

Chem. J. Chinese Universities ›› 2019, Vol. 40 ›› Issue (3): 481.doi: 10.7503/cjcu20180652
• Physical Chemistry • Previous Articles Next Articles
YAO Shouguang1,*( ), CHEN Yong1,2, CHENG Jie2,3, SHEN Yaju2,3, DING Dapei1, YANG Yusheng2,3
), CHEN Yong1,2, CHENG Jie2,3, SHEN Yaju2,3, DING Dapei1, YANG Yusheng2,3
Received:2019-09-26
Online:2019-01-24
Published:2019-01-24
Contact:
YAO Shouguang
E-mail:zjyaosg@126.com
Supported by:CLC Number:
TrendMD:
YAO Shouguang,CHEN Yong,CHENG Jie,SHEN Yaju,DING Dapei,YANG Yusheng. Effect of Potassium Stannate on the Deposition/Dissolution Behavior of Zinc Anode for Zinc-nickel Single-flow Battery†[J]. Chem. J. Chinese Universities, 2019, 40(3): 481.
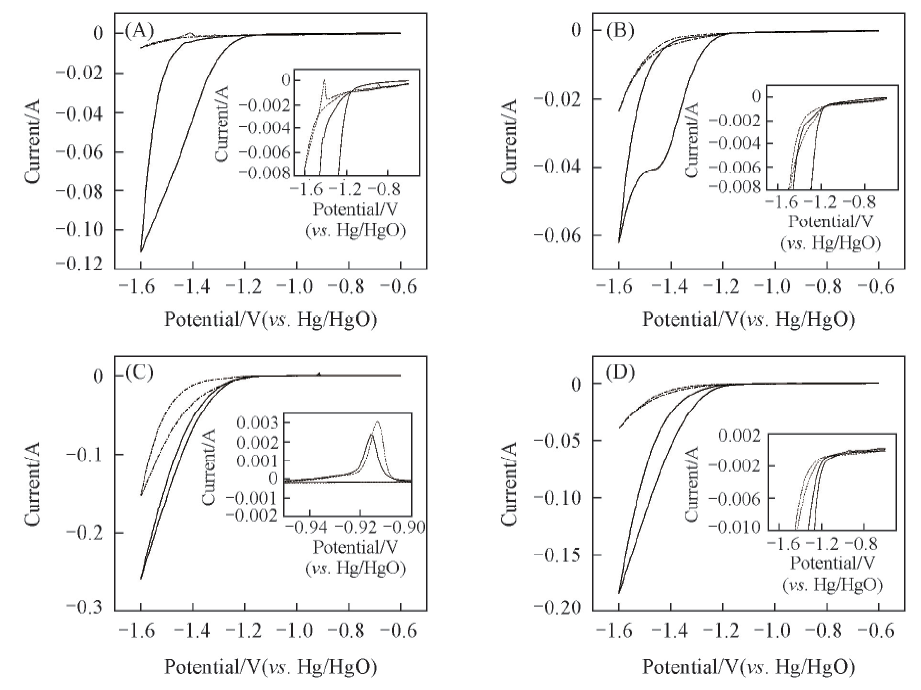
Fig.1 Cyclic voltammetry curves of nickel-plated steel strip in alkaline solution at different scan rates(A), (B): The solution is 8 mol/L KOH+5 g/L LiOH+0.05 mol/L K2SnO3; (C), (D): the solution is 8 mol/L KOH+5 g/L LiOH; (A), (C) at 1 mV/s; (B), (D) at 5 mV/s. — First cycle; fifth cycle.
