

Chem. J. Chinese Universities ›› 2018, Vol. 39 ›› Issue (6): 1178.doi: 10.7503/cjcu20170710
• Analytical Chemistry • Previous Articles Next Articles
CUI Shengfeng1,*( ), WAN Jingwei1, ZHOU Chenghe2
), WAN Jingwei1, ZHOU Chenghe2
Received:2017-11-08
Online:2018-06-10
Published:2018-05-08
Contact:
CUI Shengfeng
E-mail:cuishengfeng@rpc.edu.cn
Supported by:CLC Number:
TrendMD:
CUI Shengfeng, WAN Jingwei, ZHOU Chenghe. Fluorescence Spectroscopic Analysis on the Synergistic Mechanism of Diazepam and Ethanol†[J]. Chem. J. Chinese Universities, 2018, 39(6): 1178.
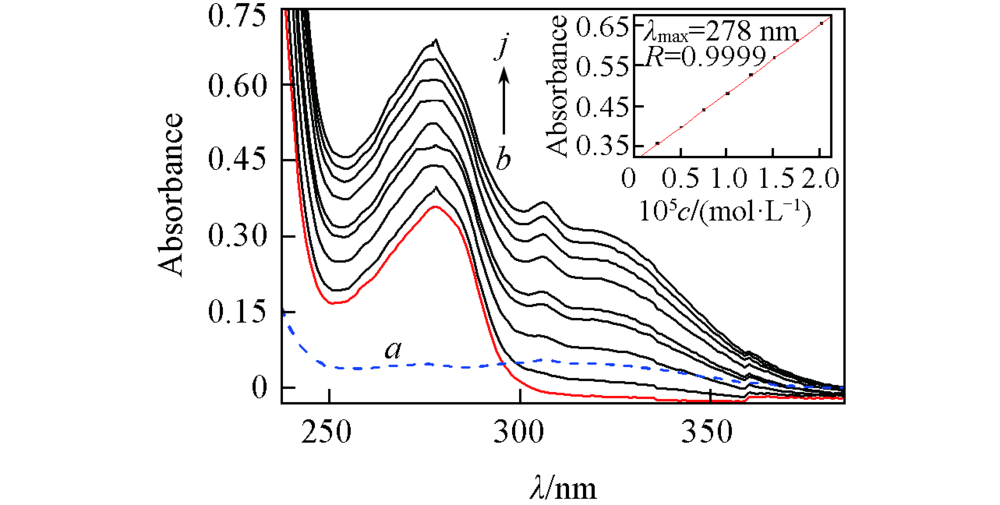
Fig.1 UV-Vis absorption spectra of HSA in the presence of various concentrations of diazepam(298 K, pH=7.4) c(HSA)=1.5×10-5 mol/L; a. diazepam only; concentrations of diazepam are 0—1.6×10-5 mol/L for curves b—j at increment of 0.2. The inset corresponds to the absorbance at 278 nm with different concentrations of diazepam.
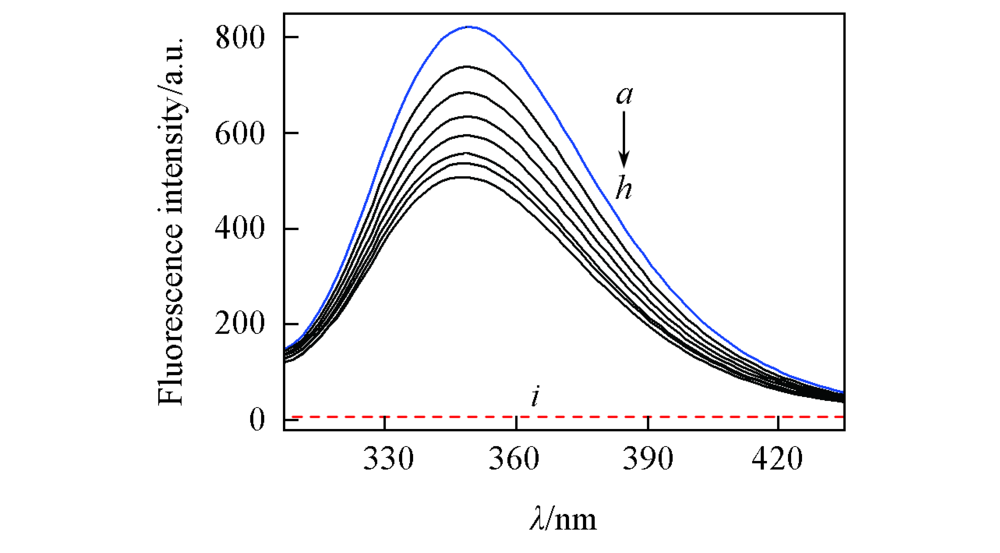
Fig.2 Emission spectra of HSA in the presence of diazepam(T=298 K)c(HSA)=1.5×10-5 mol/L; concentrations of diazepam are 0—1.4×105 mol/L for curves a—h at increment of 0.2; i. diazepam only.
