

Chem. J. Chinese Universities ›› 2015, Vol. 36 ›› Issue (5): 899.doi: 10.7503/cjcu20141138
• Organic Chemistry • Previous Articles Next Articles
ZHENG Minyan*( ), WEI Yongsheng, GENG Wei
), WEI Yongsheng, GENG Wei
Received:2014-12-29
Online:2015-05-10
Published:2015-04-13
Contact:
ZHENG Minyan
E-mail:zmy71@126.com
Supported by:CLC Number:
TrendMD:
ZHENG Minyan, WEI Yongsheng, GENG Wei. Synthesis, Photosensitive and Electrochemical Properties of Asymmetric Liquid Crystals Based on Tri-ring Azo-benzoic Acid†[J]. Chem. J. Chinese Universities, 2015, 36(5): 899.
| Compd. | Appea- rance | Yield (%) | m.p./℃ | IR(KBr), | MS([M+], calcd.), m/z | Elemental analysis(%, calcd.) | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| N | C | H | ||||||||
| 3a | Orange needle | 82.4 | 211.2—212.6 | 3200—2500(m, O—H), 3069, 2994, 2922, 2882(s, C—H), 1734, 1684(vs, C═O), 1602(m, N═N), 1594, 1559, 1541, 1449(m, ArH), 1428, 1276, 1222, 1201, 1178, 1074, 1010(vs, C—O—C), 879(s, trans-R—N═N—R), 807(w, 1,4-Ar), 688 [w,(CH2)n] | 366 (366.41) | 68.94 (68.84) | 6.30 (6.05) | 7.83 (7.65) | ||
| Compd. | Appea- rance | Yield (%) | m.p./℃ | IR(KBr), ν/cm-1 | MS([M+], calcd.), m/z | Elemental analysis(%, calcd.) | ||||
| N | C | H | ||||||||
| 3b | Orange needle | 80.2 | 221.7—222.8 | 3200—2500(m, O—H), 2950, 2930, 2864(s, C—H), 1755, 1697(vs, C═O), 1603(m, N═N), 1563, 1499(m, ArH), 1427, 1290, 1223, 1200, 1141, 1121, 1009(vs, C—O—C), 868(s, trans-R—N═N—R), 809(w, 1, 4-Ar), 690[w,(CH2)n] | 380 (380.44) | 69.67 (69.46) | 6.54 (6.36) | 7.49 (7.36) | ||
| 3c | Orange needle | 84.0 | 208.9—209.5 | 3000—2500(m, O—H), 2960, 2931(s, C—H), 1755, 1680(vs, C═O), 1603(m, N═N), 1592, 1500, 1449(m, ArH), 1428, 1290, 1224, 1201, 1142, 1122, 1101, 1076, 1010(vs, C—O—C), 869(s, trans-R—N═N—R), 808(w, 1, 4-Ar), 765 [w,(CH2)n] | 394 (394.46) | 70.22 (70.03) | 6.73 (6.64) | 7.15 (7.10) | ||
| 3d | Orange needle | 81.1 | 198.0—199.7 | 3000—2500(m, O—H), 3069, 2994, 2922, 2882(s, C—H), 1734, 1684(vs, C═O), 1602(m, N═N), 1594, 1559(m, ArH), 1498, 1428, 1276, 1222, 1201, 1178, 1141, 1074, 1010(vs, C—O—C), 879(s, trans-R—N═N—R), 812(w, 1, 4-Ar), 776 [w,(CH2)n] | 360 (360.11) | 70.14 (69.99) | 4.60 (4.48) | 7.93 (7.77) | ||
| 3e | Orange needle | 80.7 | 200.4—201.9 | 3000—2500(m, O—H), 3097, 3071, 2968, 2853(s, C—H), 1735, 1685(vs, C═O), 1602(m, N═N), 1559, 1541, 1496, 1458(m, ArH), 1266, 1223, 1195, 1177, 1142, 1068, 1009(vs, C—O—C), 882(s, trans-R—N═N—R), 811(w, 1,4-Ar), 766 [w,(CH2)n] | 374 (374.39) | 70.77 (70.58) | 4.90 (4.85) | 7.66 (7.48) | ||
