

Chem. J. Chinese Universities ›› 2014, Vol. 35 ›› Issue (4): 831.doi: 10.7503/cjcu20130797
• Physical Chemistry • Previous Articles Next Articles
JIANG Jianhong1, LI Xu1, XIAO Shengxiong1, GU Huiwen2, LI Chuanhua1, YANG Ping1, WEI Deliang1, HE Dugui1, LI Aitao1, LI Xia1, YAO Feihong1, LI Qiangguo1,*( )
)
Received:2013-08-16
Online:2014-04-10
Published:2014-02-27
Contact:
LI Qiangguo
E-mail:liqiangguo@163.com
Supported by:CLC Number:
TrendMD:
JIANG Jianhong, LI Xu, XIAO Shengxiong, GU Huiwen, LI Chuanhua, YANG Ping, WEI Deliang, HE Dugui, LI Aitao, LI Xia, YAO Feihong, LI Qiangguo. Interaction of 2-{[4-Amino-5-(3,4,5-trimethoxy-benzyl)-pyrimidin-2-ylimino]-methyl}-6-methoxy-phenol with S. pombe Cells and BSA†[J]. Chem. J. Chinese Universities, 2014, 35(4): 831.
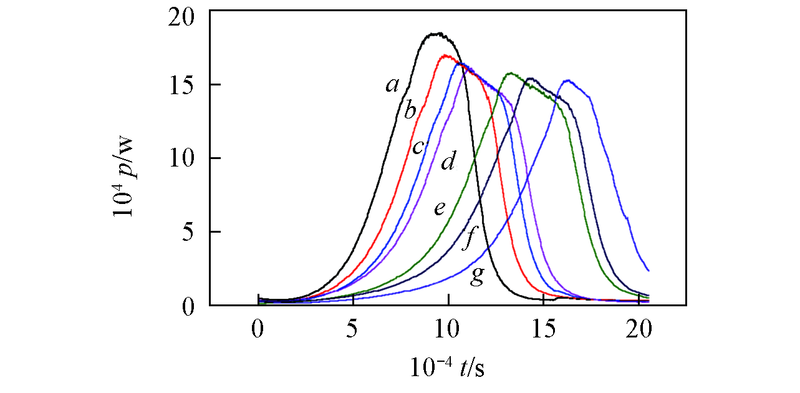
Fig.1 Power-time curves for the growth of S. pombe affected by APYPConcentrations of APYP/(mg·L-1): a. 0; b. 40; c. 60; d. 80; e. 100; f. 120; g. 140.
| c(APYP)/(mg·L-1) | k*/s-1 | R | I | IC50/(mg·L-1) |
|---|---|---|---|---|
| 0 | 7.20×10-5±5×10-8 | 0.9994 | 0 | |
| 40 | 6.26×10-5±4×10-8 | 0.9992 | 13.1 | |
| 60 | 5.37×10-5±4×10-8 | 0.9993 | 25.4 | |
| 80 | 4.76×10-5±2×10-8 | 0.9998 | 33.9 | 123.2 |
| 100 | 4.10×10-5±7×10-9 | 1.0000 | 43.1 | |
| 120 | 3.63×10-5±1×10-8 | 0.9995 | 49.6 | |
| 140 | 3.40×10-5±2×10-8 | 0.9991 | 52.8 |
