

高等学校化学学报 ›› 2023, Vol. 44 ›› Issue (5): 20220729.doi: 10.7503/cjcu20220729
收稿日期:2022-11-19
出版日期:2023-05-10
发布日期:2023-02-06
通讯作者:
付永柱
E-mail:yfu@zzu.edu.cn
基金资助:
REN Siyuan, GUO Wei, FU Yongzhu( )
)
Received:2022-11-19
Online:2023-05-10
Published:2023-02-06
Contact:
FU Yongzhu
E-mail:yfu@zzu.edu.cn
Supported by:摘要:
近年来, 有机硫化物因具有结构灵活、 种类繁多、 资源丰富和环境友好的特点而在储能领域得到广泛应用, 并展示出较大的发展前景. 有机硫化物在充放电过程中会发生硫-硫(S—S)键的可逆断裂和形成, 分子中 S—S键的数目决定了电子转移的多少, 而有机基团可以影响电池的电化学行为. 本文综合评述了有机硫化物在可充电电池中的研究进展, 主要包括有机硫小分子和聚合物正极材料在可充电锂电池中的应用, 有机硫电解液添加剂对锂-硫(Li-S)和锂-硒(Li-Se)电池性能的影响及有机硫材料在其它可充电电池中的应用等4个方面. 这些结构可调的有机硫化物展示出优异的循环稳定性, 改变了传统Li-S和Li-Se电池的氧化-还原路径, 参与正负极固体电解质界面的形成, 能抑制多硫化物的穿梭效应, 阻止锂枝晶的生长. 最后, 讨论了有机硫化物在可充电电池领域未来研究发展中面临的挑战.
中图分类号:
TrendMD:
任思远, 郭玮, 付永柱. 有机硫化物在可充电电池中的研究进展. 高等学校化学学报, 2023, 44(5): 20220729.
REN Siyuan, GUO Wei, FU Yongzhu. Research Progress of Organosulfur in Rechargeable Batteries. Chem. J. Chinese Universities, 2023, 44(5): 20220729.
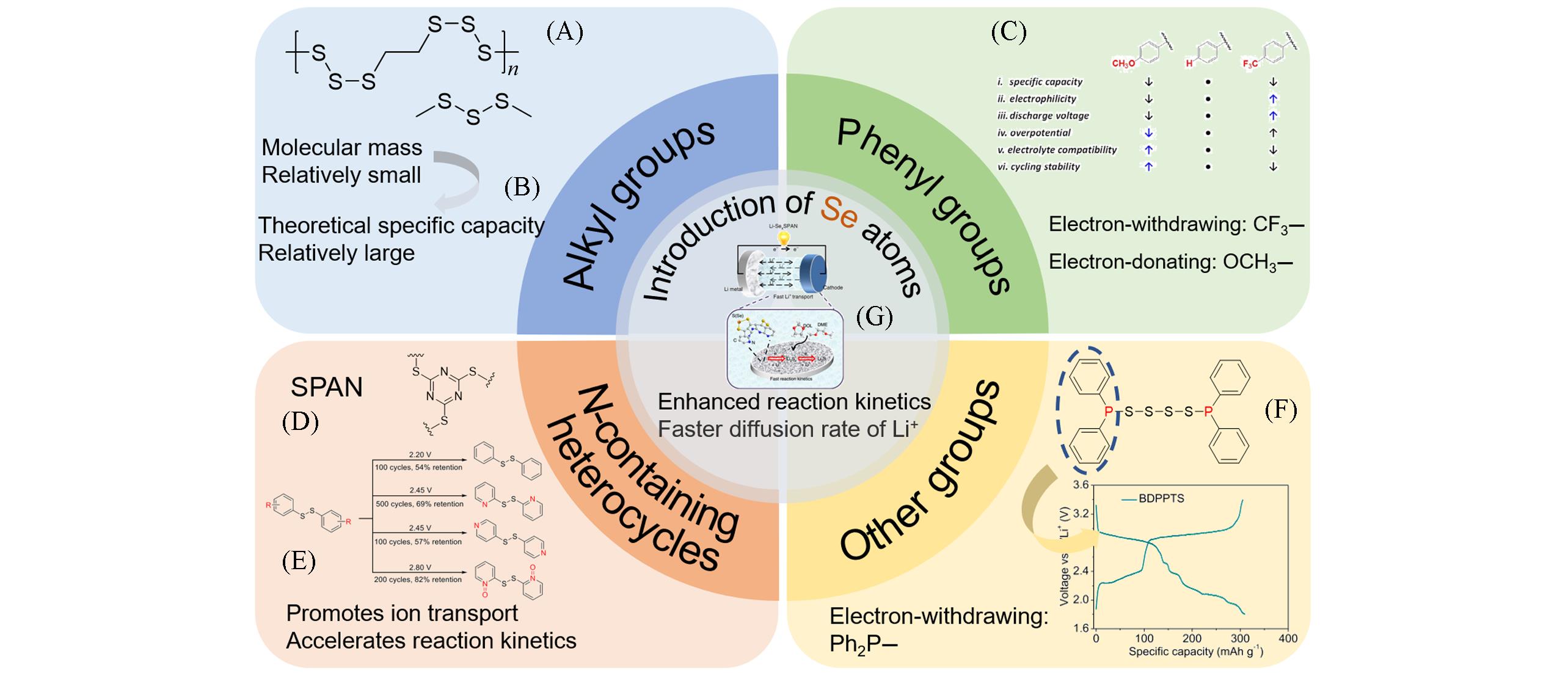
Fig.2 Relationship diagram between properties of organic groups and electrochemical behavior of batteries(A) Copyright 2019, American Chemical Society[20]; (B) Copyright 2016, Wiley-VCH[21]; (C) Copyright 2017, Wiley-VCH[22]; (D) Copyright 2015, Nature Publishing Group[23]; (E) Copyright 2019, the Royal Society of Chemistry[24]; (F) Copyright 2021, Nature Publishing Group[25]; (G) Copyright 2019, Nature Publishing Group[26].
