

高等学校化学学报 ›› 2020, Vol. 41 ›› Issue (5): 1018.doi: 10.7503/cjcu20190621
收稿日期:2019-12-02
出版日期:2020-05-10
发布日期:2020-02-07
通讯作者:
穆晓清
E-mail:xqmu@jiangnan.edu.cn
基金资助:
WU Tao1,2,MU Xiaoqing1,2,*( ),NIE Yao1,XU Yan1
),NIE Yao1,XU Yan1
Received:2019-12-02
Online:2020-05-10
Published:2020-02-07
Contact:
Xiaoqing MU
E-mail:xqmu@jiangnan.edu.cn
Supported by:摘要:
基于同源建模建立了Bacillus cereus胺脱氢酶(BcAmDH)的三维结构, 采用半理性设计方法, 对底物结合口袋附近的8个氨基酸残基(L42, G43, M67, A115, E116, T136, V293和V296)分别进行单点饱和突变, 通过显色法筛选出3个正向突变位点(116, 136和293). 进一步采用迭代饱和突变策略对这3个正向位点进行组合突变, 获得最优突变株V293A/E116V/T136S, 其对苯乙酮还原反应的催化效率达到2.54 L·min-1·mmol-1, 比BcAmDH提高了719%; 与BcAmDH相比, 最优突变株在催化苯乙酮的不对称还原反应时, 底物浓度由100 mmol/L提高至300 mmol/L, 转化率由42.1%提高至80.2%. 分子对接结果表明, 突变株底物结合口袋的位阻减小和底物进出通道的扩大是提高催化效率的主要原因.
中图分类号:
TrendMD:
吴涛,穆晓清,聂尧,徐岩. 迭代饱和突变提高Bacillus cereus胺脱氢酶对苯乙酮还原的催化效率. 高等学校化学学报, 2020, 41(5): 1018.
WU Tao,MU Xiaoqing,NIE Yao,XU Yan. Improving Catalytic Efficiency of Bacillus Cereus Amine Dehydrogenase for Acetophenone Reduction by Iterative Saturation Mutagenesis †. Chem. J. Chinese Universities, 2020, 41(5): 1018.
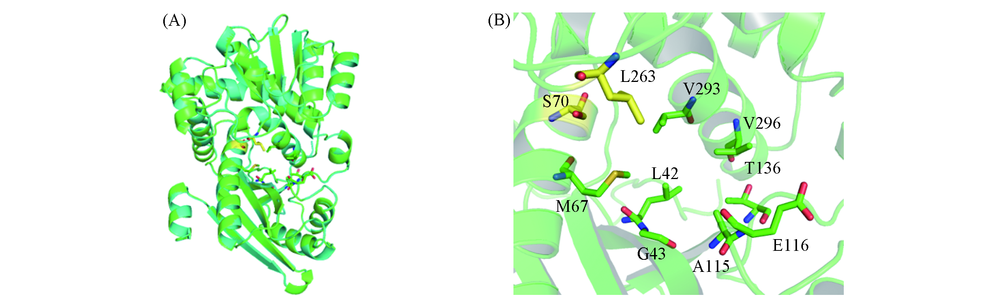
Fig.1 3D structure of GcLeuDH and BcAmDH(A) and distribution of selected amino acid(B) 3D structure of GcLeuDH and BcAmDH are shown as cyan and green cartoon, respectively. The eight selected amino acid residues(L42, G43, M67, A115, E116, T136, V293, V296) and the two residues initially mutated to generate AmDH activity(S70, L263) are shown as green and yellow sticks, respectively.
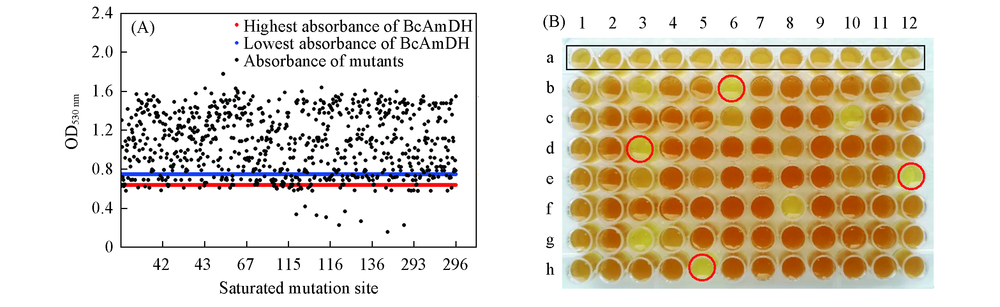
Fig.2 High-throughput screening results of site-directed saturation mutagenesis library(A) and color-rendering results of 116-site saturation mutagenesis library(B) Color-rendering results of BcAmDH and positive mutants are shown in black rectangle and red circle, respectively.
