

高等学校化学学报 ›› 2020, Vol. 41 ›› Issue (5): 901.doi: 10.7503/cjcu20190634
• 庆祝《高等学校化学学报》复刊40周年专栏 • 上一篇 下一篇
收稿日期:2019-12-09
出版日期:2020-05-10
发布日期:2020-02-10
通讯作者:
王亚培
E-mail:yapeiwang@ruc.edu.cn
基金资助:
GAO Naiwei,MA Qiang,HE Yonglin,WANG Yapei( )
)
Received:2019-12-09
Online:2020-05-10
Published:2020-02-10
Contact:
Yapei WANG
E-mail:yapeiwang@ruc.edu.cn
Supported by:摘要:
基于离子液体的绿色液体电子器件可回收性强, 且具备柔性、 自修复性、 可重塑与可重构性等性能, 拓宽了液体电子器件的应用范围, 为绿色环保的多功能电子器件的开发提供了新途径. 本文围绕离子液体基的电子器件进行了总结, 并阐述了该类器件广阔的应用前景.
中图分类号:
TrendMD:
高乃伟, 马强, 贺泳霖, 王亚培. 基于离子液体的绿色电子器件. 高等学校化学学报, 2020, 41(5): 901.
GAO Naiwei, MA Qiang, HE Yonglin, WANG Yapei. Green Electronic Devices Based on Ionic Liquids †. Chem. J. Chinese Universities, 2020, 41(5): 901.
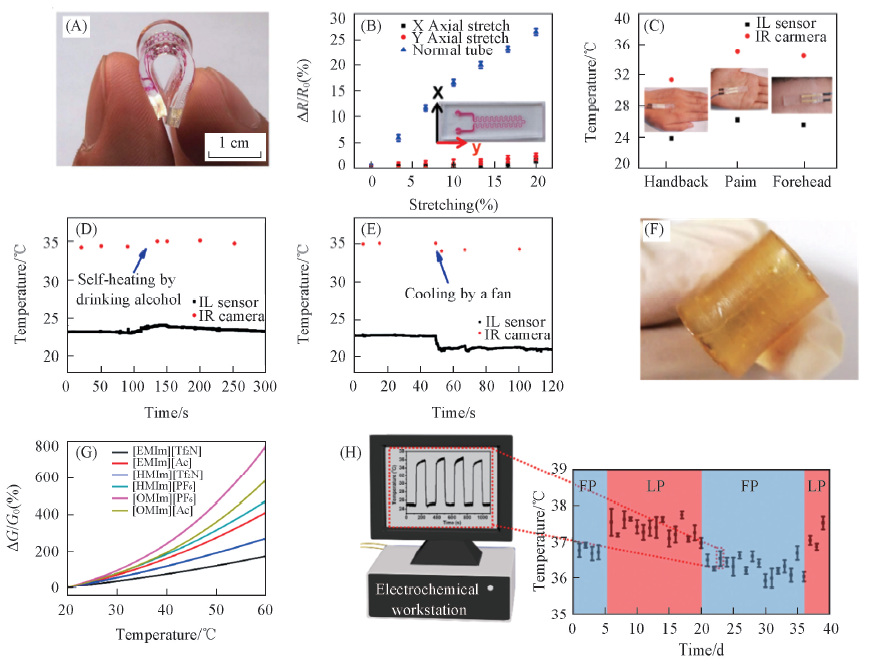
Fig.1 Liquid electronic materials and healthy monitor of liquid temperature sensors[24,27,28] (A) Optical image of flexible temperature sensor[24]. (B) The measurement of resistance stability of flexible temperature sensor at different stress[24]. (C—E) Flexible ionic liquid-based temperature sensor and the monitoring of body temperature[24]. Copyright 2015, John Wiley and Sons. (F) Optical image of self-healing temperature sensor[27]. (G) The electrical response of different ionic liquids against temperature change[27]. Copyright 2015, John Wiley and Sons. (H) Measurement of periodic temperature change of female volunteer by the CaCl2/EG-based temperature sensor[28]. Copyright 2018, American Chemical Society.