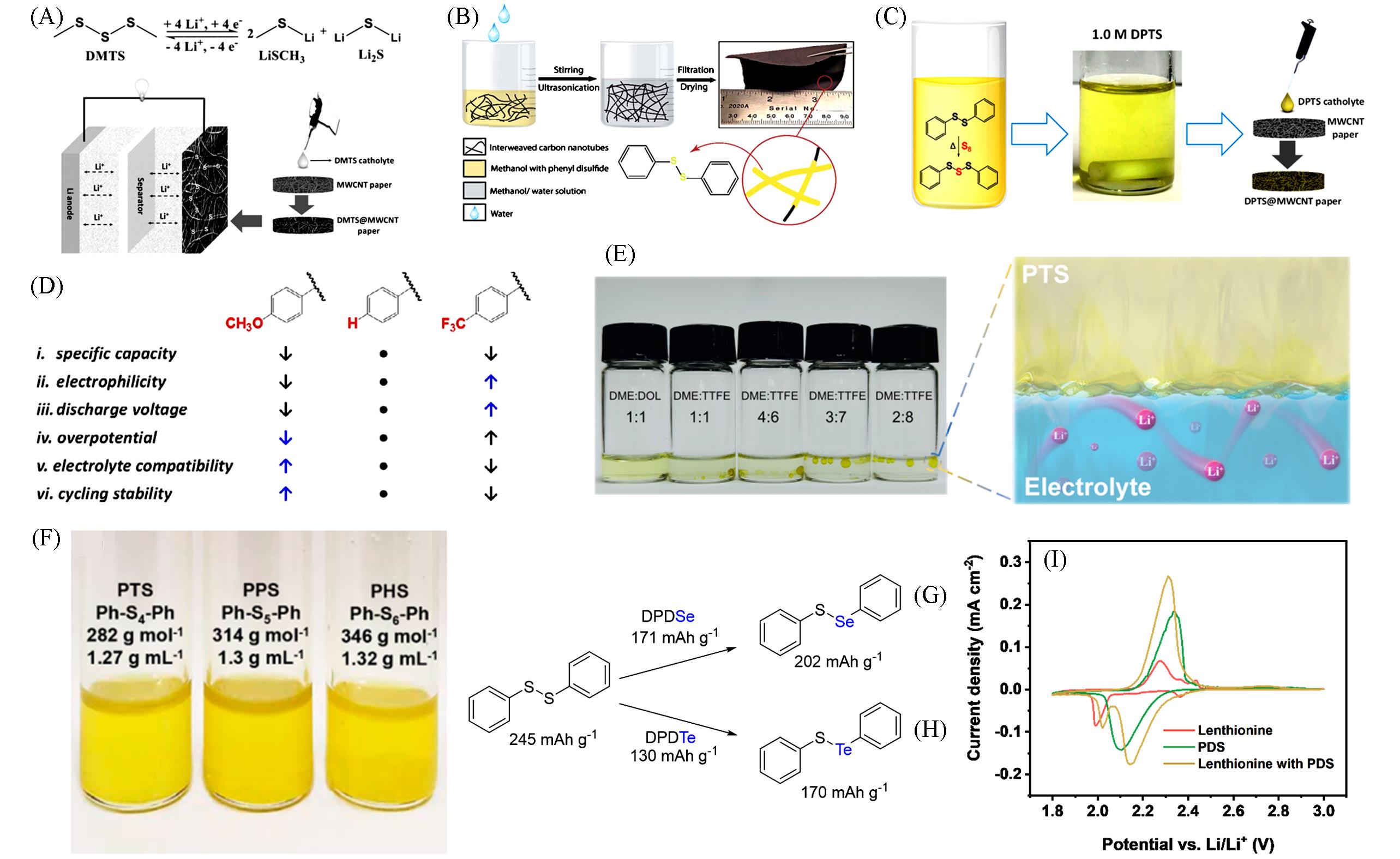
Fig.3 Redox reaction of DMTS in the lithium cell and schematic illustration of the cell configuration and the composition of the cathode(A)[21], schematic diagram of the “phase extraction” method for preparing DPDS@CNT cathode(B)[35], schematic illustration of the synthesis of DPTS in liquid electrolyte at 70 ℃(C)[36], effects of functional groups on phenyl rings on the properties and electrochemical performances of bis(aryl) tetrasulfides(D)[22], digital photos and interface diagrams for the mixture of PTS and electrolytes(E)[37], optical image and the properties of the PTS, PPS and PHS(F)[38], the synthetic route of PhS—SePh(G)[39], the synthetic route of PhS—TePh(H)[40] and CV profile of lenthionine, DPDS, and lenthionine with DPDS(I)[41](A) Copyright 2016, Wiley-VCH; (B) Copyright 2017, the Royal Society of Chemistry; (C) Copyright 2016, American Chemical Society; (D) Copyright 2017, Wiley-VCH; (E) Copyright 2022, Wiley-VCH; (F) Copyright 2018, American Chemical Society; (G) Copyright 2018, the Royal Society of Chemistry; (H) Copyright 2022, the Royal Society of Chemistry; (I) Copyright 2022, Wiley-VCH.
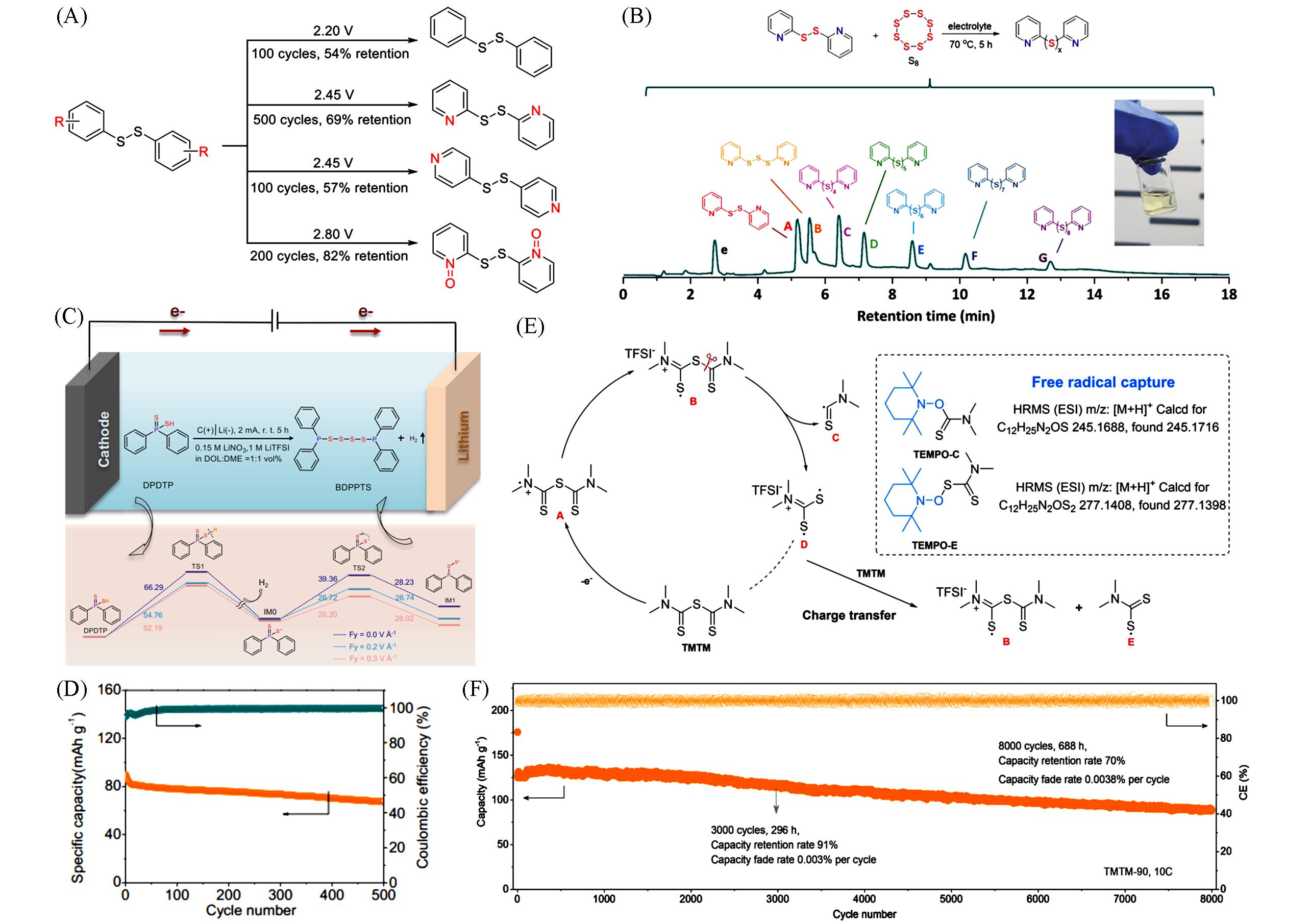
Fig.4 Electrochemical behavior of DPDS, 2,2′⁃DpyDS, 4,4′⁃DpyDS and DpyDSDO(A)[24】, the synthesis of Py2S x in electrolyte and total ion chromatogram(TIC) spectrum of the mixture catholyte(B)[44], electrochemical synthesis and reaction process simulation of BDPPTS(C), cycling performance of the Li/BDPPTS cell at 0.2C(D)[25], proposed molecular structure transformation mechanism of TMTM via electrochemical oxidation(E) and long cycle performance of TMTM⁃90 at 10C(F)[45]Inset of (B): photograph of the prepared Py2S x catholyte.(A) Copyright 2019, the Royal Society of Chemistry; (B) Copyright 2020, Wiley-VCH; (C, D) Copyright 2021, Nature Publishing Group; (E, F) Copyright 2022, American Chemical Society.
