

高等学校化学学报 ›› 2020, Vol. 41 ›› Issue (4): 716.doi: 10.7503/cjcu20190504
李康明1,李延赛1,易阳杰1,徐雷涛1,叶姣1,欧晓明2,李建明2,胡艾希1,*( )
)
收稿日期:2019-09-27
出版日期:2020-04-10
发布日期:2020-02-07
通讯作者:
胡艾希
E-mail:axhu@hnu.edu.cn
基金资助:
LI Kangming1,LI Yansai1,YI Yangjie1,XU Leitao1,YE Jiao1,OU Xiaoming2,LI Jianming2,HU Aixi1,*( )
)
Received:2019-09-27
Online:2020-04-10
Published:2020-02-07
Contact:
Aixi HU
E-mail:axhu@hnu.edu.cn
Supported by:摘要:
以2种5-吡唑甲酸为原料, 分别与噻唑胺和苯并呋喃胺发生缩合反应, 制备了23个新的5-吡唑甲酰胺类化合物; 采用核磁共振波谱和质谱等对目标化合物结构进行了表征; 对化合物B1和B3进行了量子化学计算、 前沿分子轨道及分子范德华表面静电势分析. 生物活性实验结果表明, 在500 mg/L浓度下部分化合物对黏虫和蚜虫有良好的杀虫活性, 其中化合物A1和B1对黏虫的致死率均为100%, 化合物B3, B4和C9对蚜虫的致死率分别为100%, 100%和95.54%; 在25 mg/L浓度下化合物A3对烟草赤星病菌的抑制率、 化合物C7对小麦赤霉病菌的抑制率以及化合物A1和A4对油菜菌核病菌的抑制率均大于50%.
中图分类号:
TrendMD:
李康明, 李延赛, 易阳杰, 徐雷涛, 叶姣, 欧晓明, 李建明, 胡艾希. 5-吡唑甲酰胺类衍生物的设计、 合成与生物活性. 高等学校化学学报, 2020, 41(4): 716.
LI Kangming, LI Yansai, YI Yangjie, XU Leitao, YE Jiao, OU Xiaoming, LI Jianming, HU Aixi. Design, Synthesis and Biological Activity of 5-Pyrazole Carboxamides †. Chem. J. Chinese Universities, 2020, 41(4): 716.
| Compd. | Reaction time/h | Feature | m. p./℃ | Yield(%) | HRMS(calcd.), m/z* |
|---|---|---|---|---|---|
| A1 | 4.0 | Grey solid | 148—149 | 68.8 | 364.1012(364.1310) |
| A2 | 1.0 | White solid | 149—150 | 66.0 | 378.1209(378.1507) |
| A3 | 1.0 | White solid | 157—158 | 59.5 | 392.1405(392.1707) |
| A4 | 0.5 | Yellow solid | 138—139 | 61.4 | 440.0810(440.1714) |
| A5 | 0.5 | White solid | 173—174 | 32.7 | 522.1002(522.0701) |
| A6 | 8.0 | Yellow solid | 250—252 | 48.2 | 491.0907(491.0421) |
Table 1 Physical data and HRMS results of compounds A
| Compd. | Reaction time/h | Feature | m. p./℃ | Yield(%) | HRMS(calcd.), m/z* |
|---|---|---|---|---|---|
| A1 | 4.0 | Grey solid | 148—149 | 68.8 | 364.1012(364.1310) |
| A2 | 1.0 | White solid | 149—150 | 66.0 | 378.1209(378.1507) |
| A3 | 1.0 | White solid | 157—158 | 59.5 | 392.1405(392.1707) |
| A4 | 0.5 | Yellow solid | 138—139 | 61.4 | 440.0810(440.1714) |
| A5 | 0.5 | White solid | 173—174 | 32.7 | 522.1002(522.0701) |
| A6 | 8.0 | Yellow solid | 250—252 | 48.2 | 491.0907(491.0421) |
| Compd. | Reaction time/h | Feature | m. p./℃ | Yield(%) | HRMS(calcd.), m/z* |
|---|---|---|---|---|---|
| B1 | 4 | White solid | 177—179 | 28.1 | 447.0534(447.1229) |
| B2 | 10 | White solid | 185—187 | 30.9 | 461.0247(461.1349) |
| B3 | 10 | White solid | 135—138 | 31.6 | 560.0925(560.0139) |
| B4 | 10 | White solid | 218—220 | 52.4 | 574.0826(574.0229) |
Table 2 Physical data and HRMS results of compounds B
| Compd. | Reaction time/h | Feature | m. p./℃ | Yield(%) | HRMS(calcd.), m/z* |
|---|---|---|---|---|---|
| B1 | 4 | White solid | 177—179 | 28.1 | 447.0534(447.1229) |
| B2 | 10 | White solid | 185—187 | 30.9 | 461.0247(461.1349) |
| B3 | 10 | White solid | 135—138 | 31.6 | 560.0925(560.0139) |
| B4 | 10 | White solid | 218—220 | 52.4 | 574.0826(574.0229) |
| Compd. | Reaction time/h | Feature | m. p./℃ | Yield(%) | HRMS(calcd.), m/z* |
|---|---|---|---|---|---|
| C1 | 8 | White solid | 126—128 | 43.6 | 327.1037(327.0640) |
| C2 | 16 | White solid | 151—152 | 69.4 | 356.0103(356.0902) |
| C3 | 4 | White solid | 126—128 | 62.0 | 357.0416(357.0717) |
| C4 | 4 | Grey solid | 164—166 | 44.2 | 394.0312(394.1113) |
| C5 | 4 | Grey solid | 177—179 | 33.5 | 432.0509(432.9812) |
| C6 | 5 | White solid | 92—94 | 58.6 | 357.0615(357.0718) |
| C7 | 4 | Yellow solid | 166—168 | 35.6 | 447.0609(447.1213) |
| C8 | 10 | White solid | 174—176 | 42.6 | 461.0913(461.1319) |
| C9 | 4 | White solid | 220—222 | 57.3 | 470.0309(469.9612) |
| C10 | 6 | White solid | 222—224 | 52.1 | 507.0629(507.0031) |
| C11 | 4 | White solid | 147—150 | 40.7 | 470.0425(469.9617) |
| C12 | 10 | White solid | 158—160 | 50.2 | 560.0523(560.0121) |
| C13 | 10 | White solid | 131—133 | 63.4 | 574.0009(574.0213) |
Table 3 Physical data and HRMS results of compounds C