Table 1 Thermokinetic parameters of the growth of S.pombe affected by different concentrations of APYP at 301.15 K
| c(APYP)/(mg·L-1) | k*/s-1 | R | I | IC50/(mg·L-1) |
|---|---|---|---|---|
| 0 | 7.20×10-5±5×10-8 | 0.9994 | 0 | |
| 40 | 6.26×10-5±4×10-8 | 0.9992 | 13.1 | |
| 60 | 5.37×10-5±4×10-8 | 0.9993 | 25.4 | |
| 80 | 4.76×10-5±2×10-8 | 0.9998 | 33.9 | 123.2 |
| 100 | 4.10×10-5±7×10-9 | 1.0000 | 43.1 | |
| 120 | 3.63×10-5±1×10-8 | 0.9995 | 49.6 | |
| 140 | 3.40×10-5±2×10-8 | 0.9991 | 52.8 |
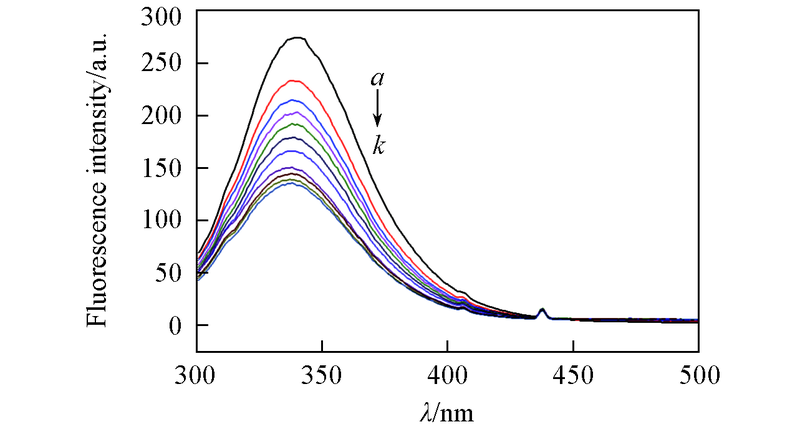
Fig.4 Quenching effects of APYP on BSA fluore-scence intensity at 298.15 Kλex=280 nm, λem=340 nm, c(BSA)=2.0×10-6 mol/L, c(APYP)=2.0×10-6 mol/L. c(APYP)/c(BSA), a—k: 0, 1.0, 2.0, 3.0, 4.0, 5.0, 6.0, 7.0, 8.0, 9.0, 10.0.
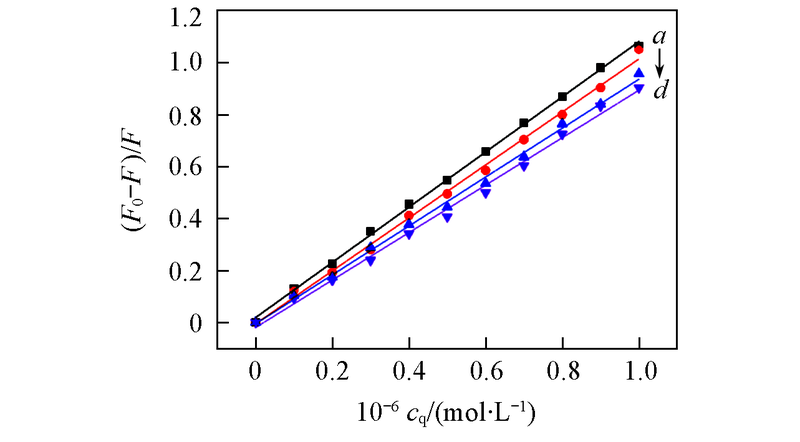
Fig.5 Stem-Volmer plots for the fluorescence quenching of BSA by APYP at different temperaturesa. 298.15 K, slope=1.0622, R=0.9996; b. 302.15 K, slope=1.0194, R=0.9985; c. 306.15 K, slope=0.9414, R=0.9987; d. 310.15 K, slope=0.9133, R=0.9977.
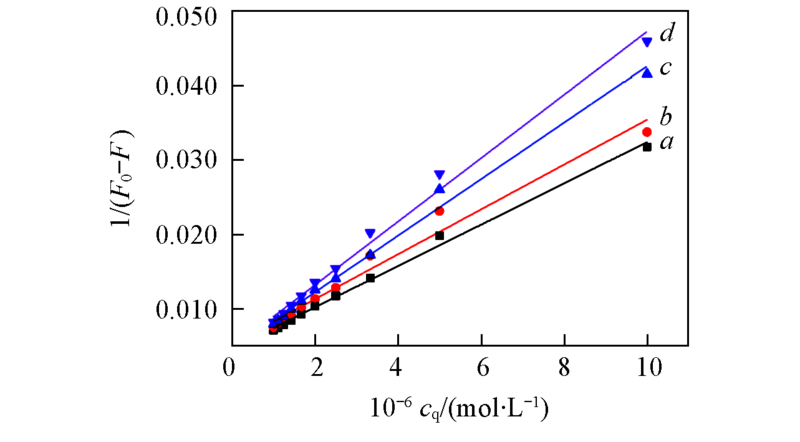
Fig.6 Lineweaver-Burk plots for the fluorescence quenching of BSA by APYP at different temperaturesa. 298.15 K, slope=0.00277, R=0.9976; b. 302.15 K, slope=0.00301, R=0.9875; c. 306.15 K, slope=0.00379, R=0.9962; d. 310.15 K, slope=0.00425, R=0.09960.