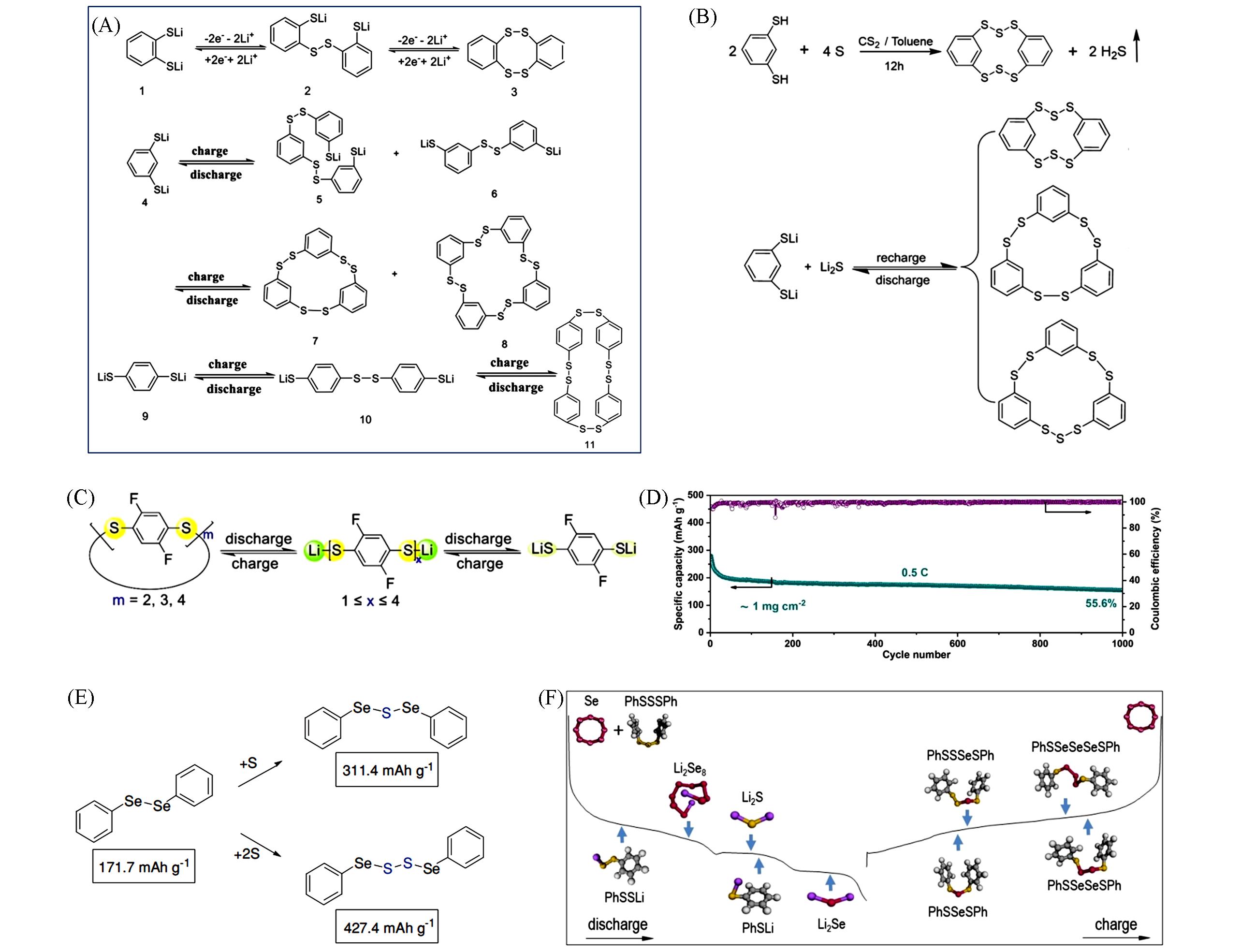
Fig.5 Redox reactions of 1,2⁃, 1,3⁃ and 1,4⁃LBDTs during the charge and discharge(A)[46], synthesis of the HDBCO and the redox reactions of ICPSs during cycling(B)[47], the synthesis process of F⁃MCDS(C), long⁃term cycling performance of the Li/F⁃MCDS cell at 0.5C rate(D)[48], synthesis of DPDSe⁃S and DPDSe⁃S2(E)[50] and the redox mechanism of the DPTS⁃Se cathode in a lithium cell(F)[51](A) Copyright 2019, Wiley-VCH; (B) Copyright 2020, the Royal Society of Chemistry; (C, D) Copyright 2022, the Royal Society of Chemistry; (E) Copyright 2018, Wiley-VCH; (F) Copyright 2019, Wiley-VCH.
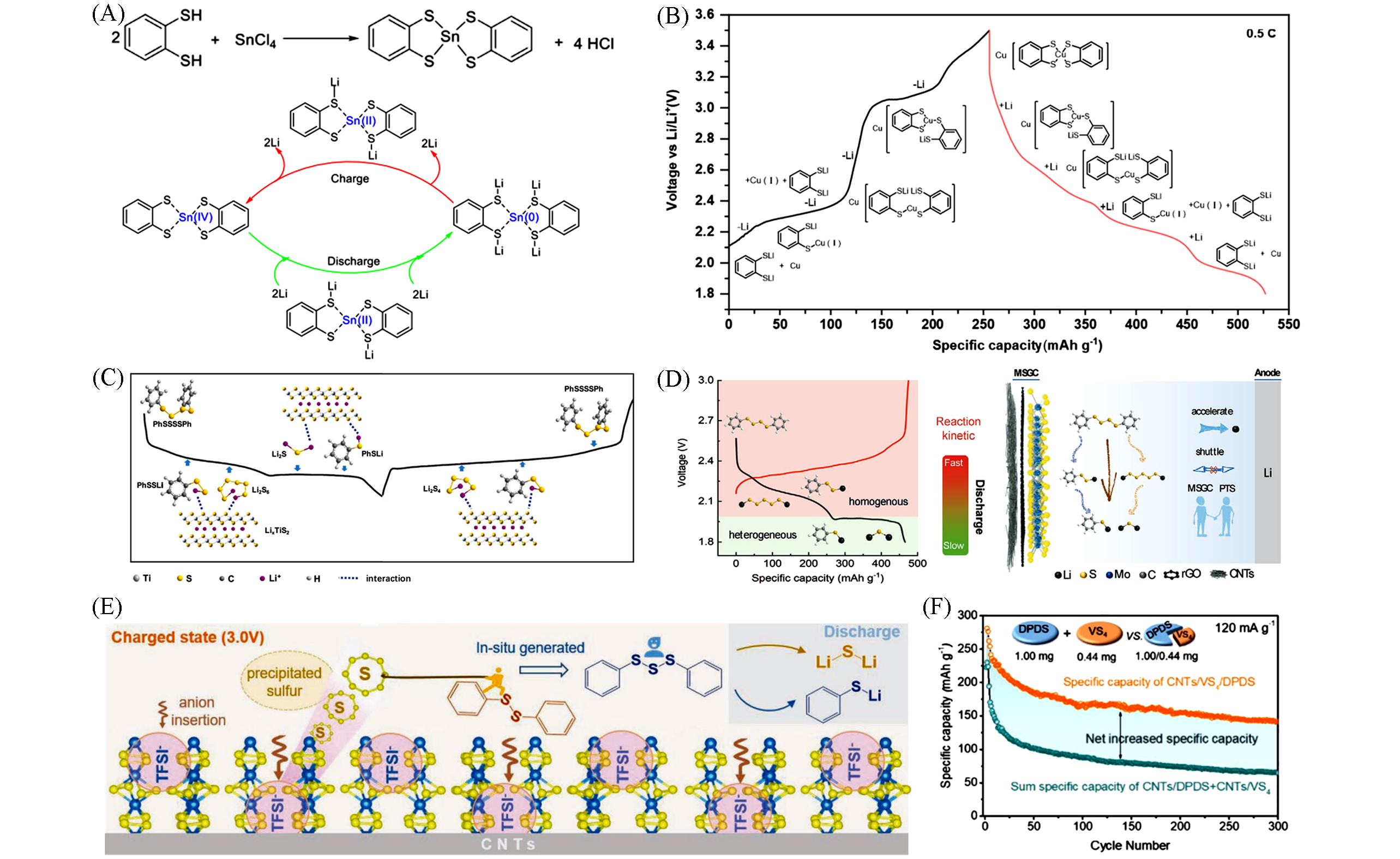
Fig.6 Redox mechanism of Sn⁃(BDT)2 in a lithium cell(A)[28], schematic of the redox reactions of 1,2⁃LBDT@Cu NWs in a lithium cell(B)[29], redox reactions of the TiS2 NSs@MWCNT⁃PTS electrode(C)[27], illustration of Li⁃PTS battery performance enhancement by the MSGC substrate(D)[31], schematic diagram of the charge⁃discharge mechanism of VS4/DPDS(E) and comparison of discharge capacity between CNTs/VS4/DPDS and sum specific capacity of CNTs/DPDS and CNTs/VS4(F)[30](A) Copyright 2021, Elsevier; (B) Copyright 2023, Elsevier; (C) Copyright 2020, Wiley-VCH; (D) Copyright 2022, Wiley-VCH; (E, F) Copyright 2021, Wiley-VCH.