| Compd. | Reaction time/h | Feature | m. p./℃ | Yield(%) | HRMS(calcd.), m/z* |
|---|---|---|---|---|---|
| C1 | 8 | White solid | 126—128 | 43.6 | 327.1037(327.0640) |
| C2 | 16 | White solid | 151—152 | 69.4 | 356.0103(356.0902) |
| C3 | 4 | White solid | 126—128 | 62.0 | 357.0416(357.0717) |
| C4 | 4 | Grey solid | 164—166 | 44.2 | 394.0312(394.1113) |
| C5 | 4 | Grey solid | 177—179 | 33.5 | 432.0509(432.9812) |
| C6 | 5 | White solid | 92—94 | 58.6 | 357.0615(357.0718) |
| C7 | 4 | Yellow solid | 166—168 | 35.6 | 447.0609(447.1213) |
| C8 | 10 | White solid | 174—176 | 42.6 | 461.0913(461.1319) |
| C9 | 4 | White solid | 220—222 | 57.3 | 470.0309(469.9612) |
| C10 | 6 | White solid | 222—224 | 52.1 | 507.0629(507.0031) |
| C11 | 4 | White solid | 147—150 | 40.7 | 470.0425(469.9617) |
| C12 | 10 | White solid | 158—160 | 50.2 | 560.0523(560.0121) |
| C13 | 10 | White solid | 131—133 | 63.4 | 574.0009(574.0213) |
| Compd. | 1H NMR(400 MHz, CDCl3), δ | 13C NMR(100 MHz, CDCl3), δ |
|---|---|---|
| A1 | 8.28(s, 1H, NH), 7.05(s, 1H, benzofuran-H), 7.00(s, 1H, benzofuran-H), 4.16(s, 3H, NCH3), 3.89(s, 3H, OCH3), 3.04(s, 2H, CH2), 2.67(q, J=7.6 Hz, 2H, CH2), 1.51(s, 6H, 2CH3), 1.26(t, J=7.6 Hz, 3H, CH3) | 156.42, 149.64, 144.90, 144.30, 131.29, 129.89, 128.13, 110.45, 107.36, 104.92, 87.94, 56.02, 43.41, 40.78, 28.19, 19.29, 12.82 |
| A2 | 8.25(s, 1H, NH), 7.02(s, 2H, benzofuran-H), 4.14(d, J=6.8 Hz, 2H, CH2), 4.16(s, 3H, CH3), 3.03(s, 2H, CH2), 2.67(q, J=7.5 Hz, 2H, CH2), 1.51(s, 6H, 2CH3), 1.44(t, J=7.0 Hz, 3H, CH3), 1.27(t, J=7.5 Hz, 3H, CH3) | 156.38, 149.62, 145.28, 143.50, 131.33, 129.78, 128.42, 110.45, 107.33, 106.51, 87.74, 64.66, 43.47, 40.77, 28.18, 19.30, 14.85, 12.83 |
| A3 | 8.25(s, 1H, NH), 7.01(s, 2H, benzofuran-H), 4.17(s, 3H, NCH3), 4.03(t, J=6.9 Hz, 2H, OCH2), 3.02(s, 2H, CH2), 2.67(q, J=7.6 Hz, 2H, CH2), 1.90—1.80(m, 2H, CH2), 1.51(s, 6H, 2 CH3), 1.27(t, J=7.6 Hz, 3H, CH3), 1.02(t, J=7.4 Hz, 3H, CH3) | |
| Compd. | 1H NMR(400 MHz, CDCl3), δ | 13C NMR(100 MHz, CDCl3), δ |
| A4 | 8.19(s, 1H, NH), 7.45(d, J=7.5 Hz, 2H, Ph-H), 7.38—7.27(m, 3H, Ph-H), 7.08(s, 1H, benzofuran-H), 6.99(s, 1H, benzofuran-H), 5.19(s, 2H, OCH2), 4.15(s, 3H, NCH3), 3.03(s, 2H, CH2), 2.66(q, J=7.6 Hz, 2H, CH2), 1.52(s, 6H, 2CH3), 1.26(t, J=7.6 Hz, 3H, CH3) | 156.45, 149.72, 145.69, 143.24, 137.13, 131.42, 129.88, 128.92, 128.59, 128.01, 127.73, 111.25, 107.99, 107.45, 87.99, 71.43, 43.49, 40.84, 28.29, 19.40, 12.95 |
| A5 | 8.32(s, 1H, NH), 8.07(d, J=8.4 Hz, 1H, C6H3), 7.54(s, 1H, benzofuran-H), 7.37(d, J=8.4 Hz, 2H, C6H3), 7.26(s, 1H, benzofuran-H), 4.16(s, 3H, NCH3), 3.09(s, 2H, CH2), 2.67(q, J=7.5 Hz, 2H, CH2), 1.49(s, 6H, 2CH3), 1.26(t, J=7.5 Hz, 3H, CH3) | 162.03, 156.55, 149.80, 147.93, 136.04, 133.67, 133.47, 131.39, 130.52, 129.67, 127.24, 116.42, 114.81, 107.62, 89.30, 43.36, 40.97, 28.18, 19.42, 12.96 |
| A6 | 8.47—7.39(m, 3H, pyridine-H), 6.91(s, 1H, benzofuran-H), 6.83(s, 1H, pyrazole-H), 6.78(s, 1H, benzofuran-H), 4.01(q, J=7.0 Hz, 2H, CH2), 2.94(s, 2H, CH2), 1.47(s, 6H, 2CH3), 1.37(t, J=7.0 Hz, 3H, CH3) | 155.50, 148.85, 146.66, 145.22, 143.34, 139.99, 139.52, 129.40, 129.20, 128.27, 127.98, 125.80, 110.66, 110.14, 106.42, 87.80, 64.49, 43.38, 28.15, 14.79 |
| B1 | 10.11(s, 1H, NH), 7.27—7.03(m, 3H, benzofuran-H+thiazole-H), 4.21(s, 3H, NCH3), 3.95(s, 3H, OCH3), 3.07(s, 2H, CH2), 2.68(q, J=7.6 Hz, 2H, CH2), 1.54(s, 6H, 2CH3), 1.27(t, J=7.5 Hz, 3H, CH3) | 156.08, 155.58, 150.89, 150.07, 147.58, 144.54, 129.15, 128.41, 127.39, 115.47, 109.30, 108.94, 106.20, 88.18, 55.95, 43.26, 41.14, 28.25, 19.22, 12.71 |
| B2 | 10.38(s, 1H, NH), 7.30(s, 1H, benzofuran-H), 7.28(s, 1H, benzofuran-H), 7.02(s, 1H, thiazole-H), 4.22(t, 2H, J=7.2 Hz, CH2), 4.21(s, 3H, CH3), 3.06(s, 2H, CH2), 2.69(q, J=7.6 Hz, 2H, CH2), 1.54(s, 6H, 2CH3), 1.47(t, J=7.0 Hz, 3H, CH3), 1.28(t, J=7.6 Hz, 3H, CH3) | 156.10, 155.62, 154.05, 150.97, 150.12, 148.07, 143.79, 129.23, 128.74, 127.29, 115.55, 111.16, 106.14, 88.02, 64.59, 43.36, 41.17, 28.28, 19.21, 14.95, 12.69 |