| 3f | Orange needle | 85.2 | 199.5—200.9 | 3100—2500(m, O—H), 2958, 2929, 2871(s, C—H), 1734, 1684(vs, C═O), 1604(m, N═N), 1495, 1464(m, ArH), 1309, 1267, 1223, 1196, 1141, 1099, 1061, 1010(vs, C—O—C), 882(s, trans-R—N═N—R), 831(w, 1, 4-Ar), 776 [w,(CH2)n] | 388 (388.42) | 71.33 (71.12) | 5.30 (5.19) | 7.39 (7.21) | ||
| 3g | Orange needle | 88.6 | 186.3—187.8 | 3000—2500(m, O—H), 2956, 2927, 2857(s, C—H), 1734, 1681(vs, C═O), 1603(m, N═N), 1595, 1495(m, ArH), 1416, 1379, 1270, 1223, 1198, 1176, 1141, 1100, 1068, 1010(vs, C—O—C), 946, 880(s, trans-R—N═N—R), 846(w, 1, 4-Ar), 775 [w,(CH2)n] | 402 (402.44) | 71.78 (71.63) | 5.66 (5.51) | 6.75 (6.96) | ||
| 3h | Orange needle | 84.3 | 187.1—188.5 | 3000—2500(m, O—H), 2929, 2857(s, C—H), 1786, 1685(vs, C═O), 1602(m, N═N), 1541, 1468(m, ArH), 1417, 1358, 1287, 1255, 1220, 1142, 1101, 1041, 1008(vs, C—O—C), 943, 882(s, trans-R—N═N—R), 817(w, 1, 4-Ar), 721 [w,(CH2)n] | 416 (416.47) | 72.33 (72.10) | 5.99 (5.81) | 6.88 (6.73) | ||
Table 1 Appearance, yields, melting points, IR, MS data and elemental analysis of target compounds
| Compd. | Appea- rance | Yield (%) | m.p./℃ | IR(KBr), | MS([M+], calcd.), m/z | Elemental analysis(%, calcd.) | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| N | C | H | ||||||||
| 3a | Orange needle | 82.4 | 211.2—212.6 | 3200—2500(m, O—H), 3069, 2994, 2922, 2882(s, C—H), 1734, 1684(vs, C═O), 1602(m, N═N), 1594, 1559, 1541, 1449(m, ArH), 1428, 1276, 1222, 1201, 1178, 1074, 1010(vs, C—O—C), 879(s, trans-R—N═N—R), 807(w, 1,4-Ar), 688 [w,(CH2)n] | 366 (366.41) | 68.94 (68.84) | 6.30 (6.05) | 7.83 (7.65) | ||
| Compd. | Appea- rance | Yield (%) | m.p./℃ | IR(KBr), ν/cm-1 | MS([M+], calcd.), m/z | Elemental analysis(%, calcd.) | ||||
| N | C | H | ||||||||
| 3b | Orange needle | 80.2 | 221.7—222.8 | 3200—2500(m, O—H), 2950, 2930, 2864(s, C—H), 1755, 1697(vs, C═O), 1603(m, N═N), 1563, 1499(m, ArH), 1427, 1290, 1223, 1200, 1141, 1121, 1009(vs, C—O—C), 868(s, trans-R—N═N—R), 809(w, 1, 4-Ar), 690[w,(CH2)n] | 380 (380.44) | 69.67 (69.46) | 6.54 (6.36) | 7.49 (7.36) | ||
| 3c | Orange needle | 84.0 | 208.9—209.5 | 3000—2500(m, O—H), 2960, 2931(s, C—H), 1755, 1680(vs, C═O), 1603(m, N═N), 1592, 1500, 1449(m, ArH), 1428, 1290, 1224, 1201, 1142, 1122, 1101, 1076, 1010(vs, C—O—C), 869(s, trans-R—N═N—R), 808(w, 1, 4-Ar), 765 [w,(CH2)n] | 394 (394.46) | 70.22 (70.03) | 6.73 (6.64) | 7.15 (7.10) | ||
| 3d | Orange needle | 81.1 | 198.0—199.7 | 3000—2500(m, O—H), 3069, 2994, 2922, 2882(s, C—H), 1734, 1684(vs, C═O), 1602(m, N═N), 1594, 1559(m, ArH), 1498, 1428, 1276, 1222, 1201, 1178, 1141, 1074, 1010(vs, C—O—C), 879(s, trans-R—N═N—R), 812(w, 1, 4-Ar), 776 [w,(CH2)n] | 360 (360.11) | 70.14 (69.99) | 4.60 (4.48) | 7.93 (7.77) | ||
