

Chem. J. Chinese Universities ›› 2015, Vol. 36 ›› Issue (12): 2415.doi: 10.7503/cjcu20150508
• Organic Chemistry • Previous Articles Next Articles
LI Xiuyun1, WAN Chuan1, DU Shijie1, LI Hong1, YUAN Huizhu2, JIANG Jiazhen1, XIAO Yumei1, QIN Zhaohai1,*( )
)
Received:2015-06-30
Online:2015-12-10
Published:2015-11-19
Contact:
QIN Zhaohai
E-mail:qinzhaohai@263.net
Supported by:CLC Number:
TrendMD:
LI Xiuyun, WAN Chuan, DU Shijie, LI Hong, YUAN Huizhu, JIANG Jiazhen, XIAO Yumei, QIN Zhaohai. Synthesis and Fungicidal Activities of Biaryl Methanone O-Benzyl Oximes†[J]. Chem. J. Chinese Universities, 2015, 36(12): 2415.
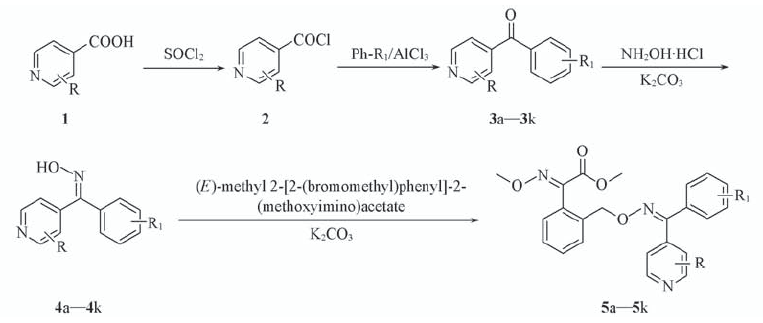
Scheme 1 Synthetic routes of compound 5 2a: R=H; 2b: R=Cl; 3a, 4a, 5a: R=H, R1=H; 3b, 4b, 5b: R=H, R1=4-Me; 3c, 4c, 5c: R=H, R1=4-OMe; 3d, 4d, 5d: R=H, R1=3-Cl-4-Me; 3e, 4e, 5e: R=H, R1=i-Pr; 3f, 4f, 5f: R=H, R1=4-Br; 3g, 4g, 5g: R=H, R1=2,3-2Cl; 3h, 4h, 5h: R=H, R1=4-t-Bu; 3i, 4i, 5i: R=2-Cl, R1=4-Me; 3j, 4j, 5j: R=2-Cl, R1=4-t-Bu; 3k, 4k, 5k: R=2-Cl, R1=4-Ph.
| Compd. | Appearance | Yield(%) | m.p./℃ | 1H NMR(300 MHz, CDCl3, δ) |
|---|---|---|---|---|
| 4c | White solid | 89 | 165—166 | 3.79, 3.81(m, 3H), 6.85(dd, J=8.9, 5.9 Hz, 2H), 7.15(d, J=8.9 Hz, 1H), |
| 7.35(dd, J=13.5, 7.5 Hz, 4H), 8.72(d, J=6.0 Hz, 2H), 10.04(s, 1H) | ||||
| 4d | White solid | 87 | 191—193 | 2.34—2.44(m, 3H), 7.14—7.49(m, 5H), 8.60(d, J=3.0 Hz, 1H), 8.76 |
| (d, J=6.0 Hz, 1H), 10.62(s, 1H) | ||||
| 4e | White solid | 89 | 178—179 | 1.23(s, 3H), 1.26(s, 3H), 2.86—3.00(m, 1H), 7.21(d, J=8.5 Hz, 2H), |
| 7.29—7.49(m, 5H), 8.75(d, J=5.8 Hz, 2H) | ||||
| 4i | White solid | 84 | 179—180 | 2.40(s, 3H), 7.11—7.46(m, 6H), 8.36(d, J=5.1 Hz, 1H), 8.52(d, J= |
| 3.0 Hz, 1H), 9.42(s, 1H) | ||||
| 4j | White solid | 79 | 187—188 | 1.34(s, 9H), 7.30—7.41(m, 4H), 7.51(d, J=8.5 Hz, 1H), 8.37(d, J= |
| 5.1 Hz, 1H), 8.52(d, J=6.0 Hz, 1H), 8.73(s, 1H) |
Table 1 Appearance, yields, melting points and 1H NMR data of compounds 4c—4e, 4i and 4j
| Compd. | Appearance | Yield(%) | m.p./℃ | 1H NMR(300 MHz, CDCl3, δ) |
|---|---|---|---|---|
| 4c | White solid | 89 | 165—166 | 3.79, 3.81(m, 3H), 6.85(dd, J=8.9, 5.9 Hz, 2H), 7.15(d, J=8.9 Hz, 1H), |
| 7.35(dd, J=13.5, 7.5 Hz, 4H), 8.72(d, J=6.0 Hz, 2H), 10.04(s, 1H) | ||||
| 4d | White solid | 87 | 191—193 | 2.34—2.44(m, 3H), 7.14—7.49(m, 5H), 8.60(d, J=3.0 Hz, 1H), 8.76 |
| (d, J=6.0 Hz, 1H), 10.62(s, 1H) | ||||
| 4e | White solid | 89 | 178—179 | 1.23(s, 3H), 1.26(s, 3H), 2.86—3.00(m, 1H), 7.21(d, J=8.5 Hz, 2H), |
| 7.29—7.49(m, 5H), 8.75(d, J=5.8 Hz, 2H) | ||||