| B3 | 8.54—7.44(m, 3H, pyridine-H), 6.98(s, 1H, pyrazole-H), 7.19—6.86(m, 3H, benzofuran-H+thiazole-H), 3.90(s, 3H, CH3), 3.06(s, 2H, CH2), 1.52(s, 6H, 2CH3) | 157.53, 154.97, 150.65, 148.37, 147.80, 146.87, 144.58, 139.56, 137.26, 128.86, 128.51, 128.05, 126.92, 125.89, 115.56, 111.40, 109.32, 106.60, 88.27, 55.83, 43.17, 28.23 |
| B4 | 10.83(s, 1H, NH), 8.55—7.43(m, 3H, pyridine-H), 6.97(s, 1H, pyrazole-H), 7.18—6.76(m, 3H, benzofuran-H+ thiazole-H), 4.14(q, J=7.0 Hz, 2H, CH2), 3.04(s, 2H, CH2), 1.50(s, 6H, 2CH3), 1.43(t, J=7.0 Hz, 3H, CH3) | 157.61, 155.02, 150.71, 148.41, 148.16, 146.90, 143.80, 139.54, 137.24, 128.87, 128.77, 128.02, 126.87, 125.88, 115.55, 111.38, 111.06, 106.54, 88.08, 64.45, 43.22, 28.22, 14.92 |
| C1 | 10.00(s, 1H, NH), 4.22(s, 3H, NCH3), 2.69(t, 2H, J=7.6 Hz, CH2), 2.67(s, 3H, CH3), 2.54(s, 3H, COCH3), 1.27(t, J=7.6 Hz, 3H, CH3) | 190.46, 157.35, 155.72, 155.51, 150.25, 128.64, 126.17, 109.39, 41.26, 30.43, 19.17, 18.09, 12.65 |
| C2 | 4.21(s, 3H, NCH3), 3.96—3.97[2s, (2.25+0.75)H, OCH3], 2.69(t, 2H, J=7.6 Hz, CH2), 2.46—2.51[2s, (0.75+2.25)H, CH3], 2.25, 2.31[2s, (2.25+0.75) H, CH3], 1.27(t, J=7.6 Hz, 3H, CH3) | |
| C3 | 10.06(s, 1H, NH), 4.32(q, J=6.9 Hz, 2H, OCH2), 4.20(s, 3H, NCH3), 2.69—2.65(m, 5H, CH3+CH2), 1.36(t, J=7.0 Hz, 3H, CH3), 1.26(t, J=7.4 Hz, 3H, CH3) | 162.52, 157.92, 156.72, 155.70, 150.22, 128.72, 116.74, 109.31, 60.96, 41.25, 19.18, 17.10, 14.34, 12.67 |
| C4 | 9.88(s, 1H, NH), 8.27(s, 1H, 1,2,4-triazole-H), 8.11(s, 1H, 1,2,4-triazole-H), 4.18(s, 3H, NCH3), 2.69(q, J=7.5 Hz, 2H, CH2), 1.27(t, J=7.6 Hz, 3H, CH3), 1.16(s, 9H, 3CH3) | 156.73, 155.70, 152.53, 152.48, 150.22, 147.36, 128.68, 119.78, 109.18, 41.19, 35.95, 29.58, 19.20, 12.70 |
| C5 | 10.16(s, 1H, NH), 7.82(d, 1H, C6H2), 7.50(d, 1H, C6H2), 7.26(s, 1H, thiazole-H), 4.20(s, 3H, NCH3), 2.68(q, J=7.6 Hz, 2H, CH2), 1.27(t, J=7.6 Hz, 3H, CH3) | 158.02, 155.64(d), 155.55, 150.17, 144.75, 132.77(d), 131.91, 128.90, 126.87(d), 120.87(d), 12.67, 19.21, 41.15, 109.14, 114.53, 118.55(d) |
| C6 | 9.98(s, 1H, NH), 6.89(s, 1H, thiazole-H), 4.20(s, 5H, COCH2+NCH3), 3.73—3.70(m, 2H, CH2), 2.71—2.61(m, 2H, CH2), 1.32—1.22(m, 6H, CH3+CH3) | 170.17, 156.39, 155.51, 150.10, 144.01, 129.08, 111.54, 109.03, 61.13, 41.12, 37.01, 19.1, 14.18, 12.68 |
| Compd. | 1H NMR(400 MHz, CDCl3), δ | 13C NMR(100 MHz, CDCl3), δ |
| C7 | 10.06(s, 1H, NH), 7.89(d, J=8.5 Hz, 1H, benzofuran-H), 7.57(s, 1H, thiazole-H), 6.48(d, J=8.5 Hz, 1H, benzofuran-H), 4.22(s, 3H, NCH3), 3.86(s, 3H, OCH3), 3.00(s, 2H, CH2), 2.68(q, J=7.3 Hz, 2H, CH2), 1.55(s, 6H, 2CH3), 1.27(t, J=7.4 Hz, 3H, CH3) | 157.30, 156.15, 155.52, 155.15, 150.04, 146.24, 129.31, 127.67, 114.18, 111.09, 109.72, 108.87, 102.80, 88.27, 55.41, 41.14, 40.08, 28.67, 19.23, 12.73 |
| C8 | 10.02(s, 1H, NH), 7.88(d, J=8.6 Hz, 1H, benzofuran-H), 7.57(s, 1H, thiazole-H), 6.47(d, J=8.7 Hz, 1H, benzofuran-H), 4.22(s, 3H, CH3), 4.09(q, J=7.0 Hz, 2H, CH2), 3.01(s, 2H, CH3), 2.69(q, J=7.6 Hz, 2H, CH2), 1.55(s, 6H, 2CH3), 1.41(t, J=7.0 Hz, 3H, CH3), 1.28(t, J=7.6 Hz, 3H, CH3) | 157.33, 155.59, 155.56, 155.20, 150.04, 146.31, 129.38, 127.57, 114.4, 110.86, 109.61, 108.88, 103.77, 88.19, 63.59, 41.10, 40.19, 28.69, 19.23, 14.96, 12.72 |
| C9 | 8.58—7.48(m, 3H, pyridine-H), 6.95(s, 1H, pyrazole-H), 4.27(q, J=7.0 Hz, 2H, CH2), 2.61(s, 3H, CH3), 1.31(t, J=7.1 Hz, 3H, CH3) | 162.25, 158.79, 155.90, 155.24, 148.22, 147.00, 140.00, 137.25, 128.98, 128.35, 126.08, 116.73, 111.84, 61.02, 16.92, 14.27 |
| C10 | 8.47(s, 1H, 1,2,4-triazole-H), 8.09(s, 1H, 1,2,4-triazole-H), 8.77—7.52(m, 3H, pyridine-H), 6.86(s, 1H, pyrazole-H), 1.10(s, 9H, 3CH3) | |
