

Chem. J. Chinese Universities ›› 2021, Vol. 42 ›› Issue (4): 1299.doi: 10.7503/cjcu20200630
• Article • Previous Articles Next Articles
WANG Renheng1( ), XIAO Zhe1, LI Yan1, SUN Yiling1, FAN Shuting1, ZHENG Junchao2, QIAN Zhengfang1(
), XIAO Zhe1, LI Yan1, SUN Yiling1, FAN Shuting1, ZHENG Junchao2, QIAN Zhengfang1( ), HE Zhenjiang2(
), HE Zhenjiang2( )
)
Received:2020-08-31
Online:2021-04-10
Published:2021-01-05
Contact:
WANG Renheng,QIAN Zhengfang,HE Zhenjiang
E-mail:wangrh@szu.edu.cn;zq001@szu.edu.cn;hzjcsu@csu.edu.cn
Supported by:CLC Number:
TrendMD:
WANG Renheng, XIAO Zhe, LI Yan, SUN Yiling, FAN Shuting, ZHENG Junchao, QIAN Zhengfang, HE Zhenjiang. Synthesis of Li2FeP2O7 Cathode Material at Different Temperatures and Its Electrochemical Performance for Lithium Ion Batteries[J]. Chem. J. Chinese Universities, 2021, 42(4): 1299.
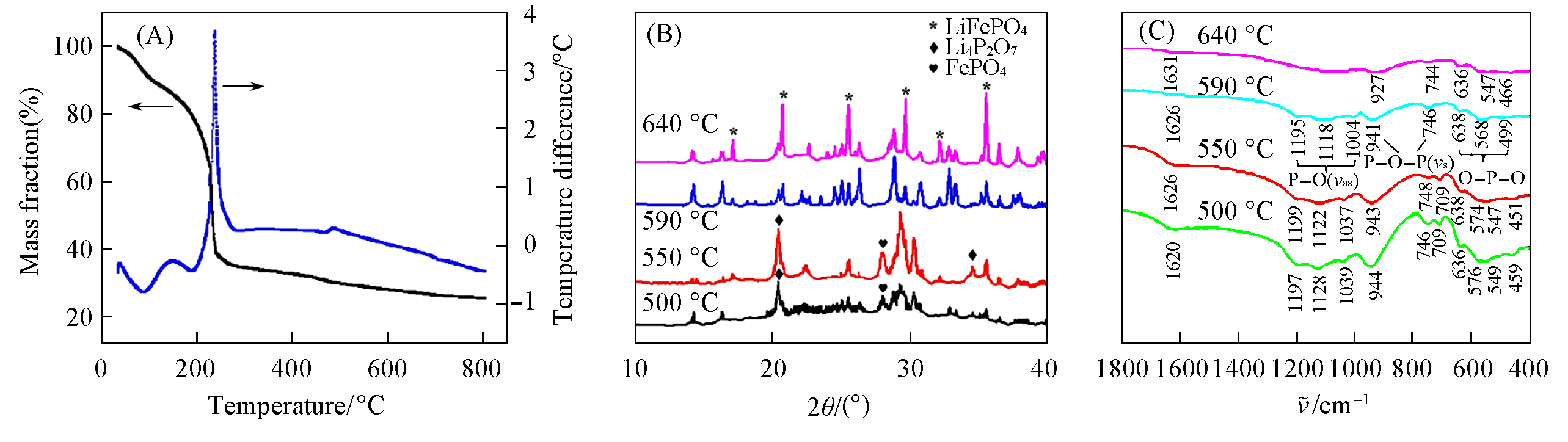
Fig.1 TG?DSC curves of the freeze?dried precursor of Li2FeP2O7(A) and XRD patterns(B) and FTIR spectra(C) of Li2FeP2O7 sintered at 500, 550, 590 and 640 ℃
| Temperature/℃ | Rl/Ω | ωb/Hz | Rr/Ω | 104I0/(mA·cm-2) |
|---|---|---|---|---|
| 500 | 1.695 | 175.8 | 225.81 | 1.14 |
| 550 | 13.04 | 683.6 | 71.13 | 3.61 |
| 590 | 2.504 | 463.9 | 85.55 | 3.00 |
Table 1 Results of electrochemical impedance and exchange current density
| Temperature/℃ | Rl/Ω | ωb/Hz | Rr/Ω | 104I0/(mA·cm-2) |
|---|---|---|---|---|
| 500 | 1.695 | 175.8 | 225.81 | 1.14 |
| 550 | 13.04 | 683.6 | 71.13 | 3.61 |
| 590 | 2.504 | 463.9 | 85.55 | 3.00 |
| 1 | Tarascon J. M., Armand M., Nature, 2001, 414(6861), 359—367 |
| 2 | Choi J. W., Aurbach D., Nat. Rev. Mater., 2016, 1(4), 16013 |
| 3 | Zhang W., Sun X. L., Tang Y. X., Xia H. R., Zeng Y., Qiao L., Zhu Z. Q., Lv Z. S., Zhang Y.Y., Ge X., Xi S. B., Wang Z. G., Du Y. H., Chen X. D., J. Am. Chem. Soc., 2019, 141(36), 14038—14042 |
| 4 | Xiao Z., Wang R. H., Li Y., Sun Y. L., Fan S. T., Xiong K. Y., Zhang H., Qian Z. F., Front. Chem., 2019, 7, 1—8 |
| 5 | Wang R. H., Sun Y. L., Yang K. S., Zheng J. C., Li Y., Qian Z. F., He Z. J., Zhong S. K., J. Energy Chem., 2020, 50, 271—279 |
| 6 | Zhang X. Q., Li T., Li B. Q., Zhang R., Shi P., Yan C., Huang J. Q., Zhang Q., Angew. Chem. Int. Ed., 2020, 59(8), 3252—3257 |