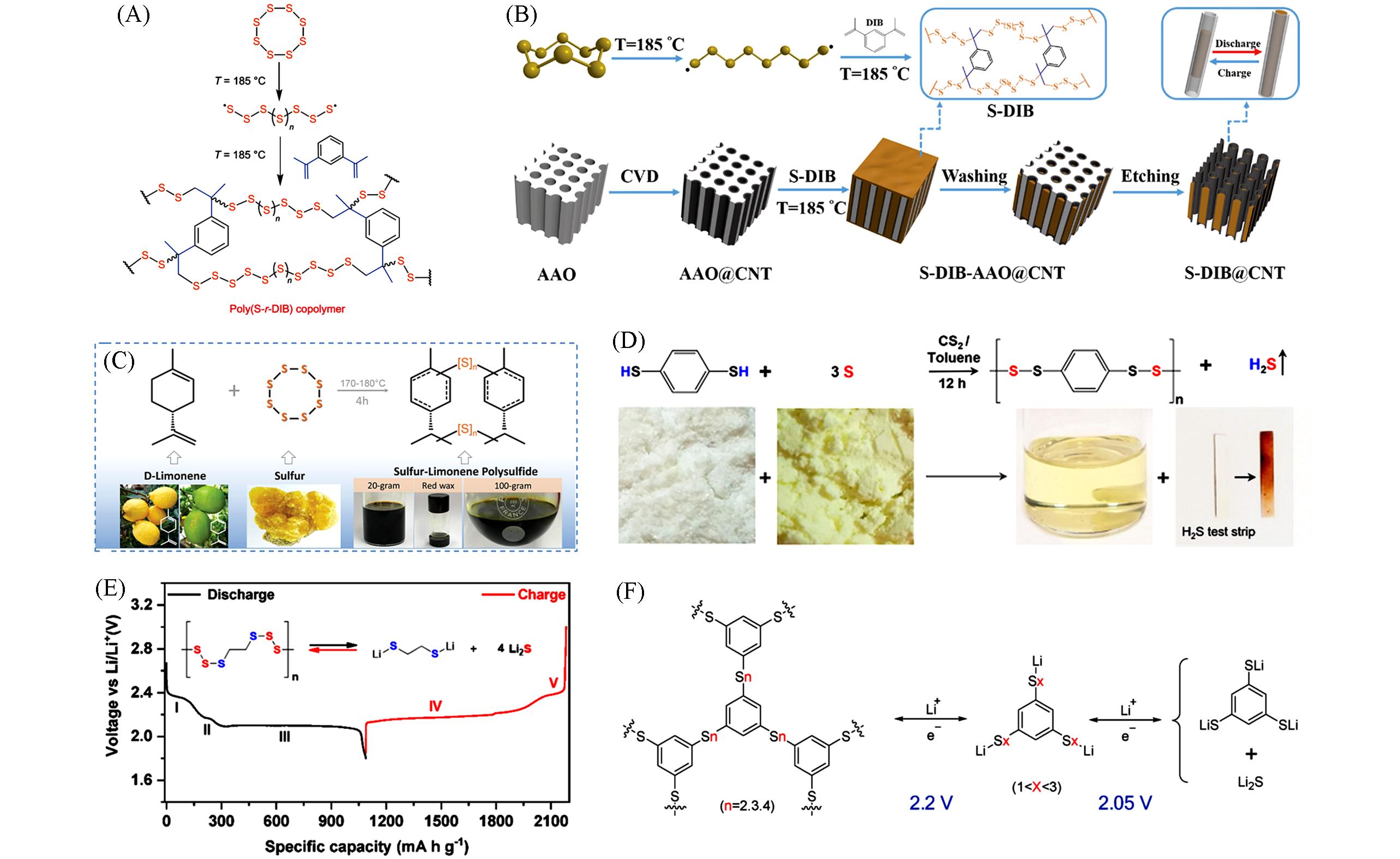
Fig.7 Synthesis scheme of the inverse vulcanization between S8 and DIB monomer(A)[57], synthesis scheme of the S⁃DIB@CNT copolymer(B)[60], one⁃pot synthesis of SLP from D⁃limonene and sulfur as raw materials(C)[61], synthesis scheme and the corresponding visual representation of PPTS, color change of lead acetate⁃based test strip verifies the production of H2S gas(D)[62], the charge⁃discharge curve and redox reaction of Li/PEHS batteries(E)[20] and the proposed redox process of Li/BTTP cell(F)[63](A) Copyright 2013, Nature Publishing Group; (B) Copyright 2017, Wiley-VCH; (C) Copyright 2018, Wiley-VCH; (D) Copyright 2018, American Chemical Society; (E) Copyright 2019, American Chemical Society; (F) Copyright 2021, Elsevier.
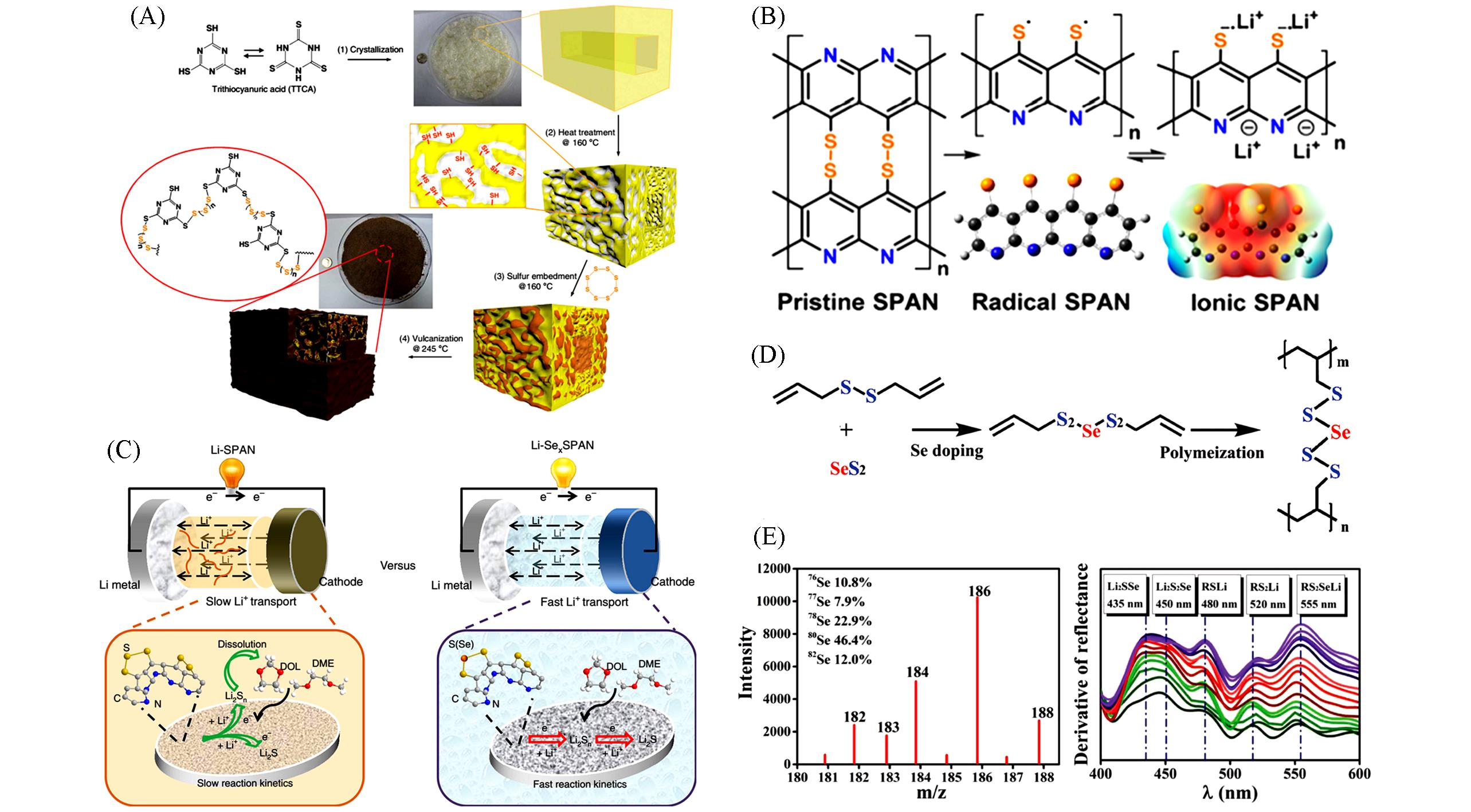
Fig.8 Synthesis of S⁃TTCA(A)[23], the lithiation mechanism of SPAN(B)[72], the proposed reaction mechanism of Li/SPAN and Li/Se x SPAN(C)[26], the synthetic route of PDATtSSe polymer(D), LC⁃MS of PDATtSSe and homologous first⁃order derivatives of the Li/PDATtSSe cell(E)[73](A) Copyright 2015, Nature Publishing Group; (B) Copyright 2018, American Chemical Society; (C) Copyright 2019, Nature Publishing Group; (D, E) Copyright 2017, Wiley-VCH.