| 3e | Orange needle | 80.7 | 200.4—201.9 | 3000—2500(m, O—H), 3097, 3071, 2968, 2853(s, C—H), 1735, 1685(vs, C═O), 1602(m, N═N), 1559, 1541, 1496, 1458(m, ArH), 1266, 1223, 1195, 1177, 1142, 1068, 1009(vs, C—O—C), 882(s, trans-R—N═N—R), 811(w, 1,4-Ar), 766 [w,(CH2)n] | 374 (374.39) | 70.77 (70.58) | 4.90 (4.85) | 7.66 (7.48) | ||
| 3f | Orange needle | 85.2 | 199.5—200.9 | 3100—2500(m, O—H), 2958, 2929, 2871(s, C—H), 1734, 1684(vs, C═O), 1604(m, N═N), 1495, 1464(m, ArH), 1309, 1267, 1223, 1196, 1141, 1099, 1061, 1010(vs, C—O—C), 882(s, trans-R—N═N—R), 831(w, 1, 4-Ar), 776 [w,(CH2)n] | 388 (388.42) | 71.33 (71.12) | 5.30 (5.19) | 7.39 (7.21) | ||
| 3g | Orange needle | 88.6 | 186.3—187.8 | 3000—2500(m, O—H), 2956, 2927, 2857(s, C—H), 1734, 1681(vs, C═O), 1603(m, N═N), 1595, 1495(m, ArH), 1416, 1379, 1270, 1223, 1198, 1176, 1141, 1100, 1068, 1010(vs, C—O—C), 946, 880(s, trans-R—N═N—R), 846(w, 1, 4-Ar), 775 [w,(CH2)n] | 402 (402.44) | 71.78 (71.63) | 5.66 (5.51) | 6.75 (6.96) | ||
| 3h | Orange needle | 84.3 | 187.1—188.5 | 3000—2500(m, O—H), 2929, 2857(s, C—H), 1786, 1685(vs, C═O), 1602(m, N═N), 1541, 1468(m, ArH), 1417, 1358, 1287, 1255, 1220, 1142, 1101, 1041, 1008(vs, C—O—C), 943, 882(s, trans-R—N═N—R), 817(w, 1, 4-Ar), 721 [w,(CH2)n] | 416 (416.47) | 72.33 (72.10) | 5.99 (5.81) | 6.88 (6.73) | ||
| Compd. | 1H NMR(400 MHz, CDCl3), δ |
|---|---|
| 3a | 0.90(s, 3H, CH3), 1.40—2.97(m, 10H, 10cyclohexyl-H), 7.53, 7.97, 8.01, 8.23(d, J=8.0, 8.4, 8.8, 8.0 Hz, 2H of each, 8Ph-H), 12.96(s, 1H, COOH) |
| 3b | 0.89(t, 3H, J=7.6 Hz, CH3), 1.25—1.26(m, 2H, CH2), 1.25—2.18(m, 10H, 10cyclohexyl-H), 7.27, 7.64, 7.80, 8.25(d, J=8.0, 9.2, 8.8, 8.0 Hz, 2H of each, 8Ph-H), 12.81(s, 1H, COOH) |
| Compd. | 1H NMR(400 MHz, CDCl3), δ |
| 3c | 0.88(t, 3H, J=7.2 Hz, CH3), 1.20—1.27(m, 2H, CH2), 1.24(m, 2H, CH2), 1.57—2.60(m, 10H, 10cyclohexyl-H), 7.30, 7.48, 8.01, 8.31(d, J=8.0, 8.4, 8.4, 8.0 Hz, 2H of each, 8Ph-H), 12.73(s, 1H, COOH) |
| 3d | 2.41(s, 3H, CH3), 7.39, 7.41, 8.01, 8.04, 8.32, 8.41(d, J=7.2, 7.2, 8.0, 8.0, 7.2, 7.2 Hz, 2H of each, 12Ph-H), 13.11(s, 1H, COOH) |
| 3e | 1.27(t, J=7.2 Hz, 3H, CH3), 2.74(q, J=7.2 Hz, 2H, CH2), 7.29, 7.35, 7.41, 8.03, 8.12, 8.25(d, J=8.0, 8.4, 8.4, 8.0, 8.0, 8.0 Hz, 2H of each, 12Ph-H), 12.91(s, 1H, COOH) |
| 3f | 0.97(t, 3H, J=7.2 Hz, CH3), 1.70(m, 2H, CH2), 2.68(t, J=7.2 Hz, 2H, CH2), 7.32, 7.34, 7.98, 8.01, 8.11, 8.35(d, J=8.0, 8.4, 8.0, 8.0, 8.0, 8.4 Hz, 2H of each, 12Ph-H), 12.86(s, 1H, COOH) |
| 3g | 0.90(t, 3H, J=7.2 Hz, CH3), 1.29—1.37(m, 2H, CH2), 1.54—1.65(m, 2H, CH2), 2.66(t, J=7.2 Hz, 2H, CH2), 7.29, 7.36, 7.97, 8.02, 8.09, 8.22(d, J=8.4, 8.8, 8.4, 8.8, 8.0, 8.4 Hz, 2H of each, 12Ph-H), 13.01(s, 1H, COOH) |
