

Chem. J. Chinese Universities ›› 2017, Vol. 38 ›› Issue (6): 1033.doi: 10.7503/cjcu20170068
• Analytical Chemistry • Previous Articles Next Articles
ZHANG Na1, WAN Xin1, SONG Xuyan2, HUANG Long2, ZHENG Jingjing3, XING Jun1,*( )
)
Received:2017-02-03
Online:2017-06-10
Published:2017-05-23
Contact:
XING Jun
E-mail:gc-ms@263.net
Supported by:CLC Number:
TrendMD:
ZHANG Na, WAN Xin, SONG Xuyan, HUANG Long, ZHENG Jingjing, XING Jun. Application of Octadecyl Ionic Liquid Hybrid Solid-phase Microextraction Monolithic Column in the Detection of Polycyclic Aromatic Hydrocarbons in Coffee†[J]. Chem. J. Chinese Universities, 2017, 38(6): 1033.
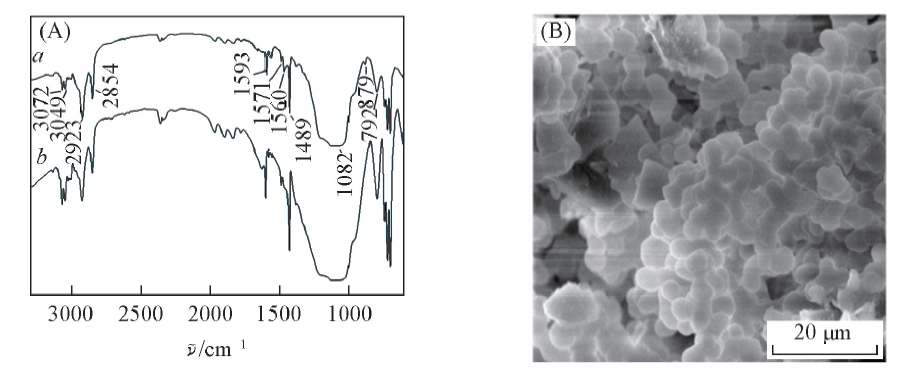
Fig.1 FTIR spectra(A) and SEM image(B) of C18IL in-tube SPME a. Ionic liquids hybrid monolithic column; b. hybrid monolithic column without ionic liquid.
| PAHs | RSD(%) | |
|---|---|---|
| Repeatability(n=3) | Column-to-column repeatability(n=3) | |
| Nap | 10.1 | 12.2 |
| Flu | 7.4 | 7.9 |
| Phe | 6.9 | 7.2 |
| FL | 4.8 | 11.7 |
| Pyr | 4.9 | 9.4 |
Table 1 Repeatability of the C18IL in-tube SPME
| PAHs | RSD(%) | |
|---|---|---|
| Repeatability(n=3) | Column-to-column repeatability(n=3) | |
| Nap | 10.1 | 12.2 |
| Flu | 7.4 | 7.9 |
| Phe | 6.9 | 7.2 |
| FL | 4.8 | 11.7 |
| Pyr | 4.9 | 9.4 |
| PAHs | Linear range/ (μg·L-1) | Linear regression | LOD/ (μg·L-1) | LOQ/ (μg·L-1) | RSD(%, n=5) | |||
|---|---|---|---|---|---|---|---|---|
| Slope | Intercept | R2 | Intra-day | Inter-day | ||||
| Nap | 0.4—200 | 605.28 | 1192.7 | 0.9998 | 0.072 | 0.240 | 7.9 | 9.6 |
| Flu | 0.1—200 | 1192.00 | -152.13 | 0.9994 | 0.016 | 0.053 | 6.3 | 9.7 |
| Phe | 0.1—200 | 1081.40 | 480.43 | 0.9997 | 0.008 | 0.027 | 5.3 | 12.6 |
| FL | 0.1—200 | 845.05 | -424.32 | 0.9992 | 0.007 | 0.023 | 2.0 | 8.4 |
| Pyr | 0.1—200 | 770.26 | 120.27 | 0.9995 | 0.013 | 0.043 | 2.2 | 4.9 |
Table 2 Analytical performance of the C18IL in-tube-SPME-GC-FID method for 5 PAHs analysis
| PAHs | Linear range/ (μg·L-1) | Linear regression | LOD/ (μg·L-1) | LOQ/ (μg·L-1) | RSD(%, n=5) | |||
|---|---|---|---|---|---|---|---|---|
| Slope | Intercept | R2 | Intra-day | Inter-day | ||||
| Nap | 0.4—200 | 605.28 | 1192.7 | 0.9998 | 0.072 | 0.240 | 7.9 | 9.6 |
| Flu | 0.1—200 | 1192.00 | -152.13 | 0.9994 | 0.016 | 0.053 | 6.3 | 9.7 |
| Phe | 0.1—200 | 1081.40 | 480.43 | 0.9997 | 0.008 | 0.027 | 5.3 | 12.6 |
| FL | 0.1—200 | 845.05 | -424.32 | 0.9992 | 0.007 | 0.023 | 2.0 | 8.4 |
| Pyr | 0.1—200 | 770.26 | 120.27 | 0.9995 | 0.013 | 0.043 | 2.2 | 4.9 |
| Method | Sample | LOD/(μg·L-1) | Ref. | |||||
|---|---|---|---|---|---|---|---|---|
| Nap | Flu | Phe | FL | Pyr | Nap | |||
| C18IL in-tube SPME(10 cm)-GC-FID | Coffee | 0.072 | 0.016 | 0.008 | 0.007 | 0.013 | 0.072 | This work |
| LDS-DLLMEa-GC-MS | Water | 0.058 | 0.023 | 0.028 | 0.038 | 0.037 | 0.058 | [ |
| Polyester fibers-in-tube(30 cm) SPME-HPLC-DAD | Water | 0.01 | 0.01 | 0.01 | 0.03 | 0.01 | 0.01 | [ |