| T/K | 104 KSV/(L·mol-1) | 10-12 Kq/(L·mol-1·s-1) | R | SD |
|---|---|---|---|---|
| 298 | 4.32 | 6.75 | 0.998 | 0.015 |
| 303 | 4.03 | 6.30 | 0.997 | 0.017 |
| 310 | 3.62 | 5.66 | 0.999 | 0.004 |
Table 1 Stern-Volmer constants of the diazepam-HSA system*
| T/K | 104 KSV/(L·mol-1) | 10-12 Kq/(L·mol-1·s-1) | R | SD |
|---|---|---|---|---|
| 298 | 4.32 | 6.75 | 0.998 | 0.015 |
| 303 | 4.03 | 6.30 | 0.997 | 0.017 |
| 310 | 3.62 | 5.66 | 0.999 | 0.004 |
| T/K | 104 Ka/(L·mol-1) | R | SD |
|---|---|---|---|
| 298 | 8.68 | 0.999 | 0.06 |
| 303 | 6.74 | 0.999 | 0.08 |
| 310 | 4.42 | 0.999 | 0.06 |
Table 2 Modified Stern-Volmer constants of diazepam-HSA system(pH=7.4)
| T/K | 104 Ka/(L·mol-1) | R | SD |
|---|---|---|---|
| 298 | 8.68 | 0.999 | 0.06 |
| 303 | 6.74 | 0.999 | 0.08 |
| 310 | 4.42 | 0.999 | 0.06 |
| T/K | 104Kb/(L·mol-1) | n | SD | R |
|---|---|---|---|---|
| 298 | 8.21 | 1.08 | 0.003 | 0.997 |
| 303 | 7.08 | 1.14 | 0.001 | 0.997 |
| 310 | 4.47 | 1.12 | 0.001 | 0.995 |
Table 3 Binding constants and sites of diazepam-HSA system(pH=7.4)
| T/K | 104Kb/(L·mol-1) | n | SD | R |
|---|---|---|---|---|
| 298 | 8.21 | 1.08 | 0.003 | 0.997 |
| 303 | 7.08 | 1.14 | 0.001 | 0.997 |
| 310 | 4.47 | 1.12 | 0.001 | 0.995 |
| Concentration of ethanol/(mg·mL-1) | 104Kb/(L·mol-1) | Kb/K0(%) | R | SD |
|---|---|---|---|---|
| 0.2 | 3.25 | 39.6 | 0.992 | 0.005 |
| 0.4 | 1.30 | 15.8 | 0.991 | 0.002 |
| 0.6 | 2.88 | 35.1 | 0.993 | 0.004 |
| 0.8 | 6.37 | 77.6 | 0.997 | 0.006 |
| 1.0 | 3.82 | 46.5 | 0.993 | 0.005 |
| 1.2 | 2.81 | 34.2 | 0.993 | 0.004 |
| 1.4 | 4.62 | 56.3 | 0.992 | 0.007 |
| 1.6 | 3.78 | 46.0 | 0.994 | 0.005 |
| 1.8 | 4.36 | 53.1 | 0.991 | 0.006 |
Table 4 Effect of ethanol on the Kb value of diazepam-HSA system*
| Concentration of ethanol/(mg·mL-1) | 104Kb/(L·mol-1) | Kb/K0(%) | R | SD |
|---|---|---|---|---|
| 0.2 | 3.25 | 39.6 | 0.992 | 0.005 |
| 0.4 | 1.30 | 15.8 | 0.991 | 0.002 |
| 0.6 | 2.88 | 35.1 | 0.993 | 0.004 |
| 0.8 | 6.37 | 77.6 | 0.997 | 0.006 |
| 1.0 | 3.82 | 46.5 | 0.993 | 0.005 |
| 1.2 | 2.81 | 34.2 | 0.993 | 0.004 |
| 1.4 | 4.62 | 56.3 | 0.992 | 0.007 |
| 1.6 | 3.78 | 46.0 | 0.994 | 0.005 |
| 1.8 | 4.36 | 53.1 | 0.991 | 0.006 |
| Concentration of ethanol/ (mg·kg-1) | Concentration of diazepam/(μg·mL-1) | Concentration of nordiazepam/(μg·mL-1) | ||||
|---|---|---|---|---|---|---|
| 20 min | 40 min | 60 min | 20 min | 40 min | 60 min | |
| 0 | 1.245 | 0.873 | 1.096 | 4.345 | 6.486 | 7.135 |
| 600 | 0.773 | 0.606 | 0.801 | 3.752 | 6.738 | 11.365 |