| 3h | 0.90(t, 3H, J=6.8 Hz, CH3), 1.25—1.44(m, 6H, CH2CH2CH2), 1.64(t, 2H, J=7.2 Hz, CH2), 7.28, 7.32, 8.03, 8.06, 8.22, 8.28(d, J=8.4, 8.4, 8.0, 8.4, 8.0, 8.4 Hz, 2H of each, 12Ph-H), 13.02(s, 1H, COOH) |
Table 2 1H NMR data for target compounds
| Compd. | 1H NMR(400 MHz, CDCl3), δ |
|---|---|
| 3a | 0.90(s, 3H, CH3), 1.40—2.97(m, 10H, 10cyclohexyl-H), 7.53, 7.97, 8.01, 8.23(d, J=8.0, 8.4, 8.8, 8.0 Hz, 2H of each, 8Ph-H), 12.96(s, 1H, COOH) |
| 3b | 0.89(t, 3H, J=7.6 Hz, CH3), 1.25—1.26(m, 2H, CH2), 1.25—2.18(m, 10H, 10cyclohexyl-H), 7.27, 7.64, 7.80, 8.25(d, J=8.0, 9.2, 8.8, 8.0 Hz, 2H of each, 8Ph-H), 12.81(s, 1H, COOH) |
| Compd. | 1H NMR(400 MHz, CDCl3), δ |
| 3c | 0.88(t, 3H, J=7.2 Hz, CH3), 1.20—1.27(m, 2H, CH2), 1.24(m, 2H, CH2), 1.57—2.60(m, 10H, 10cyclohexyl-H), 7.30, 7.48, 8.01, 8.31(d, J=8.0, 8.4, 8.4, 8.0 Hz, 2H of each, 8Ph-H), 12.73(s, 1H, COOH) |
| 3d | 2.41(s, 3H, CH3), 7.39, 7.41, 8.01, 8.04, 8.32, 8.41(d, J=7.2, 7.2, 8.0, 8.0, 7.2, 7.2 Hz, 2H of each, 12Ph-H), 13.11(s, 1H, COOH) |
| 3e | 1.27(t, J=7.2 Hz, 3H, CH3), 2.74(q, J=7.2 Hz, 2H, CH2), 7.29, 7.35, 7.41, 8.03, 8.12, 8.25(d, J=8.0, 8.4, 8.4, 8.0, 8.0, 8.0 Hz, 2H of each, 12Ph-H), 12.91(s, 1H, COOH) |
| 3f | 0.97(t, 3H, J=7.2 Hz, CH3), 1.70(m, 2H, CH2), 2.68(t, J=7.2 Hz, 2H, CH2), 7.32, 7.34, 7.98, 8.01, 8.11, 8.35(d, J=8.0, 8.4, 8.0, 8.0, 8.0, 8.4 Hz, 2H of each, 12Ph-H), 12.86(s, 1H, COOH) |
| 3g | 0.90(t, 3H, J=7.2 Hz, CH3), 1.29—1.37(m, 2H, CH2), 1.54—1.65(m, 2H, CH2), 2.66(t, J=7.2 Hz, 2H, CH2), 7.29, 7.36, 7.97, 8.02, 8.09, 8.22(d, J=8.4, 8.8, 8.4, 8.8, 8.0, 8.4 Hz, 2H of each, 12Ph-H), 13.01(s, 1H, COOH) |
| 3h | 0.90(t, 3H, J=6.8 Hz, CH3), 1.25—1.44(m, 6H, CH2CH2CH2), 1.64(t, 2H, J=7.2 Hz, CH2), 7.28, 7.32, 8.03, 8.06, 8.22, 8.28(d, J=8.4, 8.4, 8.0, 8.4, 8.0, 8.4 Hz, 2H of each, 12Ph-H), 13.02(s, 1H, COOH) |
| Entry | Compd. | λmax/nm | f(cis)t(%) | Time of isomerization/min | ||||
|---|---|---|---|---|---|---|---|---|
| Trans isomer | Cis isomer | |||||||
| Ⅰ | Ⅱ | Ⅰ | Ⅱ | Solution | Mesophase | |||
| 1 | 3a | 233 | 347 | 235 | 354 | 11 | 40 | 50 |
| 3b | 223 | 330 | 223 | 327 | 55 | 40 | 70 | |
| 3c | 230 | 334 | 229 | 335 | 38 | 50 | 80 | |
| 2 | 3d | 238 | 340 | 238 | 334 | 37 | 70 | 90 |
| 3e | 239 | 337 | 240 | 334 | 50 | 70 | 90 | |
| 3f | 239 | 339 | 239 | 338 | 37 | 60 | 80 | |
| 3g | 238 | 336 | 239 | 333 | 29 | 60 | 70 | |
| 3h | 239 | 338 | 238 | 333 | 16 | 50 | 70 | |
Table 3 UV spectrum of target compounds and the maximum time of isomerization
| Entry | Compd. | λmax/nm | f(cis)t(%) | Time of isomerization/min | ||||
|---|---|---|---|---|---|---|---|---|
| Trans isomer | Cis isomer | |||||||
| Ⅰ | Ⅱ | Ⅰ | Ⅱ | Solution | Mesophase | |||
| 1 | 3a | 233 | 347 | 235 | 354 | 11 | 40 | 50 |