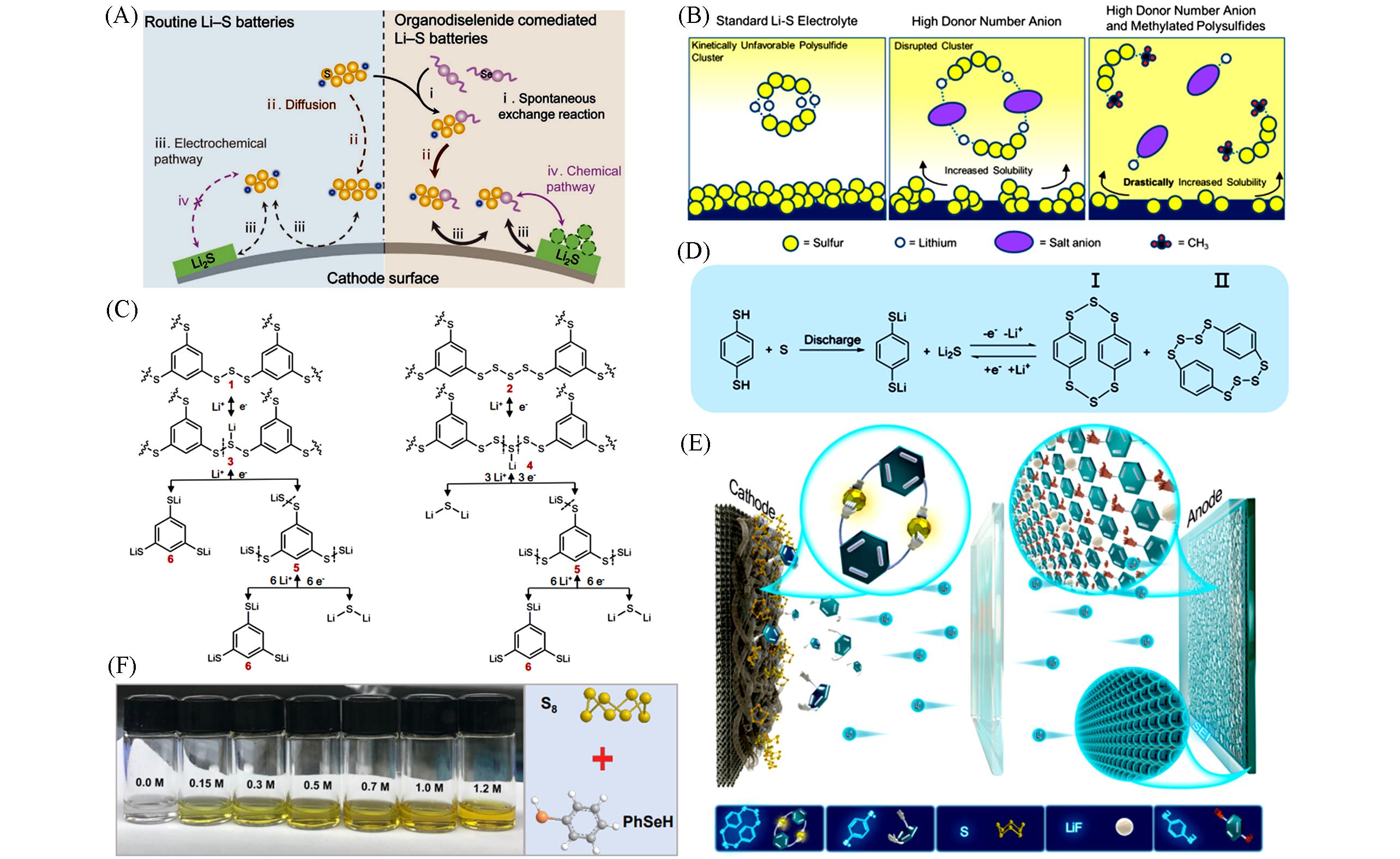
Fig.9 Schematic of routine and organodiselenide⁃comediated reaction pathway for Li⁃S batteries(A)[75], illustration of the theorized improvements to the Li⁃S electrolyte enabled through the presence of high donor number compounds in solution, as well as high donor compounds in conjunction with organosulfur active material(B)[76], reaction mechanism for the Li⁃S battery with BTT electrolyte(C)[33], reaction mechanism(D) and schematic illustration(E) for the Li⁃S battery with 1,4⁃BDT electrolyte[34] and the optical images and the structure of S8 and PhSeH(F)[77](A) Copyright 2021, Wiley-VCH; (B) Copyright 2021, American Chemical Society; (C) Copyright 2021, Nature Publishing Group; (D, E) Copyright 2021, American Chemical Society; (F) Copyright 2022, Tsinghua University Press.