| Temperature/K | KLB | |||
|---|---|---|---|---|
| 298.15 | 1.317×106 | -22.997 | -34.929 | 40.020 |
| 302.15 | 1.241×106 | -22.997 | -35.248 | 40.546 |
| 306.15 | 1.013×106 | -22.997 | -35.198 | 39.853 |
| 310.15 | 9.198×105 | -22.997 | -35.409 | 40.019 |
Table 2 Relative thermodynamic parameters of APYP-BSA system
| Temperature/K | KLB | |||
|---|---|---|---|---|
| 298.15 | 1.317×106 | -22.997 | -34.929 | 40.020 |
| 302.15 | 1.241×106 | -22.997 | -35.248 | 40.546 |
| 306.15 | 1.013×106 | -22.997 | -35.198 | 39.853 |
| 310.15 | 9.198×105 | -22.997 | -35.409 | 40.019 |
| Temperature/K | KA/(L·mol-1) | n | R | Temperature/K | KA/(L·mol-1) | n | R |
|---|---|---|---|---|---|---|---|
| 298.15 | 3.3´105 | 0.9026(≈1) | 0.9968 | 306.15 | 7.5´105 | 0.9820(≈1) | 0.9980 |
| 302.15 | 5.6´105 | 0.9521(≈1) | 0.9951 | 310.15 | 1.0´106 | 1.0139(≈1) | 0.9971 |
Table 3 Binding constants and number of binding sites of APYP-BSA system
| Temperature/K | KA/(L·mol-1) | n | R | Temperature/K | KA/(L·mol-1) | n | R |
|---|---|---|---|---|---|---|---|
| 298.15 | 3.3´105 | 0.9026(≈1) | 0.9968 | 306.15 | 7.5´105 | 0.9820(≈1) | 0.9980 |
| 302.15 | 5.6´105 | 0.9521(≈1) | 0.9951 | 310.15 | 1.0´106 | 1.0139(≈1) | 0.9971 |
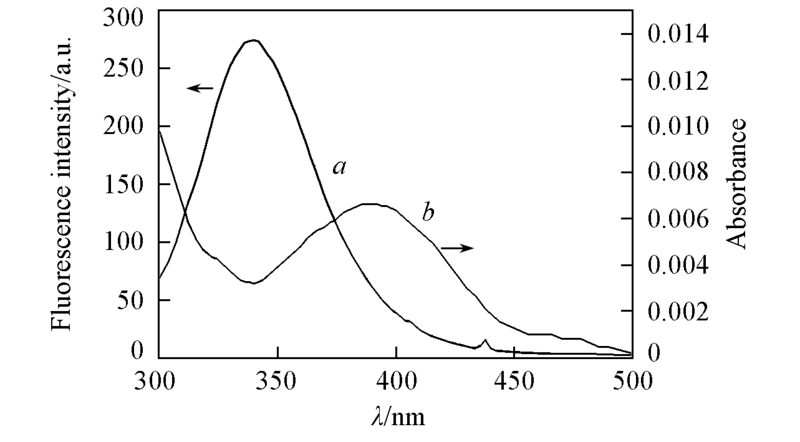
Fig.8 Spectral overlap between the absorption spectrum of APYP(a) and fluorescence emission spectrum of BSA(b) at 298.15 Kc(BSA)=2.0×10-6 mol/L; c(APYP)=2.0×10-6, 5 mmol/L. Tris-HCl buffer, pH=7.4. Overlapped region is highlighted.