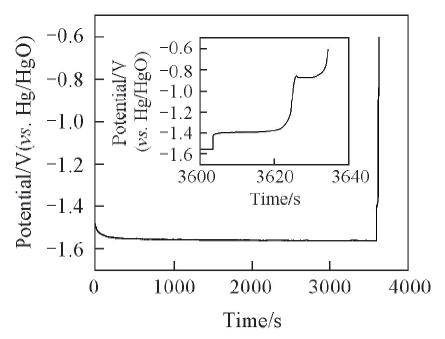
Fig.2 Constant-current charge-discharge curves of nickel-plated steel strip in 8 mol/L KOH+5 g/L LiOH+0.05 mol/L K2SnO3 aqueous solution at a current density of 5 mA/cm2
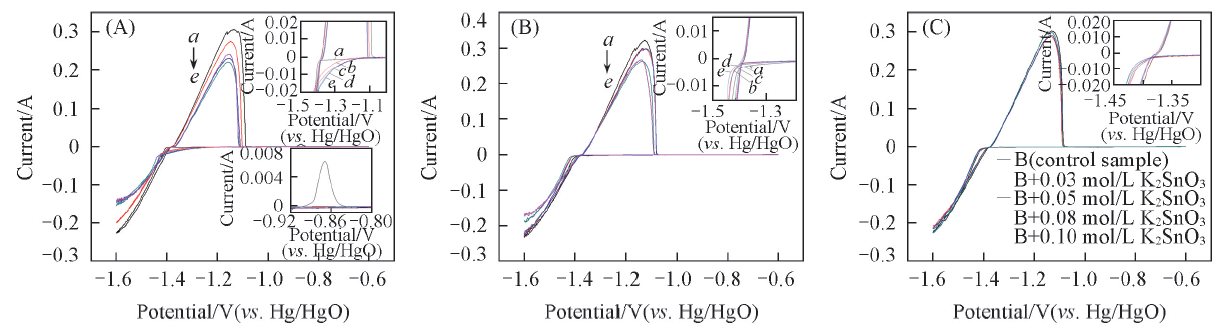
Fig.3 CV curves of the first cycle at scan rates of 1 mV/s(A), 5 mV/s(B) and 10 mV/s(C) on the nickel-plated steel strip electrodesElectrolyte: a. B; b. B+0.03 mol/L K2SnO3; c. B+0.05 mol/L K2SnO3; d. B+0.08 mol/L K2SnO3; e. B+0.1 mol/L K2SnO3. Enlarged curves in the insets, up: initial electroplate of the electrodes; down: another anode peak of control sample.
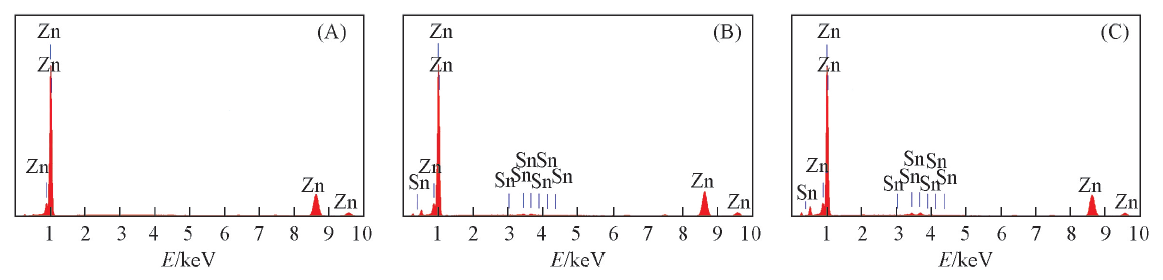
Fig.4 EDS of the surface of nickel-plated steel strip charged at current density of 10 mA/cm2 for 20 min in different electrolytesElectrolyte: (A) B; (B) B+0.05 mol/L K2SnO3; (C) B+0.1 mol/L K2SnO3.
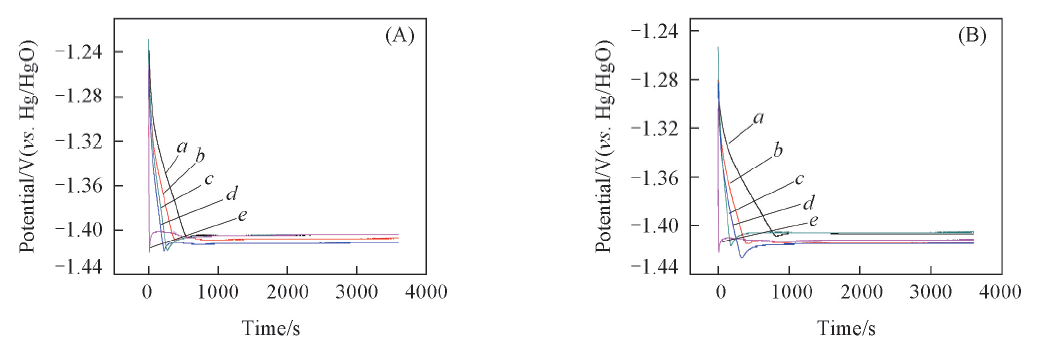
Fig.5 The first charging curves of the nickel-plated steel strip electrodes in five different electrolytes(A) 5 mA/cm2, 1 h; (B) 10 mA/cm2, 0.5 h. Electrolyte: a. B; b. B+0.03 mol/L K2SnO3; c. B+0.05 mol/L K2SnO3; d. B+0.08 mol/L K2SnO3; e. B+0.1 mol/L K2SnO3.