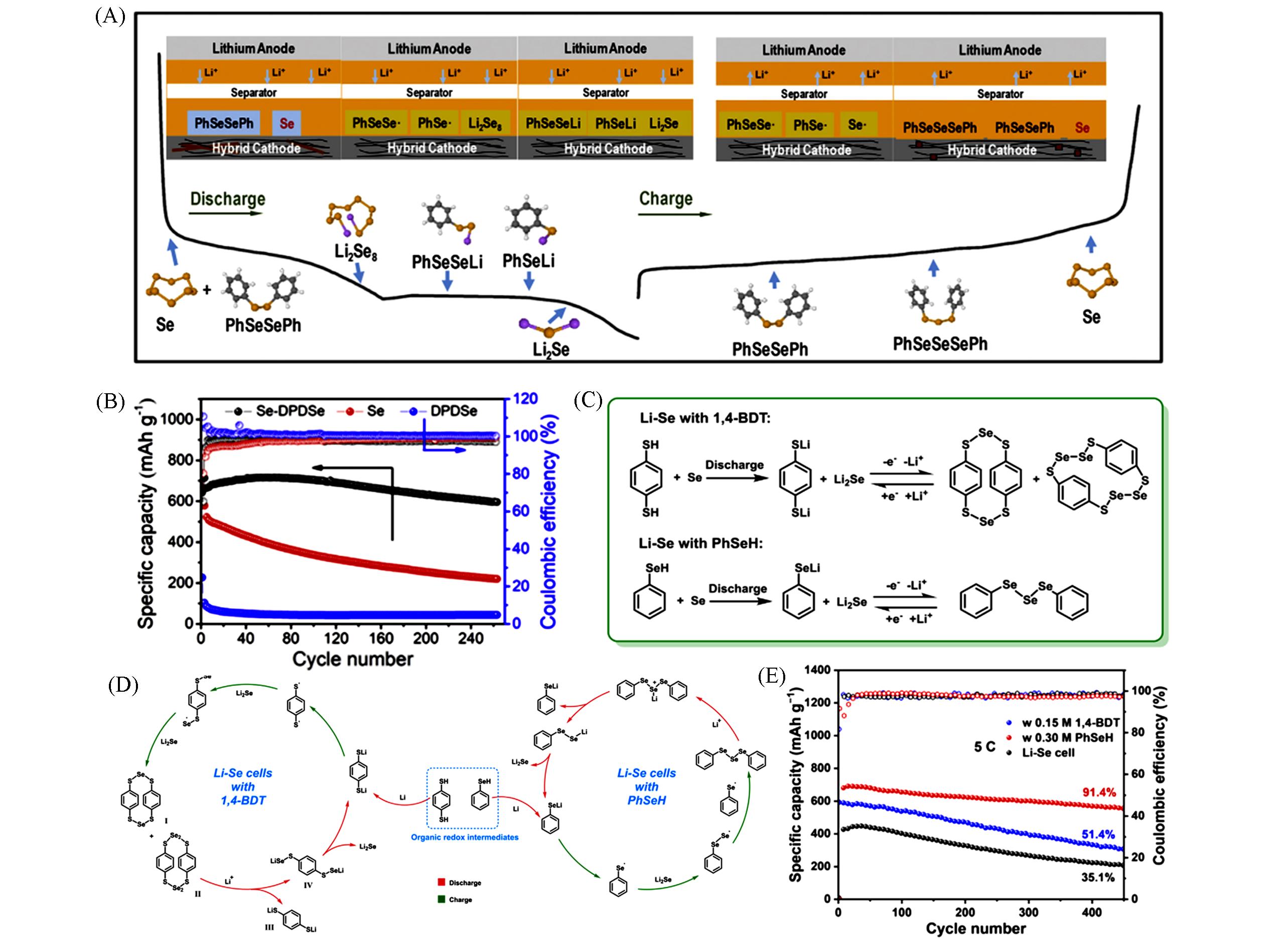
Fig.10 Scheme of proposed redox reactions of the Li/Se⁃DPDSe cell(A), cycling performance of Li/DPDSe, Li/Se, and Li/Se⁃DPDSe cells(B)[78], proposed electrochemical reaction pathways of the Li⁃Se cells with 1,4⁃BDT and PhSeH(C), reaction mechanism for the Li⁃Se cells with 1,4⁃BDT and PhSeH electrolyte(D) and long⁃term cycle performance of the Li⁃Se cells with/without additives at 5C(E)[79](A, B) Copyright 2020, Elsevier; (C—E) Copyright 2022, American Institute of Physics.
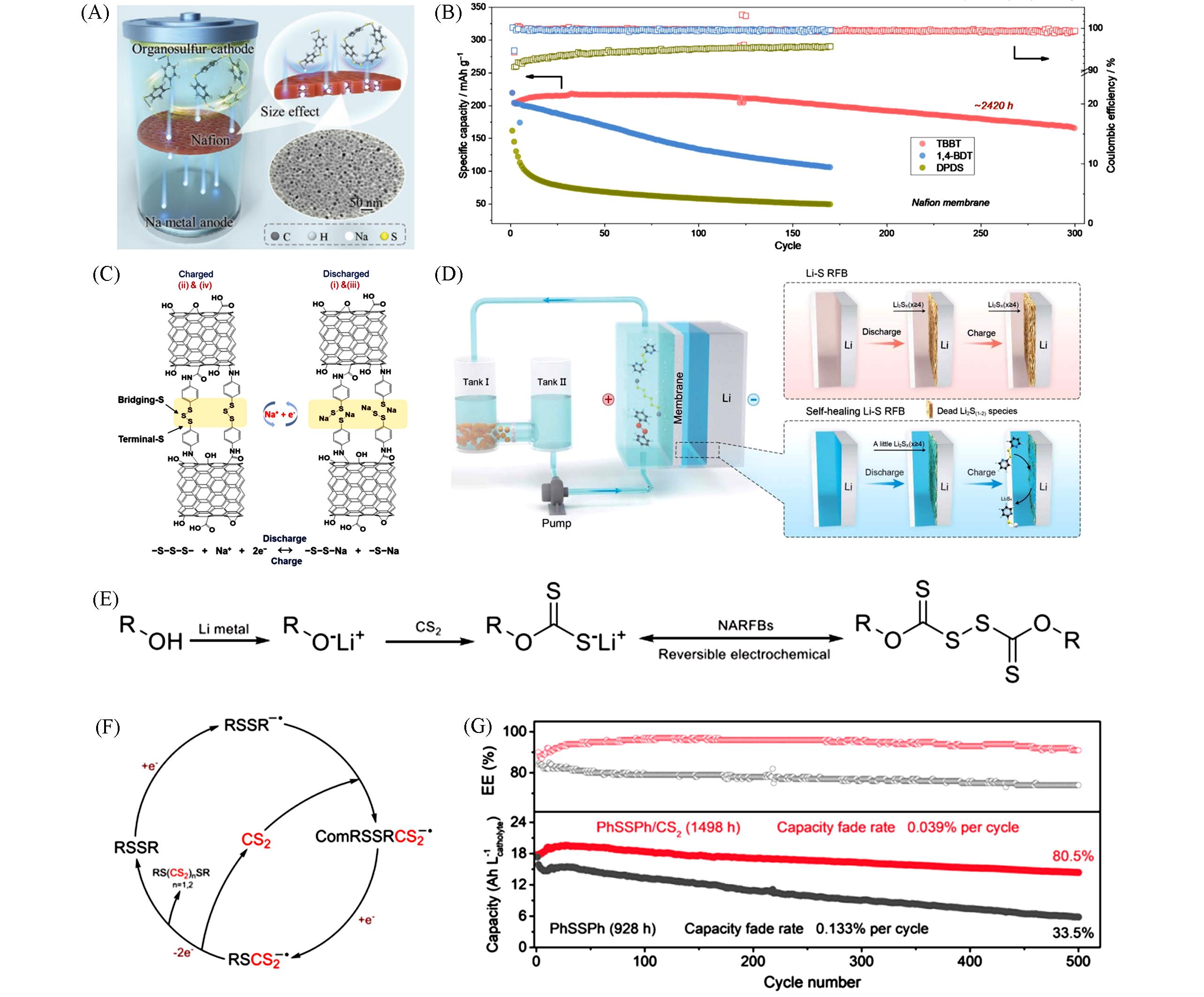
Fig.11 Cell configuration with the size effect of organosulfur(A), the long⁃term cycle performance of the three Na⁃organosulfur cells with Nafion membranes(B)[86], proposed redox mechanism of SOS⁃OCNT(C)[87], schematic illustration of a Li⁃S RFB with DpyDS and DPDSe as adjuvants(D)[89], SFE and application of xanthogen disulfides in NARFBs(E)[90], the redox⁃mediating mechanism of CS2 for RSSR(F) and cycling performance of PhSSPh and PhSSPh/CS2 at 0.2 mA/cm2(G)[91](A, B) Copyright 2021, Wiley-VCH; (C) Copyright 2020, American Chemical Society; (D) Copyright 2021, the Royal Society of Chemistry; (E) Copyright 2022, Wiley-VCH; (F, G) Copyright 2022, National Academy of Sciences.