| 3b | 223 | 330 | 223 | 327 | 55 | 40 | 70 | |
| 3c | 230 | 334 | 229 | 335 | 38 | 50 | 80 | |
| 2 | 3d | 238 | 340 | 238 | 334 | 37 | 70 | 90 |
| 3e | 239 | 337 | 240 | 334 | 50 | 70 | 90 | |
| 3f | 239 | 339 | 239 | 338 | 37 | 60 | 80 | |
| 3g | 238 | 336 | 239 | 333 | 29 | 60 | 70 | |
| 3h | 239 | 338 | 238 | 333 | 16 | 50 | 70 | |
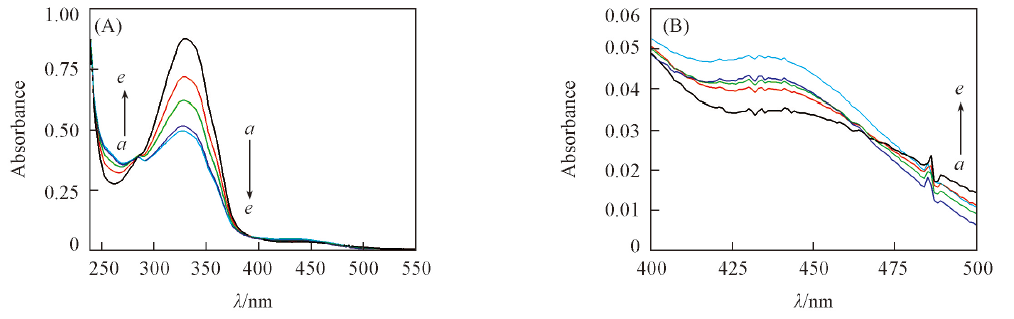
Fig.1 UV-Vis absorption spectra of compound 3b in methanol during trans-to-cis isomerization Fig.1(B) illustrates the small increase of absorption in 430 nm. Time/min: a. 0; b. 10; c. 20; d. 30; e. 40.
| Entry | Compd. | Eox/eV | Φp/eV | Ered/eV | Φn/eV | EHOMO/eV | ELUMO/eV | Eg/eV | ||
|---|---|---|---|---|---|---|---|---|---|---|
| Ⅰ | Ⅱ | Ⅰ | Ⅱ | |||||||
| 1 | 3a | 0.69 | 0.66 | 0.66 | -0.49 | -0.62 | -0.49 | -5.06 | -3.91 | 1.15 |
| 3b | 0.76 | 0.76 | -0.42 | -0.60 | -0.42 | -5.16 | -3.98 | 1.18 | ||
| 3c | 0.76 | 0.63 | 0.63 | -0.53 | -0.64 | -0.53 | -5.03 | -3.87 | 1.16 | |
| 2 | 3d | 0.82 | 0.65 | 0.65 | -0.47 | -0.56 | -0.47 | -5.05 | -3.93 | 1.12 |
| 3e | 0.88 | 0.88 | -0.51 | -0.65 | -0.51 | -5.28 | -3.89 | 1.39 | ||
| 3f | 0.82 | 0.82 | -0.45 | -0.54 | -0.45 | -5.22 | -3.95 | 1.27 | ||
| 3g | 0.83 | 0.83 | -0.54 | -0.64 | -0.54 | -5.23 | -3.86 | 1.37 | ||
| 3h | 0.81 | 0.58 | 0.58 | -0.50 | -0.62 | -0.50 | -4.98 | -3.90 | 1.08 | |
Table 4 Redox potentials, EHOMO, ELUMO and energy gaps of target compounds
| Entry | Compd. | Eox/eV | Φp/eV | Ered/eV | Φn/eV | EHOMO/eV | ELUMO/eV | Eg/eV | ||
|---|---|---|---|---|---|---|---|---|---|---|
| Ⅰ | Ⅱ | Ⅰ | Ⅱ | |||||||
| 1 | 3a | 0.69 | 0.66 | 0.66 | -0.49 | -0.62 | -0.49 | -5.06 | -3.91 | 1.15 |
| 3b | 0.76 | 0.76 | -0.42 | -0.60 | -0.42 | -5.16 | -3.98 | 1.18 | ||
| 3c | 0.76 | 0.63 | 0.63 | -0.53 | -0.64 | -0.53 | -5.03 | -3.87 | 1.16 | |
| 2 | 3d | 0.82 | 0.65 | 0.65 | -0.47 | -0.56 | -0.47 | -5.05 | -3.93 | 1.12 |
| 3e | 0.88 | 0.88 | -0.51 | -0.65 | -0.51 | -5.28 | -3.89 | 1.39 | ||
| 3f | 0.82 | 0.82 | -0.45 | -0.54 | -0.45 | -5.22 | -3.95 | 1.27 | ||
| 3g | 0.83 | 0.83 | -0.54 | -0.64 | -0.54 | -5.23 | -3.86 | 1.37 | ||
| 3h | 0.81 | 0.58 | 0.58 | -0.50 | -0.62 | -0.50 | -4.98 | -3.90 | 1.08 | |
| Entry | Compd. | L/Wa | m.p./℃ | ΔH of m.p./(J·g-1) | d.p.b/℃ |
|---|---|---|---|---|---|
| 1 | 3a | 4.362 | 211.2 | 28.72 | 218—369 |
| 3b | 4.638 | 221.0 | 26.54 | 232—419 | |
| 3c | 4.967 | 208.2 | 23.17 | 226—425 | |
| 2 | 3d | 4.110 | 198.7 | 25.62 | 257—413 |
| 3e | 4.447 | 200.3 | 26.76 | 246—388 | |
| 3f | 4.497 | 199.1 | 21.82 | 230—429 | |
| 3g | 4.549 | 186.8 | 40.39 | 240—393 | |
| 3h | 4.722 | 187.9 | 27.67 | 230—349 |
Table 5 DSC measurement results of target compounds