| 7 | Manthiram A., JOM, 1997, 49(3), 43—46 |
| 8 | Wang C. J., Zhu Y. L., Gao F., Qi C., Zhao P. L., Meng Q. F., Wang J. Y., Wu Q. B., Int. J. Energy Res., 2020, 44(7), 5477—5487 |
| 9 | Wang H. B., Liu L. J., Wang R. W., Yan X., Wang Z. Q., Hu J. T., Chen H. B., Jiang S., Ni L., Qiu H. L., Tang H. T., Wei Y. J., Zhang Z. T., Qiu S. L., Pan F., ChemSusChem, 2018, 11(13), 2255—2261 |
| 10 | Liu Y., Gu Y. J., Luo G. Y., Chen Z. L., Wu F. Z., Dai X. Y., Mai Y., Li J. Q., Ceram. Int., 2020, 46(10), 14857—14863 |
| 11 | Zhong Z. Q., Chen L. Z., Zhu C. B., Ren W. B., Kong L. Y., Wan Y. X., J. Power Sources, 2020, 464, 228235 |
| 12 | Chen Y. Q., Xiang K. X., Zhou W., Zhu Y. R., Bai N. B., Chen H., J. Alloys Compd., 2018, 749, 1063—1070 |
| 13 | Wang X. F., Feng Z. J., Huang J. T., Deng W., Li X. B., Zhang H. S., Wen Z. H., Carbon, 2018, 127, 149—157 |
| 14 | Jang H., Taniguchi I., J. Alloys Compd., 2017, 709, 557—565 |
| 15 | Nishimura S., Nakamura M., Natsui R., Yamada A., J. Am. Chem. Soc., 2010, 132(39), 13596—13597 |
| 16 | Zhang J. F., Ji G. J., Zhao R. R., Li D. M., Zhang J. Y., Liu J., Wang C. H., Zhang B., Ou X., Appl. Surf. Sci., 2020, 513, 145854 |
| 17 | Barpanda P., Nishimura S. I., Yamada A., Adv. Energy Mater., 2012, 2(7), 841—859 |
| 18 | Rodríguez R. A., Montoro L. A., Ávila Santos M., Mohallem N. D. S., Laffita Y. M., Pérez⁃Cappe E. L., Electrochim. Acta, 2020, 337, 135712 |
| 19 | Kosova N. V., Tsapina A. M., Slobodyuk A. B., Petrov S. A., Electrochim. Acta, 2015, 174, 1278—1289 |
| 20 | Lee Y., Jo J. H., Park H., Ko W., Kang J., Myung S. T., Sun Y. K., Kim J., Small Methods, 2020, 4(3), 1900847 |
| 21 | Deng C., Zhang S., Wang H. F., Zhang G. M., Nano Energy, 2018, 49, 419—433 |
| 22 | Blidberg A., Häggström L., Ericsson T., Tengstedt C., Gustafsson T., Björefors F., Chem. Mater., 2015, 27(11), 3801—3804 |
| 23 | Nagano H., Taniguchi I., J. Power Sources, 2015, 298, 280—284 |
| 24 | Tan L., Zhang S., Deng C., J. Power Sources, 2015, 275, 6—13 |
| 25 | Jung H. W., Kim Y. J., Ryu K. S., J. Electroceramics, 2020, 44, 78—86 |
| 26 | Jang H., Taniguchi I., J. Alloys Compd., 2017, 709, 557—565 |