| 4i | White solid | 84 | 179—180 | 2.40(s, 3H), 7.11—7.46(m, 6H), 8.36(d, J=5.1 Hz, 1H), 8.52(d, J= |
| 3.0 Hz, 1H), 9.42(s, 1H) | ||||
| 4j | White solid | 79 | 187—188 | 1.34(s, 9H), 7.30—7.41(m, 4H), 7.51(d, J=8.5 Hz, 1H), 8.37(d, J= |
| 5.1 Hz, 1H), 8.52(d, J=6.0 Hz, 1H), 8.73(s, 1H) |
| Compd. | m.p./℃ | Yield(%) | HRMS(calcd.), m/z(M+H)+ | Compd. | m.p./℃ | Yield(%) | HRMS(calcd.), m/z(M+H)+ |
|---|---|---|---|---|---|---|---|
| 5a | 85—87 | 78 | 404.1605(404.1605) | 5g | 103—104 | 70 | 472.0907(472.0825) |
| 5b | —* | 75 | 418.1710(418.1761) | 5h | —* | 75 | 460.2232(460.2231) |
| 5c | —* | 65 | 434.1710(434.1710) | 5i | 84—85 | 71 | 452.1374(452.1372) |
| 5d | 88—90 | 72 | 452.1374(452.1372) | 5j | —* | 74 | 494.1844(494.1841) |
| 5e | —* | 80 | 446.2078(446.2074) | 5k | —* | 67 | 514.1528(514.1528) |
| 5f | 104—106 | 68 | 482.0710(482.0710) |
Table 2 Melting points, yields and HRMS data of compounds 5a—5k
| Compd. | m.p./℃ | Yield(%) | HRMS(calcd.), m/z(M+H)+ | Compd. | m.p./℃ | Yield(%) | HRMS(calcd.), m/z(M+H)+ |
|---|---|---|---|---|---|---|---|
| 5a | 85—87 | 78 | 404.1605(404.1605) | 5g | 103—104 | 70 | 472.0907(472.0825) |
| 5b | —* | 75 | 418.1710(418.1761) | 5h | —* | 75 | 460.2232(460.2231) |
| 5c | —* | 65 | 434.1710(434.1710) | 5i | 84—85 | 71 | 452.1374(452.1372) |
| 5d | 88—90 | 72 | 452.1374(452.1372) | 5j | —* | 74 | 494.1844(494.1841) |
| 5e | —* | 80 | 446.2078(446.2074) | 5k | —* | 67 | 514.1528(514.1528) |
| 5f | 104—106 | 68 | 482.0710(482.0710) |
| Compd. | 1H NMR(300 MHz, CDCl3, δ) | 13C NMR(75 MHz, CDCl3, δ) |
|---|---|---|
| 5a | 3.70(d, J=2.1 Hz, 3H), 3.98(d, J=0.9 Hz, 3H), 5.11(s, 2H), 7.20—7.13(m, 1H), 7.23(dd, J=4.4, 1.6 Hz, 1H), 7.44—7.30(m, 9H), 8.56(dd, J=4.4, 1.7 Hz, 1H), 8.69(dd, J=4.5, 1.5 Hz, 1H) | 52.8, 63.2, 75.1, 122.1, 123.8, 127.6, 128.5, 129.3, 129.8, 131.6, 134.8, 135.8, 141.2, 144.0, 149.3, 149.7, 154.8, 163.7 |
| 5b | 2.37(d, J=15.0 Hz, 3H), 3.71(d, J=0.5 Hz, 3H), 3.98(d, J=3.1 Hz, 3H), 5.10(s, 2H), 7.13—7.22(m, 5H), 7.26(s, 1H), 7.28—7.42(m, 4H), 8.51(d, J=6.2 Hz, 1H), 8.67(d, J=5.1 Hz, 1H) | 21.4, 52.8(s), 63.7(s), 75.0, 127.2—128.1(m), 128.1— 130.0(m), 132.0, 135.9, 139.4, 140.0, 141.3, 149.3, 149.7, 154.9, 163.2 |
| 5c | 3.69(s, 3H), 3.83(s, 3H), 3.99(s, 3H), 5.12(s, 2H), 6.88—6.97(m, 2H), 7.12—7.20(m, 1H), 7.28—7.48(m, 7H), 8.55(d, J =5.6 Hz, 2H) | 52.7, 55.2, 63.7, 75.1, 113.7, 113.8, 122.4, 123.7, 123.8, 127.7, 128.4, 128.8, 129.0, 129.4, 129.7, 131.2, 135.9, 141.4, 144.4, 149.3, 149.8, 154.5, 160.2, 160.9, 163.1 |