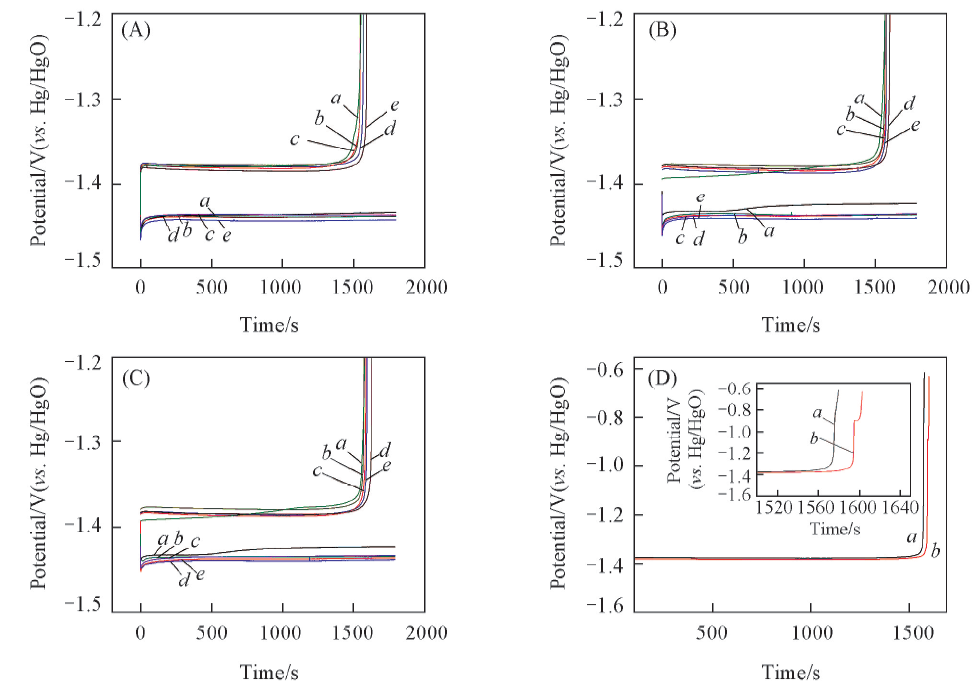
Fig.6 Charge-discharge curves of nickel-plated steel strip electrodes in different electrolytes at 10 mA/cm2(A) 3rd Cycle; (B) 6th cycle; (C) 9th cycle; (D) amplification of the tin deposition potential of the negative electrode on the 9th cycle. Electrolyte: (A)—(C) a. B(control sample); b. B+0.03 mol/L K2SnO3; c. B+0.05 mol/L K2SnO3; d. B+0.08 mol/L K2SnO3; e. B+0.10 mol/L K2SnO3. (D) a. B; b. B+0.08 mol/L K2SnO3.

Fig.7 SEM images of the surface of the nickel-plated steel strip electrode charged at a current density of 10 mA/cm2 for 500 s in different electrolytesElectrolyte: (A) B; (B) B+0.03 mol/L K2SnO3; (C) B+0.05 mol/L K2SnO3; (D) B+0.08 mol/L K2SnO3; (E) B+0.10 mol/L K2SnO3.