| 27 | Jugović D., Mitrić M., Milović M., Ivanovski V. N., Škapin S., Dojčinović B., Uskoković D., J. Alloys Compd., 2019, 786, 912—919 |
| 28 | Jung H. W., Int. J. Electrochem. Sci., 2018, 13(12), 12024—12039 |
| 29 | Ryu D. J., Lee S. H., Ryu K. S., J. Electrochem. Soc., 2017, 164(13), A2930—A2939 |
| 30 | Zhang B., Ou X., Zheng J. C., Shen C., Ming L., Han Y. D., Wang J. L., Qin S. E., Electrochim. Acta, 2014, 133, 1—7 |
| 31 | Guo S. N., Bai Y., Geng Z. F., Wu F., Wu C., J. Energy Chem., 2019, 32, 159—165 |
| 32 | Ma C., Jiang J. L., Xu T. T., Ji H. M., Yang Y., Yang G., ChemElectroChem, 2018, 5(17), 2387—2394 |
| 33 | Wang S. L., Wang R. H., Zhao Q. N., Ren L., Wen J., Chang J., Fang X. L., Hu N., Xu C. H., J. Colloid Interface Sci., 2019, 544, 37—45 |
| 34 | Wang R. H., Dai X. Y., Qian Z. F., Sun Y. L., Fan S. T., Xiong K. Y., Zhang H., Wu F. X., ACS Mater. Lett., 2020, 2(4), 280—290 |
| 35 | Li Y., Zhang H., Xiao Z., Wang R. H., Front. Chem., 2019, 7, 1—8 |
| [1] | ZHANG Shiyu, HE Runhe, LI Yongbing, WEI Shijun, ZHANG Xingxiang. Fabrication of Lithium-sulfur Battery Cathode with Radiation Crosslinked Low Molecular Weight of Polyacrylonitrile and the Mechanism of Sulfur Storage [J]. Chem. J. Chinese Universities, 2022, 43(3): 20210632. |
| [2] | GAO Xiaole, WANG Jiaxin, LI Zhifang, LI Yanchun, YANG Donghua. Synthesis of NiOx-ZSM-5 Composite Materials and Its Electrocatalytic Hydrogen Evolution Performance in Microbial Electrolysis Cell [J]. Chem. J. Chinese Universities, 2021, 42(9): 2886. |
| [3] | BAO Junquan, ZHENG Shibing, YUAN Xuming, SHI Jinqiang, SUN Tianjiang, LIANG Jing. An Organic Salt PTO(KPD)2 with Enhanced Performance as a Cathode Material in Lithium-ion Batteries [J]. Chem. J. Chinese Universities, 2021, 42(9): 2911. |
| [4] | LI Huiyang, ZHU Siying, LI Sha, ZHANG Qiaobao, ZHAO Jinbao, ZHANG Li. Influencing Factors and Promotion Strategies of the First-cycle Coulombic Efficiency of Silicon Suboxide Anodes in Lithium-ion Batteries [J]. Chem. J. Chinese Universities, 2021, 42(8): 2342. |
| [5] | FAN Xiaoyong, WU Yan, SUN Ruibo, GOU Lei, LI Donglin. Construction and Zn Storage Performance of Three Dimensional Porous MnOx@In2O3 Cubes [J]. Chem. J. Chinese Universities, 2021, 42(6): 1816. |