| 5d | 2.32—2.41(m, 3H), 3.70(s, 3H), 3.97(d, J=2.1 Hz, 3H), 5.11(s, 2H), 7.12—7.18(m, 2H), 7.21(dd, J=4.4, 1.6 Hz, 1H), 7.26— 7.47(m, 6H), 8.56(dd, J=4.7, 1.4 Hz, 1H), 8.69(dd, J=4.5, 1.5 Hz, 1H) | 20.1, 52.8, 63.7, 75.2, 122.0, 123.8, 126.3, 128.0, 128.1, 128.5, 128.8, 129.1, 129.4, 129.9, 131.5, 133.3, 135.6, 136.3, 140.9, 143.7, 149.2, 149.8, 154.0, 163.1 |
| 5e | 1.25(d, J=6.9 Hz, 6H), 2.77—3.05(m, 1H), 3.68(d, J=4.7 Hz, 3H), 3.97(s, 3H), 5.11(s, 2H), 7.10—7.52(m, 11H), 8.54(d, J=5.5 Hz, 1H), 8.67(d, J=5.5 Hz, 1H) | 23.8, 33.9, 52.7, 63.7, 75.0, 122.2, 123.8, 126.4, 127.7, 128.6, 129.4, 132.4, 135.9, 141.2, 144.2, 149.3, 149.8, 150.1, 150.8, 154.8, 163.1 |
| 5f | 3.71(d, J=1.7 Hz, 3H), 3.98(d, J=2.7 Hz, 3H), 5.10(s, 2H), 7.22—7.13(m, 3H), 7.31—7.23(m, 3H), 7.59—7.35(m, 5H), 8.57(dd, J=4.5, 1.6 Hz, 1H), 8.68(dd, J=3.0, 1.6 Hz, 1H) | 52.8, 63.7, 75.1, 122.1, 123.9, 127.7, 128.1—130.0(m), 131.6, 134.8, 135.8, 141.2, 144.0, 149.3, 149.7, 154.8, 163.2 |
| 5g | 3.71(d, J=2.6 Hz, 3H), 3.97(d, J=1.6 Hz, 3H), 5.11(s, 2H), 7.11—7.20(m, 3H), 7.25—7.30(m, 1H), 7.38(m, 4H), 7.50(dd, J= 5.2, 2.9 Hz, 1H), 8.57(dd, J=4.5, 1.5 Hz, 1H), 8.70(dd, J=3.0, 1.5 Hz, 1H) | 52.8, 63.7, 75.5, 121.7, 123.5, 126.8, 127.9, 128.6, 129.1, 129.4, 130.4, 131.1, 132.8, 133.6, 133.9, 134.8, 135.4, 139.9, 142.8, 149.2, 150.0, 152.7, 163.1 |
| 5h | 1.29(s, 9H), 3.62(d, J=5.5 Hz, 3H), 3.91(s, 3H), 5.09(s, 2H), 7.09—7.25(m, 3H), 7.26—7.45(m, 7H), 8.57(m, 2H) | 31.2, 34.7, 52.7, 63.6, 75.1, 77.0, 122.2, 123.7, 125.2, 127.5, 128.6, 129.3, 129.6, 132.0, 135.9, 141.1, 144.1, 149.3, 149.8, 152.3, 153.0, 154.7, 163.1 |
| 5i | 2.38(d, J=15.5 Hz, 3H), 3.73(d, J=4.6 Hz, 3H), 3.99(d, J=3.1 Hz, 3H), 5.10(s, 2H), 7.10—7.30(m, 7H), 7.33—7.49(m, 4H), 8.31(d, J=5.2 Hz, 1H), 8.45(d, J=5.1 Hz, 1H) | 21.4, 52.8, 63.8, 75.2, 120.9, 122.6, 124.2, 127.4, 127.8, 128.8, 129.2, 129.4, 131.4, 135.7, 139.7, 140.3, 144.3, 147.2, 149.2, 149.6, 151.7, 153.6, 154.0, 163.2 |
| 5j | 1.31(s, 9H), 3.72(s, 3H), 3.99(s, 3H), 5.09(s, 2H), 7.16(s, 2H), 7.24(dd, J=1.2, 0.7 Hz, 1H), 7.28—7.47(m, 7H), 8.45(dd, J=5.0, 0.6 Hz, 1H) | 31.2, 31.2, 31.3, 34.8, 52.8, 63.7, 75.0, 122.2, 123.9, 125.2, 127.3, 127.7, 128.4, 128.7, 128.9, 129.2, 129.3, 129.4, 131.9 , 135.9, 141.5, 149.6, 152.4, 153.2, 154.7, 163.2 |
| 5k | 3.71(s, 3H), 3.97(s, 3H), 5.19(s, 2H), 7.19(t, J=7.0 Hz, 2H), 7.27—7.52(m, 10H), 7.57(dd, J=7.8, 3.5 Hz, 4H), 8.38(m, 1H) | 52.8, 63.7, 75.4, 122.7, 124.3, 127.0, 128.0, 129.0, 129.5, 129.8, 133.1, 135.7, 140.0, 142.7, 144.1, 149.3, 149.9, 151.7, 153.4, 163.1 |
Table 3 NMR data of compounds 5a—5k
| Compd. | 1H NMR(300 MHz, CDCl3, δ) | 13C NMR(75 MHz, CDCl3, δ) |
|---|---|---|