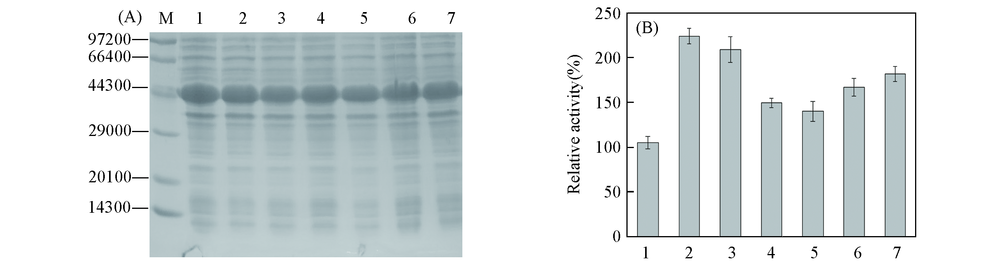
Fig.3 SDS-PAGE analysis(A) and relative activity(B) of positive mutants obtained by site-directed saturation mutagenesis (A) M: low molecular protein marker; lane 1—7 represent crude enzyme for BcAmDH, V293A, T136G, T136M, E116L, E116C and E116V, respectively. (B) 1. BcAmDH; 2. V293A; 3. T136G; 4. T136M; 5. E116L; 6. E116C; 7. E116V.
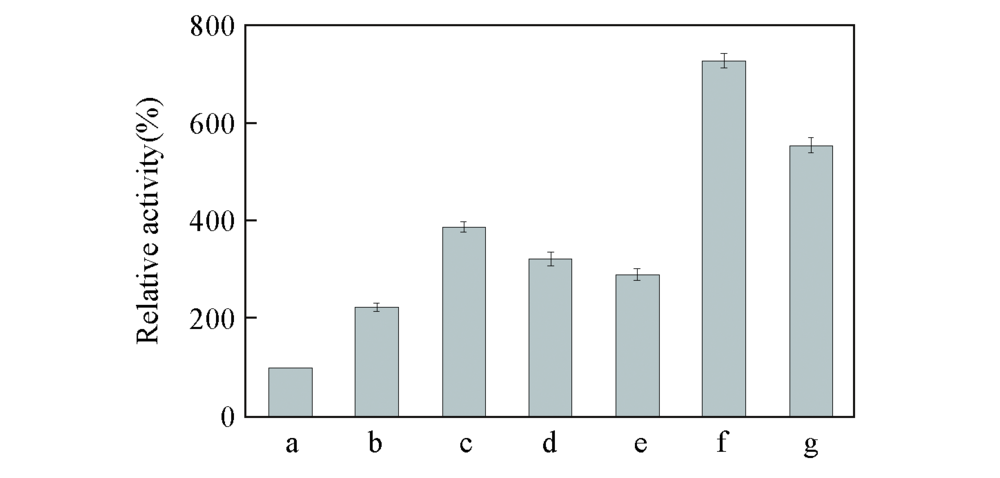
Fig.4 Relative activity of positive mutants obtained by iterative saturation mutagenesis a. BcAmDH; b. V293A; c. V293A/E116V; d. V293A/E116C; e. V293A/E116K; f. V293A/E116V/T136S; g. V293A/E116V/T136C.