| C11 | 8.54—7.45(m, 3H, pyridine-H), 6.91(s, 1H, pyrazole-H), 6.82(s, 1H, thiazole-H), 4.18(q, J=7.1 Hz, 2H, CH2), 3.67(s, 2H, CH2), 1.26(t, J=7.0 Hz, 3H, CH3) | 170.21, 157.07, 154.83, 148.35, 146.99, 143.50, 139.67, 137.67, 128.91, 128.23, 126.02, 111.72, 111.44, 61.25, 36.90, 14.15 |
| C12 | 7.69(d, J=8.6 Hz, 1H, benzofuran), 8.55—7.43(m, 3H, pyridine-H), 7.40(s, 1H, thiazole-H), 6.92(s, 1H, pyrazole-H), 6.47(d, J=8.7 Hz, 1H, benzofuran-H), 3.84(s, 3H, OCH3), 2.98(s, 2H, CH2), 1.54(s, 6H, 2CH3) | 157.71, 157.13, 156.37, 155.90, 148.62, 146.87, 144.74, 139.49, 138.17, 129.00, 128.08, 127.23, 125.84, 114.38, 111.49, 110.02, 109.05, 103.08, 88.55, 55.42, 40.02, 28.64 |
| C13 | 10.74(s, 1H, NH), 7.65(d, J=8.6 Hz, 1H, benzofuran-H), 8.55—7.43(m, 3H, pyridine-H), 7.39(s, 1H, thiazole-H), 6.85(s, 1H, pyrazole-H), 6.43(d, J=8.7 Hz, 1H, benzofuran-H), 4.07(q, J=7.0 Hz, 2H, CH2), 2.99(s, 2H, CH2), 1.54(s, 6H, 2CH3), 1.40(t, J=7.0 Hz, 3H, CH3) | 157.85, 157.10, 156.09, 155.85, 148.65, 146.85, 146.48, 139.49, 138.45, 129.03, 128.12, 127.10, 125.83, 114.62, 111.51, 109.66, 108.69, 104.05, 88.54, 63.63, 40.12, 28.66, 14.93 |
Table 4 1H NMR and 13C NMR data of compounds A—C
| Compd. | 1H NMR(400 MHz, CDCl3), δ | 13C NMR(100 MHz, CDCl3), δ |
|---|---|---|
| A1 | 8.28(s, 1H, NH), 7.05(s, 1H, benzofuran-H), 7.00(s, 1H, benzofuran-H), 4.16(s, 3H, NCH3), 3.89(s, 3H, OCH3), 3.04(s, 2H, CH2), 2.67(q, J=7.6 Hz, 2H, CH2), 1.51(s, 6H, 2CH3), 1.26(t, J=7.6 Hz, 3H, CH3) | 156.42, 149.64, 144.90, 144.30, 131.29, 129.89, 128.13, 110.45, 107.36, 104.92, 87.94, 56.02, 43.41, 40.78, 28.19, 19.29, 12.82 |
| A2 | 8.25(s, 1H, NH), 7.02(s, 2H, benzofuran-H), 4.14(d, J=6.8 Hz, 2H, CH2), 4.16(s, 3H, CH3), 3.03(s, 2H, CH2), 2.67(q, J=7.5 Hz, 2H, CH2), 1.51(s, 6H, 2CH3), 1.44(t, J=7.0 Hz, 3H, CH3), 1.27(t, J=7.5 Hz, 3H, CH3) | 156.38, 149.62, 145.28, 143.50, 131.33, 129.78, 128.42, 110.45, 107.33, 106.51, 87.74, 64.66, 43.47, 40.77, 28.18, 19.30, 14.85, 12.83 |
| A3 | 8.25(s, 1H, NH), 7.01(s, 2H, benzofuran-H), 4.17(s, 3H, NCH3), 4.03(t, J=6.9 Hz, 2H, OCH2), 3.02(s, 2H, CH2), 2.67(q, J=7.6 Hz, 2H, CH2), 1.90—1.80(m, 2H, CH2), 1.51(s, 6H, 2 CH3), 1.27(t, J=7.6 Hz, 3H, CH3), 1.02(t, J=7.4 Hz, 3H, CH3) | |
| Compd. | 1H NMR(400 MHz, CDCl3), δ | 13C NMR(100 MHz, CDCl3), δ |
| A4 | 8.19(s, 1H, NH), 7.45(d, J=7.5 Hz, 2H, Ph-H), 7.38—7.27(m, 3H, Ph-H), 7.08(s, 1H, benzofuran-H), 6.99(s, 1H, benzofuran-H), 5.19(s, 2H, OCH2), 4.15(s, 3H, NCH3), 3.03(s, 2H, CH2), 2.66(q, J=7.6 Hz, 2H, CH2), 1.52(s, 6H, 2CH3), 1.26(t, J=7.6 Hz, 3H, CH3) | 156.45, 149.72, 145.69, 143.24, 137.13, 131.42, 129.88, 128.92, 128.59, 128.01, 127.73, 111.25, 107.99, 107.45, 87.99, 71.43, 43.49, 40.84, 28.29, 19.40, 12.95 |
| A5 | 8.32(s, 1H, NH), 8.07(d, J=8.4 Hz, 1H, C6H3), 7.54(s, 1H, benzofuran-H), 7.37(d, J=8.4 Hz, 2H, C6H3), 7.26(s, 1H, benzofuran-H), 4.16(s, 3H, NCH3), 3.09(s, 2H, CH2), 2.67(q, J=7.5 Hz, 2H, CH2), 1.49(s, 6H, 2CH3), 1.26(t, J=7.5 Hz, 3H, CH3) | 162.03, 156.55, 149.80, 147.93, 136.04, 133.67, 133.47, 131.39, 130.52, 129.67, 127.24, 116.42, 114.81, 107.62, 89.30, 43.36, 40.97, 28.18, 19.42, 12.96 |
| A6 | 8.47—7.39(m, 3H, pyridine-H), 6.91(s, 1H, benzofuran-H), 6.83(s, 1H, pyrazole-H), 6.78(s, 1H, benzofuran-H), 4.01(q, J=7.0 Hz, 2H, CH2), 2.94(s, 2H, CH2), 1.47(s, 6H, 2CH3), 1.37(t, J=7.0 Hz, 3H, CH3) | 155.50, 148.85, 146.66, 145.22, 143.34, 139.99, 139.52, 129.40, 129.20, 128.27, 127.98, 125.80, 110.66, 110.14, 106.42, 87.80, 64.49, 43.38, 28.15, 14.79 |
| B1 | 10.11(s, 1H, NH), 7.27—7.03(m, 3H, benzofuran-H+thiazole-H), 4.21(s, 3H, NCH3), 3.95(s, 3H, OCH3), 3.07(s, 2H, CH2), 2.68(q, J=7.6 Hz, 2H, CH2), 1.54(s, 6H, 2CH3), 1.27(t, J=7.5 Hz, 3H, CH3) | 156.08, 155.58, 150.89, 150.07, 147.58, 144.54, 129.15, 128.41, 127.39, 115.47, 109.30, 108.94, 106.20, 88.18, 55.95, 43.26, 41.14, 28.25, 19.22, 12.71 |