| Poly(SAM-DVB) monolithic(50 cm)-CEC-APCI-MS | Seafood | 26.7 | 2.7 | 1.1 | [ | |||
| MSPEb-HPLC-FLD | Coffee and tea | 0.005 | [ | |||||
| Si-MWCNTs monolithic(3 cm)-SPME-GC-MS | Water | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | [ | |
Table 3 LODs of PHAs by different analysis methods
| Method | Sample | LOD/(μg·L-1) | Ref. | |||||
|---|---|---|---|---|---|---|---|---|
| Nap | Flu | Phe | FL | Pyr | Nap | |||
| C18IL in-tube SPME(10 cm)-GC-FID | Coffee | 0.072 | 0.016 | 0.008 | 0.007 | 0.013 | 0.072 | This work |
| LDS-DLLMEa-GC-MS | Water | 0.058 | 0.023 | 0.028 | 0.038 | 0.037 | 0.058 | [ |
| Polyester fibers-in-tube(30 cm) SPME-HPLC-DAD | Water | 0.01 | 0.01 | 0.01 | 0.03 | 0.01 | 0.01 | [ |
| Poly(SAM-DVB) monolithic(50 cm)-CEC-APCI-MS | Seafood | 26.7 | 2.7 | 1.1 | [ | |||
| MSPEb-HPLC-FLD | Coffee and tea | 0.005 | [ | |||||
| Si-MWCNTs monolithic(3 cm)-SPME-GC-MS | Water | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | [ | |
| PAHs | Concentration/(μg·L-1) | Recovery(%) | RSD(%, n=5) |
|---|---|---|---|
| Nap | 1 | 94.68 | 8.4 |
| 10 | 100.00 | 4.8 | |
| 100 | 90.10 | 8.2 | |
| Flu | 1 | 101.63 | 10.2 |
| 10 | 92.98 | 10.0 | |
| 100 | 85.79 | 12.1 | |
| Phe | 1 | 97.63 | 4.3 |
| 10 | 98.98 | 8.8 | |
| 100 | 87.20 | 17.7 | |
| FL | 1 | 103.42 | 8.8 |
| 10 | 97.13 | 6.8 | |
| 100 | 91.80 | 12.9 | |
| Pyr | 1 | 98.65 | 9.8 |
| 10 | 99.78 | 8.9 | |
| 100 | 93.40 | 11.8 |
Table 4 Recoveries of 5 PHAs in the coffee samples
| PAHs | Concentration/(μg·L-1) | Recovery(%) | RSD(%, n=5) |
|---|---|---|---|
| Nap | 1 | 94.68 | 8.4 |
| 10 | 100.00 | 4.8 | |
| 100 | 90.10 | 8.2 | |
| Flu | 1 | 101.63 | 10.2 |
| 10 | 92.98 | 10.0 | |
| 100 | 85.79 | 12.1 | |
| Phe | 1 | 97.63 | 4.3 |
| 10 | 98.98 | 8.8 | |
| 100 | 87.20 | 17.7 | |
| FL | 1 | 103.42 | 8.8 |
| 10 | 97.13 | 6.8 | |
| 100 | 91.80 | 12.9 | |
| Pyr | 1 | 98.65 | 9.8 |
| 10 | 99.78 | 8.9 | |
| 100 | 93.40 | 11.8 |
| [1] | Orecchio S., Ciotti V.P., Culotta L., Food Chem. Toxicol., 2009, 47(4), 819—826 |
| [2] | Chu Y.F., Coffee: Emerging Health Effects and Disease Prevention, John Wiley & Sons, Ames, 2012, 197—205 |
| [3] | Shi Y., Wu H., Wang C., Guo X., Du J., Du J., Food Chem., 2016, 199, 75—80 |
| [4] | Ballesteros R., Hernández J.J., Lyons L. L., Atmos. Environ., 2009, 43(3), 655—662 |
| [5] | Dasgupta S, Banerjee K, Utture S, Kusan P., Wagh S., Dhumal K., Kolekar S., Adsule P.G., J. Chromatogr. A, 2011, 1218(38), 6780—6791 |
| [6] | Xu H., Ding Z.Q., Feng Y. Q., Anal. Chim. Acta, 2009, 636(1), 28—33 |
| [7] | Castro D., Slezakova K., Oliva-Teles M.T., Delerue-Matos C., Alvim-Ferraz M. C., Morais S., Pereira M. C., J. Sep. Sci., 2009, 32(4), 501—510 |
| [8] | Navarro P., Etxebarria N., Arana G., Anal. Chim. Acta, 2009, 648(2), 178—182 |
| [9] | Kouzayha A., Al Iskandarani M., Mokh S., Rabaa A.R., Budzinski H., Jaber F., J. Agr. Food Chem., 2011, 59(14), 7592—7600 |