| Mutant | Acetophenone | NADH | ||||
|---|---|---|---|---|---|---|
| Km/ (mmol·L-1) | kcat/ min-1 | (kcat/Km)/ (L·min-1·mmol-1) | Km/ (mmol·L-1) | kcat/ min-1 | (kcat/Km)/ (L·min-1·mmol-1) | |
| BcAmDH | 31.11±1.74 | 9.58±0.51 | 0.31 | 0.026±0.0025 | 4.13±0.32 | 158.85 |
| V293A | 36.18±1.65 | 26.02±1.48 | 0.72 | 0.031±0.0023 | 8.77±0.76 | 282.90 |
| V293A/E116V | 31.52±1.92 | 44.89±3.37 | 1.42 | 0.028±0.0034 | 16.09±0.97 | 574.64 |
| V293A/E116V/T136S | 28.87±1.22 | 73.25±6.21 | 2.54 | 0.027±0.0031 | 21.14±1.31 | 782.96 |
Table 1 Kinetic parameters of BcAmDH and three mutants
| Mutant | Acetophenone | NADH | ||||
|---|---|---|---|---|---|---|
| Km/ (mmol·L-1) | kcat/ min-1 | (kcat/Km)/ (L·min-1·mmol-1) | Km/ (mmol·L-1) | kcat/ min-1 | (kcat/Km)/ (L·min-1·mmol-1) | |
| BcAmDH | 31.11±1.74 | 9.58±0.51 | 0.31 | 0.026±0.0025 | 4.13±0.32 | 158.85 |
| V293A | 36.18±1.65 | 26.02±1.48 | 0.72 | 0.031±0.0023 | 8.77±0.76 | 282.90 |
| V293A/E116V | 31.52±1.92 | 44.89±3.37 | 1.42 | 0.028±0.0034 | 16.09±0.97 | 574.64 |
| V293A/E116V/T136S | 28.87±1.22 | 73.25±6.21 | 2.54 | 0.027±0.0031 | 21.14±1.31 | 782.96 |
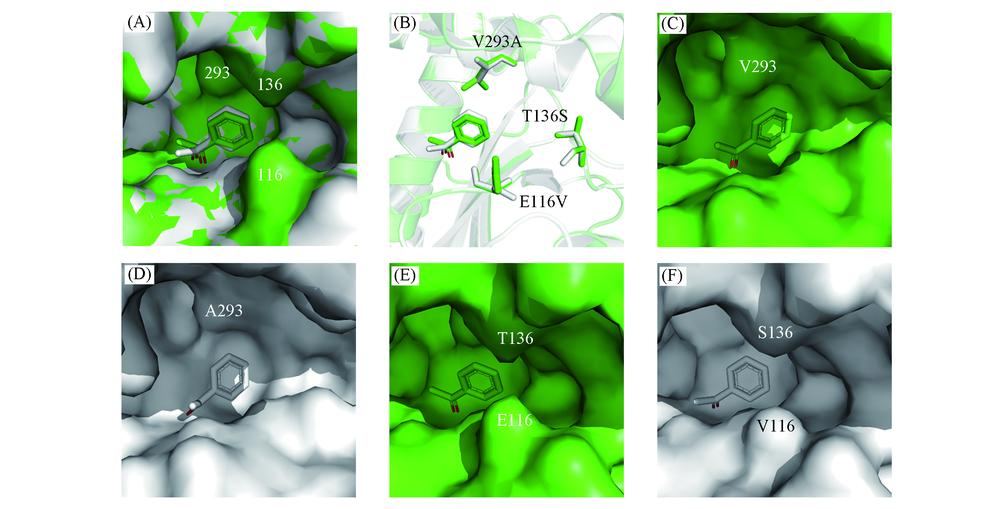
Fig.5 Molecular docking results of BcAmDH and mutant V293A/E116V/T136S with acetophenone (A) Molecular docking results of BcAmDH and mutant V293A/E116V/T136S with acetophenone, 3D structures of BcAmDH and mutant V293A/E116V/T136S are shown as green and white surface, 116, 136 and 293 are the numbering of amino acid residues; (B) molecular docking results of BcAmDH and mutant V293A/E116V/T136S with acetophenone, 3D structures of BcAmDHand mutant V293A/E116V/T136S are shown as green and white cartoon, E116V, T136S and V293A refer to mutation results; molecular docking results of BcAmDH(C) and mutant V293A/E116V/T136S with acetophenon(D), showing the effect of V293A mutation near the substrate side chain phenyl group on the shape of the substrate binding pocket; molecular docking results of BcAmDH(E) and mutant V293A/E116V/T136S with acetophenon(F), showing the effect of E116V and T136S mutations at the substrate entry channel on the shape of the substrate binding pocket. The substrate acetophenone is shown as green sticks with BcAmDH and white sticks with mutant V293A/E116V/T136S.
