

Chem. J. Chinese Universities ›› 2016, Vol. 37 ›› Issue (5): 938.doi: 10.7503/cjcu20150943
• Physical Chemistry • Previous Articles Next Articles
YAO Huiqin1,*( ), HUANG Shan1, SU Qiaoling1, SHI Keren2,*, GAN Qianqian1, WANG Mingke1
), HUANG Shan1, SU Qiaoling1, SHI Keren2,*, GAN Qianqian1, WANG Mingke1
Received:2015-12-10
Online:2016-05-10
Published:2016-04-22
Contact:
YAO Huiqin,SHI Keren
E-mail:huiqin_yao@163.com
CLC Number:
TrendMD:
YAO Huiqin, HUANG Shan, SU Qiaoling, SHI Keren, GAN Qianqian, WANG Mingke. Loading of Myoglobin into Layer-by-layer Films Assembled Through Boronic Acid-diol Specific Recognition and Its Electrochemical Study†[J]. Chem. J. Chinese Universities, 2016, 37(5): 938.
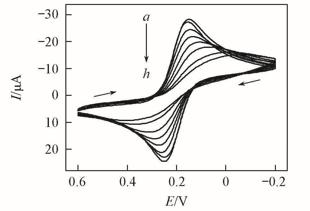
Fig.1 CVs of 1.0 mmol/L Fe(CN)63- at a scan rate of 0.1 V/s in pH=7.0 buffers at bare PG electrode(a), PG/CS film electrode(b) and PG/CS/{PAA-PBA/Dex}n film electrodes with n=1—7(c—h)
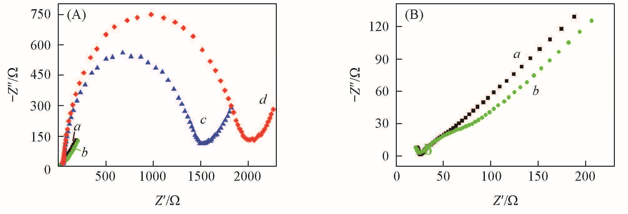
Fig.2 EIS responses of bare PG(a), PG/CS film(b), PG/CS {PAA-PBA/Dex}6 film(c) and PG/CS{PAA-PBA/Dex}6-Mb film(d) at 0.17 V in pH=7.0 buffers containing 5.0 mmol/L Fe(CN)3-/4-(A) and magnification of curves a and b of (A) (B)
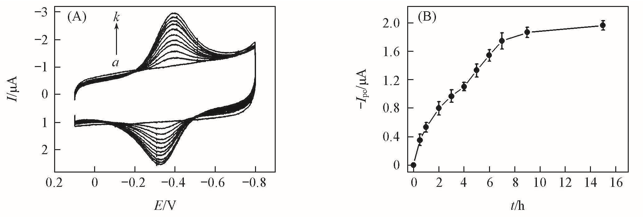
Fig.3 CVs in pH=7.0 buffers at a scan rate of 0.2 V/s for {PAA-PBA/Dex}6-Mb film after {PAA-PBA/Dex}6 film was immersed in 1 mg/mL Mb solutions at pH=5.0 for different time(A) and effect of the immersing time in Mb solution on reduction peak currents of electroactive Mb(Ipc) for {PAA-PBA/Dex}6-Mb film(B)(A) t/h: a. 0; b. 0.5; c. 1.0; d. 2.0; e. 3.0; f. 4.0; g. 5.0; h. 6.0; i. 7.0; j. 9.0; k. 15.0.
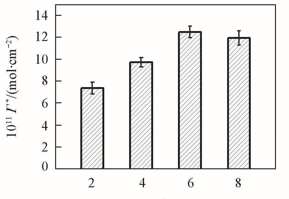
Fig.4 Effect of different number of bilayers(n) on the surface concentration of electroactive Mb(Γ*) after {PAA-PBA/Dex}n films were immersed in 1 mg/mL Mb solutions for 9 h
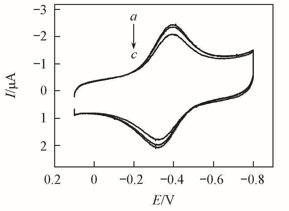
Fig.5 CVs at a scan rate of 0.2 V/s for {PAA-PBA/Dex}6-Mb films in pH=7.0 buffersa. The first cycle; b. after 200 cycles; c. after the films were immersed in pH=7.0 buffers for 7 d.