| B2 | 10.38(s, 1H, NH), 7.30(s, 1H, benzofuran-H), 7.28(s, 1H, benzofuran-H), 7.02(s, 1H, thiazole-H), 4.22(t, 2H, J=7.2 Hz, CH2), 4.21(s, 3H, CH3), 3.06(s, 2H, CH2), 2.69(q, J=7.6 Hz, 2H, CH2), 1.54(s, 6H, 2CH3), 1.47(t, J=7.0 Hz, 3H, CH3), 1.28(t, J=7.6 Hz, 3H, CH3) | 156.10, 155.62, 154.05, 150.97, 150.12, 148.07, 143.79, 129.23, 128.74, 127.29, 115.55, 111.16, 106.14, 88.02, 64.59, 43.36, 41.17, 28.28, 19.21, 14.95, 12.69 |
| B3 | 8.54—7.44(m, 3H, pyridine-H), 6.98(s, 1H, pyrazole-H), 7.19—6.86(m, 3H, benzofuran-H+thiazole-H), 3.90(s, 3H, CH3), 3.06(s, 2H, CH2), 1.52(s, 6H, 2CH3) | 157.53, 154.97, 150.65, 148.37, 147.80, 146.87, 144.58, 139.56, 137.26, 128.86, 128.51, 128.05, 126.92, 125.89, 115.56, 111.40, 109.32, 106.60, 88.27, 55.83, 43.17, 28.23 |
| B4 | 10.83(s, 1H, NH), 8.55—7.43(m, 3H, pyridine-H), 6.97(s, 1H, pyrazole-H), 7.18—6.76(m, 3H, benzofuran-H+ thiazole-H), 4.14(q, J=7.0 Hz, 2H, CH2), 3.04(s, 2H, CH2), 1.50(s, 6H, 2CH3), 1.43(t, J=7.0 Hz, 3H, CH3) | 157.61, 155.02, 150.71, 148.41, 148.16, 146.90, 143.80, 139.54, 137.24, 128.87, 128.77, 128.02, 126.87, 125.88, 115.55, 111.38, 111.06, 106.54, 88.08, 64.45, 43.22, 28.22, 14.92 |
| C1 | 10.00(s, 1H, NH), 4.22(s, 3H, NCH3), 2.69(t, 2H, J=7.6 Hz, CH2), 2.67(s, 3H, CH3), 2.54(s, 3H, COCH3), 1.27(t, J=7.6 Hz, 3H, CH3) | 190.46, 157.35, 155.72, 155.51, 150.25, 128.64, 126.17, 109.39, 41.26, 30.43, 19.17, 18.09, 12.65 |
| C2 | 4.21(s, 3H, NCH3), 3.96—3.97[2s, (2.25+0.75)H, OCH3], 2.69(t, 2H, J=7.6 Hz, CH2), 2.46—2.51[2s, (0.75+2.25)H, CH3], 2.25, 2.31[2s, (2.25+0.75) H, CH3], 1.27(t, J=7.6 Hz, 3H, CH3) | |
| C3 | 10.06(s, 1H, NH), 4.32(q, J=6.9 Hz, 2H, OCH2), 4.20(s, 3H, NCH3), 2.69—2.65(m, 5H, CH3+CH2), 1.36(t, J=7.0 Hz, 3H, CH3), 1.26(t, J=7.4 Hz, 3H, CH3) | 162.52, 157.92, 156.72, 155.70, 150.22, 128.72, 116.74, 109.31, 60.96, 41.25, 19.18, 17.10, 14.34, 12.67 |
| C4 | 9.88(s, 1H, NH), 8.27(s, 1H, 1,2,4-triazole-H), 8.11(s, 1H, 1,2,4-triazole-H), 4.18(s, 3H, NCH3), 2.69(q, J=7.5 Hz, 2H, CH2), 1.27(t, J=7.6 Hz, 3H, CH3), 1.16(s, 9H, 3CH3) | 156.73, 155.70, 152.53, 152.48, 150.22, 147.36, 128.68, 119.78, 109.18, 41.19, 35.95, 29.58, 19.20, 12.70 |
| C5 | 10.16(s, 1H, NH), 7.82(d, 1H, C6H2), 7.50(d, 1H, C6H2), 7.26(s, 1H, thiazole-H), 4.20(s, 3H, NCH3), 2.68(q, J=7.6 Hz, 2H, CH2), 1.27(t, J=7.6 Hz, 3H, CH3) | 158.02, 155.64(d), 155.55, 150.17, 144.75, 132.77(d), 131.91, 128.90, 126.87(d), 120.87(d), 12.67, 19.21, 41.15, 109.14, 114.53, 118.55(d) |
| C6 | 9.98(s, 1H, NH), 6.89(s, 1H, thiazole-H), 4.20(s, 5H, COCH2+NCH3), 3.73—3.70(m, 2H, CH2), 2.71—2.61(m, 2H, CH2), 1.32—1.22(m, 6H, CH3+CH3) | 170.17, 156.39, 155.51, 150.10, 144.01, 129.08, 111.54, 109.03, 61.13, 41.12, 37.01, 19.1, 14.18, 12.68 |
| Compd. | 1H NMR(400 MHz, CDCl3), δ | 13C NMR(100 MHz, CDCl3), δ |
| C7 | 10.06(s, 1H, NH), 7.89(d, J=8.5 Hz, 1H, benzofuran-H), 7.57(s, 1H, thiazole-H), 6.48(d, J=8.5 Hz, 1H, benzofuran-H), 4.22(s, 3H, NCH3), 3.86(s, 3H, OCH3), 3.00(s, 2H, CH2), 2.68(q, J=7.3 Hz, 2H, CH2), 1.55(s, 6H, 2CH3), 1.27(t, J=7.4 Hz, 3H, CH3) | 157.30, 156.15, 155.52, 155.15, 150.04, 146.24, 129.31, 127.67, 114.18, 111.09, 109.72, 108.87, 102.80, 88.27, 55.41, 41.14, 40.08, 28.67, 19.23, 12.73 |
| C8 | 10.02(s, 1H, NH), 7.88(d, J=8.6 Hz, 1H, benzofuran-H), 7.57(s, 1H, thiazole-H), 6.47(d, J=8.7 Hz, 1H, benzofuran-H), 4.22(s, 3H, CH3), 4.09(q, J=7.0 Hz, 2H, CH2), 3.01(s, 2H, CH3), 2.69(q, J=7.6 Hz, 2H, CH2), 1.55(s, 6H, 2CH3), 1.41(t, J=7.0 Hz, 3H, CH3), 1.28(t, J=7.6 Hz, 3H, CH3) | 157.33, 155.59, 155.56, 155.20, 150.04, 146.31, 129.38, 127.57, 114.4, 110.86, 109.61, 108.88, 103.77, 88.19, 63.59, 41.10, 40.19, 28.69, 19.23, 14.96, 12.72 |
| C9 | 8.58—7.48(m, 3H, pyridine-H), 6.95(s, 1H, pyrazole-H), 4.27(q, J=7.0 Hz, 2H, CH2), 2.61(s, 3H, CH3), 1.31(t, J=7.1 Hz, 3H, CH3) | 162.25, 158.79, 155.90, 155.24, 148.22, 147.00, 140.00, 137.25, 128.98, 128.35, 126.08, 116.73, 111.84, 61.02, 16.92, 14.27 |