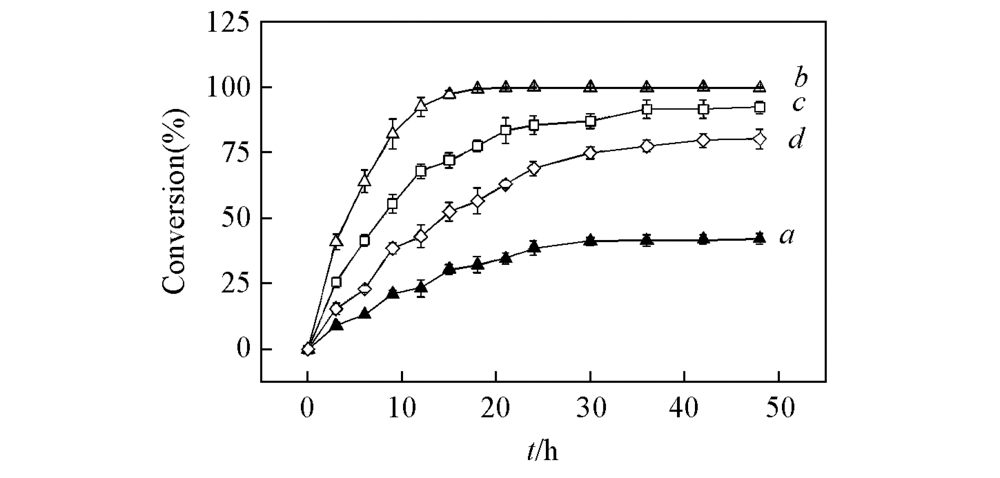
Fig.6 Time course of asymmetric reduction of acetophenone with different concentrations by BcAmDH and mutant V293A/E116V/T136S a. 100 mmol/L Acetophenone with BcAmDH; b. 100 mmol/L acetophenone with V293A/E116V/T136S; c. 200 mmol/L acetophenone with V293A/E116V/T136S; d. 300 mmol/L acetophenone with V293A/E116V/T136S.
| [1] |
Tseliou V., Knaus T., Masman M. F., Corrado M. L., Mutti F. G., Nat. Commun., 2019,10(1), 1—11
doi: 10.1038/s41467-018-07882-8 URL pmid: 30602773 |
| [2] |
Jann M. W., Pharmacotherapy, 2000,20(1), 1—12
doi: 10.1592/phco.20.1.1.34664 URL pmid: 10641971 |
| [3] | Zhang H ., The Preparation and Analysis of α-Phenylethylamine, Sichuan University, Chengdu, 2004 |
| ( 张华 . 手性物α-苯乙胺的制备和分析研究, 成都: 四川大学, 2004) | |
| [4] |
Tan X., Gao S., Zeng W., Xin S., Yin Q., Zhang X ., J. Am. Chem. Soc., 2018,140(6), 2024—2027
doi: 10.1021/jacs.7b12898 URL pmid: 29377687 |
| [5] | Hoben C. E., Tetrahedron Lett., 2008,49(6), 977—979 |
| [6] |
Parvulescu A. N., Jacobs P. A., Vos D. E., Chem. Eur. J., 2010,13(7), 2034—2043
doi: 10.1002/chem.200600899 URL pmid: 17152100 |
| [7] | Allwein S. P., McWilliams J. C., Secord E. A., Mowrey D. R., Nelson T. D., Kress M. H., Tetrahedron Lett., 2006,47(36), 6409—6412 |
| [8] | Torres Gavilán A., Escalante J., Regla I., López Munguía A., Castillo E ., Tetrahedron: Asymmetry, 2007,18(22), 2621—2624 |
| [9] | Falus P. D., Boros Z., Hornyánszky G., Nagy J., Darvas F., Ürge L., Poppe L., Stud. U. Babes-Bol. Che., 2010,55(4), 289—296 |
| [10] | Leisch H., Grosse S., Iwaki H., Hasegawa Y., Lau P. C., Can. J. Chem., 2011,90(1), 39—45 |
| [11] | Fesko K., Steiner K., Breinbauer R., Schwab H., Schürmann M., Strohmeier G. A., J. Mol. Catal. B: Enzym., 2013,96, 103—110 |
| [12] | Malik M. S., Park E. S., Shin J. S., Green Chem., 2012,14(8), 2137—2140 |
| [13] | Ye L. J., Toh H. H., Yang Y., Adams J. P., Snajdrova R., Li Z., ACS Catal., 2015,5(2), 1119—1122 |
| [14] | Abrahamson M. J., Wong J. W., Bommarius A. S., Adv. Synth. Catal., 2013,355(9), 1780—1786 |
| [15] |
Knaus T., Böhmer W., Mutti F. G., Green Chem., 2017,19(2), 453—463