| 5a | 3.70(d, J=2.1 Hz, 3H), 3.98(d, J=0.9 Hz, 3H), 5.11(s, 2H), 7.20—7.13(m, 1H), 7.23(dd, J=4.4, 1.6 Hz, 1H), 7.44—7.30(m, 9H), 8.56(dd, J=4.4, 1.7 Hz, 1H), 8.69(dd, J=4.5, 1.5 Hz, 1H) | 52.8, 63.2, 75.1, 122.1, 123.8, 127.6, 128.5, 129.3, 129.8, 131.6, 134.8, 135.8, 141.2, 144.0, 149.3, 149.7, 154.8, 163.7 |
| 5b | 2.37(d, J=15.0 Hz, 3H), 3.71(d, J=0.5 Hz, 3H), 3.98(d, J=3.1 Hz, 3H), 5.10(s, 2H), 7.13—7.22(m, 5H), 7.26(s, 1H), 7.28—7.42(m, 4H), 8.51(d, J=6.2 Hz, 1H), 8.67(d, J=5.1 Hz, 1H) | 21.4, 52.8(s), 63.7(s), 75.0, 127.2—128.1(m), 128.1— 130.0(m), 132.0, 135.9, 139.4, 140.0, 141.3, 149.3, 149.7, 154.9, 163.2 |
| 5c | 3.69(s, 3H), 3.83(s, 3H), 3.99(s, 3H), 5.12(s, 2H), 6.88—6.97(m, 2H), 7.12—7.20(m, 1H), 7.28—7.48(m, 7H), 8.55(d, J =5.6 Hz, 2H) | 52.7, 55.2, 63.7, 75.1, 113.7, 113.8, 122.4, 123.7, 123.8, 127.7, 128.4, 128.8, 129.0, 129.4, 129.7, 131.2, 135.9, 141.4, 144.4, 149.3, 149.8, 154.5, 160.2, 160.9, 163.1 |
| 5d | 2.32—2.41(m, 3H), 3.70(s, 3H), 3.97(d, J=2.1 Hz, 3H), 5.11(s, 2H), 7.12—7.18(m, 2H), 7.21(dd, J=4.4, 1.6 Hz, 1H), 7.26— 7.47(m, 6H), 8.56(dd, J=4.7, 1.4 Hz, 1H), 8.69(dd, J=4.5, 1.5 Hz, 1H) | 20.1, 52.8, 63.7, 75.2, 122.0, 123.8, 126.3, 128.0, 128.1, 128.5, 128.8, 129.1, 129.4, 129.9, 131.5, 133.3, 135.6, 136.3, 140.9, 143.7, 149.2, 149.8, 154.0, 163.1 |
| 5e | 1.25(d, J=6.9 Hz, 6H), 2.77—3.05(m, 1H), 3.68(d, J=4.7 Hz, 3H), 3.97(s, 3H), 5.11(s, 2H), 7.10—7.52(m, 11H), 8.54(d, J=5.5 Hz, 1H), 8.67(d, J=5.5 Hz, 1H) | 23.8, 33.9, 52.7, 63.7, 75.0, 122.2, 123.8, 126.4, 127.7, 128.6, 129.4, 132.4, 135.9, 141.2, 144.2, 149.3, 149.8, 150.1, 150.8, 154.8, 163.1 |
| 5f | 3.71(d, J=1.7 Hz, 3H), 3.98(d, J=2.7 Hz, 3H), 5.10(s, 2H), 7.22—7.13(m, 3H), 7.31—7.23(m, 3H), 7.59—7.35(m, 5H), 8.57(dd, J=4.5, 1.6 Hz, 1H), 8.68(dd, J=3.0, 1.6 Hz, 1H) | 52.8, 63.7, 75.1, 122.1, 123.9, 127.7, 128.1—130.0(m), 131.6, 134.8, 135.8, 141.2, 144.0, 149.3, 149.7, 154.8, 163.2 |
| 5g | 3.71(d, J=2.6 Hz, 3H), 3.97(d, J=1.6 Hz, 3H), 5.11(s, 2H), 7.11—7.20(m, 3H), 7.25—7.30(m, 1H), 7.38(m, 4H), 7.50(dd, J= 5.2, 2.9 Hz, 1H), 8.57(dd, J=4.5, 1.5 Hz, 1H), 8.70(dd, J=3.0, 1.5 Hz, 1H) | 52.8, 63.7, 75.5, 121.7, 123.5, 126.8, 127.9, 128.6, 129.1, 129.4, 130.4, 131.1, 132.8, 133.6, 133.9, 134.8, 135.4, 139.9, 142.8, 149.2, 150.0, 152.7, 163.1 |
| 5h | 1.29(s, 9H), 3.62(d, J=5.5 Hz, 3H), 3.91(s, 3H), 5.09(s, 2H), 7.09—7.25(m, 3H), 7.26—7.45(m, 7H), 8.57(m, 2H) | 31.2, 34.7, 52.7, 63.6, 75.1, 77.0, 122.2, 123.7, 125.2, 127.5, 128.6, 129.3, 129.6, 132.0, 135.9, 141.1, 144.1, 149.3, 149.8, 152.3, 153.0, 154.7, 163.1 |