| [6] | LIU Tiefeng, ZHANG Ben, SHENG Ouwei, NAI Jianwei, WANG Yao, LIU Yujing, TAO Xinyong. Research Progress of the Binders for the Silicon Anode [J]. Chem. J. Chinese Universities, 2021, 42(5): 1446. |
| [7] | WANG Yimeng, LIU Kai, WANG Baoguo. Coating Strategies of Ni-rich Layered Cathode in LIBs [J]. Chem. J. Chinese Universities, 2021, 42(5): 1514. |
| [8] | ZHANG Huishuang, GAO Yanxiao, WANG Qiuxian, LI Xiangnan, LIU Wenfeng, YANG Shuting. High-low Temperature Properties of Ni-rich LiNi0.6Co0.2Mn0.2O2 Cathode Material by Hydrothermal Synthesis with CTAB Assisted [J]. Chem. J. Chinese Universities, 2021, 42(3): 819. |
| [9] | HAN Muyao, ZHAO Lina, SUN Jie. Advances in Silicon and Silicon-based Anode Materials [J]. Chem. J. Chinese Universities, 2021, 42(12): 3547. |
| [10] | HUANG Yongfeng, HUANG Wenting, LIU Wenbao, LIU Yuefeng, LIU Wei, XU Chengjun. Mechanism of Storage and Capacity Attenuation of V2O5 as Cathode of Zinc-ion Battery [J]. Chem. J. Chinese Universities, 2020, 41(8): 1859. |
| [11] | LU Di,ZHENG Chunman,CHEN Yufang,LI Yujie,ZHANG Hongmei. Synthesis of Li-rich Layers/Spinel/Carbon Composite Cathode Materials with Phenol Formaldehyde Resin and Its Electrochemical Performance† [J]. Chem. J. Chinese Universities, 2020, 41(7): 1684. |
| [12] | ZHANG Chenyang,WEN Yuehua,ZHAO Pengcheng,CHENG Jie,QIU Jingyi,SUN Yanzhi. Effect of Organic Carbon Source on Performance of LiTi2(PO4)3/C Composite Electrodes in Aqueous Solutions † [J]. Chem. J. Chinese Universities, 2020, 41(6): 1352. |
| [13] | JI Tianyi, LIU Xiaoxu, ZHAO Jiupeng, LI Yao. Synthesis and Lithium-storage Characteristics of Three-dimensional Cross-linked Graphene Nanofibers † [J]. Chem. J. Chinese Universities, 2020, 41(4): 821. |
| [14] | LI Xin, CHEN Liang, MA Xiaotao, ZHANG Ding, XU Shoudong, ZHOU Xianxian, DUAN Donghong, LIU Shibin. Preparation of V2O3 Hollow Spheres for Lithium Sulfur Batteries † [J]. Chem. J. Chinese Universities, 2019, 40(9): 1972. |
| [15] | YANG Jinge, LI Yujie, LU Di, CHEN Yufang, SUN Weiwei, ZHENG Chunman. Morphology Control and Lithium Storage Performance of Micro/nano Li-rich Cathode Material† [J]. Chem. J. Chinese Universities, 2019, 40(7): 1495. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||