| Entry | Compd. | L/Wa | m.p./℃ | ΔH of m.p./(J·g-1) | d.p.b/℃ |
|---|---|---|---|---|---|
| 1 | 3a | 4.362 | 211.2 | 28.72 | 218—369 |
| 3b | 4.638 | 221.0 | 26.54 | 232—419 | |
| 3c | 4.967 | 208.2 | 23.17 | 226—425 | |
| 2 | 3d | 4.110 | 198.7 | 25.62 | 257—413 |
| 3e | 4.447 | 200.3 | 26.76 | 246—388 | |
| 3f | 4.497 | 199.1 | 21.82 | 230—429 | |
| 3g | 4.549 | 186.8 | 40.39 | 240—393 | |
| 3h | 4.722 | 187.9 | 27.67 | 230—349 |

Fig.2 POM images of compounds 3a—3h during heating process(A)—(H) Schlieren textures of 3a, 3b, 3c, 3d, 3e, 3f, 3g and 3h taken at 214, 227, 228, 221, 220, 210, 220 and 220 ℃, respectively.
| [1] | 陈然, 安忠维, 陈新兵, 陈沛. 高等学校化学学报, 2014, 35( 7), 1433- 1438 |
| Chen, R. , An Z., W. , Chen X., B. , Chen, P. , Chem. J. Chinese Universities, 2014, 35( 7), 1433- 1438 | |
| [2] | Ikeda, T. , Tsutsumi, O. , Tsutsumi, O. , Science, 1995, 268( 30), 1873- 1875 |
| [3] | Eich, M. , Wendorff J., H. , Makromol. Chem. Rapid Commun., 1987, 8( 10), 467- 471 |
| [4] | Zhou D., W. , Liang L., Y. , Lu M., G. , Polym. Bull., 2011, 66( 8), 1111- 1123 |
| [5] | Wu, L.F. , Tuo X., L. , Cheng, H. , Chen, Z. , Wang X., G. , Macromolecules, 2001, 34( 23), 8005- 8013 |
| [6] | Krohn, K. , John, M. , Demikhov E., I. , Russian Chemical Bulletin(International Edition ), 2001, 50( 7), 1248- 1254 |
| [7] | Luo Y., N. , Xu X., Z. , Sun F., X. , Yu X., Y. , Zhang, X. , Zhang, T. , Yu L., Y. , Chem. Res. Chinese Universities, 2014, 30( 1), 27- 31 |
| [8] | Lee H., K. , Doi, K. , Harada, H. , Tsutsumi, O. , Kanazawa, A. , Shiono, T. , Ikeda, T. , J. Phys. Chem. B, 2000, 104( 30), 7023- 7028 |
| [9] | Tong, X. , Zhao, Y. , Chem. Mater., 2009, 21( 17), 4047- 4054 |
| [10] | Rau H. , Photochemistry and Photophysics Vol. Ⅱ, CRC Press, Boca Raton, FL, 1990, 119— 141 |
| [11] | Kang X., L. , Zhao J., R. , Li H., Y. , He S., J. , Colloid Polym. Sci., 2013, 291( 9), 2245- 2251 |
| [12] | Yao J., L. , You Y., J. , Liu, H. , Dong L., J. , Xiong C., X. , J. Mater. Sci., 2011, 46( 10), 3343- 3348 |
| [13] | Circu, V. , Mocanu A., S. , Rosu, C. , Manaila-Maximean, D. , Dumitrascu, F. , J. Therm. Anal. Calorim., 2012, 107( 3), 877- 886 |
| [14] | Kaš, par M. , Novotná, V. , Hamplová, V. , Podoliak, N. , Nonnenmacher, D. , Glogarová, F. , Giesselmann, M. , Liq. Cryst., 2011, 38( 5), 309- 315 |
| [15] | 郑敏燕, 魏永生, 安忠维. 有机化学, 2008, 28( 12), 1948- 1953 |
| Zheng M., Y. , Wei Y., S. , An Z., W. , Chinese Journal of Organic Chemistry, 2008, 28( 12), 1948- 1953 | |
| [16] | Luo Y., N. , Xu X., Z. , Zhang, X. , Yu X., Y. , Qu X., S. , Yang Y., Y. , Shen Q., H. , Chem. Res. Chinese Universities, 2013, 29( 6), 1045- 1049 |
