

高等学校化学学报 ›› 2019, Vol. 40 ›› Issue (9): 1937.doi: 10.7503/cjcu20190124
收稿日期:2019-02-28
出版日期:2019-09-10
发布日期:2019-08-09
通讯作者:
陈爱民
E-mail:amchen@zjut.edu.cn
基金资助:
ZHAO Yun,WEI Jinjing,ZHOU Yu,ZHU Jixiu,WANG Houyong,CHEN Aimin( )
)
Received:2019-02-28
Online:2019-09-10
Published:2019-08-09
Contact:
CHEN Aimin
E-mail:amchen@zjut.edu.cn
Supported by:摘要:
以十六烷基三甲基溴化铵(CTAB)为模板剂, 钛酸异丙酯为钛源, 通过水热法合成了Ti-MCM-41分子筛催化剂并对其进行了表征, 结果表明, Ti-MCM-41催化剂具有介孔结构, 其孔径约2.0~4.0 nm. 考察了无溶剂条件下Ti-MCM-41对环氧化合物与胺的开环反应的催化性能. 结果表明, 当反应温度和压力分别为145 ℃和2.5 MPa时, Ti-MCM-41(5)对环氧乙烷(EO)与二甲胺(DMA)的反应具有良好的催化性能, 环氧乙烷的转化率和N,N-二甲基乙醇胺的选择性分别为98.37%和86.68%. 表征结果表明, 催化剂骨架中四配位结构的Ti使其表面具有丰富的路易斯酸性位, 该酸性位对环氧化合物与胺的开环反应具有重要促进作用. 此外, 还对Ti-MCM-41催化环氧乙烷与二甲胺开环反应的机理进行了初步探讨.
中图分类号:
TrendMD:
赵韵, 魏金晶, 周煜, 朱继秀, 王后勇, 陈爱民. Ti-MCM-41催化环氧化合物与胺的开环反应. 高等学校化学学报, 2019, 40(9): 1937.
ZHAO Yun, WEI Jinjing, ZHOU Yu, ZHU Jixiu, WANG Houyong, CHEN Aimin. Ti-MCM-41 Catalysts for the Ring Opening Reaction of Epoxy Compounds with Amines †. Chem. J. Chinese Universities, 2019, 40(9): 1937.
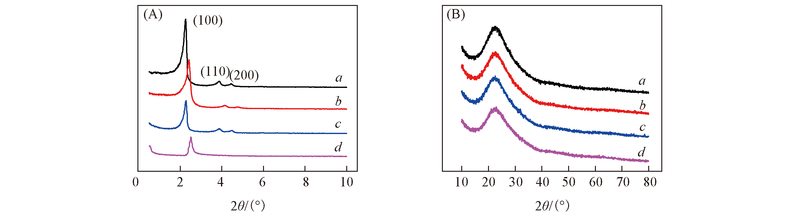
Fig.1 Low-angle(A) and wide-angle(B) XRD patterns of Ti-MCM-41(40)(a), Ti-MCM-41(20)(b), Ti-MCM-41(10)(c) and Ti-MCM-41(5)(d) calcined at 550 ℃ for 6 h
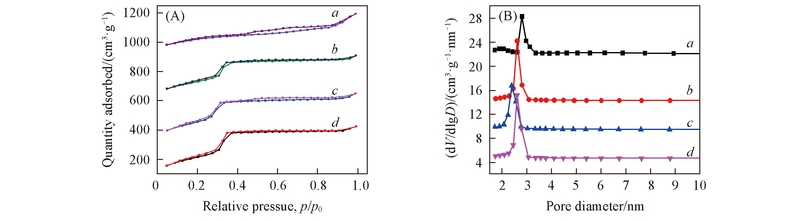