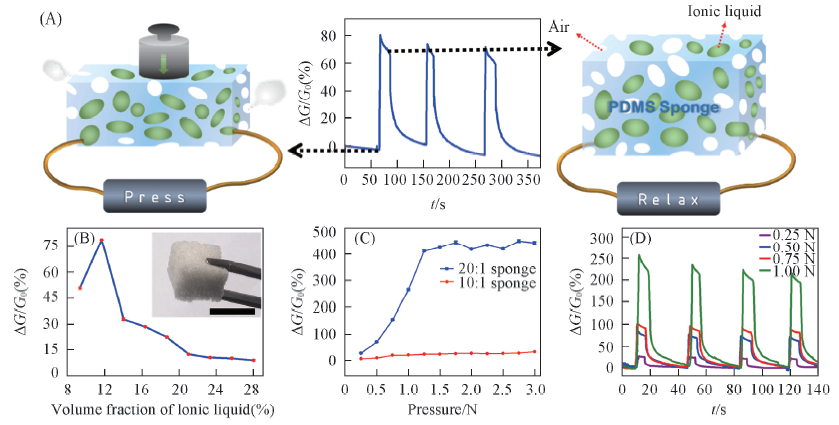
Fig.2 Mechanism and measurement of liquid strain sensors based on ionic liquids[24] (A) Schematic illustration of the sensing principle of liquid strain sensors via partially filling [OMIm]Ac into a PDMS sponge. (B) the relationship between volume fraction of ionic liquid and the conductivity change at a given pressure of 0.5 N. (C) Pressure sensing performance of PDMS sponge with different crosslinking degrees, the pressure ranges from 0.25 to 3.00 N with an interval of 0.25 N. (D) Pressure sensing measurements of PDMS sponge with crosslinking degree of 20:1 at different pressure forces. Copyright 2015, John Wiley and Sons.
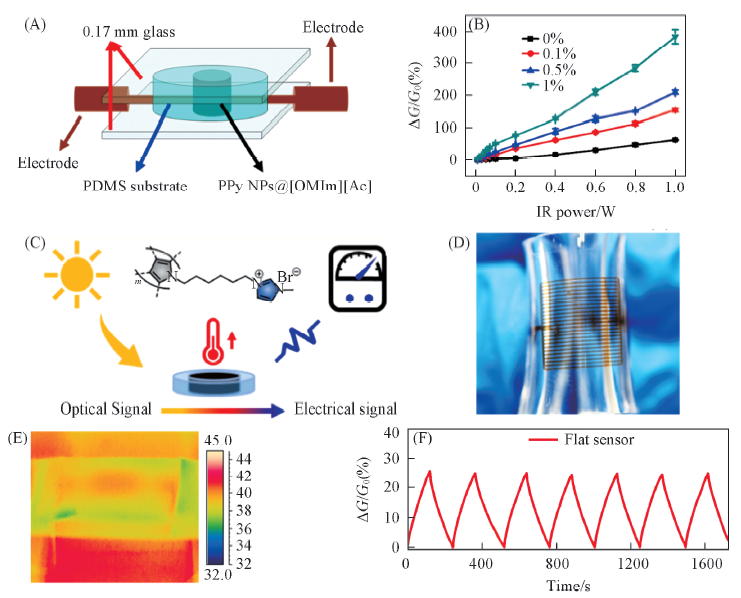
Fig.3 Near infrared response measurement of liquid NIR sensors based on ionic liquids[24,37] (A) Structure of the flexible NIR sensor based on PPyNPs@[OMIm][Ac][24]. (B) NIR sensing of flexible NIR sensor doping with different mass fraction of PPy NPs at different NIR power[24]. Copyright 2015, John Wiley and Sons. (C) Mechanism of light sensing by poly-[Py-Cn-MIm]Br on the basis of photothermal conversion[37]. (D) Optical image of multi-channel light sensor[37]. (E, F) IR image and electrical responses of flexible light sensors[37]. Copyright 2019, American Chemical Society.