| [10] | Martendal E., Carasek E., J. Chromatogr. A, 2011, 1218(13), 1707—1714 |
| [11] | Liu W., Qi J., Yan L Jia Q., Yu C., J. Chromatogr. B, 2011, 879(28), 3012—3016 |
| [12] | Guan X., Zhao C., Liu X., Zhang H., J. Chromatogr. A, 2013, 1302, 28—33 |
| [13] | Han D., Row K.H., Molecules, 2010, 15(4), 2405—2426 |
| [14] | Tian M., Liu J., Row K.H., Molecules, 2009, 14, 2127—2134 |
| [15] | Zhou Q., Zhang X., Xiao J., J. Chromatogr. A, 2009, 1216(20), 4361—4365 |
| [16] | Zhang M., Tu X.N., Wang J. Y., Fang T., Wang Y. L., Xu X. D., Zhang M. L., Chen Y. T., Chem. Res. Chinese Universities, 2016, 32(4), 530—533 |
| [17] | Yu F.L., Xie P. H., Zhu G. Q., Yuan B., Xie C. X., Yu S. T., Chem. J. Chinese Universities, 2016, 37(12), 2184—2190 |
| (于凤丽, 谢盼辉, 朱国强, 袁冰, 解从霞, 于世涛. 高等学校化学学报, 2016, 37(12), 2184—2190) | |
| [18] | Abdolmohammad-Zadeh H., Galeh-Assadi M., Shabkhizan S., Mousazadeh H., Arab. J. Chem., 2016, 9, S587—S594 |
| [19] | Zhang H., Bai L.G., Wei Z., Liu S., Liu H. Y., Yan H. Y., Talanta, 2016, 149, 62—68 |
| [20] | Shan Y.H., QiaoL. Z., Shi X. Z., Xu G. W., J. Chromatogr. A, 2015, 1375, 101—109 |
| [21] | Litschauer M., Neouze M.A., J. Mater. Chem., 2008, 18(6), 640—646 |
| [22] | Cazin C.S. J., Veith M., Braunstein P., Bedford R. B., Synthesis, 2005, 2005(4), 622—626 |
| [23] | Likhanova N.V., Domínguez-Aguilar M. A., Olivares-Xometl O., Nava-Entzana N., Arce E., Dorantes H., Corros. Sci., 2010, 52(6), 2088—2097 |
| [24] | Zheng M.M., Lin B., Feng Y Q., J. Chromatogr. A, 2007, 1164(1), 48—55 |
| [25] | Yan L.J., Zhang Q. H., Feng Y. Q., Zhang W. B., Li T., Zhang L. H., Zhang Y. K., J. Chromatogr. A, 2006, 1121(1), 92—98 |
| [26] | Jeong B., Kim S.W., Bae Y. H., Adv. Drug Deliver. Rev., 2012, 64, 154—162 |
| [27] | Guo L., Tan S., Li X., Lee H.K., J. Chromatogr. A, 2016, 1438, 1—9 |
| [28] | Bu Y., Feng J., Sun M., Zhou C.L., Luo C. N., Anal. Bionanl. Chem., 2016, 1—12 |
| [29] | Cheng Y.J., Huang S. H., Chiu J. Y., Liu W. L., Huang H. Y., J. Chromatogr. A, 2013, 1313, 132—138 |
| [30] | Shi Y., Wu H., Wang C., Guo X., Du J., Du L., Food Chem., 2016, 199, 75—80 |
| [31] | Fresco-Cala B., Cárdenas S., Valcárcel M., J. Chromatogr. A, 2016, 1468, 55—63 |
| [32] | Houessou J.K., Benac C., Delteil C., Camel V., J. Agr. Food Chem., 2005, 53(4), 871—879 |
| [1] | CUI Wei, ZHAO Deyin, BAI Wenxuan, ZHANG Xiaodong, YU Jiang. CO2 Absorption in Composite of Aprotic Solvent and Iron-based Ionic Liquid [J]. Chem. J. Chinese Universities, 2022, 43(8): 20220120. |
| [2] | PENG Kuilin, LI Guilin, JIANG Chongyang, ZENG Shaojuan, ZHANG Xiangping. Research Progress for the Role of Electrolytes in the CO2 Electrochemical Reduction [J]. Chem. J. Chinese Universities, 2022, 43(7): 20220238. |
| [3] | JI Shuangqi, JIN Zhao, GUAN Wenna, PAN Xiangyu, GUAN Tong. Preparation and Chromatographic Performance of Mixed-mode Silica Stationary Phase Modified by Double Cationic Ionic Liquid and Octadecyl Group [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220008. |
