

Chem. J. Chinese Universities ›› 2016, Vol. 37 ›› Issue (3): 454.doi: 10.7503/cjcu20150703
• Analytical Chemistry • Previous Articles Next Articles
LI Lanjie1, LI Xuwen1, DING Jian2, LIU Ying1, WU Qian1, WANG Xiaozhong1, LI Min1, JIN Yongri1,*( )
)
Received:2015-09-09
Online:2016-03-10
Published:2016-01-24
Contact:
JIN Yongri
E-mail:jinyr@jlu.edu.cn
CLC Number:
TrendMD:
LI Lanjie, LI Xuwen, DING Jian, LIU Ying, WU Qian, WANG Xiaozhong, LI Min, JIN Yongri. Ionic Liquid Surfactant-mediated Ultrasonic-assisted Extraction Coupled with HPLC for the Determination of Five Rare Ginsenosides in Panax notoginseng (Burk.) F. H. Chen†[J]. Chem. J. Chinese Universities, 2016, 37(3): 454.
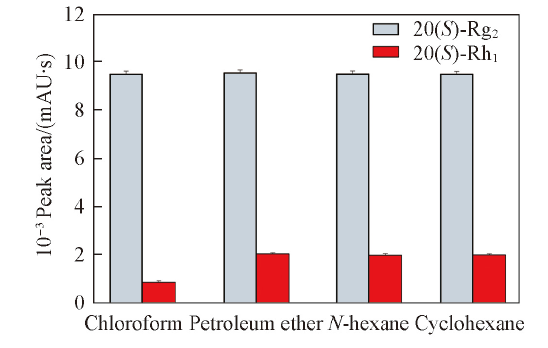
Fig.3 Effects of different degreasants on the extraction efficiencyConditions: 1.00 mL ionic liquid, 5.0 mL water, 7.00 g K2HPO4, 0.60 g Sanqi, ultrasonic time was 20 min. Error bars represent SDs.
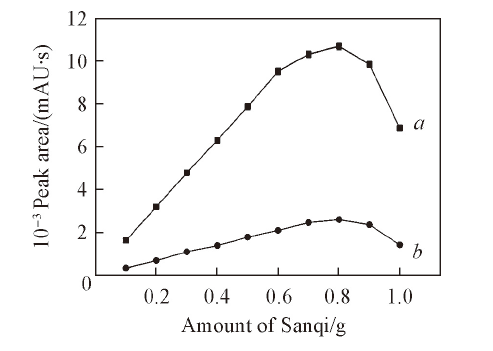
Fig.5 Effects of different amounts of Sanqi on the extraction efficiencies of 20(S)-Rg2(a) and 20(S)-Rh1(b)The degreasant was petroleum ether. Conditions: 1.00 mL ionic liquid, 5.0 mL water, 7.00 g K2HPO4, ultrasonic time was 20 min. Each data point represents two analytes.
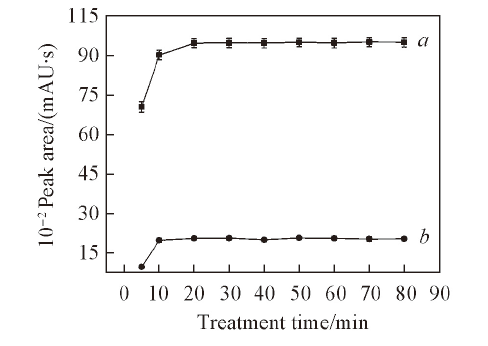
Fig.6 Effects of the ultrasonic treatment time on the extraction efficiencies of 20(S)-Rg2(a) and 20(S)-Rh1(b)The degreasant was petroleum ether. Conditions: 1.00 mL ionic liquid. 5.0 mL water, 7.00 g K2HPO4, 0.60 g Sanqi. Each data point represents two analytes. Error bars represent SDs.