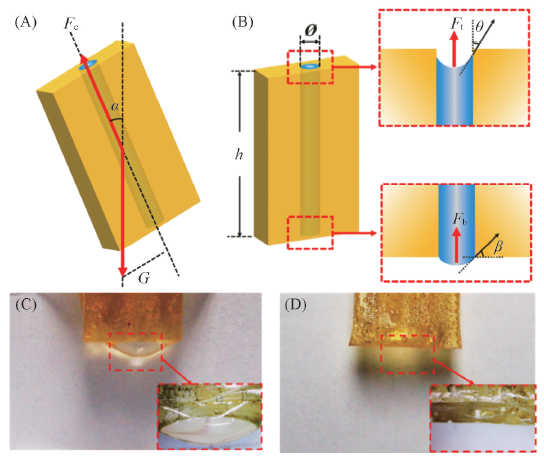
Fig.4 Mechanicalmodel of liquid sensors based on ionic liquids confined by straight-shape capillary channel[27] (A), (B) The mechanical analysis of ionic liquids filled in a straight-shape capillary channel. (C), (D) Optical images of a vertically placed microchannel filled with [OMIm]PF6. The diameters of microchannel are 1.615 mm(C) and 0.709 mm(D), respectively. Copyright 2015, John Wiley and Sons.
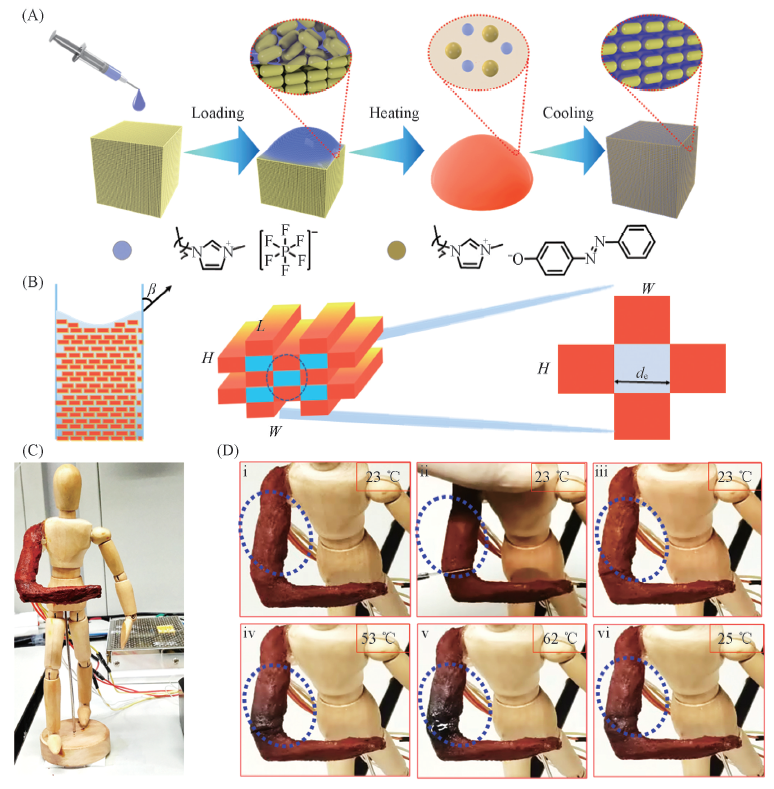
Fig.5 Preparation and repairable measurement of crystal-confined freestanding ionic liquids[41] (A) Preparation of crystal-confined ionic liquids as a complex of [OMIm]PF6 and [OMIm]AzoO through a super-saturated solution cooling method. (B) A mathematical model to demonstrate the pinning capillary effect by [OMIm]AzoO crystals. (C) Optical images of self-healing robotic arms made of crystal-confined ionic liquids. (D) Repairable measurement through the process of cutting and healing of the robotic arm at different status. Copyright 2019, Springer Nature.