| [17] | Kreger, K. , Wolfer, P. , Audorff, H. , Kador, L. , Stingelin-Stutzmann, N. , Smith, P. , Schmidt H., W. , J. Am. Chem. Soc., 2010, 132( 2), 509- 516 |
| [18] | Sevigny, S. , Bouchard, L. , Motallebi, S. , Zhao, Y. , Macromolecules, 2003, 36( 24), 9033- 9041 |
| [19] | Leclair, S. , Mathew, L. , Giguere, M. , Motallebi, S. , Zhao, Y. , Macromolecules, 2003, 36( 24), 9024- 9032 |
| [20] | Hunter C., A. , Sarson L., D. , Tetrahedron Lett., 1996, 37( 5), 699- 702 |
| [21] | Cevasco, G. , Thea, S. , J. Org. Chem., 1999, 64( 15), 5422- 5429 |
| [22] | 樊冬丽, 翟岩, 张妍, 涂伟, 黄耀东. 高等学校化学学报, 2014, 35( 11), 2447- 2454 |
| Fan D., L. , Zhai, Y. , Zhang, Y. , Tu, W. , Huang Y., D. , Chem. J. Chinese Universities, 2014, 35( 11), 2447- 2454 | |
| [23] | Wang W., Y. , Wang M., Z. , Polymer Bulletin, 2007, 59( 4), 537- 544 |
| [24] | Ya, Q. , Dong X., Z. , Chen W., Q. , Duan X., M. , Dyes and Pigments, 2008, 79( 2), 159- 165 |
| [25] | Arias, J. , Bardaj, M. , Espinet, P. , Folcia C., L. , Ortega, J. , Etxebarria, J. Inorg. Chem., 2009, 48( 13), 6205- 6210 |
| [26] | 向能军, 李狄豪, 梁万里, 苏树江, 石建新, 龚孟濂. 化学学报, 2006, 64( 11), 1157- 1162 |
| Xiang N., J. , Lee T., H. , Leung L., M. , So S., K. , Shi J., X. , Gong M., L. , Acta Chimica Sinica, 2006, 64( 11), 1157- 1162 | |
| [27] | Gao, L. , Zhang, J. , He, C. , Zhang, Y. , Sun Q., J. , Li Y., F. , Science China, 2014, 57( 7), 966- 972 |
| [28] | 金长清, 苏树江, 梁万里, 马凤英, 王立军, 刘云, 张春玉. 发光学报, 2004, 25( 5), 541- 545 |
| Jin C., Q. , So S., K. , Leung L., M. , Ma F., Y. , Wang L., J. , Liu, Y. , Zhang C., Y. , Chinese Journal of Luminescence, 2004, 25( 5), 541- 545 | |
| [29] | Karim M., R. , Yahya, R. , Sheikh M. R., K. , Salleh N., M. , Hassan, A. , Ekramul M. H. N., M. , Journal of Polymer Research, 2014, 21( 6), 487- 496 |
| [1] | XIE Fan, CHEN Shanshan, ZHUO Longhai, LU Zhaoqing, GAO Kun, DAI Qiyang. Fabrication of Poly(p-xylene) Nanofiber Arrays by CVD Liquid Crystal Template Method and Their Degradability [J]. Chem. J. Chinese Universities, 2021, 42(8): 2643. |
| [2] | FAN Ye, LI Qian, FANG Yun, XIA Yongmei. Fabrication of Lamellar Liquid Crystals of Conjugated Linoleic Acid as Drug Delivery Systems † [J]. Chem. J. Chinese Universities, 2020, 41(4): 750. |
| [3] | WEI Fuzhi, FENG Wei, XIA Yan, LI Dongfeng, HOU Ruibin. Synthesis and Mesocrystalline Properties of Glassy Liquid Crystal Compounds Based on Tetrathiofuvalene and Cyanobenzene† [J]. Chem. J. Chinese Universities, 2018, 39(7): 1549. |
| [4] | WANG Yi, ZHANG Ping, WU Shengxiu, SUN Yuanyuan, ZHAO Tong, LIU Shixi. Fragmentation Mechanism of Two Kinds of Violated Even-electron Rule Compounds with Doubly Charged Ions in Mass Spectrometry† [J]. Chem. J. Chinese Universities, 2018, 39(3): 435. |