Fig.2 N2 adsorption-desorption isotherms(A) and pore size distributions(B) of Ti-MCM-41(5)(a), Ti-MCM-41(10)(b), Ti-MCM-41(20)(c) and Ti-MCM-41(40)(d) calcined at 550 ℃
| Entry | Catalyst | SBET/(m2·g-1) | Vt/(cm3·g-1) | D/nm |
|---|---|---|---|---|
| 1 | MCM-41 | 790 | 0.75 | 3.8 |
| 2 | Ti-MCM-41(5) | 444 | 0.43 | 3.9 |
| 3 | Ti-MCM-41(10) | 682 | 0.56 | 3.3 |
| 4 | Ti-MCM-41(20) | 869 | 0.62 | 2.9 |
| 5 | Ti-MCM-41(40) | 811 | 0.63 | 3.1 |
Table 1 Textural properties of Ti-MCM-41 samples with different Si/Ti molar ratios*
| Entry | Catalyst | SBET/(m2·g-1) | Vt/(cm3·g-1) | D/nm |
|---|---|---|---|---|
| 1 | MCM-41 | 790 | 0.75 | 3.8 |
| 2 | Ti-MCM-41(5) | 444 | 0.43 | 3.9 |
| 3 | Ti-MCM-41(10) | 682 | 0.56 | 3.3 |
| 4 | Ti-MCM-41(20) | 869 | 0.62 | 2.9 |
| 5 | Ti-MCM-41(40) | 811 | 0.63 | 3.1 |
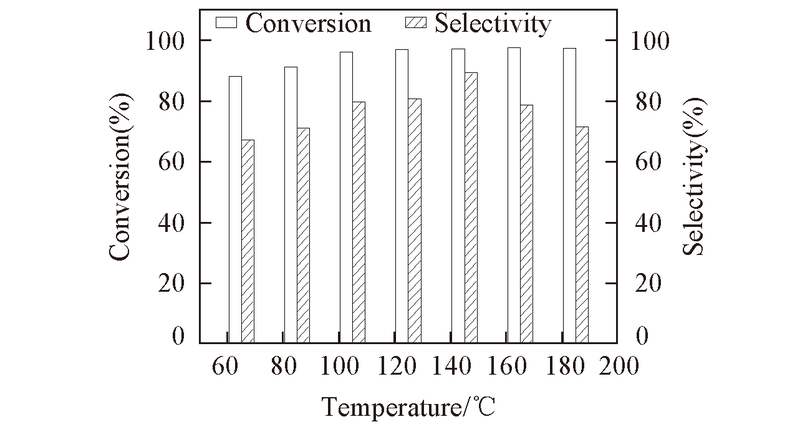
Fig.9 Effect of the reaction temperature on the conversion and selectivity Reaction condition: catalyst: Ti-MCM-41(5) calcined at 550 ℃; 2.2 MPa for 35 min; 5%(mass fraction) of catalyst with respect to EO, EO/DMA molar ratio: 1∶3.5.
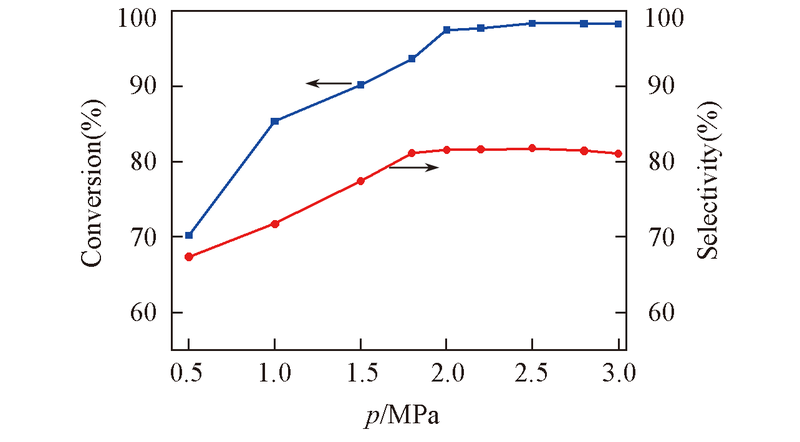
Fig.10 Effect of the reaction pressure on the conversion and selectivity Reaction condition: catalyst: Ti-MCM-41(5) calcined at 550 ℃, 135 ℃ for 35 min, 5%(mass fraction) of catalyst with respect to EO, EO/DMA molar ratio: 1∶3.5.