doi: 10.1039/C6GC01987K URL pmid: 28663713 |
| [16] | Au S. K., Bommarius B. R., Bommarius A. S., ACS Catal., 2014,4(11), 4021—4026 |
| [17] | Mayol O., David S., Darii E., Debard A., Mariage A., Pellouin V., Petit J. L., Salanoubat M., Berardinis V., Zaparucha A., Catal. Sci. Technol., 2016,6(20), 7421—7428 |
| [18] | Itoh N., Yachi C., Kudome T ., J. Mol. Catal. B: Enzym., 2000,10(1—3), 281—290 |
| [19] |
Abrahamson M. J., Vázquez F. E., Woodall N. B., Moore J. C., Bommarius A. S., Angew. Chem. Int. Ed., 2012,51(16), 3969—3972
doi: 10.1002/anie.201107813 URL pmid: 22396126 |
| [20] | Chen F. F., Liu Y. Y., Zheng G. W., Xu J. H., ChemCatChem, 2015,7(23), 3838—3841 |
| [21] |
Löwe J., Ingram A. A., Gröger H., Biorg. Med. Chem., 2018,26(7), 1387—1392
doi: 10.1016/j.bmc.2017.12.005 URL pmid: 29548785 |
| [22] |
Bommarius B. R., Schürmann M., Bommarius A. S., Chem. Commun., 2014,50(95), 14953—14955
doi: 10.1039/c4cc06527a URL pmid: 25347124 |
| [23] |
Chen F. F., Zheng G. W., Liu L., Li H., Chen Q., Li F. L., Li C. X., Xu J. H., ACS Catal., 2018,8(3), 2622—2628
doi: 10.1021/acscatal.7b04135 URL |
| [24] |
Yamaguchi H., Kamegawa A., Nakata K., Kashiwagi T., Mizukoshi T., Fujiyoshi Y., Tani K ., J. Struct. Biol., 2019,205(1), 11—21
doi: 10.1016/j.jsb.2018.12.001 URL pmid: 30543982 |
| [25] |
Waterhouse A., Bertoni M., Bienert S., Studer G., Tauriello G., Gumienny R., Heer F. T., Beer T. A. P., Rempfer C., Bordoli L., Nucleic Acids Res., 2018,46(W1), W296—W303
doi: 10.1093/nar/gky427 URL pmid: 29788355 |
| [26] |
Lenka S., Jan S., Tomas M., David B., Jiri D., Nucleic Acids Res., 2018,46(W1), W356—W362
doi: 10.1093/nar/gky417 URL pmid: 29796670 |
| [27] |
Wei T., Chang C., Xue L., Zhao J., Jie L., Nucleic Acids Res., 2018,46(W1), W363—W367
doi: 10.1093/nar/gky473 URL pmid: 29860391 |
| [28] | Yin X., Liu Y., Meng L., Zhou H., Wu J., Yang L., Adv. Synth. Catal, 2019,361(4), 803—812 |
| [29] | Rao L., Bi Y. F., Qiu X. G., Zhang F., Wang Y., Cao S. G., Chem. Res. Chinese Universities, 2009,25(3), 353—356 |
