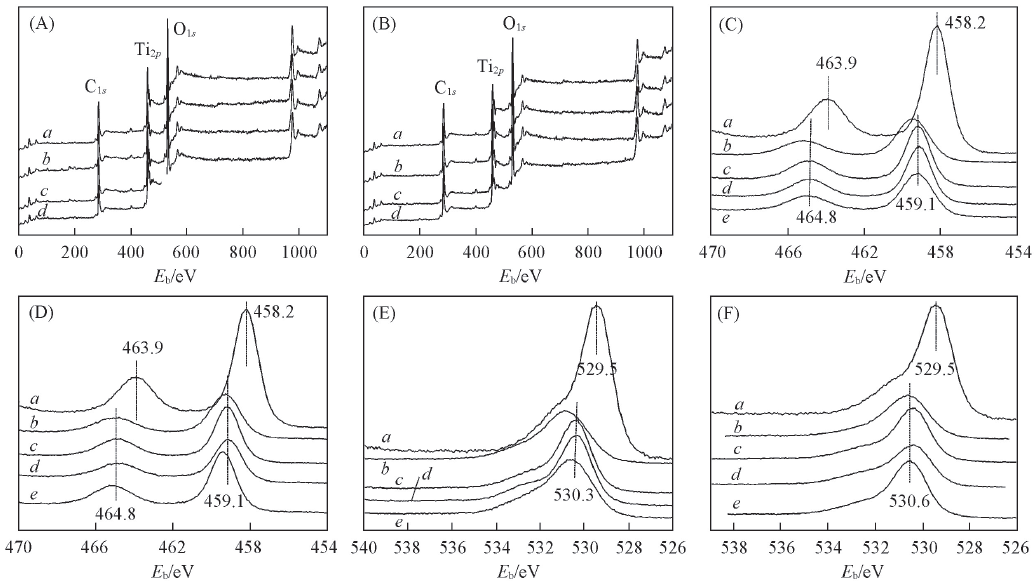
Fig.6 XPS spectra of MPT and MPT/CCF(A,B) Full spectra; (C,D) Ti2p; (E, F) O1s. (A) a. 2-MPT/CCF-400; b. 2-MPT/CCF-500; c. 2-MPT/CCF-600; d. 2-MPT/CCF-700. (B) a. 1-MPT/CCF-500; b. 2-MPT/CCF-500; c. 3-MPT/CCF-500; d. 4-MPT/CCF-500. (C), (E) a. MPT; b. 2-MPT/CCF-400; c. 2-MPT/CCF-500; d. 2-MPT/CCF-600; e. 2-MPT/CCF-700. (D), (F) a. MPT; b. 1-MPT/CCF-500; c. 2-MPT/CCF-500; d. 3-MPT/CCF-500; e. 4-MPT/CCF-500.
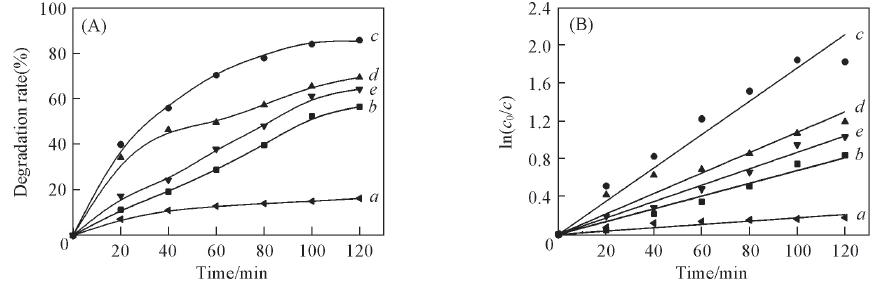
Fig.8 Effects of degradation time on degradation rate(A) and ln(c0/c)(B) with different catalysts(A, B) a. CCF; b. 1-MPT/CCF-500; c. 2-MPT/CCF-500; d. 3-MPT/CCF-500; e. 4-MPT/CCF-500.(B) a. y=0.0017x, R2=0.94, t1/2=407.73 min; b. y=0.0067x, R2=0.98, t1/2=103.45 min; c. y=0.0176x, R2=0.98, t1/2=39.38 min; d. y=0.0107x, R2=0.97, t1/2=64.78 min; e. y=0.0086x, R2=0.99, t1/2=80.69 min.
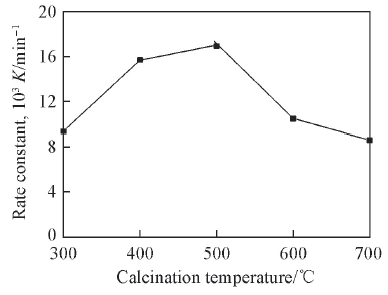
Fig.9 Effects of calcination temperature of the catalyst on degradation rate constantDegradation conditions: 35 ℃; relative humidity:55%; bias voltage: 10 V.