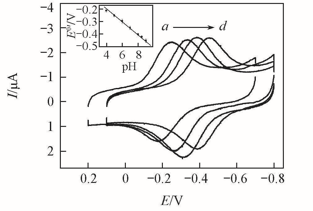
Fig.7 CVs of {PAA-PBA/Dex}6-Mb films at a scan rate of 0.2 V/s in buffers at different pHpH: a. 4.0; b. 6.0; c. 7.0; d. 8.5. Inset: influence of pH of testing solutions on the formal potential(E°') estimated by CV at a scan rate of 0.2 V/s for {PAA-PBA/Dex}6-Mb film.
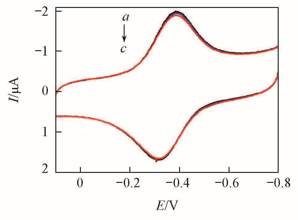
Fig.8 CVs at before and after different volume of air was injected 0.2 V/s for {PAA-PBA/Dex}6-Mb films in pH=7.0 buffers containing 0.1 mol/L different supporting electrolytesa. NaCl; b. KCl; c. NaBr.
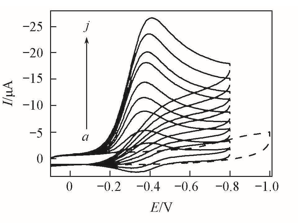
Fig.9 CVs at 0.2 V/s in 10 mL of pH=7.0 buffers for {PAA-PBA/Dex}6(a), {PAA-PBA/Dex}6-Mb films before(b) and {PAA-PBA}6-Mb films(c—j) after different volume of air was injectedV(air)/nL: b. 60; c. 50; d. 10; e. 15; f. 20; g. 30; h. 40; i. 60; j. 90.
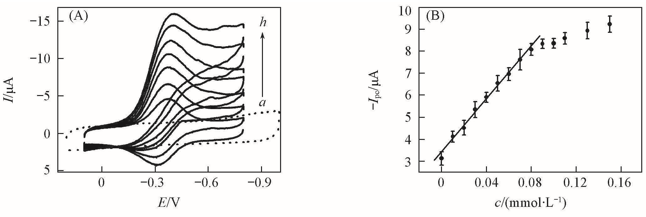
Fig.10 CVs at a scan rate of 0.2 V/s for {PAA-PBA/Dex}6 films in buffers(pH=7) containing 100 μmol/L H2O2(a), {PAA-PBA/Dex}6-Mb films(b), {PAA-PBA/Dex}6-Mb films with 20(c), 40(d), 60(e), 80(f), 100(g) and 150 μmol/L(h) H2O2 in pH=7.0 buffers, respectively(A) and dependence of CV Ipc on the concentration of H2O2 for {PAA-PBA/Dex}6-Mb film at 0.2 V/s(B)
| [1] | Chaplin M.F., Bucke C., Enzyme Technology, Cambridge University Press, Cambridge, 1990 |
| [2] | Rusling J. F., Acc. Chem.Res., 1998, 31, 363—369 |
| [3] | Decher G., Schneloff J.B., Multilayer Thin Films: Sequential Assembly of Nano-composite Materials, Wiley-VCH, Weinheim, Germany, 2012, 23—36 |
| [4] | Duan Y., An Q., Zhang Q., Zhang Y., Chem. Res. ChineseUniversities, 2015, 31(4), 674—679 |
| [5] | Jiang M., Eisenberg A., Liu G.J., Zhang X., Macromolecular Self-Assembly, Scientific Press, Beijing, 2008, 150—175 |
| [6] | Sun J., Zhu Z. Y., Lai J. P., Luo J., Liu X. Y., Chem. J. Chinese Universities,2015, 36(3), 581—588 |
| (孙军, 朱正意, 赖健平, 罗静, 刘晓亚. 高等学校化学学报, 2015, 36(3), 581—588) | |
| [7] | Eaawa Y., Miki R., Seki T., Materials, 2014, 7, 1201—1220 |
| [8] | Iwami Y., Yamamoto H., Kanekiyo Y., Chem.Lett., 2013, 42(10), 1214—1216 |
| [9] | Liu L., Xia N., Xing Y., Deng D., Int. J. Electrochem.Sci., 2013, 8, 11161—11174 |
| [10] | Lacina K., Skladal P., James T., Chem. CentralJ., 2014, 8, 60—77 |
| [11] | Levy T., Dejugnat C., Sukhorukov G. B., Adv. Funct.Mater., 2008, 18, 1586—1594 |
| [12] | Song S. Y., Yoon H. C., Sens. ActuatorsB, 2009, 140, 233—239 |
| [13] | James T.D., Phillips M. D., Shinkai S., Boronic Acids in Saccharide Recognition, RSC Publishing, Cambridge, 2006, 13—33 |
| [14] | Li S., Davis E. N., Anderson J., Lin Q., Wang Q., Biomacromolecules,2009, 10, 113—118 |