| C10 | 8.47(s, 1H, 1,2,4-triazole-H), 8.09(s, 1H, 1,2,4-triazole-H), 8.77—7.52(m, 3H, pyridine-H), 6.86(s, 1H, pyrazole-H), 1.10(s, 9H, 3CH3) | |
| C11 | 8.54—7.45(m, 3H, pyridine-H), 6.91(s, 1H, pyrazole-H), 6.82(s, 1H, thiazole-H), 4.18(q, J=7.1 Hz, 2H, CH2), 3.67(s, 2H, CH2), 1.26(t, J=7.0 Hz, 3H, CH3) | 170.21, 157.07, 154.83, 148.35, 146.99, 143.50, 139.67, 137.67, 128.91, 128.23, 126.02, 111.72, 111.44, 61.25, 36.90, 14.15 |
| C12 | 7.69(d, J=8.6 Hz, 1H, benzofuran), 8.55—7.43(m, 3H, pyridine-H), 7.40(s, 1H, thiazole-H), 6.92(s, 1H, pyrazole-H), 6.47(d, J=8.7 Hz, 1H, benzofuran-H), 3.84(s, 3H, OCH3), 2.98(s, 2H, CH2), 1.54(s, 6H, 2CH3) | 157.71, 157.13, 156.37, 155.90, 148.62, 146.87, 144.74, 139.49, 138.17, 129.00, 128.08, 127.23, 125.84, 114.38, 111.49, 110.02, 109.05, 103.08, 88.55, 55.42, 40.02, 28.64 |
| C13 | 10.74(s, 1H, NH), 7.65(d, J=8.6 Hz, 1H, benzofuran-H), 8.55—7.43(m, 3H, pyridine-H), 7.39(s, 1H, thiazole-H), 6.85(s, 1H, pyrazole-H), 6.43(d, J=8.7 Hz, 1H, benzofuran-H), 4.07(q, J=7.0 Hz, 2H, CH2), 2.99(s, 2H, CH2), 1.54(s, 6H, 2CH3), 1.40(t, J=7.0 Hz, 3H, CH3) | 157.85, 157.10, 156.09, 155.85, 148.65, 146.85, 146.48, 139.49, 138.45, 129.03, 128.12, 127.10, 125.83, 114.62, 111.51, 109.66, 108.69, 104.05, 88.54, 63.63, 40.12, 28.66, 14.93 |
| Compd. | Inhibition rate(%) | Mortality rate(%) | |||
|---|---|---|---|---|---|
| Aa | Gz | Ss | Ms | Af | |
| A1 | 45.00 | 40.00 | 56.30 | 100 | 50.68 |
| A2 | 30.00 | 14.30 | 45.80 | 0 | 22.93 |
| A3 | 50.00 | 17.10 | 33.30 | 0 | 49.69 |
| A4 | 30.00 | 11.40 | 54.20 | 0 | 19.64 |
| A5 | 45.00 | 22.90 | 31.30 | 0 | 58.91 |
| A6 | 0 | 0 | | 0 | 54.16 |
| B1 | 6.82 | 40.58 | 12.98 | 100 | 18.21 |
| B2 | 0.00 | 5.80 | | 0 | 62.32 |
| B3 | 7.50 | 23.19 | | 0 | 100 |
| B4 | 0.00 | 0 | | 0 | 100 |
| C1 | 6.82 | 14.49 | 9.87 | 0 | 41.85 |
| C2 | | | | | |
| C3 | 22.50 | 28.99 | | 0 | 18.27 |
| C4 | 13.64 | 40.58 | 42.36 | 4.55 | 12.66 |
| C5 | 0.00 | 47.83 | 25.41 | 5.56 | 19.93 |
| C6 | 15.91 | 33.33 | 42.81 | 0 | 36.09 |
| C7 | 0.00 | 53.62 | 37.68 | 3.85 | 16.90 |
| C8 | 0.00 | 8.70 | | 0 | 47.00 |
| C9 | 0.00 | 11.59 | | 0 | 95.54 |
| C10 | 5.00 | 10.14 | | 0 | 67.21 |
| C11 | 7.50 | 0 | | 0 | 56.04 |
| C12 | | | | | |
| C13 | | | | | |
| Boscalid | | 37.00 | 100 | | |
| Azoxystrobin | | | 93.50 | | |
| Dimethachlon | 100 | | | | |
| Chlorantraniliprole | | | | 100 | |
| Imidacloprid | | | | | 100 |
Table 5 Bactericidal and insecticidal activities of compounds A—C*
| Compd. | Inhibition rate(%) | Mortality rate(%) | |||
|---|---|---|---|---|---|
| Aa | Gz | Ss | Ms | Af | |
| A1 | 45.00 | 40.00 | 56.30 | 100 | 50.68 |
| A2 | 30.00 | 14.30 | 45.80 | 0 | 22.93 |
| A3 | 50.00 | 17.10 | 33.30 | 0 | 49.69 |
| A4 | 30.00 | 11.40 | 54.20 | 0 | 19.64 |
| A5 | 45.00 | 22.90 | 31.30 | 0 | 58.91 |
| A6 | 0 | 0 | | 0 | 54.16 |
| B1 | 6.82 | 40.58 | 12.98 | 100 | 18.21 |
| B2 | 0.00 | 5.80 | | 0 | 62.32 |
| B3 | 7.50 | 23.19 | | 0 | 100 |
| B4 | 0.00 | 0 | | 0 | 100 |
| C1 | 6.82 | 14.49 | 9.87 | 0 | 41.85 |
| C2 | | | | | |
| C3 | 22.50 | 28.99 | | 0 | 18.27 |
| C4 | 13.64 | 40.58 | 42.36 | 4.55 | 12.66 |
| C5 | 0.00 | 47.83 | 25.41 | 5.56 | 19.93 |
| C6 | 15.91 | 33.33 | 42.81 | 0 | 36.09 |
| C7 | 0.00 | 53.62 | 37.68 | 3.85 | 16.90 |
| C8 | 0.00 | 8.70 | | 0 | 47.00 |
| C9 | 0.00 | 11.59 | | 0 | 95.54 |
| C10 | 5.00 | 10.14 | | 0 | 67.21 |
| C11 | 7.50 | 0 | | 0 | 56.04 |
| C12 | | | | | |
| C13 | | | | | |
| Boscalid | | 37.00 | 100 | | |
| Azoxystrobin | | | 93.50 | | |
| Dimethachlon | 100 | | | | |
| Chlorantraniliprole | | | | 100 | |
| Imidacloprid | | | | | 100 |
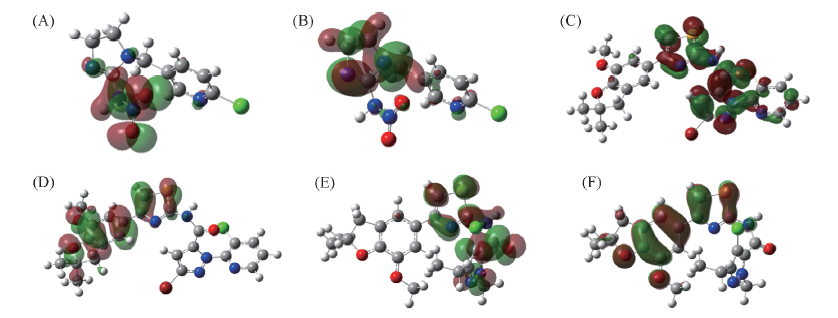
Fig.1 LUMO(A, C, E) and LUMO(B, D, F) maps for imidacloprid(A, B), B3(C, D) and B1(E, F) The green parts represent positive molecular orbital, and the red parts represent negative molecular orbital.