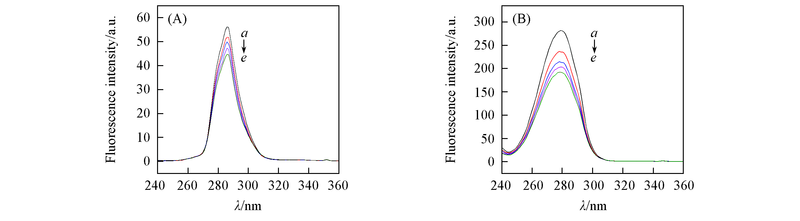
Fig.9 Synchronous fluorescence spectra of BSA at 298.15 K(A) Δλ=15 nm; (B) Δλ=60 nm. c(BSA)=c(APYP)=2.0×10-6 mol/L. c(APYP)/c(BSA), a—e: 0, 1.0, 2.0, 4.0, 5.0.
| [1] | Torres A., Garedew A., Schmolz E., Lamprecht I., Thermochim. Acta,2004, 415(1/2), 107—113 |
| [2] | Montanari M., Andricopulo A., Montanari C., Thermochim. Acta,2004, 417(2), 283—294 |
| [3] | Panneerselvam P., Nair R. R., Vijayalakshmi G., Subramanian E. H., Sridhar S. K., Eur. J. Med. Chem., 2005, 40, 225—229 |
| [4] | Prabhakaran R., Geetha A., Thilagavathi M., Karvembu R., Krishnan V., Bertagnolli H., Natarajan K., J. Inorg. Biochem., 2004, 98, 2131—2140 |
| [5] | Assegid G., Lamprecht I., Thermochim. Acta,1997, 290(2), 155—166 |
| [6] | Jeewoth T., Bhowon M. G., Wah H. L. K., Transit. Met. Chem., 1999, 24(4), 445—448 |
| [7] | Wang Z., Chemical Dictionary, Chemical Industry Press, Beijing, 2010, 445 |
| (王箴. 化工辞典, 北京:化学工业出版社, 2010, 445) | |
| [8] | Yang D. J., Li X., Hu J. L., Yao F. H., Xiao S. X., Yang L. L., Xiang Y. L., Li Q. G., Acta Chim. Sinica,2010, 68, 2373—2380 |
| (杨德俊, 李旭, 胡吉林, 姚飞虹, 肖圣雄, 杨丽兰, 向宥霖, 李强国. 化学学报, 2010, 68, 2373—2380) | |
| [9] | Li Q. G., Yang D. J., Li X., Ye L. J., Wei D. L., Xiao S. X., Acta Chim. Sinica,2008, 66, 2686—2692 |
| (李强国, 杨德俊, 李旭, 叶丽娟, 魏得良, 肖圣雄. 化学学报, 2008, 66, 2686—2692) | |
| [10] | Chen G.Z., Huang X. Z., Xu J. G., Zheng Z. Z., Wang Z. B., Fluorescence Analytical Methods, Science Press, Beijing, 1990, 495 |
| (陈国珍, 黄贤智, 许金钩, 郑朱梓, 王尊本. 荧光分析法(2版), 北京: 科学出版社, 1990, 495) | |
| [11] | Cheng P. F., Xu K. X., Yao W. Y., Kong H. J., Kou L., Ma X. D., Wang C. J., Chem. Res. Chinese Universities,2013, 29(4), 642—646 |
| [12] | Lakowicz J.R., Principles of Fluorescence Spectroscopy, Plenum Press, New York, 1983, 257—295 |
| [13] | Jiang C. Q., Wang J. Z., Wang H. J., He X. C., Chinese J. Anal. Chem., 1999, 27(8), 894—898 |
| (江崇球, 王敬政, 王洪鉴, 贺笑春. 分析化学, 1999, 27(8), 894—898) | |
| [14] | Braun A.M., Maurette M. T., Photochemical Technology, Ed.: Oliveros E., Wiley, Newyork, 1991, 41 |
| [15] | Guo Q. L., Li R., Jiang F. L., Tu J. C., Li L. W., Liu Y. , Physico-Chimica Sinica,2009, 25(10), 2147—2154 |
| (郭清莲, 李冉, 蒋风雷, 涂建成, 李林尉, 刘义. 物理化学学报, 2009, 25(10), 2147—2154) | |
| [16] | Demant E. J., Friche E., Biochem. Pharmacol., 1998, 55, 27—32 |
| [17] | Yang S. P., Han L. J., Pan Y., Wang D. Q., Zhao C., Wang B., Chem. J. Chinese Universities,2012, 33(1), 14—21 |