| [1] |
Yin L., Cheng H., Mao S., Haasch R., Liu Y., Xie X., Hwang S. W., Jain H., Kang S. K., Su Y., Li R., Huang Y., Rogers J. A., Adv. Funct. Mater., 2014,24(5), 645—658
doi: 10.1002/adfm.201301847 URL |
| [2] |
Hwang S. W., Song J. K., Huang X., Cheng H., Kang S. K., Kim B. H., Kim J. H., Yu S., Huang Y., Rogers J. A., Adv. Mater., 2014,26(23), 3905—3911
doi: 10.1002/adma.201306050 URL pmid: 24692101 |
| [3] |
Zhu H., Luo W., Ciesielski P. N., Fang Z., Zhu J. Y., Henriksson G., Himmel M. E., Hu L., Chem. Rev., 2016,116(16), 9305—9374
doi: 10.1021/acs.chemrev.6b00225 URL pmid: 27459699 |
| [4] |
Torimoto T., Tsuda T., Okazaki K., Kuwabata S., Adv. Mater, 2010,22(11), 1196—1221
doi: 10.1002/adma.200902184 URL pmid: 20437507 |
| [5] |
Hao L., Zhao Y., Yu B., Yang Z., Zhang H., Han B., Gao X., Liu Z., ACS Catal., 2015,5(9), 4989—4993
doi: 10.1021/acscatal.5b01274 URL |
| [6] | Hu X. Y., Liu Q. G., Zhuo K. L., Wang J. J., Chem. J. Chinese Universities, 2013,34(2), 324—330 |
| ( 胡晓宇, 刘千阁, 卓克磊, 王键吉 . 高等学校化学学报, 2013,34(2), 324—330) | |
| [7] | Li G. H., Zhang S. J., Li Z. X., Li M. X., Zhang X. P., Chem. J. Chinese Universities, 2004,25(6), 1137—1138 |
| ( 李桂花, 张锁江, 李增喜, 李明岫, 张香平 . 高等学校化学学报, 2004,25(6), 1137—1138) | |
| [8] | Liu Z., Acta Phys. Chim. Sin., 2019,35(8), 792—793 |
| ( 刘志敏 . 物理化学学报, 2019,35(8), 792—793) | |
| [9] | Hu D. L., Zhang D. Y., Zhang C. L., Ding S. J., Yang Z. Z., Chem. J. Chinese Universities, 2012,33(10), 2345—2350 |
| ( 胡大林, 张东阳, 张成亮, 丁书江, 杨振忠 . 高等学校化学学报, 2012,33(10), 2345—2350) | |
| [10] | Earle M. J., McCormac P. B., Seddon K. R., Green Chem., 2000,2(6), 261—262 |
| [11] | Zhu H. P., Yang F., Tang J., He M. Y., Green Chem., 2003,5(1), 38—39 |
| [12] | Fukaya Y., Iizuka Y., Sekikawa K., Ohno H., Green Chem., 2007,9(11), 1155—1157 |
| [13] |
Dong K., Liu X., Dong H., Zhang X., Zhang S., Chem. Rev, 2017,117(10), 6636—6695
doi: 10.1021/acs.chemrev.6b00776 URL pmid: 28488441 |
| [14] | Zhang S., Sun J., Zhang X., Xin J., Miao Q., Wang J., Chem. Soc. Rev, 2014,43(22), 7386—7869 |
| [15] |
Goubaidoulline I., Vidrich G., Johannsmann D., Anal. Chem, 2005,77(2), 615—619
doi: 10.1021/ac048436a URL pmid: 15649061 |
| [16] | He Y., Gui Q., Wang Y., Wang Z., Liao S., Wang Y ., Small, 2018,14(19), 1800934 |
| [17] |
Gui Q., He Y., Gao N. W., Tao X. L., Wang Y., Adv. Funct. Mater., 2017,27(36), 1702050
doi: 10.1002/adfm.v27.36 URL |
| [18] |
Gui Q., Zhou Y., Liao S., He Y., Tang Y., Wang Y ., Soft Matter, 2019,15(3), 393—398
doi: 10.1039/c8sm02234h URL pmid: 30570632 |
| [19] |