| [30] | Zhou J., Xu G., Han R., Dong J., Zhang W., Zhang R., Ni Y., Catal. Sci. Technol, 2016,6(16), 6320—6327 |
| [31] |
Sun Z. T., Lonsdale R., Kong X. D., Xu J. H., Zhou J. H., Reetz M. T., Angew. Chem. Int. Ed., 2015,54(42), 12410—12415
doi: 10.1002/anie.201501809 URL pmid: 25891639 |
| [32] |
Reetz M. T., Kahakeaw D., Sanchis J., Mol. Biosyst., 2009,5(2), 115—122
doi: 10.1039/b814862g URL pmid: 19156255 |
| [33] |
Luo X., Wang Y. J., Shen W., Zheng Y. G., J. Biotechnol., 2016,224, 20—26
doi: 10.1016/j.jbiotec.2016.03.008 URL pmid: 26959479 |
| [34] | Huang L., Ma H. M., Yu H. L., Xu J. H., Adv. Synth. Catal., 2014,356(9), 1943—1948 |
| [1] | 罗磊, 穆晓清, 吴涛, 聂尧, 徐岩. 一锅法合成去甲基麻黄素[J]. 高等学校化学学报, 2021, 42(8): 2458. |
| [2] | 程景, 娄文勇, 宗敏华. 有机溶剂/缓冲液双相体系中固定化Acetobacter sp.CCTCC M209061细胞催化1-(4-甲氧基)-苯基乙醇不对称氧化反应[J]. 高等学校化学学报, 2014, 35(7): 1529. |
| [3] | 梁建, 何霞, 董海亮, 刘海瑞, 张华, 许并社. ZnO/ZnSe复合纳米结构的制备及可见光光催化性能[J]. 高等学校化学学报, 2014, 35(3): 455. |
| [4] | 刘晓晶, 周宏勇, 刘兵, 李小娜, 宋沙沙, 李云庆, 王家喜. 氨基葡萄糖衍生物钌络合物催化苯乙酮氢化反应[J]. 高等学校化学学报, 2014, 35(11): 2335. |
| [5] | 杨俊, 赵文波, 杨朝芬, 孙晓东, 王齐, 朱艳琴, 陈华. (1S,2S)-DPEN修饰的Ir/Al2O3催化苯乙酮及其衍生物的不对称加氢反应[J]. 高等学校化学学报, 2013, 34(7): 1679. |
| [6] | 袁剑, 张波, 汤明慧, 卢晗锋, 陈银飞. 苯乙酮氢转移反应中ZrO2 /MCM-41和 ZrO2 /AC的催化性能对比[J]. 高等学校化学学报, 2012, 33(06): 1326. |
| [7] | 明燕花, 樊爱丽, 金子林, 蒋景阳. 温控Noyori配体的合成及在苯乙酮不对称氢转移反应中的应用[J]. 高等学校化学学报, 2010, 31(8): 1548. |
| [8] | 张凯, 吴腊霞, 胡磊磊, 丁小燕, 王欢, 陆嘉星. 电化学方法固定CO2用于2-羟基-2-(4-甲氧基苯基)-丙酸甲酯的合成[J]. 高等学校化学学报, 2010, 31(7): 1410. |
| [9] | 王梦亮, 杜刚, 刘滇生 . 光控生物不对称还原苯乙酮的研究[J]. 高等学校化学学报, 2006, 27(9): 1686. |
| [10] | 刘万毅, 李金夫, 马永祥, 梁永民. 1,5-二芳基二酮衍生物的制备-无溶剂状态下苯乙酮与芳基烯酮的共轭加成反应研究[J]. 高等学校化学学报, 2001, 22(S1): 141. |
| [11] | 杨大成, 章国林, 杨宇, 钟裕国. 对-甲苯乙酮与芳香醛和芳香胺的Mannich反应[J]. 高等学校化学学报, 2000, 21(11): 1694. |
| [12] | 许宜铭. 苯乙酮的TiO2光催化降解及其影响因素[J]. 高等学校化学学报, 2000, 21(10): 1539. |
| [13] | 谷文祥, 王洪星, 还振威. 二溴卡宾与取代苯乙酮反应机理的研究[J]. 高等学校化学学报, 1999, 20(7): 1068. |
| [14] | 许翩翩, 高景星, 郑荣辉, 彭蔚萍, 黄培强, 万惠霖, 王文国, 丁开宁, 陈守正. 多齿胺膦钌配合物催化苯乙酮的氢转移氢化[J]. 高等学校化学学报, 1998, 19(3): 442. |
| [15] | 邹建平, 刘现军, 许勇, 陆忠娥. 1-苯甲酰基-1-甲硫基甲醇与硫脲和芳胺的反应研究[J]. 高等学校化学学报, 1998, 19(10): 1589. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||