| [1] | Wang S. B., Ang H. M., Tade M. O., Environ. Int., 2007, 33(5), 694—705 |
| [2] | Loh M. M., Levy J. I., Spengler J. D., Houseman E. A., Bennett D. H., Environ. Health Perspect., 2007, 115(8), 1160—1168 |
| [3] | Mo J., Zhang Y., Xu Q., Lamson J. J., Zhao R., Atmos. Environ., 2009, 43(14), 2229—2246 |
| [4] | Kontos A. G., Katsanaki A., Maggos T., Likodimos V., Ghicov A., Kim D., Kunze J., Vasilakos C., Schmuki P., Falaras P., Chem. Phys. Lett., 2010, 290(1—3), 58—62 |
| [5] | Sin J. C., Lam S. M., Lee K. T., Mohamed A. R., Ceram. Int., 2014, 40(4), 5431—5440 |
| [6] | Zhang Z. F., Liu H. R., Zhang H., Liu X. G., Jia H. S., Xu B. S., Chem. J. Chinese Universities, 2013, 34(12), 2827—2833 |
| (张振飞, 刘海瑞, 张华, 刘旭光, 贾虎生, 许并社.高等学校化学学报, 2013, 34(12), 2827—2833) | |
| [7] | Yu C. L., Zhou W. Q., Yu J. M., Yang J. G., Fan Q. Z., Chem. Res. Chinese Universities, 2012, 28(1), 124—128 |
| [8] | Maury A., Ramirez N., Belie D., J. Adv. Oxidation Technol., 2009, 12(1), 100—104 |
| [9] | Diamanti M. V., Ormellese M., Pedeferri M., Cement Concrete Res., 2008, 38(11), 1349—1353 |
| [10] | Ramirez A. M., Demeestere K., Belie N. D., Build. Environ., 2010, 45(4), 832—838 |
| [11] | Xu P., Li Y. J., Liu C., Li M., Deng R. C., Chem. J. Chinese Universities, 2014, 35(9), 1954—1961 |
| (徐鹏, 李佑稷, 刘晨, 李铭, 邓瑞成.高等学校化学学报, 2014, 35(9), 1954—1961) | |
| [12] | Kohtani S., Nishioka S., Yoshioka E., Miyabe H., Catal. Commun., 2014, 43, 61—65 |
| [13] | Ji T. H., Liu Y., Dong L. Y., Han P., Sun J. Y., Chem. Res. Chinese Universities, 2012, 28(4), 721—726 |
| [14] | Huang D., Luo G. S., Wang Y. J., Microporous Mesoporous Mater., 2005, 84(1—3), 27—33 |
| [15] | Liu C., Li Y. J., Xu P., Li M., Zen M. X., Microporous Mesoporous Mater., 2014, 195(1), 319—329 |
| [16] | Li Y. J., Song Z. J., Li Z. P., Ouyang Y. Z., Yan W. B., Chem. J. Chinese Universities, 2007, 28(9), 1710—1715 |
| (李佑稷, 宋志娟, 李志平, 欧阳玉祝, 颜文斌. 高等学校化学学报, 2007, 28(9), 1710—1715) | |
| [17] | Ye S. Y., Tian Q. M., Song X. L., Lou S. C., J. Photoch. Photobio. A, 2009, 208(1), 27—35 |
| [18] | Okpalugo T. I. T., Papakonstantinou P., Murphy H., Mclaughlin J., Brown N. M. D., Carbon, 2005, 43(14), 2951—2959 |
| [19] | Hu G., Meng X., Feng X., Ding Y., Zhang S., Yang M., J. Mater. Sci., 2007, 42(17), 7162—7170 |
| [20] | Gregory J. W., Geoffrey D. W., Ray L. F., Simon A. M., J. Mater. Chem., 2002, 12, 1787—1791 |
| [21] | Bu X. Z., Zhang G. K., Gao Y. Y., Yang Y. Q., Microporous Mesoporous Mater., 2010, 136(1—3), 132—137 |
| [22] | Mohamed A. A., Qais B. A., Shaker H., Nader A. B., Desalination, 2007, 206(1—3), 407—413 |
| [23] | Yap P. S., Lim T. T., Appl. Cata. B-Environ., 2011, 101(3/4), 709—717 |
| [24] | Yin L. S., Shen H., Zhang J. X., Acta Electronica Sinica, 2002, 30(6), 808—810 |
| (尹荔松, 沈辉, 张进修, 电子学报, 2002, 30(6), 808—810) | |
| [25] | Su W. Y., Fu X. Z., Wei K. M., Zhang H. H., Lin H. X., Wang X. C., Li D. Z., Spectrosc. Spect. Anal., 2001, 21(1), 32—34 |
| (苏文悦, 付贤智, 魏可镁, 张汉辉, 林华香, 王心晨, 李旦振. 光谱学与光谱分析, 2001, 21(1), 32—34) | |
| [26] | Wang Y. J., Shi R., Lin J., Zhu Y. F., Appl. Catal. B-Environ., 2010, 100(1/2), 179—183 |
| [27] | Uner D., Tapan N.., Üner M., Appl. Catal. A; Gen., 2003, 251(2), 225—234 |
| [28] | Huang J.C., Preparation and Photocatalytic Performance of TiO2 and Pt/TiO2 Nano Composite Photocatalyst Based on Thermocatalytic Synergetic Effect, Wuhan University of Technology, Wuhan, 2010 |
| (黄济超. 具有光热协同作用的纳米TiO2及Pt/TiO2复合光催化剂的制备和气相光催化性能, 武汉: 武汉理工大学, 2010) | |
| [29] | Li D., Experimental Study on Fixed TiO2 Preparation & Application in Degradation of H2S in Low Concentration, Xi’an University of Architecture and Technology, Xi’an, 2007 |
| (李东. TiO2负载型催化剂的制备及其降解低浓度H2S的实验研究, 西安: 西安建筑科技大学, 2007) | |
| [30] | Leng W. H., Zhang Z., Cheng S. A., Zhang J. Q., Cao C. N., Acta Scientiae Circum Stantiae, 2001, 21(6), 710—714 |
| (冷文华, 张昭, 成少安, 张鉴清, 曹楚男. 环境科学学报, 2001, 21(6), 710—714) | |
| [31] | Fang T., Yang C., Liao L. X., Jing Y. Q., Technology of Water Treatment, 2012, 38(2), 45—48 |
| (方涛, 杨超, 廖丽霞, 景瑛琦. 水处理技术, 2012, 38(2), 45—48) |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||