| Entry | Catalyst | EO conversion(%) | DMA selectivity(%) | DEMA yield(%) |
|---|---|---|---|---|
| 1 | TiO2 | 97.37 | 81.21 | 79.07 |
| 2 | MCM-41 | 97.44 | 83.53 | 81.40 |
| 3 | Ti-MCM-41(5) | 98.37 | 86.68 | 85.27 |
| 4 | Ti-MCM-41(10) | 97.56 | 73.84 | 75.60 |
| 5 | Ti-MCM-41(20) | 97.28 | 72.46 | 71.22 |
| 6 | Ti-MCM-41(40) | 97.23 | 76.52 | 74.40 |
Table 2 Reaction results of EO with DMA over various catalysts*
| Entry | Catalyst | EO conversion(%) | DMA selectivity(%) | DEMA yield(%) |
|---|---|---|---|---|
| 1 | TiO2 | 97.37 | 81.21 | 79.07 |
| 2 | MCM-41 | 97.44 | 83.53 | 81.40 |
| 3 | Ti-MCM-41(5) | 98.37 | 86.68 | 85.27 |
| 4 | Ti-MCM-41(10) | 97.56 | 73.84 | 75.60 |
| 5 | Ti-MCM-41(20) | 97.28 | 72.46 | 71.22 |
| 6 | Ti-MCM-41(40) | 97.23 | 76.52 | 74.40 |
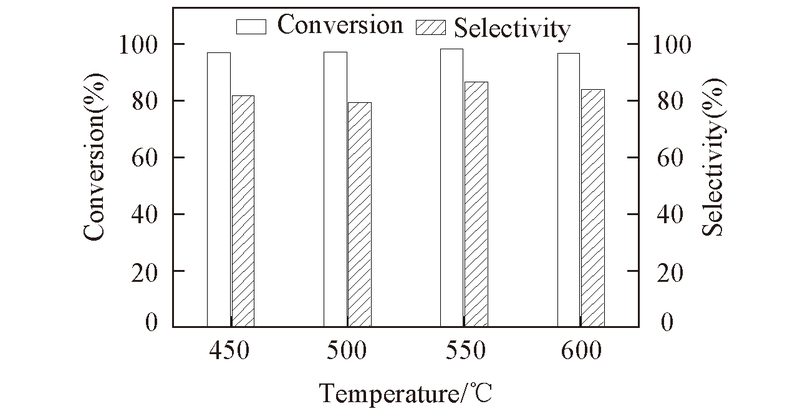
Fig.11 Effect of the calcined temperatures of Ti-MCM-41(5) sample on the conversion and selectivity under 145 ℃ and 2.5 MPa for 40 min Ti-MCM-41(5) catalyst was added 5%(mass fraction) with respect to EO; the molar ratio EO to DMA is 1∶3.5.
| [1] | Corey E.J., Zhang F. Y.,Angew. Chem. Int. Ed. Engl., 1999, 38, 1931—1934 |
| [2] | Johannes C. W., Visser M. S., Weatherhead G. S., Hoveyda A. H., ,J. Am. Chem. Soc., 1998, 120, 8340— 8347 |
| [3] | Ager D. J., Prakash I., Schaad D. R., ,Chem. Rev., 1996, 96, 835— 875 |
| [4] | Brien P. O ., Angew. Chem. Int. Ed. Engl. 1999, 38, 326— 329 |
| [5] | Tu Y. Q., Xia W. J., ,Chem. J. Chinese Universities, 1999,20( 4), 587— 589 |
| (涂永强,夏吾炯. 高等学校化学学报, 1999, 20(4), 587— 589) | |
| [6] | Clarkson C., Musonda C. C., Chibale K., Campbell W. E., Smith P., ,Bioorg. Med. Chem., 2003, 11, 4417— 4422 |
| [7] | Palomo C., Oiarbide M., Laso A., ,Angew. Chem. Int. Ed., 2005, 44, 3881— 3884 |
| [8] | Pujala B., Rana S., Chakraborti A. K., ,J. Org. Chem., 2011, 76( 21), 8768— 8780 |
| [9] | Cai P. Y., Zhang E. S., Wu Y. S., Fang T. B., Li Q. Q., Yang C., Wang J., Shang Y. J., ,ACS Omega., 2018, 3( 11), 14575— 14584 |
| [10] | He W., Wang L., Sun C., Wu K., He S., Chen J., Wu P., Yu Z., ,. Chem. Eur. J. 2011, 17, 13308— 13317 |
| [11] | Cui X. J., Dai X. C., Deng Y. Q., Shi F., ,. Chem. Eur. J. 2013, 19, 3665— 3675 |
| [12] | Jumde V. R., Petricci E., Petrucci C., Santillo N., Taddei M., Vaccaro L., ,Org. Lett., 2015, 17( 16), 3990— 3993 |