| (杨树平, 韩立军, 潘燕, 王大奇, 赵翠, 王波. 高等学校化学学报, 2012, 33(1), 14—21) | |
| [18] | Ma G. B., Gao F., Ren B. Z., Yang P., Acta Chim. Sinica,1995, 53(12), 1193—1196 |
| (马贵斌, 高飞, 任斌知, 杨频. 化学学报, 1995, 53(12), 1193—1196) | |
| [19] | Xie M. X., Xu X. Y., Wang Y. D., Liu Y., Acta Chim. Sinica,2005, 63(22), 2055—2062 |
| (谢孟峡, 徐晓云, 王英典, 刘媛. 化学学报, 2005, 63(22), 2055—2062) | |
| [20] | Chen G.Z., Huang X. Z., Xu J. G., Zheng Z. Z., Wang Z. B., Fluorescence Analytical Methods, Science Press, Beijing, 1990, 122 |
| (陈国珍, 黄贤智, 许金钩, 郑朱梓, 王尊本. 荧光分析法(2版), 北京: 科学出版社, 1990, 122) | |
| [21] | Yang M. M., Yang P., Zhang L. W., Chin. Sci. Bull., 1994, 39(1), 31 |
| (杨曼曼, 杨频, 张立伟. 科学通报, 1994, 39(1), 31) | |
| [22] | Huang B., Zou G. L., Yang T. M., Acta Chim. Sinica,2002, 60(10), 1867—1871 |
| (黄波, 邹国林, 杨天鸣. 化学学报, 2002, 60(10), 1867—1871) | |
| [23] | Huang Y., Wang J., Guo G. Y., Tao Z., Xue S. F., Zhu Q. J., Zhou Q. D., Chem. J. Chinese Universities,2013, 34(2), 375—380 |
| (黄英, 王娟, 郭改英, 陶朱, 薛赛凤, 祝黔江, 周清娣. 高等学校化学学报, 2013, 34(2), 375—380) | |
| [24] | Yu Z. W., Gao W. Y., Progress in Chemistry,2006, 18(7/8), 1049—1055 |
| (尉志武, 高文颖. 化学进展, 2006, 18(7/8), 1049—1055) |
| [1] | LI Mengshuo, ZHANG Jing, LIU Dan, ZHU Yaxian, ZHANG Yong. Interactions of Pyrene with Human Serum Albumin and Bovine Serum Albumin: Microenvironmental Polarity Differences at Binding Sites [J]. Chem. J. Chinese Universities, 2021, 42(3): 731. |
| [2] | HAO Yuanyuan, WU Qi, LI Ji, GE Chao, MA Chaoying, QIAN Yong, SU Zhi, LIU Hongke. Novel OsⅡ -arene Complexes Based on Bipyridyl Derivative Ligands: Synthesis, Crystal Structure, Anticancer Activity and Interaction with DNA/BSA† [J]. Chem. J. Chinese Universities, 2018, 39(4): 614. |
| [3] | AN Pengjiao, YU Nannan, SUN Ruisheng, SUI Xiaofang, SONG Yuguang. Characterization of the Interaction Between Esterified TAM Radical and Bovine Serum Albumin [J]. Chem. J. Chinese Universities, 2017, 38(8): 1354. |
| [4] | XU Guoqing, HAO Changchun, HE Jianzhen, ZHANG Lei, SUN Runguang. Effect of Bovine Serum Albumin on the Structure of DSPE Monolayer† [J]. Chem. J. Chinese Universities, 2017, 38(12): 2238. |
| [5] | ZHANG Jing, CHEN Linfeng, ZHU Yaxian, ZHANG Yong. Effects of Hydroxypropyl-β-cyclodextrin(HPCD) on the Interaction of 1-Hydroxypyrene with Bovine Serum Albumin† [J]. Chem. J. Chinese Universities, 2017, 38(1): 28. |