| 5i | 2.38(d, J=15.5 Hz, 3H), 3.73(d, J=4.6 Hz, 3H), 3.99(d, J=3.1 Hz, 3H), 5.10(s, 2H), 7.10—7.30(m, 7H), 7.33—7.49(m, 4H), 8.31(d, J=5.2 Hz, 1H), 8.45(d, J=5.1 Hz, 1H) | 21.4, 52.8, 63.8, 75.2, 120.9, 122.6, 124.2, 127.4, 127.8, 128.8, 129.2, 129.4, 131.4, 135.7, 139.7, 140.3, 144.3, 147.2, 149.2, 149.6, 151.7, 153.6, 154.0, 163.2 |
| 5j | 1.31(s, 9H), 3.72(s, 3H), 3.99(s, 3H), 5.09(s, 2H), 7.16(s, 2H), 7.24(dd, J=1.2, 0.7 Hz, 1H), 7.28—7.47(m, 7H), 8.45(dd, J=5.0, 0.6 Hz, 1H) | 31.2, 31.2, 31.3, 34.8, 52.8, 63.7, 75.0, 122.2, 123.9, 125.2, 127.3, 127.7, 128.4, 128.7, 128.9, 129.2, 129.3, 129.4, 131.9 , 135.9, 141.5, 149.6, 152.4, 153.2, 154.7, 163.2 |
| 5k | 3.71(s, 3H), 3.97(s, 3H), 5.19(s, 2H), 7.19(t, J=7.0 Hz, 2H), 7.27—7.52(m, 10H), 7.57(dd, J=7.8, 3.5 Hz, 4H), 8.38(m, 1H) | 52.8, 63.7, 75.4, 122.7, 124.3, 127.0, 128.0, 129.0, 129.5, 129.8, 133.1, 135.7, 140.0, 142.7, 144.1, 149.3, 149.9, 151.7, 153.4, 163.1 |
| Compd. | CL | BB | RS | PC | PA | PI | BC |
|---|---|---|---|---|---|---|---|
| 5a | 51.6 | 31.5 | 50.3 | 42.7 | 32.2 | 62.3 | 9.6 |
| 5b | 46.2 | 32.6 | 48.3 | -5.7 | 33.6 | 68.6 | 27.4 |
| 5c | 43.0 | 23.2 | 45.1 | 50.0 | 30.6 | 59.1 | 26.0 |
| 5d | 66.2 | 54.5 | 61.3 | 59.0 | 49.9 | 76.6 | 32.9 |
| 5e | 48.1 | 13.9 | 45.1 | 47.2 | 21.2 | 66.2 | 9.6 |
| 5f | 52.2 | 34.7 | 44.0 | -6.2 | 33.3 | 68.1 | 37.0 |
| 5g | 50.8 | 31.5 | 46.0 | 36.5 | 43.3 | 71.3 | 27.4 |
| 5h | 49.8 | 26.7 | 33.3 | 44.4 | 36.6 | 41.0 | 4.1 |
| 5i | 43.3 | 5.3 | 26.1 | 11.7 | 34.4 | 39.9 | 5.5 |
| 5j | 33.3 | 1.6 | 9.9 | 17.9 | 26.4 | 25.6 | - |
| 5k | 6.8 | -25.7 | -7.2 | 6.1 | -8.3 | 8.0 | 2.7 |
| Azoxystrobin | 54.3 | 33.7 | 52.6 | 29.2 | 35.0 | 81.9 | 37.0 |
| Trifloxystrobin | 60.8 | 11.7 | 40.5 | 24.7 | 42.1 | 70.5 | 60.3 |
Table 4 In vitro fungicidal activity(inhibition ratio, %) of compounds 5a—5k(50 mg/L)*
| Compd. | CL | BB | RS | PC | PA | PI | BC |
|---|---|---|---|---|---|---|---|
| 5a | 51.6 | 31.5 | 50.3 | 42.7 | 32.2 | 62.3 | 9.6 |
| 5b | 46.2 | 32.6 | 48.3 | -5.7 | 33.6 | 68.6 | 27.4 |
| 5c | 43.0 | 23.2 | 45.1 | 50.0 | 30.6 | 59.1 | 26.0 |
| 5d | 66.2 | 54.5 | 61.3 | 59.0 | 49.9 | 76.6 | 32.9 |
| 5e | 48.1 | 13.9 | 45.1 | 47.2 | 21.2 | 66.2 | 9.6 |
| 5f | 52.2 | 34.7 | 44.0 | -6.2 | 33.3 | 68.1 | 37.0 |
| 5g | 50.8 | 31.5 | 46.0 | 36.5 | 43.3 | 71.3 | 27.4 |
| 5h | 49.8 | 26.7 | 33.3 | 44.4 | 36.6 | 41.0 | 4.1 |
| 5i | 43.3 | 5.3 | 26.1 | 11.7 | 34.4 | 39.9 | 5.5 |
| 5j | 33.3 | 1.6 | 9.9 | 17.9 | 26.4 | 25.6 | - |
| 5k | 6.8 | -25.7 | -7.2 | 6.1 | -8.3 | 8.0 | 2.7 |
| Azoxystrobin | 54.3 | 33.7 | 52.6 | 29.2 | 35.0 | 81.9 | 37.0 |
| Trifloxystrobin | 60.8 | 11.7 | 40.5 | 24.7 | 42.1 | 70.5 | 60.3 |
| Compd. | 400 mg/L(Spraying assay) | 25 mg/L(Spores germ. assay) | ||||