| 1200 | 0.783 | 0.736 | 0.752 | 3.124 | 6.906 | 12.653 |
| 1800 | 0.521 | 0.504 | 0.691 | 3.426 | 7.094 | 12.974 |
Table 5 Concentrations of diazepam and nordiazepam in the heart blood of mice
| Concentration of ethanol/ (mg·kg-1) | Concentration of diazepam/(μg·mL-1) | Concentration of nordiazepam/(μg·mL-1) | ||||
|---|---|---|---|---|---|---|
| 20 min | 40 min | 60 min | 20 min | 40 min | 60 min | |
| 0 | 1.245 | 0.873 | 1.096 | 4.345 | 6.486 | 7.135 |
| 600 | 0.773 | 0.606 | 0.801 | 3.752 | 6.738 | 11.365 |
| 1200 | 0.783 | 0.736 | 0.752 | 3.124 | 6.906 | 12.653 |
| 1800 | 0.521 | 0.504 | 0.691 | 3.426 | 7.094 | 12.974 |
| [1] | Askgaarda G., Hallasb J., Fink-Jensenc A., Molanderc A. C., Madsenb K. G., Pottegård A., Drug Alcohol Depen., 2016, 161, 258—264 |
| [2] | Rehm J., Anderson P., Barry J., Dimitrov P., Elekes Z., Feijão F., Frick U., Gual A., Gmel G., Kraus L., Marmet S., Raninen J., Rehm M. X., Scafato E., Shield K. D., Trapencieris M., Gmel G., Eur. Addict. Res., 2015, 21(1), 6—18 |
| [3] | Chavez L. J., Liu C. F., Tefft N., Hebert P. L., Clark B. J., Rubinsky A. D., Lapham G. T., Bradley K. A., Drug Alcohol Depen., 2016, 158, 94—101 |
| [4] | Mintzer M. Z., Griffiths R. R., Behav. Pharmacol., 2002, 13(8), 653—658 |
| [5] | Kendler K. S., Ohlsson H., Edwards A. C., Sundquist J., Sundquist K., Drug Alcohol Depen., 2017, 179, 220—228 |
| [6] | Chiara D. C., Jayakar S. S., Zhou X., Zhang X., Savechenkov P. Y., Bruzik K. S., Miller K. W., Cohen J. B., J. Biol. Chem., 2013, 288, 19343—19357 |
| [7] | Enkema M. C., Bowen S., Drug Alcohol Depen., 2017, 179, 1—7 |
| [8] | Griesa D. A., Condouris G. A., Shey Z., Houpt M., Life Sci., 2005, 76(15), 1667—1674 |
| [9] | Hendey G. W., Dery R., Barnes R. L., Snowden B., Mentler P., Am. J. Emerg. Med., 2011, 29(4), 382—385 |
| [10] | Poulos C. X., Zack M., Behav. Pharmacol., 2004, 15(7), 503—512 |
| [11] | Addolorato G., Leggio L., Abenavoli L., Agabio R., Caputo F., Capristo E., Colombo G., Gessa G. L., Gasbarrini G., Am. J. Med., 2006, 119(3), 13—18 |
| [12] | Lader M., Addiction, 2011, 106(12), 2086—2109 |
| [13] | Bartlett G., Abrahamowicz M., Grad R., Sylvestre M. P., Tamblyn R., BMC Fam. Pract., 2009, 10, 1—8 |
| [14] | Cui S. F., Ren Y., Zhang S. L., Peng X. M., Damu G. L. V., Geng R. X., Zhou C. H., Bioorg. Med. Chem. Lett., 2013, 23(11), 3267—3272 |
| [15] | Cui S. F., Peng L. P., Zhang H. Z., Rasheed S., Kumar K. V., Zhou C. H.,Eur. J. Med. Chem., 2014, 86, 318—334 |