Tang S. K., Baker G. A., Ravula S., Jones J. E., Zhao H., Green Chem., 2012,14(10), 2922—2932
doi: 10.1039/c2gc35631g URL |
| [20] |
Guo P., Zhang H., Liu X., Sun J., ACS Appl. Mater. Interfaces, 2018,10(2), 2105—2113
doi: 10.1021/acsami.7b16880 URL pmid: 29264915 |
| [21] |
Tao X., Jia H., He Y., Liao S., Wang Y., ACS Sens., 2017,2(3), 449—454
doi: 10.1021/acssensors.7b00060 URL pmid: 28723205 |
| [22] |
Leys J., Rajesh R. N., Menon P. C., Glorieux C., Longuemart S., Nochemann P., Pallens M., Binnemans K., J. Chem. Phys., 2010,133(3), 034503
doi: 10.1063/1.3455892 URL pmid: 20649333 |
| [23] |
Liao H., Liao S., Tao X., Liu C., Wang Y., J. Mater. Chem. C, 2018,6(47), 12992—12999
doi: 10.1039/C8TC04699A URL |
| [24] |
Jia H., He Y., Zhang X., Du W., Wang Y., Adv. Electron. Mater, 2015,1(3), 1500029
doi: 10.1002/aelm.201500029 URL |
| [25] |
Bubel B., Menyo M. S., Mates T. E., Waite J. H., Chabinyc M. L., Adv. Mater., 2015,27(21), 3331—3335
doi: 10.1002/adma.201500556 URL pmid: 25903475 |
| [26] |
Pitsch F., Krull F. F., Agel F., Schulz P., Wasserscheid P., Melin T., Wessling M., Adv. Mater., 2012,24(31), 4306—4310
doi: 10.1002/adma.201201832 URL pmid: 22718429 |
| [27] |
He Y., Liao S., Jia H., Cao Y., Wang Z., Wang Y., Adv. Mater, 2015,27(31), 4622—4627
doi: 10.1002/adma.201501436 URL pmid: 26153404 |
| [28] |
Tao X., Liao S., Wang S., Wu D., Wang Y., ACS Sens., 2018,3(7), 1338—1346
doi: 10.1021/acssensors.8b00249 URL pmid: 29905463 |
| [29] |
Lei Z., Wu P., Nat. Commun, 2018,9, 1134
doi: 10.1038/s41467-018-03456-w URL pmid: 29555905 |
| [30] |
Gao N., Zhang X., Liao S., Jia H., Wang Y., ACS Macro Lett., 2016,5(7), 823—827
doi: 10.1021/acsmacrolett.6b00338 URL |
| [31] |
Xu X. Q., Wang Z., Li R., He Y., Wang Y., Chem. Eur. J., 2018,24(39), 9769—9772
doi: 10.1002/chem.v24.39 URL |
| [32] |
Wang J., Zhao J., Li Y. B., Yang M., Chang Y. Q., Zhang J. P., Sun Z. W., Wang Y., ACS Macro Lett., 2015,4(4), 392—397
doi: 10.1021/acsmacrolett.5b00089 URL |
| [33] |
Shen X., Ji M., Zhang S., Qin Y., Zhang P., Wang Y., Guo Z. X., Pan M., Zhang Z., Compos. Sci. Technol., 2019,170(20), 77—84
doi: 10.1016/j.compscitech.2018.11.029 URL |
| [34] |
Cao Y., Dou J. H., Zhao N. J., Zhang S., Zheng Y. Q., Zhang J. P., Wang J. Y., Pei J., Wang Y., Chem. Mater., 2017,29(2), 718—725
doi: 10.1021/acs.chemmater.6b04405 URL |
| [35] |
He Y., Cao Y., Wang Y., Asian J. Org. Chem, 2018,7(11), 2201—2212
doi: 10.1002/ajoc.201800450 URL |
| [36] |
Jiao Y., Liu K., Wang G., Wang Y., Zhang X., Chem. Sci, 2015,6(7), 3975—3980