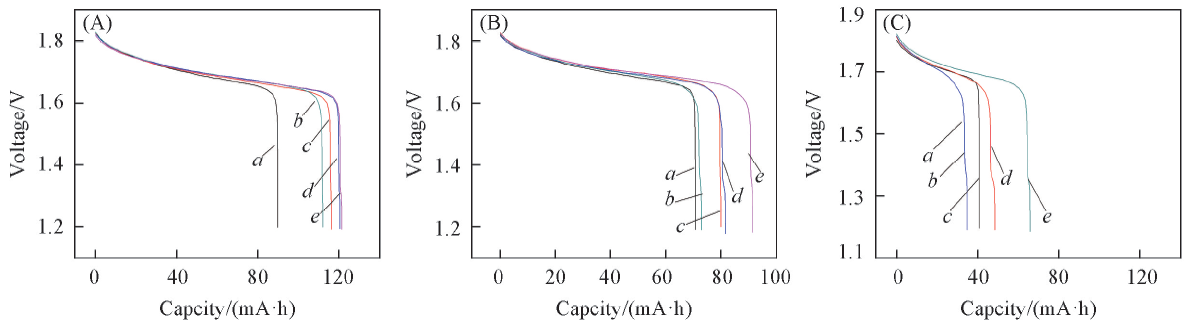
Fig.8 Discharge curves of zinc-nickel single flow battery with nickel-plated steel strip as negative electrode after charging in different electrolytes and resting for different timeCharging and discharging current density is 10 mA/cm2, charged to capacity of 150 mA·h. (A) Discharging after resting of 12 h; (B) discharging after resting of 24 h; (C) discharging after resting of 48 h. Electrolyte: a. B; b. B+0.03 mol/L K2SnO3; c. B+0.05 mol/L K2SnO3; d. B+0.08 mol/L K2SnO3; e. B+0.10 mol/L K2SnO3.
| [1] | Lee C.W.., Sathiyanarayanan K., Eom S. W., Yun M. S.,J. Power Sources, 2006, 160(2), 1436—1441 |
| [2] | SzczeSniak B., Cyrankowska M., Nowacki A., J. Power Sources, 1998, 75(1), 130—138 |
| [3] | Li F., Liu H. J., Wang Y. G., Li H. Q., Xia Y. Y., Chem. J. Chinese Universities, 2007, 28(11), 2133—2137 |
| (李峰, 刘海晶, 王永刚, 李会巧, 夏永姚. 高等学校化学学报, 2007, 28(11), 2133—2137 | |
| [4] | Deng R. R., Fang C., Liu Y. B., Wu Q. Q., Chen L. Z., Battery Bimonthly, 2004, 34(1), 50—52 |
| (邓润荣, 方春, 刘勇标, 吴前麒, 陈力中. 电池, 2004, 34(1), 50—52) | |
| [5] | Wang S. W., Yang Z. H., Zeng L. H., Zhao Y. B., Wang X. H., Chinese J. Power Sources, 2008, 32(4), 58—60 |
| (王升威, 杨占红, 曾利辉, 赵玉彬, 王小花. 电源技术, 2008, 32(4), 58—60) | |
| [6] | Zhu W.H.., Flanzer M. E., Tatarchuk B. J.,J. Power Sources, 2002, 112(2), 353—366 |
| [7] | Yuan Y.F.., Tu J. P., Wu H. H., Li Y., Zhao X. B.,J. Power Sources, 2006, 159(1), 357—360 |
| [8] | Zeng L. H., Yang Z. H., Sang S. B., Zhao Y. B., Battery Bimonthly, 2007, 37(4), 289—291 |
| (曾利辉, 杨占红, 桑商斌, 赵玉彬. 电池, 2007, 37(4), 289—291) | |
| [9] | Zhang C., Wang J. M., Zhang Z., Zhang J. Q., Cao C. N., Chinese Journal of Nonferrous Metals, 2012, 11(5), 780—784 |
| (张春, 王建明, 张昭, 张鉴清, 曹楚南. 中国有色金属学报, 2012, 11(5), 780—784) | |
| [10] | Gao C.Q.., Luo L. C., Shi J. Z., Zhou Y. H.,[J].Wuhan University(Natural Science Edition), 1999, 45(4), 427—431 |
| (高翠琴, 罗列超, 石建珍, 周运鸿. 武汉大学学报(自然科学版), 1999, 45(4), 427—431) | |
| [11] | Liu K. L., He P., Bai H. M., Chen J. C., Dong F. Q., Wang S. B., He M. Q., Yuan S. P., Materials Chemistry and Physics, 2017, 199, 73—78 |
| [12] | Lan C. J., Lee C. Y., Chin T. S., Electrochim Acta, 2007, 52(17), 5407—5416 |
| [13] | Shivkumar R., Kalaignan G.P.., Vasudevan T.,J. Power Sources, 1995, 55(1), 53—62 |
| [14] | Cheng J., Zhang L., Yang Y. S., Wen Y. H., Cao G. P., Wang X. D., Electrochem. Commun., 2007, 9, 2639—2642 |
| [15] | Zhang L., Cheng J., Yang Y.S.., Wen Y. H., Wang X. D., Cao G. P.,J. Power Sources, 2008, 179(1), 381—387 |
| [16] | Ito Y., Nyce M., Plivelich R., Klein M., Banerjee S., J. Power Sources, 2011, 196, 6583—6587 |
| [17] | Cheng J., Wen Y.H.., Cao G. P., Yang Y. S.,J. Power Sources, 2011, 196, 1589—1592 |