| [6] | ZHANG Jing, CHEN Weixiao, ZHANG Wei, DUAN Ying, ZHU Yuxiu, ZHU Yaxian, ZHANG Yong. Interaction of 1-Hydroxypyrene with BSA Using Fluorescence Anisotropy and Synchronous Fluorescence Analysis Methods† [J]. Chem. J. Chinese Universities, 2015, 36(8): 1511. |
| [7] | YANG Shuilan, SONG Pan, SHE Wenjie, YANG Tianlin. Mechanism of the Interaction Between a Phosphorus-containing Tripod Ligand Europium(Ⅲ) Complex and Bovine Serum Albumin† [J]. Chem. J. Chinese Universities, 2015, 36(7): 1254. |
| [8] | LI Xu, JIANG Jianhong, HAN Buxing, GU Huiwen, XIE Zhaofeng, CHEN Lan, XIAO Shengxiong, LI Chuanhua, LI Aitao, LI Xia, YAO Feihong, WANG Qun, LI Qiangguo. Synthesis and Biological Activities of o-Vanillin-histidine Schiff-base and Its Lanthanum Complex† [J]. Chem. J. Chinese Universities, 2015, 36(5): 856. |
| [9] | WANG Manyuan, ZHANG Chao, LI Jing, LI Zhaoxia, GONG Muxin. Interaction of Antimalarial Components Combination from Artemisia annua L. with Bovine Serum Albumin† [J]. Chem. J. Chinese Universities, 2014, 35(2): 309. |
| [10] | LIU Xiaoxia, DENG Hao, WANG Yanying, LU Zhiwei, ZENG Xianyin, WANG Xianxiang, ZOU Ping, RAO Hanbing. Thermodynamics Studies on the BSA Adsorption onto Zinc Oxide Surfaces with Different Morphologies† [J]. Chem. J. Chinese Universities, 2014, 35(10): 2156. |
| [11] | LIN Hai-Bin, ZHENG Lin, LIN Yu-Qin, ZHOU Zhao-Hui. Study of Interaction Between Bovine Serum Albumin and Cobalt Complexes of Phenanthroline and Nitrilotriacetate by Fluorescence Spectrometry [J]. Chem. J. Chinese Universities, 2013, 34(8): 1818. |
| [12] | YANG Shu-Ping, HAN Li-Jun, PAN Yan, WANG Da-Qi, WANG Nan-Nan, WANG Ting. Synthesis, Characterization, Biological Activity and Interaction with Bovine Serum Albumin of 8- or 6-(3-Chlorobenzoyl)coumarin Derivatives [J]. Chem. J. Chinese Universities, 2013, 34(2): 364. |
| [13] | HUANG Ying, WANG Juan, GUO Gai-Ying, TAO Zhu, XUE Sai-Feng, ZHU Qian-Jiang, ZHOU Qing-Di. Interaction of 6-Thioguanine with Cucurbit[7]uril and Bovine Serum Albumin by Spectroscopic Method [J]. Chem. J. Chinese Universities, 2013, 34(2): 375. |
| [14] | LV Jian-Quan, HU Qin-Qin, DING Ran, ZHANG Xia, ZHOU Xing-Wang. Synthesis and Fluorescence Property of Novel AgInS2@ZnS Quantum Dots [J]. Chem. J. Chinese Universities, 2013, 34(11): 2478. |
| [15] | WANG Shan-Shan, WANG Xue-Ting, GUO Ming-Ming, YU Jun-Sheng. Ligand Effects on Selective Interaction of CdTe Quantum Dots with BSA [J]. Chem. J. Chinese Universities, 2012, 33(06): 1195. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||