| DFT | Δ | ΔELUMO/Hartree | ΔEHOMO/Hartree | ΔEb/Hartree | Mortality rate(%) |
|---|---|---|---|---|---|
| Imidacloprid | -4613.41817 | -0.06741 | -0.23952 | 0.17211 | 100 |
| B1 | -2117.50553782 | -0.06094 | -0.19678 | 0.13584 | 18.21 |
| B4 | -4859.53075761 | -0.06146 | -0.20170 | 0.14024 | 100 |
| B3 | -4820.20321025 | -0.06211 | -0.20260 | 0.14049 | 100 |
Table 6 Frontier molecular orbital energies and acaricidal activities of compounds B1, B3, B4 and imidacloprid
| DFT | Δ | ΔELUMO/Hartree | ΔEHOMO/Hartree | ΔEb/Hartree | Mortality rate(%) |
|---|---|---|---|---|---|
| Imidacloprid | -4613.41817 | -0.06741 | -0.23952 | 0.17211 | 100 |
| B1 | -2117.50553782 | -0.06094 | -0.19678 | 0.13584 | 18.21 |
| B4 | -4859.53075761 | -0.06146 | -0.20170 | 0.14024 | 100 |
| B3 | -4820.20321025 | -0.06211 | -0.20260 | 0.14049 | 100 |
| [1] | Liu T., Tian H K.,. TongY. L., Yang C. L.,Ni Y. P Modern Agrochemicals, 2009,8(6), 5— 13 |
| ( 刘婷,田辉凯,童益利,杨春龙,倪珏萍.现代农药, 2009, 8(6), 5— 13) | |
| [2] | Kees K. L., Caggiano T., Steiner K., Fitzgerald J. J., Kates M. J., Christos T. E., Kulishoff J. M., Moore R. D., Mccaleb M. L., J. Med. Chem., 1995, 38( 4), 617— 628 |
| [3] | Zhou Q., Zhen D. D., Shi Y. J., Yao W., Qian H. W., Ding Y., Wei Z. H., Shen A. B., Feng X., Shi J., Dai H., Chin. J. Org. Chem., 2018, 38, 3318— 3325 |
| ( 周钱, 郑丹丹, 石玉军, 姚炜, 钱宏炜, 丁颖, 魏中昊, 沈爱宝, 冯霞, 石健, 戴红 . 有机化学, 2018, 38, 3318— 3325) | |
| [4] | Suresh L., Sagar V. K. P., Poornachandra Y., Ganesh K. C., Chandramouli G., Bioorg. Med. Chem. Lett., 2017, 27( 6), 1451— 1457 |
| [5] | Ma H. J., Li Y. H., Zhao Q. F., Zhang T., Xie R. L., Mei X. D., Ning J., J. Agric. Food Chem., 2010, 58, 4356— 4360 |
| [6] | Zhang Y. B., World Pesticides, ,2003, 25(6) 43— 45 |
| ( 张一宾.世界农药, 2003, 25(6), 43— 45) | |
| [7] | Bai Y. L., Agrochemicals Today, 2018, 10(11), 36— 40 |
| ( 柏亚罗.今日农药, 2018, 10(11), 36— 40) | |
| [8] | Yang G. Q., Huang Q., Chen L., Yang H. B., Yu H. B., Li B., World Pesticides, 2012, 34 6), 19— 21 |
| ( 杨桂秋,黄琦,陈霖,杨辉斌,于海波,李斌.世界农药, 2012, 34(6), 19— 21) | |
| [9] | Nagahori H., Yoshino H., Tomigahara Y., Isobe N., Kaneko H., Nakatsuka I., J. Agric. Food. Chem., 2000, 48( 11), 5760— 5770 |
| [10] | Zhang Q. K., World Pesticides, 2009, 31 3), 53— 53 |
| ( 张庆宽.世界农药, 2009, 31(3), 53— 53) | |
| [11] | Liu A. C., Yu C. H., Zhang S. K., Feng J. L., He X. L., Modern Agrochemicals, 2016, 15 1), 16— 18 |
| ( 刘安昌,余彩虹,张树康,冯佳丽,贺晓露.现代农药, 2016, 15(1), 16— 18) | |
| [12] | Wang H. Y., Mao M. Z., Wang W ., Agrochemicals, 2018, 57 4), 255— 258 |
| ( 王海洋,毛明珍,王威.农药, 2018, 57(4), 255— 258) | |
| [13] | Shi Y. J., Zhou Q., Wang Y., Qian H. W., Ye L. Y., Feng X., Chen H., Li Y. T., Dai H., Wei Z. H., Wu J. M., Chin. J. Org. Chem., 2018, 38 9), 2450— 2457 |
| ( 石玉军,周钱,王杨,钱宏炜,叶林玉,冯霞,陈辉,李雅婷,戴红,魏中昊,吴锦明.有机化学, 2018, 38(9), 2450— 2457) | |
| [14] | Zhong L. K., Jiang T., Zhang F., Fu Q., Liu X. H., Xu T. M., Ding C. R., Chen J., Yuan J., Tan C. X., Chin. J. Org. Chem., 2019, 39, 2655— 2662 |
| ( 钟良坤, 江涛, 张帆, 付庆, 刘幸海, 许天民, 丁成荣, 陈杰, 袁静, 谭成侠 . 有机化学, 2019, 39, 2655— 2662) | |
| [15] | Hong Y., Dai H., Ye L. Y., Zhong S. L., Cao X. F., Shi Y. J., Li C. J., Shi J., Shi L., Chin. J. Org. Chem., 2017, 37 11), 3006— 3012 |
| ( 洪宇,戴红,叶林玉,仲苏林,曹雄飞,石玉军,李春建,石健,施磊.有机化学, 2017, 37(11), 3006— 3012) | |
| [16] | Yan Z. Z., Liu A. P., Huang M. Z., Liu M. H., Pei H., Huang L., Yi H. B., Liu W. D., Hu A. X ., Eur. J. Med. Chem. , 2018, 149, 170— 181 |
| [17] | Luo X. F., Hu A. X., Wang Y., Ye J., Wang X. G., Ou X. M., Chem. J. Chinese Universities, 2011, 32 12), 2800— 2805 |
| ( 罗先福,胡艾希,王宇,叶姣,王晓光,欧晓明.高等学校化学学报, 2011, 32(12), 2800— 2805) | |
| [18] | Hu A. X., Lin D., Dai M. C., Luo X. F., Ye J., Ou X. M., Application of 4-(Benzofuran-5-yl)-2-phenylaminothiazole as a Bactericide, CN103141486A, 2013-06-12 |
| ( 胡艾希, 林定, 戴明崇, 罗先福, 叶姣, 欧晓明 . 4-(苯并呋喃-5-基)-2-苯氨基噻唑作为杀菌剂的应用, CN103141486A, 2013 -06-12) | |
| [19] | Yang Z. H., Li B. B., Ye J., Hu A. X., Chem. J. Chinese Universities, 2016, 37 8), 1442— 1450 |