doi: 10.1039/c5sc01167a URL pmid: 29218167 |
| [37] |
He Y., Xu X. Q., Lv S., Liao H., Wang Y., Langmuir, 2019,35(5), 1192—1198
doi: 10.1021/acs.langmuir.8b02125 URL pmid: 30086640 |
| [38] |
Li M., Zhu W., Zhang P., Chao Y., He Q., Yang B., Li H., Borisevich A., Dai S ., Small, 2016,12(26), 3535—5542
doi: 10.1002/smll.201600358 URL pmid: 27225944 |
| [39] |
Zhang S., Zhang J., Zhang Y., Deng Y., Chem. Rev, 2017,117(10), 6755—6833
doi: 10.1021/acs.chemrev.6b00509 URL pmid: 28032993 |
| [40] |
Zhang J., Bai Y., Sun X. G., Li Y., Guo B., Chen J., Veith G. M., Hensley D. K., Paranthaman M. P., Goodenough J. B., Dai S., Nano Lett., 2015,15(5), 3398—3402
doi: 10.1021/acs.nanolett.5b00739 URL pmid: 25844598 |
| [41] |
Gao N., He Y., Tao X., Xu X. Q., Wu X., Wang Y., Nat. Commun., 2019,10, 547
doi: 10.1038/s41467-019-08433-5 URL pmid: 30710100 |
| [42] | Wang Y., Jia H ., Chem., 2017,80(2), 123—131 |
| ( 王亚培, 贾晗钰 . 化学通报, 2017,80(2), 123—131) | |
| [43] | He Y., Wang Y., Acta Polym . Sin., 2019,50(2), 102—108 |
| ( 贺泳霖, 王亚培 . 高分子学报, 2019,50(2), 102—108) |
| [1] | 崔伟, 赵德银, 白文轩, 张晓东, 余江. CO2在非质子溶剂与铁基离子液体复合体系中的吸收[J]. 高等学校化学学报, 2022, 43(8): 20220120. |
| [2] | 彭奎霖, 李桂林, 江重阳, 曾少娟, 张香平. 电解液调控CO2电催化还原性能微观机制的研究进展[J]. 高等学校化学学报, 2022, 43(7): 20220238. |
| [3] | 季双琦, 靳钊, 观文娜, 潘翔宇, 关彤. 双阳离子型离子液体和十八烷基修饰的混合模式硅胶固定相的制备及色谱性能[J]. 高等学校化学学报, 2022, 43(6): 20220008. |
| [4] | 常斯惠, 陈涛, 赵黎明, 邱勇隽. 离子液体增塑生物基聚丁内酰胺的热分解机理[J]. 高等学校化学学报, 2022, 43(11): 20220353. |
| [5] | 万仁, 宋璠, 彭昌军, 刘洪来. 水溶液中非常规离子无限稀释摩尔电导率的基团贡献法[J]. 高等学校化学学报, 2021, 42(12): 3672. |
| [6] | 王蔓, 王鑫, 周静, 高国华. 聚离子液体催化碳酸乙烯酯与甲醇的酯交换反应[J]. 高等学校化学学报, 2021, 42(12): 3701. |
| [7] | 周墨林, 蒋欣, 易婷, 杨向光, 张一波. 硫化物固态电解质Li10GeP2S12与锂金属间界面稳定性的改善研究[J]. 高等学校化学学报, 2020, 41(8): 1810. |
| [8] | 程时富,胡皓,陈必华,吴海虹,高国华,何鸣元. 双离子液体基多孔炭的制备与电化学性能[J]. 高等学校化学学报, 2020, 41(5): 1048. |
| [9] | 高崇,于凤丽,解从霞,于世涛. 氨基醇杂多酸类离子液体催化环酮的Baeyer-Villiger氧化反应[J]. 高等学校化学学报, 2020, 41(5): 1101. |
| [10] | 朴惠兰,马品一,覃祖成,姜延晓,孙颖,王兴华,宋大千. 基于酸性离子液体填充注射器的泡腾辅助微萃取法测定果汁样品中三嗪类除草剂[J]. 高等学校化学学报, 2020, 41(2): 228. |
| [11] | 张丽,钱明超,刘雪珂,高帅涛,余江,谢海深,王宏斌,孙风江,苏向红. 铁基离子液体/NHD吸收氧化H2S的反应动力学[J]. 高等学校化学学报, 2020, 41(2): 317. |
| [12] | 王楠,姚开胜,赵晨晨,李添锦,卢伟伟. 离子液体辅助合成AuPd纳米海绵及催化性能[J]. 高等学校化学学报, 2020, 41(1): 62. |
| [13] | 李晨光, 花儿, 刘天霞. 异辛基乙二胺-CF3SO3型质子化离子液体作为液体石蜡添加剂的摩擦学性能[J]. 高等学校化学学报, 2019, 40(7): 1411. |
| [14] | 刘小舟, 管新宇, 方千荣, 金永日. 室温离子液体法合成新型三维共价有机骨架材料[J]. 高等学校化学学报, 2019, 40(7): 1341. |
| [15] | 丁中协, 梁金花, 刘振, 沈节灿, 张峰, 任晓乾, 姜岷. 功能特异性杂多酸离子液体催化丁二酸水溶液直接酯化[J]. 高等学校化学学报, 2019, 40(5): 1029. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||