| [15] | Zhang J., Geddes C. D., Lakowicz J. R., Anal.Biochem., 2004, 332, 253—260 |
| [16] | Ariga K., Ji Q., Hill J. P., Adv. Polym.Sci., 2010, 229, 51—87 |
| [17] | Song W., Song X., Yang C., Gao S., Klausen L. H., Zhang Y., Dong M., Kjems J., Int. J.Nanomed., 2015, 10, 2335—2346 |
| [18] | Hammond P. T., Materials, 2012, 15(5), 196—206 |
| [19] | Yao H., Guo H., Hu N., Electrochim. Acta,2009, 54, 7330—7338 |
| [20] | Guo X., Zhang H., Hu N., Nanotechnology, 2008, 19, 055709—055719 |
| [21] | Levy T., Dejugnat C., Sukhorukov G. B., Adv. Funct.Mater., 2008, 18, 1586—1594 |
| [22] | Hill H. A. O., Pure Appl.Chem., 1987, 59, 743—748 |
| [23] | Nilsen-Nygaard J., Strand S. P., Varum K. M., Draget K. I., Nordgard C. T., Polymer,2015, 7, 552—579 |
| [24] | Mittal K.L., Acid-Base Interactions: Relevance to Adhesion Science and Technology, VSP, Utrecht, Vol. 2, 2000, 18—25 |
| [25] | Laguecir A., Ulrich S., Labille J., Fatin-Rouge F., Stoll S., Buffle J., Eur. Polym. J., 2006, 42, 1135—1144 |
| [26] | Delaittre G., Nicolas J., Lefay C., Save M., Charleux B.,Chem. Commun., 2005, 614—616 |
| [27] | Gregor H. P., Luttinger L. B., Loebl E. M., J. Am. Chem.Soc., 1954, 76, 5879—5880 |
| [28] | Katz E., Willner I., Electroanalysis, 2003, 15, 913—918 |
| [29] | Sabatani E., Cohen-Boulakia J., Bruening M., Rubinstein I., Langmuir,1993, 9, 2974—2980 |
| [30] | Bellelli A., Antonini G., Brunori M., Springer B. A., Sligar S. G., J. Biol.Chem., 1990, 265, 18898—18901 |
| [31] | Lvov Y., Lu Z., Zu X., Schenkman J., Rusling J., J. Am. Chem.Soc., 1998, 120, 4073—4077 |
| [32] | Murray R.W.; Bard A. J.(Ed.); Electroanalytical Chemistry, Vol. 13, Marcel Dekker,New York, 1984, 191 |
| [33] | Lvov Y.; Lvov Y. Möhwald H.Eds.; Protein Architecture: Interfacing Molecular Assemblies and Immobilization Biotechnology, Marcel Dekker,New York, 2000, 125 |
| [34] | Bond A.M., Modern Polarographic Methods in Analytical Chemistry, Marcel Dekker,New York, 2011, 15—34 |
| [35] | Liu Y., Liu H., Guo X., Hu N., Electroanalysis, 2010, 22, 2261—2268 |
| [1] | LIN Gaoxin, WANG Jiacheng. Progress and Perspective on Molybdenum Disulfide with Single-atom Doping Toward Hydrogen Evolution [J]. Chem. J. Chinese Universities, 2022, 43(9): 20220321. |
| [2] | WANG Sicong, PANG Beibei, LIU Xiaokang, DING Tao, YAO Tao. Application of XAFS Technique in Single-atom Electrocatalysis [J]. Chem. J. Chinese Universities, 2022, 43(9): 20220487. |
| [3] | QIN Yongji, LUO Jun. Applications of Single-atom Catalysts in CO2 Conversion [J]. Chem. J. Chinese Universities, 2022, 43(9): 20220300. |