| 87 | Jana M., Park J. M., Kota M., Shin K. H., Rana H. H., Nakhanivej P., Huang J. Q., Park H. S., ACS Energy Lett., 2020, 6(1), 280—289 |
| 88 | Jia C., Pan F., Zhu Y. G., Huang Q., Lu L., Wang Q., Sci. Adv., 2015, 1(10), e1500886 |
| 89 | Chen Q., Guo W., Wang D., Fu Y., J. Mater. Chem. A, 2021, 9(21), 12652—12658 |
| 90 | Chen Q., Guo W., Fu Y., Adv. Sci., 2022, 9(1), e2104036 |
| 91 | Chen Q., Wang W., Li X., Guo W., Fu Y., Proc. Natl. Acad. Sci. USA, 2022, 119(22), e2202449119 |
| 1 | Dunn B., Kamath H., Tarascon J. M., Science, 2011, 334(6058), 928—935 |
| 2 | Goodenough J. B., Kim Y., Chem. Mater., 2010, 22(3), 587—603 |
| 3 | Manthiram A., J. Phys. Chem. Lett., 2011, 2(3), 176—184 |
| 4 | Manthiram A., Nat. Commun., 2020, 11(1), 1550 |
| 5 | Manthiram A., Fu Y., Chung S. H., Zu C., Su Y. S., Chem. Rev., 2014, 114(23), 11751—11787 |
| 6 | Manthiram A., Chung S. H., Zu C., Adv. Mater., 2015, 27(12), 1980—2006 |
| 7 | Yin Y. X., Xin S., Guo Y. G., Wan L. J., Angew. Chem. Int. Ed., 2013, 52(50), 13186—13200 |
| 8 | Cheng X. B., Zhang R., Zhao C. Z., Zhang Q., Chem. Rev., 2017, 117(15), 10403—10473 |
| 9 | Zheng G., Yang Y., Cha J. J., Hong S. S., Cui Y., Nano Lett., 2011, 11(10), 4462—4467 |
| 10 | Cheng X. B., Huang J. Q., Zhang Q., Peng H. J., Zhao M. Q., Wei F., Nano Energy, 2014,4 , 65—72 |
| 11 | Xin S., Gu L., Zhao N. H., Yin Y. X., Zhou L. J., Guo Y. G., Wan L. J., J. Am. Chem. Soc., 2012, 134(45), 18510—18513 |
| 12 | Zhang Y., Si Y., Guo W., Li X., Tang S., Zhang Z., Wang X., Fu Y., ACS Appl. Energy Mater., 2021, 4(9), 10104—10113 |
| 13 | Chen T., Zhang Z., Cheng B., Chen R., Hu Y., Ma L., Zhu G., Liu J., Jin Z., J. Am. Chem. Soc., 2017, 139(36), 12710—12715 |
| 14 | Tao Y., Wei Y., Liu Y., Wang J., Qiao W., Ling L., Long D., Energy Environ. Sci., 2016, 9(10), 3230—3239 |
| 15 | Deng D. R., Xue F., Jia Y. J., Ye J. C., Bai C. D., Zheng M. S., Dong Q. F., ACS Nano, 2017, 11(6), 6031—6039 |
| 16 | Liu X., Huang J. Q., Zhang Q., Mai L., Adv. Mater., 2017, 29(20), 1601759 |
| 17 | Visco S. J., DeJonghe L. C., J. Electrochem. Soc., 1988, 135(12), 2905—2909 |
| 18 | Wang D. Y., Guo W., Fu Y., Acc. Chem. Res., 2019, 52(8), 2290—2300 |
| 19 | Guo W., Wang D. Y., Chen Q., Fu Y., Adv. Sci., 2022, 9(4), 2103989 |
| 20 | Bhargav A., Chang C. H., Fu Y., Manthiram A., ACS Appl. Mater. Interfaces, 2019, 11(6), 6136—6142 |
| 21 | Wu M., Cui Y., Bhargav A., Losovyj Y., Siegel A., Agarwal M., Ma Y., Fu Y., Angew. Chem. Int. Ed., 2016, 55(34), 10027— 10031 |
| 22 | Guo W., Wawrzyniakowski Z. D., Cerda M. M., Bhargav A., Pluth M. D., Ma Y., Fu Y., Chem. Eur. J., 2017, 23(67), 16941— 16947 |
| 23 | Kim H., Lee J., Ahn H., Kim O., Park M. J., Nat. Commun., 2015, 6, 7278 |
| 24 | Wang D. Y., Si Y., Li J., Fu Y., J. Mater. Chem. A, 2019, 7(13), 7423—7429 |
| 25 | Wang D. Y., Si Y., Guo W., Fu Y., Nat. Commun., 2021, 12(1), 3220 |
| 26 | Chen X., Peng L., Wang L., Yang J., Hao Z., Xiang J., Yuan K., Huang Y., Shan B., Yuan L., Xie J., Nat. Commun., 2019, 10(1), 1021 |
| 27 | Fan Q., Guo W., Si Y., Wang X., Wang B., Fu Y., Adv. Funct. Mater., 2020, 31(2), 2001493 |
| 28 | Li F., Guo W., Si Y., Li Z., Fu Y., Electrochim. Acta, 2021, 370, 137757 |
| 29 | Wu H., Zhu L., Fu Y., J. Power Sources, 2023, 555, 232355 |
| 30 | Wang Z., Li X., Guo W., Fu Y., Adv. Funct. Mater., 2021, 31(16), 2009875 |
| 31 | Lv X., Guo W., Song J., Fu Y., Small, 2022, 18(1), e2105071 |
| 32 | Ren S., Sang P., Guo W., Fu Y., Polym. Chem., 2022, 13(40), 5676—5690 |
| 33 | Guo W., Zhang W., Si Y., Wang D., Fu Y., Manthiram A., Nat. Commun., 2021, 12(1), 3031 |
| 34 | Lian J., Guo W., Fu Y., J. Am. Chem. Soc., 2021, 143(29), 11063—11071 |
| 35 | Bhargav A., Patil S. V., Fu Y., Sustainable Energy Fuels, 2017, 1(5), 1007—1012 |
| 36 | Wu M., Bhargav A., Cui Y., Siegel A., Agarwal M., Ma Y., Fu Y., ACS Energy Lett., 2016, 1(6), 1221—1226 |
| 37 | Lv X., Yang Q., Zhang X., Song J., Guo W., Wang Q., Angew. Chem. Int. Ed., 2022, 61(49), e202213160 |
| 38 | Bhargav A., Bell M. E., Karty J., Cui Y., Fu Y., ACS Appl. Mater. Interfaces, 2018, 10(25), 21084—21090 |
| 39 | Guo W., Bhargav A., Ackerson J. D., Cui Y., Ma Y., Fu Y., Chem. Commun., 2018, 54(64), 8873—8876 |
| 40 | Chen Q., Si Y., Guo W., Fu Y., Chem. Commun., 2022, 58(78), 10993—10996 |
| 41 | Gao M., Lan J., Fu Y., Guo W., ChemSusChem, 2022, 15(11), e202200423 |
| 42 | Bhargav A., Ma Y., Shashikala K., Cui Y., Losovyj Y., Fu Y., J. Mater. Chem. A, 2017, 5(47), 25005—25013 |
| 43 | Fan Q., Si Y., Guo W., Fu Y., J. Phys. Chem. Lett., 2021, 12(2), 900—906 |
| 44 | Wang D. Y., Si Y., Guo W., Fu Y., Adv. Sci., 2020, 7(4), 1902646 |
| 45 | Chen Q., Li L., Wang W., Li X., Guo W., Fu Y., J. Am. Chem. Soc., 2022, 144(41), 18918—18926 |
| 46 | Li F., Si Y., Liu B., Li Z., Fu Y., Adv. Funct. Mater., 2019, 29(32), 1902223 |
| 47 | Li F., Si Y., Li Z., Guo W., Fu Y., J. Mater. Chem. A, 2020, 8(1), 87—90 |
| 48 | Pan Q., Lan J., Si Y., Guo W., Fu Y., Chem. Commun., 2022, 58(37), 5602—5605 |