| Analyte | Linear regression | Linear range/ (μg·mL-1) | r2 | Intra-day precision(%) | Inter-day precision(%) | LOD/ (μg·mL-1) | LOQ/ (μg·mL-1) |
|---|---|---|---|---|---|---|---|
| 20(S)-Rg2 | y=9.30x+9.55 | 4.05—1012 | 0.9998 | 0.98 | 1.03 | 0.32 | 1.28 |
| 20(S)-Rh1 | y=11.17x+8.52 | 5.28—528.0 | 0.9996 | 1.13 | 1.07 | 0.21 | 0.80 |
| Rk3 | y=25.30x+10.78 | 5.09—508.8 | 0.9998 | 1.65 | 1.63 | 0.18 | 7.02 |
| 20(S)-Rg3 | y=9.30x+9.82 | 5.04—504.0 | 0.9998 | 1.23 | 1.33 | 0.48 | 1.98 |
| Rk1 | y=10.80x+3.61 | 3.54—353.6 | 0.9999 | 0.89 | 1.03 | 0.29 | 0.97 |
Table 1 Regression equations, LODs, LOQs, intra-day and inter-day precision
| Analyte | Linear regression | Linear range/ (μg·mL-1) | r2 | Intra-day precision(%) | Inter-day precision(%) | LOD/ (μg·mL-1) | LOQ/ (μg·mL-1) |
|---|---|---|---|---|---|---|---|
| 20(S)-Rg2 | y=9.30x+9.55 | 4.05—1012 | 0.9998 | 0.98 | 1.03 | 0.32 | 1.28 |
| 20(S)-Rh1 | y=11.17x+8.52 | 5.28—528.0 | 0.9996 | 1.13 | 1.07 | 0.21 | 0.80 |
| Rk3 | y=25.30x+10.78 | 5.09—508.8 | 0.9998 | 1.65 | 1.63 | 0.18 | 7.02 |
| 20(S)-Rg3 | y=9.30x+9.82 | 5.04—504.0 | 0.9998 | 1.23 | 1.33 | 0.48 | 1.98 |
| Rk1 | y=10.80x+3.61 | 3.54—353.6 | 0.9999 | 0.89 | 1.03 | 0.29 | 0.97 |
| Analyte | Initial content/(μg·mL-1) | Added/(μg·mL-1) | Found/(μg·mL-1) | Recovery(%) | RSD(%, N=6) |
|---|---|---|---|---|---|
| 20(S)-Rg2 | 506.20±0.70 | 506.00 | 1006.30 | 99.42 | 3.08 |
| 20(S)-Rh1 | 90.47±0.58 | 99.24 | 180.67 | 95.23 | 3.87 |
| Rk3 | 5.51±0.09 | 7.95 | 14.88 | 110.55 | 5.43 |
| 20(S)-Rg3 | 95.93±0.32 | 63.00 | 160.36 | 100.90 | 2.38 |
| Rk1 | 10.65±0.01 | 11.05 | 19.98 | 92.07 | 4.43 |
Table 2 Recoveries of the analytes(N=6)
| Analyte | Initial content/(μg·mL-1) | Added/(μg·mL-1) | Found/(μg·mL-1) | Recovery(%) | RSD(%, N=6) |
|---|---|---|---|---|---|
| 20(S)-Rg2 | 506.20±0.70 | 506.00 | 1006.30 | 99.42 | 3.08 |
| 20(S)-Rh1 | 90.47±0.58 | 99.24 | 180.67 | 95.23 | 3.87 |
| Rk3 | 5.51±0.09 | 7.95 | 14.88 | 110.55 | 5.43 |
| 20(S)-Rg3 | 95.93±0.32 | 63.00 | 160.36 | 100.90 | 2.38 |
| Rk1 | 10.65±0.01 | 11.05 | 19.98 | 92.07 | 4.43 |
| Sample | Content(%) | ||||
|---|---|---|---|---|---|
| 20(S)-Rg2 | 20(S)-Rh1 | Rk3 | 20(S)-Rg3 | Rk1 | |
| GJ-1 | 0.1181±0.0033 | 0.02111±0.00045 | 0.001287±0.000035 | 0.02238±0.00024 | 0.002485±0.000025 |
| GJ-2 | 0.1178±0.0034 | 0.02056±0.00053 | 0.001406±0.000039 | 0.02292±0.00022 | 0.002540±0.000032 |
| JT-1 | 0.1120±0.0043 | 0.01768±0.00039 | 0.001271±0.000042 | 0.02238±0.00024 | 0.002730±0.000031 |
Table 3 Contents(%) of the rare ginsenosides in Sanqi(N=6)
| Sample | Content(%) | ||||
|---|---|---|---|---|---|
| 20(S)-Rg2 | 20(S)-Rh1 | Rk3 | 20(S)-Rg3 | Rk1 | |
| GJ-1 | 0.1181±0.0033 | 0.02111±0.00045 | 0.001287±0.000035 | 0.02238±0.00024 | 0.002485±0.000025 |
| GJ-2 | 0.1178±0.0034 | 0.02056±0.00053 | 0.001406±0.000039 | 0.02292±0.00022 | 0.002540±0.000032 |