| [13] | Kamal A., Prasad R. B., Reddy A. M., Naseer M., Khan A., ,Catal. Commun., 2007, 8, 1876— 1880 |
| [14] | Brahmam P., Shivani R., Asit K. C., ,J. Org. Chem., 2011, 76, 8768— 8780 |
| [15] | Wu X. Y., Shu F. C., ,Chem. J. Chinese Universities, 1999,20( 12), 1892— 1896 |
| (伍新燕, 舒福昌. 高等学校化学学报, 1999, 20(12), 1892— 1896) | |
| [16] | Narsaiah A. V., Sreenu D., Nagaiah K., ,Syn. Commun., 2006, 36, 3183— 3189 |
| [17] | Loukrakpam D. C., Phukan P., ,. Tetrahedron Lett. 2017, 58( 52), 4855— 4858 |
| [18] | An D. L., Song W. K., Peng Z. H., Zhang Y. J., Dong W. R ., Chemistryselect 2018, 3( 45), 12946— 12950 |
| [19] | Struble T. J., Lankswert H. M., Pink M., Johnston J. N., ,ACS. Catal., 2018, 8( 12), 11926— 11931 |
| [20] | Kamal A., Ramu R., Azhar M. A., Khanna G. B. R., ,Tetrahedron. Lett., 2005, 46, 2675— 2677 |
| [21] | Feher C., Tomasek S., Hancsok J., Skoda-Foldes R., ,. Appl. Catal. B 2018, 239, 52— 60 |
| [22] | Tamboli A. H., Bandal H. A., Kim H., ,Chem. Eng. J., 2016, 306, 826— 831 |
| [23] | Medamoni V., Kishore P., Narender P., Satyanarayana B., ,. J. Mol. Catal. A 2007, 266, 290— 293 |
| [24] | Mathew W. C., Robinson D. A., Timms S M., Williams A. E. G., ,Tetrahedron. Lett., 2007, 48, 6249— 6251 |
| [25] | Fraile J. M., García N., Mayoral J. A., Santomauro F. G., Guidotti M., ,ACS Catal., 2015, 5, 3552— 3561 |
| [26] | Martinez E. G., Balmori A., Ponton I., Sanchez G. D ., Catal. 2018, 8(12), 617-1—62 |
| [27] | Nadeem S., Mumtaz A., Mumtaz M., Mutaib M. I., Shaharun M. S., Abdullah B., ,J. Mol. Liq., 2018, 272, 656— 667 |
| [28] | Chen A., Wang H. Y., Liu R., Bo Y. Y., Hu J., ,Catal. Lett., 2016, 146( 7), 1182— 1193 |
| [29] | Selvaraj M., Lee K., Yoo K. S., Lee T. G., ,Micropor. Mesopor. Mater., 2005, 81, 343— 355 |
| [30] | Jeffrey K., Jacek J., Sharon M., Javier P. R ., Langmuir 2015, 31, 1242— 1247 |
| [31] | Peter B. W., Wang X. L., Wolfgang Z., Klaus R. L., Hui T. C., ,. Acta Phys. Chim. Sinica 2014, 118, 8350— 8358 |
| [32] | Chao C., Hua W., Jin Y. H., ,Appl. Surf. Sci., 2011, 257, 9802— 9808 |
| [33] | Qin X. Y., Zhao W. B., Han B., Liu B. A., Lian P. C., Wu S. S., ,Korean J. Chem. Eng., 2015, 32( 6), 1064— 1068 |