| 49 | Guo W., Fu Y., Chem. Eur. J., 2020, 26(59), 13322—13331 |
| 50 | Cui Y., Ackerson J. D., Ma Y., Bhargav A., Karty J. A., Guo W., Zhu L., Fu Y., Adv. Funct. Mater., 2018, 28(31), 1801791 |
| 51 | Zhao J., Si Y., Han Z., Li J., Guo W., Fu Y., Angew. Chem. Int. Ed., 2020, 59(7), 2654—2658 |
| 52 | Abouimrane A., Dambournet D., Chapman K. W., Chupas P. J., Weng W., Amine K., J. Am. Chem. Soc., 2012, 134(10), 4505—4508 |
| 53 | Ren S., Si Y., Guo W., Fu Y., ACS Sustainable Chem. Eng., 2022, 10(23), 7526—7535 |
| 54 | Clough A. J., Yoo J. W., Mecklenburg M. H., Marinescu S. C., J. Am. Chem. Soc., 2015, 137(1), 118—121 |
| 55 | Al Salem H., Babu G., Rao C. V., Arava L. M., J. Am. Chem. Soc., 2015, 137(36), 11542—11545 |
| 56 | Rout C. S., Kim B. H., Xu X., Yang J., Jeong H. Y., Odkhuu D., Park N., Cho J., Shin H. S., J. Am. Chem. Soc., 2013, 135(23), 8720—8725 |
| 57 | Chung W. J., Griebel J. J., Kim E. T., Yoon H., Simmonds A. G., Ji H. J., Dirlam P. T., Glass R. S., Wie J. J., Nguyen N. A., Guralnick B. W., Park J., Somogyi A., Theato P., Mackay M. E., Sung Y. E., Char K., Pyun J., Nat. Chem., 2013, 5(6), 518—524 |
| 58 | Simmonds A. G., Griebel J. J., Park J., Kim K. R., Chung W. J., Oleshko V. P., Kim J., Kim E. T., Glass R. S., Soles C. L., Sung Y. E., Char K., Pyun J., ACS Macro Lett., 2014, 3(3), 229—232 |
| 59 | Wei Y., Li X., Xu Z., Sun H., Zheng Y., Li S., Peng L., Li Z., Liu Z., Hu X., Zhao X., Huang T., Zhao B., Xi J., Gao C., Gao M., Polym. Chem., 2015, 6, 973—982 |
| 60 | Hu G., Sun Z., Shi C., Fang R., Chen J., Hou P., Liu C., Cheng H. M., Li F., Adv. Mater., 2017, 29(11), 1603835 |
| 61 | Wu F., Chen S., Srot V., Huang Y., Sinha S. K., Aken P. A. V., Maier J., Yu Y., Adv. Mater., 2018, 30(13), 1706643 |
| 62 | Bhargav A., Bell M. E., Cui Y., Fu Y., ACS Appl. Energy Mater., 2018, 1(11), 5859—5864 |
| 63 | Sang P., Song J., Guo W., Fu Y., Chem. Eng. J., 2021, 415, 129043 |
| 64 | Sang P., Si Y., Fu Y., Chem. Commun., 2019, 55(33), 4857—4860 |
| 65 | Li Q., Song Y., Xu R., Zhang L., Gao J., Xia Z., Tian Z., Wei N., Rummeli M. H., Zou X., Sun J., Liu Z., ACS Nano, 2018, 12(10), 10240—10250 |
| 66 | Zhang H., Zou M., Zhao W., Wang Y., Chen Y., Wu Y., Dai L., Cao A., ACS Nano, 2019, 13(4), 3982—3991 |
| 67 | Ren J., Zhou Y., Xia L., Zheng Q., Liao J., Long E., Xie F., Xu C., Lin D., J. Mater. Chem. A, 2018, 6(28), 13835—13847 |
| 68 | Ahmed M. S., Lee S., Agostini M., Jeong M. G., Jung H. G., Ming J., Sun Y. K., Kim J., Hwang J. Y., Adv. Sci., 2021, 8(21), e2101123 |
| 69 | Wang J., Yang J., Xie J., Xu N., Adv. Mater., 2002, 14(13/14), 963—965 |
| 70 | Yu X. G., Xie J. Y., Yang J., Huang H. J., Wang K., Wen Z. S., J. Electroanal. Chem., 2004, 573, 121—128 |
| 71 | Fanous J., Wegner M., Grimminger J., Andresen Ä., Buchmeiser M. R., Chem. Mater., 2011, 23(22), 5024—5028 |
| 72 | Wang W., Cao Z., Elia G. A., Wu Y., Wahyudi W., Abou⁃Hamad E., Emwas A. H., Cavallo L., Li L. J., Ming J., ACS Energy Lett., 2018, 3(12), 2899—2907 |
| 73 | Zhou J., Qian T., Xu N., Wang M., Ni X., Liu X., Shen X., Yan C., Adv. Mater., 2017, 29(33), 1701294 |
| 74 | Lee S., Lee H., Ha N., Lee J. T., Jung J., Eom K., Adv. Funct. Mater., 2020, 30(19), 2000028 |
| 75 | Zhao M., Chen X., Li X. Y., Li B. Q., Huang J. Q., Adv. Mater., 2021, 33(13), 2007298 |
| 76 | Gupta A., Bhargav A., Manthiram A., ACS Energy Lett., 2021, 6(1), 224—231 |
| 77 | Sun J., Zhang K., Fu Y., Guo W., Nano Res., 2023, 16(3), 3814—3822 |
| 78 | Zhao J., Guo W., Fu Y., Mater. Today Energy, 2020, 17, 100442 |
| 79 | Xie K., Sun J., Lian J., Fu Y., Guo W., Appl. Phys. Lett., 2022, 121(13), 133904 |
| 80 | Ghosh A., Shukla S., Monisha M., Kumar A., Lochab B., Mitra S., ACS Energy Lett., 2017, 2(10), 2478—2485 |
| 81 | Park C. W., Ahn J. H., Ryu H. S., Kim K. W., Ahnc H. J., Electrochem. Solid⁃State Lett., 2006, 9(3), A123—A125 |
| 82 | Park C. W., Ryu H. S., Kim K. W., Ahn J. H., Lee J. Y., Ahn H. J., J. Power Sources, 2007, 165(1), 450—454 |
| 83 | Wang J., Yang J., Nuli Y., Holze R., Electrochem. Commun., 2007, 9(1), 31—34 |
| 84 | Kim J. S., Ahn H. J., Kim I. P., Kim K. W., Ahn J. H., Park C. W., Ryu H. S., J. Solid State Electrochem., 2008, 12(7/8), 861— 865 |
| 85 | Zhou D., Chen Y., Li B., Fan H., Cheng F., Shanmukaraj D., Rojo T., Armand M., Wang G., Angew. Chem. Int. Ed., 2018, 57(32), 10168—10172 |
| 86 | Tang S., Chen Q., Si Y., Guo W., Mao B., Fu Y., Adv. Mater., 2021, 33(33), e2100824 |
| [1] | 赵欣悦, 汪靖伦, 闫晓丹, 张灵志. 腈基功能化有机硅电解液添加剂对LiFePO4电池低温性能的影响[J]. 高等学校化学学报, 2019, 40(6): 1258. |
| [2] | 艾淑娟, 宗成星, 吴为, 冯京京, 金灿, 付凤至, 刘靖, 孙冬兰, 郑琴, 郭也平. 环亚硫酸甘油酯衍生物作为电解液添加剂对锂离子电池电化学性能的影响[J]. 高等学校化学学报, 2018, 39(11): 2520. |
| [3] | 郭梦雅, 宗成星, 艾淑娟, 付凤至, 王琪, 刘靖, 孙冬兰, 郭艳玲, 郭也平. 新型锂离子电池电解液添加剂的合成与应用[J]. 高等学校化学学报, 2017, 38(10): 1857. |
| [4] | 姜雪, 史楠楠, 张莹, 程魁, 叶克, 王贵领, 曹殿学. LiBOB对Li1.15Ni0.68Mn1.32O4电极电化学行为的影响[J]. 高等学校化学学报, 2015, 36(4): 739. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||