| [18] | Ito Y., Nyce M., Plivelich R., Klein M., Steingart D., Banerjee S., J. Power Sources, 2011, 196, 2340—2345 |
| [19] | Ito Y., Wei X., Desai D., Steingart D., Banerjee S., J. Power Sources, 2012, 211, 119—128 |
| [20] | Song S. Y., Pan J. L., Wen Y. H., Cheng J., Pan J. Q., Cao G. P., Chem. J. Chinese Universities, 2014, 35(1), 134—139 |
| (宋世野, 潘君丽, 文越华, 程杰, 潘军青, 曹高萍. 高等学校化学学报, 2014, 35(1), 134—139) | |
| [21] | Cheng Y.H.., Zhang H. M., Lai Q. Z., Li X. F., Zheng Q., Xi X. L., Ding C.,J. Power Sources, 2014, 249, 435—439 |
| [22] | Cheng Y.H.., Zhang H. M., Lai Q. Z., Li X. F., Shi D. Q., Zhang L .Q.,J. Power Sources, 2013, 241, 196—202 |
| [23] | Wen Y.H.., Cheng J., Zhang L., Yan X., Yang S. Y.,J. Power Sources, 2009, 193, 890—894 |
| [24] | Wen Y. H., Wang T., Cheng J., Pan J. Q., Cao G. P., Yang Y. S., Electrochim Acta, 2012, 59, 64—68 |
| [25] | Yuan Y. F., Tu J. P., Wu H. M., Li Y., Zhao X. B., Acta Sci. Natur. Univ. Sunyatseni, 2005, 44(Sup.2), 46—49 |
| (袁永锋, 涂江平, 吴惠明, 黎阳, 赵新兵. 中山大学学报, 2005, 44(Sup.2), 46—49) | |
| [26] | Cheng J., Wen Y. H., Xu Y., Cao G. P., Yang Y. S., Chem. J. Chinese Universities, 2011, 32(11), 2640—2644 |
| (程杰, 文越华, 徐艳, 曹高萍, 杨裕生. 高等学校化学学报, 2011, 32(11), 2640—2644) | |
| [27] | Zeng H. L., Wu Z. D., Chen J. W., Lu P. R., Qin Y. W., Electroplating Process Manual, Machine Press, Beijing, 1997, 149—150 |
| (曾华梁, 吴仲达, 陈钧武, 吕佩仁, 秦月文. 电镀工艺手册, 北京: 机械工业出版社, 1997, 149—150) | |
| [28] | Wang L., Sun Y. H., Chemical World, 1996, 29(11), 20—21 |
| 王玲, 孙宇宏. 化学世界, 1996, 29(11), 20—21 | |
| [29] | Su Z. L., Xu C. Y., Hua Y. X., Li J., Ru J. J., Wang M. M., Xiong L., Zhang Y. D., Int. J. Electrochem. Sc., 2016, 11(5), 3311—3324 |
| [30] | Mansfeld F., Gilman S., J. Electrochem. Soc., 1970, 117(9), 588—592 |
| [1] | SUN Zhumei, FU Jie, LI Xing, WANG Haifang, LU Jing, TONG Tianxing, ZHU Mingfei, WANG Yunyan. Electrochemical Impedance Spectroscopy and Kinetics of Chloride Ion Removal by Electroadsorption [J]. Chem. J. Chinese Universities, 0, (): 20220528. |
| [2] | CHEN Jiaqi, CHENG Wanting, WEN Qiuhui, HAN Jingru, MA Fuqiu, YAN Yongde, XUE Yun. Modification of activated carbon electrode for efficient electrosorption of Co2+, Mn2+ and Ni2+ [J]. Chem. J. Chinese Universities, 0, (): 20220598. |
| [3] | LIU Jie, LI Jinsheng, BAI Jingsen, JIN Zhao, GE Junjie, LIU Changpeng, XING Wei. Constructing a Water-blocking Interlayer Containing Sulfonated Carbon Tubes to Reduce Concentration Polarization in Direct Methanol Fuel Cells [J]. Chem. J. Chinese Universities, 2022, 43(11): 20220420. |
| [4] | LIU Kun, YIN Yuan, GENG Wenqiang, XIA Haotian, LI Hua. Influence Mechanism of Filling Transition Metal Oxide Catalyst with Different Components on Nitrogen Fixation in Dielectric Barrier Discharge [J]. Chem. J. Chinese Universities, 2022, 43(11): 20220278. |