|---|---|---|---|---|---|---|
| CDM | WPM | CSR | CA | RB | CGM | |
| 5a | 0 | 0 | 0 | 0 | 100 | 0 |
| 5b | 0 | 0 | 85 | 95 | 100 | 0 |
| 5c | 0 | 0 | 70 | 60 | 80 | 0 |
| 5d | 70 | 0 | 0 | 0 | 100 | 0 |
| 5e | 0 | 95 | 50 | 0 | 100 | 0 |
| 5f | 0 | 0 | 80 | 0 | 100 | 0 |
| 5g | 35 | 0 | 0 | 0 | 100 | 0 |
| 5h | 0 | 100 | 0 | 100 | 80 | 0 |
| 5i | 0 | 0 | 80 | 0 | 80 | 0 |
| 5j | 0 | 0 | 0 | 0 | 100 | 0 |
| 5k | 0 | 50 | 0 | 0 | 0 | 0 |
| Azoxystrobin | 0 | 100 | 100 | 80 | 80 | 0 |
| Trifloxystrobin | 50 | 100 | 90b | 100 | 100 | 0 |
Table 5 In vivo fungicidal activity(inhibition ratio, %) of compounds 5a—5k*
| Compd. | 400 mg/L(Spraying assay) | 25 mg/L(Spores germ. assay) | ||||
|---|---|---|---|---|---|---|
| CDM | WPM | CSR | CA | RB | CGM | |
| 5a | 0 | 0 | 0 | 0 | 100 | 0 |
| 5b | 0 | 0 | 85 | 95 | 100 | 0 |
| 5c | 0 | 0 | 70 | 60 | 80 | 0 |
| 5d | 70 | 0 | 0 | 0 | 100 | 0 |
| 5e | 0 | 95 | 50 | 0 | 100 | 0 |
| 5f | 0 | 0 | 80 | 0 | 100 | 0 |
| 5g | 35 | 0 | 0 | 0 | 100 | 0 |
| 5h | 0 | 100 | 0 | 100 | 80 | 0 |
| 5i | 0 | 0 | 80 | 0 | 80 | 0 |
| 5j | 0 | 0 | 0 | 0 | 100 | 0 |
| 5k | 0 | 50 | 0 | 0 | 0 | 0 |
| Azoxystrobin | 0 | 100 | 100 | 80 | 80 | 0 |
| Trifloxystrobin | 50 | 100 | 90b | 100 | 100 | 0 |
| [1] | Yang G.L.,Chem. Ind., 2013, 31(12), 45—47 |
| (杨光亮. 化学工业, 2013, 31(12), 45—47) | |
| [2] | Zhang G. S., Pesticides Science and Administration, 2003, 24(12), 30—34 |
| (张国生. 农药科学与管理, 2003, 24(12), 30—34) | |
| [3] | Wang L., Shi Y. X., Li B. J., Liu C. L., Xiang W. S., Pesticides Science and Administration, 2008, 29(9), 24—27 |
| (王丽, 石延霞, 李宝聚, 刘长令, 向文胜. 农药科学与管理, 2008, 29(9), 24—27) | |
| [4] | Guan A. Y., Li H. C., Zhang J. B., Sun X. F., Wang L. Z., Fine and Specialty, 2012, 20(4), 24—28 |
| (关爱莹, 李慧超, 张金波, 孙旭峰, 王立增. 精细与专用化学品, 2012, 20(4), 24—28) | |
| [5] | Zhao P., China Agrochemicals, 2012, 1, 24—30 |
| (赵平. 中国农药. 2012, 1, 24—30) | |
| [6] | Masaji O., Chikako O., Takafumi T., Agricultural Microbicides Containing Methoxyiminoacetate Derivatives, JP 05255012A, 1993-10-05 |
| [7] | Nakafawa Y., Mitani A., Sano H., Hamamura H., Ando T., Sugiura T., Ito S., Preparation of 2-Pyridinecarboxaldehyde Oxime Heterocyclylmethyl Ether Compounds and Agricultural or Horticultural Fungicides, EP 20020700611, 2002-08-29 |
| [8] | Yuan L. P., Chen L., Sheng Z., Zhang Y. B., Huang J. M., Wang M., Qin X. S., Cao J., Li X. L., Preparation of Substituted Propiophenone Oxime Derivatives as Agricultural Bactericides, CN 1640868A, 2005-07-20 |
| (袁莉萍, 陈亮, 沈宙, 张一宾, 黄建明, 王敏, 秦旭升, 曹谨, 栗秀丽. 取代苯丙酮肟衍生物及其制备方法和应用, CN 1640868A, 2005-07-20) | |