| JT-1 | 0.1120±0.0043 | 0.01768±0.00039 | 0.001271±0.000042 | 0.02238±0.00024 | 0.002730±0.000031 |
| [1] | National Pharmacopoeia Committee.,Pharmacopoeia of the People’s Republic of China, Part 2, 2010, 11—12 |
| (国家药典委员会. 中华人民共和国药典, 二部, 中国医药科技出版社, 2010, 11—12) | |
| [2] | Bao J. C., Liu G., Cong D. L., Zhang C. X., Chin. Trad. Pat. Med., 2006, 28(2), 246—253 |
| (鲍建才, 刘刚, 丛登立, 张崇禧. 中成药,2006, 28(2), 246—253) | |
| [3] | Liu H. Z., Pang J., Wang Z. L., Acta Pharm. Sin., 1982, 17(11), 801 |
| (刘贺之, 庞健, 王增龄. 药学学报, 1982, 17(11), 801) | |
| [4] | Shi L., Fan P. S., Wu L., Acta Pharm. Sin., 1990, 11(1), 29 |
| (石琳, 范盘生, 吴玲. 中国药理学报, 1990, 11(1), 29) | |
| [5] | Zhang H. J., Cheng Y. Y., J. Pharmaceut. Biomed., 2006, 40(2), 429—432 |
| [6] | Guan J., Lai C. M., Li S. P., J. Pharmaceut. Biomed., 2007, 44(4), 996—1000 |
| [7] | Kang K. S., Kim H. Y., Yamabe N., Yokozawa T., Bioorg. Med. Chem. Lett., 2006, 16(19), 5028—5031 |
| [8] | Wang Y., Jiang Y., Wang B. X., Qiu Q. Y., Acta Pharm. Sin., 2002, 37(12), 927—929 |
| (王毅, 蒋艳, 王本祥, 邱全瑛. 药学学报, 2002, 37(12), 927—929) | |
| [9] | Kang S. K., Yokozawa T., Yamabe N., Kim Y. H., Park Y. H., Biol. Pharm. Bull., 2007, 30(5), 917—921 |
| [10] | HwangI. G., Kim H. Y., Joung E. M., Woo K. S., Jeong J. H., Yu K. W., Lee J., Jeong H. S., Food. Sci. Biotechnol., 2010, 19(4), 941—949 |
| [11] | Tian J. W., Fu F. H., Geng M. Y., Neurosci. Lett., 2005, 374(2), 92—97 |
| [12] | Kim H. S., Lee E. H., Ko S. R., Arch. Pharm. Res., 2004, 27(4), 429—435 |
| [13] | Kim Y. J., Kwon H. C., Ko H., Biol. Pharm. Bull., 2008, 31(5), 826—830 |
| [14] | Wang Y., Song H., Jia C.M.,The 2012 Congress of Chinese Medicine, 2012 |
| (汪雁, 宋航, 贾春梅. 2012年中国药学大会暨第十二届中国药师周论文集, 2012) | |
| [15] | Feng X. D., Liang F. H., Su R., Wu L. J., Li X. Y., Wang X. H., Zhang H. Q., Yu A. M., Chem. Res. Chinese Universities, 2013, 29(4), 647—652 |
| [16] | Wu K. K., Zhang Q. L., Liu Q., Tang F., Long Y. M., J. Sep. Sci., 2009, 32(24), 4220—4226 |
| [17] | Cao X. J., Ye X. M., Lu Y. B., Yu Y., Mo W. M., Anal. Chim. Acta, 2009, 640(1/2), 47—51 |
| [18] | Yao H., Jin Y. R., Yang J., Li L. J., Sun T., Shi X. L., Li X. W., Chem. J. Chinese Universities, 2014, 35(11), 2317—2323 |
| (姚华, 金永日, 杨洁, 李兰杰, 孙婷, 时晓磊, 李绪文. 高等学校化学学报, 2014, 35(11), 2317—2323) |
| [1] | CUI Wei, ZHAO Deyin, BAI Wenxuan, ZHANG Xiaodong, YU Jiang. CO2 Absorption in Composite of Aprotic Solvent and Iron-based Ionic Liquid [J]. Chem. J. Chinese Universities, 2022, 43(8): 20220120. |
| [2] | PENG Kuilin, LI Guilin, JIANG Chongyang, ZENG Shaojuan, ZHANG Xiangping. Research Progress for the Role of Electrolytes in the CO2 Electrochemical Reduction [J]. Chem. J. Chinese Universities, 2022, 43(7): 20220238. |
| [3] | JI Shuangqi, JIN Zhao, GUAN Wenna, PAN Xiangyu, GUAN Tong. Preparation and Chromatographic Performance of Mixed-mode Silica Stationary Phase Modified by Double Cationic Ionic Liquid and Octadecyl Group [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220008. |