| [5] | XIA Meng, PENG Xiongwei, GAO Hongfei, YAN Chao, CHEN Huiru, CHENG Xiaohong. Synthesis and Properties of Barbituric Acid Based Taper Shaped Rodlike Liquid Crystal Compound and Hydrogen Bonded Complex with Triazine Derivative† [J]. Chem. J. Chinese Universities, 2017, 38(7): 1203. |
| [6] | GUO Miaocai, HE Yaning, WANG Xiaogong. Effect of Anisotropic Photo-crosslinking on Liquid Crystal Alignment with Duplicated Surface-relief-gratings† [J]. Chem. J. Chinese Universities, 2015, 36(4): 751. |
| [7] | ZENG Qinghui, LI Na, TIAN Ye, WU Di, ZHAO Yaowu, LI Lihua, ZHOU Changren. Effects of Collagen Films with Liquid Crystal-liked Ordered Structure on Adhesion, Proliferation and Differentiation of Human Umbilical Cord Mesenchymal Stem Cells† [J]. Chem. J. Chinese Universities, 2014, 35(8): 1658. |
| [8] | YANG Guang, WANG Chuanming, TANG Ping, GU Songyuan. Self-assembling Behavior of Symmetric Coil-semiflexible-coil Triblock Copolymers† [J]. Chem. J. Chinese Universities, 2014, 35(8): 1799. |
| [9] | FAN Yu-Xia, TAN Hui, LIU Ling-Wei, WU Zhao-Yang, SHEN Guo-Li, YU Ru-Qin. Novel Nucleic Acid Signal Amplification Liquid Crystal Biosensor for Detection of p53 Gene Mutation [J]. Chem. J. Chinese Universities, 2013, 34(4): 806. |
| [10] | LI Guang, CHEN Long-Cong, CHEN Meng-Meng, GAO Bin, XIONG Xing-Liang. Liquid Crystal Biosensor Based on Growth of Gold Nanoparticles for Detection of Tyrosine [J]. Chem. J. Chinese Universities, 2013, 34(11): 2493. |
| [11] | WANG Yan-Wei, HE Ya-Ning, WANG Xiao-Gong. Photoinduced Orientation of Two Dendritic Azo Amorphous Molecular Materials [J]. Chem. J. Chinese Universities, 2012, 33(10): 2351. |
| [12] | LUO Bing-Hong, LI Jian-Hua, YANG Jing, XU Chong-En, TIAN Jin-Huan, ZHOU Chang-Ren. Preparation of Cholesterol-graft-poly(D,L-lactic acid)/ Poly(D,L-lactic acid) Composite Nanofiber Membranes by Electrospinning [J]. Chem. J. Chinese Universities, 2012, 33(06): 1360. |
| [13] | HUANG Qi-Mao, WANG Si-Wei, LI Qing, PAN Wei, DENG Peng-Xing, ZHOU Hong, PAN Zhi-Quan. Synthesis and Characterization of Curcumin Bridged Porphyrins as Photosensitizers [J]. Chem. J. Chinese Universities, 2012, 33(04): 732. |
| [14] | PENG Hai-Yan, BI Shu-Guang, LIAO Yong-Gui, ZHOU Xing-Ping, XIE Xiao-Lin. Photopolymerization Kinetics of Acrylate/Liquid Crystal Mixtures Initiated by Ketocoumarin/Amine/Triazine Ternary Photoinitiator [J]. Chem. J. Chinese Universities, 2012, 33(03): 640. |
| [15] | SHI Xiao-Jing, PENG Hai-Yan, LIAO Yong-Gui, XIE Xiao-Lin*. Kinetics of Photopolymerization for the Acrylate/Liquid Crystal Mixture [J]. Chem. J. Chinese Universities, 2011, 32(6): 1407. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||