| [4] | DENG Hongri, CAO Xiaomei, WANG Jingbo, LI Xiangyuan. Rate Rules for Hydrogen Abstraction Reactions of Polycyclic Aromatic Hydrocarbons and Unsaturated Radicals [J]. Chem. J. Chinese Universities, 2022, 43(2): 20210563. |
| [5] | CHANG Sihui, CHEN Tao, ZHAO Liming, QIU Yongjun. Thermal Degradation Mechanism of Bio-based Polybutylactam Plasticized by Ionic Liquids [J]. Chem. J. Chinese Universities, 2022, 43(11): 20220353. |
| [6] | WAN Ren, SONG Fan, PENG Changjun, LIU Honglai. Group Contribution Method for Infinite Dilution Molar Conductivity of Unconventional Ions in Water [J]. Chem. J. Chinese Universities, 2021, 42(12): 3672. |
| [7] | WANG Man, WANG Xin, ZHOU Jing, GAO Guohua. Efficient Synthesis of Dimethyl Carbonate via Transesterification of Methanol and Ethylene Carbonate Catalyzed by Poly(ionic liquid)s [J]. Chem. J. Chinese Universities, 2021, 42(12): 3701. |
| [8] | ZHOU Molin, JIANG Xin, YI Ting, YANG Xiangguang, ZHANG Yibo. Improvement of Interface Stability Between Sulfide Solid Electrolyte Li10GeP2S12 and Lithium Metal [J]. Chem. J. Chinese Universities, 2020, 41(8): 1810. |
| [9] | GAO Chong,YU Fengli,XIE Congxia,YU Shitao. Baeyer-Villiger Oxidation of Cyclic Ketones Catalyzed by Amino Alcohol Heteropoly Acid Ionic Liquid † [J]. Chem. J. Chinese Universities, 2020, 41(5): 1101. |
| [10] | GAO Naiwei, MA Qiang, HE Yonglin, WANG Yapei. Green Electronic Devices Based on Ionic Liquids † [J]. Chem. J. Chinese Universities, 2020, 41(5): 901. |
| [11] | CHENG Shifu,HU Hao,CHEN Bihua,WU Haihong,GAO Guohua,HE Mingyuan. Preparation and Electrochemical Performance of Porous Carbons Prepared from Binary Ionic Liquids † [J]. Chem. J. Chinese Universities, 2020, 41(5): 1048. |
| [12] | PIAO Huilan,MA Pinyi,QIN Zucheng,JIANG Yanxiao,SUN Ying,WANG Xinghua,SONG Daqian. Determination of Triazine Herbicides from Fruit Juice Samples Using Effervescence Assisted Microextraction Method Based on Acidic Ionic Liquid Packed Syringe [J]. Chem. J. Chinese Universities, 2020, 41(2): 228. |
| [13] | ZHANG Li,QIAN Mingchao,LIU Xueke,Gao Shuaitao,YU Jiang,XIE Haishen,WANG Hongbin,SUN Fengjiang,SU Xianghong. Dynamic Study of Oxidative Desulfurization by Iron-based Ionic Liquids/NHD † [J]. Chem. J. Chinese Universities, 2020, 41(2): 317. |
| [14] | WANG Nan,YAO Kaisheng,ZHAO Chenchen,LI Tianjin,LU Weiwei. Ionic Liquid-assisted Synthesis of AuPd Nanosponges and Their Catalytic Performance † [J]. Chem. J. Chinese Universities, 2020, 41(1): 62. |
| [15] | LI Chenguang, HUA Er, LIU Tianxia. Tribological Behaviour of Protic Ionic Liquid Composed of 2-Ethylhexylethylenediaminium Cation and Trifluoromethanesulfonate Anion as Liquid Paraffin Additive† [J]. Chem. J. Chinese Universities, 2019, 40(7): 1411. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||