| [9] | Dai H., Liu J. B., Miao W. K., Wu S. S., Qin X., Zhang X., Wang T. T., Fang J. X., Chin. J. Org. Chem., 2011, 31(10), 1662—1667 |
| (戴红, 刘建兵, 苗文科, 吴珊珊, 秦雪, 张欣, 王婷婷, 方建新. 有机化学, 2011, 31(10), 1662—1667) | |
| [10] | Mu C. W., Qin Z. H., Mod. Pestic., 2003, 2(2), 1—6 |
| (慕长炜, 覃兆海. 现代农药, 2003, 2(2), 1—6) | |
| [11] | Zhao W. G., Wang J. G., Yuan D. K., Luo T. J., Li Z. M., Pestic., 2002, 41(7), 8—11 |
| (赵卫光, 王建国, 袁德凯, 罗铁军, 李正名. 农药, 2002, 41(7), 8—11) | |
| [12] | Xu J. Y., Dong W. L., Xiong L. X., Li Z. M., Chem. J. Chinese Universities,2012, 33(2), 298—302 |
| (徐俊英, 董卫莉, 熊丽霞, 李正名. 高等学校化学学报,2012, 33(2), 298—302) | |
| [13] | Pei J. J., Ouyang G. P., Zou L. B., Fine Chemical Intermediates,2014, 44(1), 1—9 |
| (裴娟娟, 欧阳贵平, 邹骆波. 精细化工中间体,2014, 44(1), 1—9) | |
| [14] | Massolini G., Kitsos M., Gandini C., Caccialanza G., Pestic. Sci., 1989, 26(2), 209—214 |
| [15] | Shuman R. F., Amstutz E. D., Recueil des Travaux Chimiques des Pays-Bas, 1965, 84(4), 441—459 |
| [16] | Massolini G., Kitsos M., Gandini C., Caccialanza G., Pestic. Sci., 1989, 26(2), 209—214 |
| [17] | Otsuka K., Ishiyama N., Watanabe M., Seri K., Sanai K., Muraoka K., Pyridylketoxime Ethers as Calmodulin Antagonists and Their Preparation EP 366006A1,1990-05-02 |
| [18] | Xu J. G., Zhao X. M., Han X. W., Du Y. G., Pestic. Biochem. Physiol., 2007, 87(3), 220—228 |
| [19] | Li M., Zhang J. B., Yang J. C., Li Z. N., Liu C. L., Li Z. M., Chem. J. Chinese Universities,2009, 30(7), 1348—1352 |
| (李淼, 张金波, 杨吉春, 李志念, 刘长令, 李正名. 高等学校化学学报,2009, 30(7), 1348—1352) |
| [1] | DONG Xinrui, XIA Zhe, WANG Zhenxue, BIAN Qiang, LI Huabin. Design, Synthesis and Biological Activity of Pyrazole-4-carboxamides Compounds Containing 1,2,4,5-Tetrasubstituted Phenyl [J]. Chem. J. Chinese Universities, 2020, 41(12): 2759. |
| [2] | LIU Xiaoyu, XU Yi, TANG Liangfu. Synthesis and Biological Activity of 3-Alkylphosphonate Substituted Isoindolinone Derivatives† [J]. Chem. J. Chinese Universities, 2018, 39(11): 2433. |
| [3] | TAN Ying, XIAO Mengwu, YE Jiao, HU Aixi, ZENG Ziqing, OU Xiaoming. Synthesis, Crystal Structure and Fungicidal Activity of (Z)-3,3-Dimethyl-1-(1H-1,2,4-triazol-1-yl)butan-2-one O-[(5-Aryl-1,3,4-oxadiazol-2-yl)methyl] oxime [J]. Chem. J. Chinese Universities, 2017, 38(8): 1375. |
| [4] | JIA Changqing, YANG Dongyan, CHE Chuanliang, MA Yongqiang, RUI Changhui, YAN Xiaojing, QIN Zhaohai. Synthesis, Structural Characterization, Insecticidal and Fungicidal Activity of (1H-1,2,4-Triazol-5-yl)carbamates† [J]. Chem. J. Chinese Universities, 2016, 37(5): 892. |