| [34] | Maria S. B., Joana M. F. M., Dulce M. A., Joana M. F., Braga R. M., Marcus A. F., Fontes V. A., ,J. Therm. Anal. Calorim., 2015, 119, 197— 204 |
| [35] | Eimer G. A., Chanquia C. M., Sapag K., Herrero E. R., ,Micropor. Mesopor. Mater., 2008, 116, 670— 676 |
| [36] | Chao M. C., Lin H. P., Mou C. Y., Cheng B. W., Cheng C. F., ,Catal. Today., 2004, 97, 81— 87 |
| [37] | Thomas R., Antje P., Jurgen R., ,Synthetic. Commun., 2002, 32( 3), 457— 465 |
| [38] | Parker R. E., ,Chem. Rev., 1959, 2, 737— 799 |
| [39] | Satyarthi J. K., ,Appl. Catal. A:Gen., 2007, 330, 145— 151 |
| [40] | Ahmed. K., Rajendra. P. A., MallaR. M., Malla R. M., Naseer A. K., ,Catal. Commun., 2007, 8, 1876— 1880 |
| [41] | Boningari T., Rapelli S., Avvari N., Prasad J. K., Prashanth K., Benjaram M., ,Org. Process. Res. Dev., 2010, 14, 1457— 1463 |
| [1] | 刘启予, 范炜. 介孔分子筛制备技术新进展—二次合成、超分子自组装和介孔生成剂法[J]. 高等学校化学学报, 2021, 42(1): 60. |
| [2] | 刘鸿, 王正仁, 陈恒泽, 张锐昌, 陈超, 张宁. UMCM-1-NH2的后合成修饰及在制备β-氨基醇反应中的应用[J]. 高等学校化学学报, 2016, 37(11): 2012. |
| [3] | 王东龙, 周静红, 隋志军, 周兴贵. 四配位Ti掺杂有机杂化介孔分子筛的直接合成与表征[J]. 高等学校化学学报, 2015, 36(6): 1042. |
| [4] | 张涛, 董雪, 张宗弢. 孔道型微孔和介孔分子筛的改性及超级绝热性能[J]. 高等学校化学学报, 2014, 35(4): 689. |
| [5] | 袁剑, 张波, 汤明慧, 卢晗锋, 陈银飞. 苯乙酮氢转移反应中ZrO2 /MCM-41和 ZrO2 /AC的催化性能对比[J]. 高等学校化学学报, 2012, 33(06): 1326. |
| [6] | 陈冬冬 丁昌华 侯雪龙 戴立信. 肟醚碳负离子对氮杂环丙烷的开环反应[J]. 高等学校化学学报, 2011, 32(3): 694. |
| [7] | 曹宇 陈标华 张润铎. SBA-15介孔分子筛负载型过渡金属催化燃烧脱除乙腈废气[J]. 高等学校化学学报, 2011, 32(12): 2849. |
| [8] | 宋科, 管景奇, 吴淑杰, 徐海燕, 阚秋斌. 以微孔沸石MCM-49和MCM-56为硅铝源合成强酸性介孔分子筛[J]. 高等学校化学学报, 2009, 30(2): 344. |
| [9] | 刘秀丽,高国华,KAWI Sibudjing . 金属有机化学气相沉积法制备SnO2/MCM-41半导体传感器及其性能研究[J]. 高等学校化学学报, 2007, 28(9): 1609. |
| [10] | 曲凤玉,崔凌飞,黄世英,朱广山 . 介孔分子筛孔道结构对药物巯甲丙脯酸释放性能的影响[J]. 高等学校化学学报, 2007, 28(8): 1440. |
| [11] | 张浩然,李斌,雷炳富,李文连 . 共价嫁接钌(Ⅱ)配合物SBA-15介孔分子筛的光学氧传感行为[J]. 高等学校化学学报, 2007, 28(10): 1920. |
| [12] | 张浩然,白焱,李斌,雷炳富,傅春艳 . 共价嫁接Ru(Ⅱ)配合物杂化材料的制备及氧气传感性能研究[J]. 高等学校化学学报, 2007, 28(1): 16. |
| [13] | 曲凤玉, 朱广山, 林惠明, 张维维, 李守贵, 裘式纶. 以阴离子手性模板合成螺旋形貌的介孔分子筛[J]. 高等学校化学学报, 2006, 27(4): 602. |
| [14] | 陈悦 李晓天 古丽米娜 李楠 朱广山 裘式纶. 负载激光染料香豆素151的MCM-41粉体及其纤维的光学性质研究[J]. 高等学校化学学报, 2006, 27(3): 397. |
| [15] | 陈作锋, 姜艳霞, 许金梅, 庄全超, 黄令, 董全峰, 孙世刚 . 一种新型复合微孔聚合物电解质及其与锂离子电池负极相容性研究[J]. 高等学校化学学报, 2006, 27(10): 1937. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||