| [16] | Hu Y. J., Liu Y., Xiao X. H., Biomacromolecules, 2009, 10(3), 517—521 |
| [17] | Chang J. J., Wang Y., Zhang H. Z., Zhou C. H., Geng R. X., Ji Q. G., Chem.J. Chinese Universities, 2011, 32(9), 1970—1985 |
| (常娟娟, 王艳, 张慧珍, 周成合, 耿蓉霞, 吉庆刚. 高等学校化学学报, 2011, 32(9), 1970—1985) | |
| [18] | Wang Y. H., Zhang X., Zhou X. T., He H., Liu X. Q., J. China Pharm. Univ., 2017, 48(1), 60—65 |
| (王玉浩, 张雪, 周小庭, 何华, 柳晓泉. 中国药科大学学报, 2017, 48(1), 60—65) | |
| [19] | Suryawanshi V. D., Anbhule P. V., Gore A. H., Patil S. R., Kolekar G. B., Ind. Eng. Chem. Res., 2012, 51(1), 95—102 |
| [20] | Cui S. F., Dinesh A., Zhou C. H., J. Med. Chem., 2016, 59(10), 4488—4510 |
| [21] | Yin B. T., Yan C. Y., Peng X. M., Zhang S. L., Rasheed S., Geng R. X., Zhou C. H., Eur. J. Med. Chem., 2014, 71, 148—159 |
| [22] | Zhang L., Chang J. J., Zhang S. L., Damu G. L. V., Geng R. X., Zhou C. H., Bioorg. Med. Chem., 2013, 21(14), 4158—4169 |
| [23] | Ibrahim N., Ibrahim H., Kim S., Nallet J. P., Nepveu F., Biomacromolecules, 2010, 11(12), 3341—3351 |
| [24] | Peng X. M., Cai G. X., Zhou C. H., Curr. Top Med. Chem., 2013, 13, 1963—2010 |
| [25] | Ghuman J., Zunszain P. A., Petitpas I., Bhattacharya A. A., Otagirri M., Curry S., J. Mol. Biol., 2005, 353(1), 38—52 |
| [26] | Zhang S. L., Chang J. J., Damu G. L. V., Fang B., Zhou X. D., Geng R. X., Zhou C. H., Bioorg. Med. Chem. Lett., 2013, 23(4), 1008—1012 |
| [1] | WANG Junyang, LIU Zheng, ZHANG Qian, SUN Chunyan, LI Hongxia. Application of DNA Silver Nanoclusters in the Fluorescence Biosensors based on Functional Nucleic Acids [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220010. |
| [2] | LU Cong, LI Zhenhua, LIU Jinlu, HUA Jia, LI Guanghua, SHI Zhan, FENG Shouhua. Synthesis, Structure and Fluorescence Detection Properties of a New Lanthanide Metal-Organic Framework Material [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220037. |
| [3] | LI Qiao, ZHAO Yang, WANG Enju. Moisture Absorption Reaction and Fluorescence Property of Highly Active Michael System Based on Arylidenemalononitrile [J]. Chem. J. Chinese Universities, 2022, 43(3): 20210690. |
| [4] | TIAN Xueqin, MO Zheng, DING Xin, WU Pengyan, WANG Yu, WANG Jian. A Squaramide-containing Luminescent Metal-organic Framework as a High Selective Sensor for Histidine [J]. Chem. J. Chinese Universities, 2022, 43(2): 20210589. |
| [5] | WU Zexin, ZHU Yuanjie, WANG Hongzhong, WANG Junan, HE Ying. Methyl-modified Carbazole/Diphenyl Sulfone-based AIE-TADF Blue Emitter and Its OLEDs [J]. Chem. J. Chinese Universities, 2022, 43(11): 20220371. |