| [4] | CHANG Sihui, CHEN Tao, ZHAO Liming, QIU Yongjun. Thermal Degradation Mechanism of Bio-based Polybutylactam Plasticized by Ionic Liquids [J]. Chem. J. Chinese Universities, 2022, 43(11): 20220353. |
| [5] | WAN Ren, SONG Fan, PENG Changjun, LIU Honglai. Group Contribution Method for Infinite Dilution Molar Conductivity of Unconventional Ions in Water [J]. Chem. J. Chinese Universities, 2021, 42(12): 3672. |
| [6] | WANG Man, WANG Xin, ZHOU Jing, GAO Guohua. Efficient Synthesis of Dimethyl Carbonate via Transesterification of Methanol and Ethylene Carbonate Catalyzed by Poly(ionic liquid)s [J]. Chem. J. Chinese Universities, 2021, 42(12): 3701. |
| [7] | ZHOU Molin, JIANG Xin, YI Ting, YANG Xiangguang, ZHANG Yibo. Improvement of Interface Stability Between Sulfide Solid Electrolyte Li10GeP2S12 and Lithium Metal [J]. Chem. J. Chinese Universities, 2020, 41(8): 1810. |
| [8] | GAO Chong,YU Fengli,XIE Congxia,YU Shitao. Baeyer-Villiger Oxidation of Cyclic Ketones Catalyzed by Amino Alcohol Heteropoly Acid Ionic Liquid † [J]. Chem. J. Chinese Universities, 2020, 41(5): 1101. |
| [9] | GAO Naiwei, MA Qiang, HE Yonglin, WANG Yapei. Green Electronic Devices Based on Ionic Liquids † [J]. Chem. J. Chinese Universities, 2020, 41(5): 901. |
| [10] | CHENG Shifu,HU Hao,CHEN Bihua,WU Haihong,GAO Guohua,HE Mingyuan. Preparation and Electrochemical Performance of Porous Carbons Prepared from Binary Ionic Liquids † [J]. Chem. J. Chinese Universities, 2020, 41(5): 1048. |
| [11] | PIAO Huilan,MA Pinyi,QIN Zucheng,JIANG Yanxiao,SUN Ying,WANG Xinghua,SONG Daqian. Determination of Triazine Herbicides from Fruit Juice Samples Using Effervescence Assisted Microextraction Method Based on Acidic Ionic Liquid Packed Syringe [J]. Chem. J. Chinese Universities, 2020, 41(2): 228. |
| [12] | ZHANG Li,QIAN Mingchao,LIU Xueke,Gao Shuaitao,YU Jiang,XIE Haishen,WANG Hongbin,SUN Fengjiang,SU Xianghong. Dynamic Study of Oxidative Desulfurization by Iron-based Ionic Liquids/NHD † [J]. Chem. J. Chinese Universities, 2020, 41(2): 317. |
| [13] | WANG Nan,YAO Kaisheng,ZHAO Chenchen,LI Tianjin,LU Weiwei. Ionic Liquid-assisted Synthesis of AuPd Nanosponges and Their Catalytic Performance † [J]. Chem. J. Chinese Universities, 2020, 41(1): 62. |
| [14] | LIU Xiaozhou, GUAN Xinyu, FANG Qianrong, JIN Yongri. Three-dimensional Covalent Organic Frameworks Synthesized by Room Temperature Ionic Liquid Method† [J]. Chem. J. Chinese Universities, 2019, 40(7): 1341. |
| [15] | LI Chenguang, HUA Er, LIU Tianxia. Tribological Behaviour of Protic Ionic Liquid Composed of 2-Ethylhexylethylenediaminium Cation and Trifluoromethanesulfonate Anion as Liquid Paraffin Additive† [J]. Chem. J. Chinese Universities, 2019, 40(7): 1411. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||