| [4] | YAO Qing, YU Zhiyong, HUANG Xiaoqing. Progress in Synthesis and Energy-related Electrocatalysis of Single-atom Catalysts [J]. Chem. J. Chinese Universities, 2022, 43(9): 20220323. |
| [5] | HAN Fuchao, LI Fujin, CHEN Liang, HE Leiyi, JIANG Yunan, XU Shoudong, ZHANG Ding, QI Lu. Enhance of CoSe2/C Composites Modified Separator on Electrochemical Performance of Li-S Batteries at High Sulfur Loading [J]. Chem. J. Chinese Universities, 2022, 43(8): 20220163. |
| [6] | WANG Ruhan, JIA Shunhan, WU Limin, SUN Xiaofu, HAN Buxing. CO2-involved Electrochemical C—N Coupling into Value-added Chemicals [J]. Chem. J. Chinese Universities, 2022, 43(7): 20220395. |
| [7] | WANG Lijun, LI Xin, HONG Song, ZHAN Xinyu, WANG Di, HAO Leiduan, SUN Zhenyu. Efficient Electrocatalytic CO2 Reduction to CO by Tuning CdO-Carbon Black Interface [J]. Chem. J. Chinese Universities, 2022, 43(7): 20220317. |
| [8] | YANG Lijun, YU Yang, ZHANG Lei. Construction of Dual-functional 2D/3D Hydrid Co2P-CeO x Heterostructure Integrated Electrode for Electrocatalytic Urea Oxidation Assisted Hydrogen Production [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220082. |
| [9] | XIA Tian, WAN Jiawei, YU Ranbo. Progress of the Structure-property Correlation of Heteroatomic Coordination Structured Carbon-based Single-atom Electrocatalysts [J]. Chem. J. Chinese Universities, 2022, 43(5): 20220162. |
| [10] | ZHANG Hongwei, CHEN Wen, ZHAO Meiqi, MA Chao, HAN Yunhu. Research Progress of Single Atom Catalysts in Electrochemistry [J]. Chem. J. Chinese Universities, 2022, 43(5): 20220129. |
| [11] | WU Jun, HE Guanchao, FEI Huilong. Self-supported Film Electrodes Decorated with Single Atoms for Energy Electrocatalysis [J]. Chem. J. Chinese Universities, 2022, 43(5): 20220051. |
| [12] | CHEN Changli, MI Wanliang, LI Yujing. Research Progress of Single Atom Catalysts in Electrochemical Hydrogen Cycling [J]. Chem. J. Chinese Universities, 2022, 43(5): 20220065. |
| [13] | CHEN Zhaoyang, XUE Yurui, LI Yuliang. Synthesis and Applications of Graphdiyne Based Zerovalent Atomic Catalysts [J]. Chem. J. Chinese Universities, 2022, 43(5): 20220063. |
| [14] | WANG Zumin, MENG Cheng, YU Ranbo. Doping Regulation in Transition Metal Phosphides for Hydrogen Evolution Catalysts [J]. Chem. J. Chinese Universities, 2022, 43(11): 20220544. |
| [15] | DING Qin, ZHANG Zixuan, XU Peicheng, LI Xiaoyu, DUAN Limei, WANG Yin, LIU Jinghai. Effects of Cu, Ni and Co Hetroatoms on Constructions and Electrocatalytic Properties of Fe-based Carbon Nanotubes [J]. Chem. J. Chinese Universities, 2022, 43(11): 20220421. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||