| [6] | LIU Miao, LIU Ruibo, LIU Badi, QIAN Ying. Synthesis, Two-photon Fluorescence Imaging and Photodynamic Therapy of Lysosome-targeted Indole-BODIPY Photosensitizer [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220326. |
| [7] | MA Jianxin, LIU Xiaodong, XU Na, LIU Guocheng, WANG Xiuli. A Multi-functional Zn(II) Coordination Polymer with Luminescence Sensing, Amperometric Sensing, and Dye Adsorption Performance [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210585. |
| [8] | WU Ji, ZHANG Hao, LUO Yuhui, GENG Wuyue, LAN Yaqian. A Microporous Cationic Ga(III)-MOF with Fluorescence Properties for Selective sensing Fe3+ Ion and Nitroaromatic Compounds [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210617. |
| [9] | HAN Zongsu, YU Xiaoyong, MIN Hui, SHI Wei, CHENG Peng. A Rare Earth Metal-Organic Framework with H6TTAB Ligand [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210342. |
| [10] | LI Ran, ZHANG Xudong, MU Lidan, SUN Tong, AI Ganggang, SHA Yelong, ZHANG Yuqi, WANG Jijiang. Preparation and Application of Triplethiophene Derivative Functionalized SiO2 Inverse Opal Photonic Crystal Fluorescent Films [J]. Chem. J. Chinese Universities, 2021, 42(9): 2989. |
| [11] | YUAN Chunling, YAO Xiaotiao, XU Yuanjin, QIN Xiu, SHI Rui, CHENG Shiqi, WANG Yilin. Colorimetry/Ratio Fluorimetry Determination of Glucose with Bifunctional Carbon Dots [J]. Chem. J. Chinese Universities, 2021, 42(8): 2428. |
| [12] | ZHOU Jieqiong, HUANG Yan, ZHANG Zhiling, PANG Daiwen, TIAN Zhiquan. Water-soluble Ag2Te Quantum Dots with Emission in the Second Near-infrared Window [J]. Chem. J. Chinese Universities, 2021, 42(6): 2072. |
| [13] | CHEN Hongda, ZHANG Hua, WANG Zhenxin. Development of Small Animals in vivo Fluorescence-photothermal Dual Mode Imaging System [J]. Chem. J. Chinese Universities, 2021, 42(3): 725. |
| [14] | LI Mengshuo, ZHANG Jing, LIU Dan, ZHU Yaxian, ZHANG Yong. Interactions of Pyrene with Human Serum Albumin and Bovine Serum Albumin: Microenvironmental Polarity Differences at Binding Sites [J]. Chem. J. Chinese Universities, 2021, 42(3): 731. |
| [15] | KUANG Xiaojun, YI Jingwei, FANG Xiaoxia, LAI Dongmei, XU Hong. Preparation of Water-soluble Coumarin Fluorescent Substrate and Its Application in Droplet Based Digital Detection [J]. Chem. J. Chinese Universities, 2021, 42(11): 3537. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||