| [5] | DING Qin, ZHANG Zixuan, XU Peicheng, LI Xiaoyu, DUAN Limei, WANG Yin, LIU Jinghai. Effects of Cu, Ni and Co Hetroatoms on Constructions and Electrocatalytic Properties of Fe-based Carbon Nanotubes [J]. Chem. J. Chinese Universities, 2022, 43(11): 20220421. |
| [6] | HE Yujing, LI Jiale, WANG Dongyang, WANG Fuling, XIAO Zuoxu, CHEN Yanli. Zinc-based Activated Fe/Co/N Doped Biomass Carbon Electrocatalysts with High Oxygen Reduction Activity [J]. Chem. J. Chinese Universities, 2022, 43(11): 20220475. |
| [7] | HU Pingao, ZHANG Qi, ZHANG Huiru. Theoretical prediction on the Catalytic Effect of selenium-deficient WSe2 in lithium-sulfur batteries [J]. Chem. J. Chinese Universities, 0, (): 20220595. |
| [8] | WU Yujie, HUANG Wenzhi, PAN Junda, SHI Kaixiang, LIU Quanbing. Design, Regulation and Applications in Lithium-sulfur Battery Cathodes of Yolk-Shell Nanoreactors [J]. Chem. J. Chinese Universities, 0, (): 20220619. |
| [9] | . Progress of Hollow Carbon Materials as Anode for Sodium-Ion Battery [J]. Chem. J. Chinese Universities, 0, (): 20220620. |
| [10] | HOU Congcong, WANG Huiying, LI Tingting, ZHANG Zhiming, CHANG Chunrui, AN Libao. Preparation and Electrochemical Properties of N-CNTs/NiCo-LDH Composite [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220351. |
| [11] | JIANG Baozheng, HUANG Wenting, LIU Wenbao, GUO Rongsheng, XU Chengjun, KANG Feiyu. Preparation of Nano-copper Modified Three-dimensional Zinc Mesh Electrode and Its Performance as Anode for Zinc-ion Batteries [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220257. |
| [12] | LIU Kun, ZUO Jie, LI Hua, XIANG Hongfu, RAN Congfu, YANG Minghao, GENG Wenqiang. Effects of Electron Energy on the Chemical Products of Surface Dielectric Barrier Discharge Plasma [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220249. |
| [13] | WANG Pengfei, FU Wenhao, SUN Shaoni, CAO Xuefei, YUAN Tongqi. Preparation of Hierarchical Porous Carbon Materials Using Cellulose Nanocrystals as Templates and Their Electrochemical Properties [J]. Chem. J. Chinese Universities, 0, (): 20220497. |
| [14] | HU Shiying, SHEN Jiayan, HAN Junshan, HAO Tingting, LI Xing. Preparation of CoO Nanoparticles/Hollow Graphene Nanofiber composites and Its Electrochemical Performances [J]. Chem. J. Chinese Universities, 0, (): 20220462. |
| [15] | REN Shijie, QIAO Sicong, LIU Chongjing, ZHANG Wenhua, SONG Li. Synchrotron Radiation X-Ray Absorption Spectroscopy Research Progress on Platinum Single-atom Catalysts [J]. Chem. J. Chinese Universities, 2022, 43(9): 20220466. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||