| [5] | CHEN Wei, WEI Wei, LI Yuxin, WAN Yingying, LIU Qiaoxia, LI Yonghong, YU Shujing, LI Zhengming. Synthesis and Biological Activity of 2-Methyl-6-nitrobenzenesulfonylurea Derivatives† [J]. Chem. J. Chinese Universities, 2015, 36(5): 907. |
| [6] | XIE Ruilong, SONG Yue, YANG Xinling, WANG Meizi, LING Yun. Synthesis and Fungicidal Activity of Novel Daphneolone Analogues† [J]. Chem. J. Chinese Universities, 2014, 35(7): 1451. |
| [7] | YANG Haikui, XU Wanfu, DUAN Anna, YOU Wenwei, ZHAO Peiliang. Syntheses and Biological Activities of Novel Imine and Imide Derivatives Bearing 1,2,4-Triazole Moiety† [J]. Chem. J. Chinese Universities, 2014, 35(3): 555. |
| [8] | ZHANG Xiu-Lan, WANG Bao-Lei, MAO Ming-Zhen, XIONG Li-Xa, YU Shu-Jing, LI Zheng-Ming. Synthesis and Insecticidal Activity of 5-Chloro-N-[4-chloro- 2-(substitutedcarbamoyl)-6-methylphenyl]-1-aryl-3- (trifluoromethyl)-1H-pyrazole-4-carboxamide [J]. Chem. J. Chinese Universities, 2013, 34(1): 96. |
| [9] | YAN Tao, LIU Peng-Fei, ZHANG Ji-Feng, YU Shu-Jing, XIONG Li-Xia, LI Zheng-Ming. Design, Synthesis and Biological Activities of Novel Ortho-dicarboxyamides [J]. Chem. J. Chinese Universities, 2012, 33(08): 1745. |
| [10] | LIU Peng-Fei, ZHOU Sha, XIONG Li-Xia, YU Shu-Jing, ZHANG Xiao, LI Zheng-Ming. Design, Synthesis and Biological Activities of New Ryanodine Receptor Pesticides Based on Ugi Reaction [J]. Chem. J. Chinese Universities, 2012, 33(04): 738. |
| [11] | YAN Tao, YU Guan-Ping, XIONG Li-Xia, YU Shu-Jing, WANG Su-Hua, LI Zheng-Ming*. Design, Synthesis and Biological Activities of Novel Anthranilic Isophthaloyl Amide [J]. Chem. J. Chinese Universities, 2011, 32(8): 1750. |
| [12] | FENG Qi, LIU Zhi-Li, WANG Ming-Zhong, XIONG Li-Xia, YU Shu-Jing, LI Zheng-Ming*. Design, Synthesis and Biological Activities of Novel Analogues of Chlorantraniliprole Containing Heptafluoroisopropyl Group [J]. Chem. J. Chinese Universities, 2011, 32(1): 74. |
| [13] | LI Yang, ZHANG Zhi-Guo, CHI Hui-Wei, LUO Yan-Mei, LIU Chang-Ling, .... Synthesis and Bioactivity of 2-Aceto-cyanoacetic Acid Derivatives [J]. Chem. J. Chinese Universities, 2010, 31(9): 1798. |
| [14] | ZHANG Xiao-Yan, YANG Guang, ZHANG Jun, ZENG Guang-Tong, TANG Liang-Fu* . Synthesis and Biological Activities of Organotin 2-[(1,2,4-Triazol-1-yl)methylthio]benzoates [J]. Chem. J. Chinese Universities, 2010, 31(6): 1162. |
| [15] | LI Miao, ZHANG Jin-Bo, YANG Ji-Chun, LI Zhi-Nian, LIU Chang-Ling, LI Zheng-Ming. Design, Synthesis and Bioactivity of New N-Methoxycarbamate Containing Pyrazole [J]. Chem. J. Chinese Universities, 2009, 30(7): 1348. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||