| ( 杨子辉,李贝贝,叶姣,胡艾希.高等学校化学学报, 2016, 37(8), 1442— 1450) | |
| [20] | Iwataki I., Iwasa T., Hatano R., Moyano E. L ., Insecticidal Aminothiazole Derivatives, US 6737382B1, 2004 -03-18 |
| [21] | Zhang H. Q., Gong D. W., Xu L. Z ., Agrochemicals, 2016, 55 3), 170— 173 |
| ( 张画轻,龚大伟,许良忠.农药, 2016, 55(3), 170— 173) | |
| [22] | Ye J., Sun X. X., Qiu S. Y., Hu A. X., Chin. J. Struc. Chem., 2014, 33( 3), 429— 433 |
| [23] | Shen F., Hu A. X., Luo X. F., Ye J., Ou X. M., Chin. J. Org. Chem., 2012, 32 2), 388— 392 |
| ( 沈芳,胡艾希,罗先福,叶姣,欧晓明.有机化学, 2012, 32(2), 388— 392) | |
| [24] | Li B B., . Synthesis and Biological Activity of 2-(4-aryloxyphenoxy) Propanamide Containing Furanophenol Ether. Hunan University,Changsha, 2015 |
| ( 李贝贝 . 含呋喃酚醚的2-(4-芳氧苯氧基)丙酰胺的合成与生物活性, 长沙: 湖南大学, 2015) | |
| [25] | Ding N ., Synthesis and Antitumor Activity of the N-[4-(t-Butyl)-5-benzylthiazole-2-yl] Acetamide Derivatives, Hunan University, Changsha, 2016 |
| ( 丁娜 . N-(4-叔丁基-5-苄基噻唑-2-基)乙酰胺衍生物的合成与抗肿瘤活性, 长沙: 湖南大学, 2016) | |
| [26] | Chen N C.. , Pesticide Bioassay Technology, Beijin Agricultural University Press, Beijing, 1991 |
| ( 陈年春 . 农药生物测定技术, 北京:北京农业大学出版社, 1991) | |
| [27] | Frisch M . J., Trucks G. W., Schlegel H. B.,Scuseria G. E., Robb M. A Cheeseman J. R., Scalmani G.,Barone V.,Mennucci B., Petersson G. A., Nakatsuji H.,Caricato M.,Li X., Hratchian H. P., Izmaylov A. F.,Bloino J.,Zheng G., Sonnenberg J. L., Hada M.,Ehara M.,Toyota K., Fukuda R., Hasegawa J.,Ishida M.,Nakajima T., Honda Y., Kitao O.,Nakai H.,Vreven T., Montgomery J. J. A., Peralta, J. E.,Ogliaro F., Bearpark M Heyd J. J., Brothers E.,Kudin K. N.,Staroverov V. N., Kobayashi R., Normand J.,Raghavachari K.,Rendell A., Burant J. C., Iyengar S. S.,Tomasi J.,Cossi M., Rega N., Millam N. J.,Klene M.,Knox J. E., Cross J. B., Bakken V.,Adamo C.,Jaramillo J., Gomperts R., Stratmann R. E.,Yazyev O.,Austin A. J., Cammi R., Pomelli C.,Ochterski J. W.,Martin R. L., Morokuma K., Zakrzewski V. G.,Voth G. A.,Salvador P., Dannenberg J. J., Dapprich S.,Daniels A. D.,Farkas O., Foresman J. B., Ortiz J. V.,Cioslowski JFox D. J., Gaussian 09. Gaussian, Inc,Wallingford CT, 2009 |
| [28] | Lu T., Chen F., J. Comput. Chem., 2012, 33, 580— 592 |
| [29] | Lu T., Chen F., J. Mol. Graph. Model, 2012, 38, 314— 323 |
| [1] | 李康明,陈佳,易阳杰,闫忠忠,叶姣,龙楚云,柳爱平,胡艾希,李建明. 1-(4-氯苯基)-2-环丙基酮肟醚的设计、 合成及杀虫活性[J]. 高等学校化学学报, 2020, 41(5): 1026. |
| [2] | 董心睿, 夏喆, 王桢学, 边强, 李华斌. 含1,2,4,5-四取代苯基的吡唑-4-甲酰胺类化合物的设计、 合成及生物活性[J]. 高等学校化学学报, 2020, 41(12): 2759. |
| [3] | 李庆, 易平贵, 陶洪文, 李洋洋, 张志于, 彭文宇, 李玉茹. 溶剂效应和取代基效应对2-(2-氨基苯基)苯并噻唑光谱性质及激发态分子内质子转移的影响[J]. 高等学校化学学报, 2019, 40(7): 1425. |
| [4] | 赵邦屯, 陶晶晶, 陈小纪, 付慧敏, 朱卫民. 含噻吩和吡啶基的插烯式四硫富瓦烯衍生物的合成、 结构和电化学性质[J]. 高等学校化学学报, 2018, 39(7): 1449. |
| [5] | 李林, 李淼, 柴宝山, 杨吉春, 宋玉泉, 刘长令. 取代噻吩双酰胺类化合物的设计、 合成及生物活性[J]. 高等学校化学学报, 2016, 37(9): 1649. |
| [6] | 贾长青, 杨冬燕, 车传亮, 马永强, 芮昌辉, 闫晓静, 覃兆海. 1H-1,2,4-三唑-5-氨基甲酸酯类化合物的合成、 结构表征及杀虫、 抑菌活性[J]. 高等学校化学学报, 2016, 37(5): 892. |
| [7] | 买合木提江·杰力, 阿布来提·麦麦提, 阿里木江·艾拜都拉, 买买提·吐尔逊. 氟化氢团簇(HF)n(n=2~8)拓扑性区别氢键构型的图论列举法和量子化学计算法研究[J]. 高等学校化学学报, 2016, 37(12): 2275. |
| [8] | 陈九菊. 有机半导体Terazulene单晶双极电荷传输性质的理论研究[J]. 高等学校化学学报, 2016, 37(1): 121. |
| [9] | 向俊峰, 易平贵, 于贤勇, 陈建, 郝艳雷, 任志勇. 纳米腔限制环境下2-(2-羟苯基)苯并噻唑的质子转移[J]. 高等学校化学学报, 2015, 36(4): 654. |
| [10] | 王宽, 陈建刚, 王伯周, 吕剑, 王文亮, 刘峰毅, 周诚, 廉鹏, 刘忠文, 刘昭铁. FOX-12制备过程的反应机理及动力学[J]. 高等学校化学学报, 2015, 36(3): 531. |
| [11] | 李秀云, 万川, 杜士杰, 李红, 袁会珠, 蒋家珍, 肖玉梅, 覃兆海. 二芳基肟苄醚类化合物的合成及杀菌活性[J]. 高等学校化学学报, 2015, 36(12): 2415. |
| [12] | 杨冬燕, 王蕾, 贾长青, 李长胜, 马永强, 芮昌辉, 徐彦军, 覃兆海. 芳亚甲基硝基缩氨基胍类化合物的合成及杀虫活性[J]. 高等学校化学学报, 2014, 35(8): 1703. |
| [13] | 谢瑞龙, 宋越, 杨新玲, 汪梅子, 凌云. 新型瑞香酮类似物的合成及杀菌活性[J]. 高等学校化学学报, 2014, 35(7): 1451. |
| [14] | 杨海葵, 许万福, 段安娜, 游文玮, 赵培亮. ,4-三唑环的亚胺及酰亚胺类化合物的合成及生物活性[J]. 高等学校化学学报, 2014, 35(3): 555. |
| [15] | 高成贵, 隆正文, 谭兴凤, 龙波, 张为俊, 龙超云, 秦水介. 气相中HRnCCH和X(X=H2O,NH3)反应机理的理论研究[J]. 高等学校化学学报, 2014, 35(2): 344. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||