

高等学校化学学报 ›› 2022, Vol. 43 ›› Issue (6): 20220059.doi: 10.7503/cjcu20220059
收稿日期:2022-01-23
出版日期:2022-06-10
发布日期:2022-03-04
通讯作者:
王蕾
E-mail:wang_leicn2021@163.com
基金资助:
LIN Zhongqiao1, CHEN Peipei2, WANG Lei1( )
)
Received:2022-01-23
Online:2022-06-10
Published:2022-03-04
Contact:
WANG Lei
E-mail:wang_leicn2021@163.com
Supported by:摘要:
盐皮质激素受体的过表达或过度激活通常参与心血管疾病的发生发展. 通过拮抗盐皮质激素受体, 抑制其过度激活, 阻断盐皮质激素受体介导的生物学效应, 能够有效治疗相关疾病. 拮抗盐皮质激素受体化合物的化学结构决定了拮抗效应的强弱和临床上的针对性应用. 通过对盐皮质激素受体拮抗剂进行化学结构的优化, 改善其药理特性, 不仅增加了对盐皮质激素受体的选择性, 还降低了不良反应的发生率和严重程度, 同时在心血管疾病治疗方面也有不同侧重. 本文总结了不同化学结构的盐皮质激素受体拮抗剂螺内酯(Spironolactone)、 依普利酮(Eplerenone)和非奈利酮(Finerenone)的作用机制、 药理特性和心血管疾病临床应用的各自优势, 以期在后续临床应用以及新药研发方面提供新的思路.
中图分类号:
TrendMD:
林中樵, 陈佩佩, 王蕾. 不同化学结构盐皮质激素受体拮抗剂在心血管疾病中的应用. 高等学校化学学报, 2022, 43(6): 20220059.
LIN Zhongqiao, CHEN Peipei, WANG Lei. Application of Mineralocorticoid Receptor Antagonists with Different Chemical Structures in Cardiovascular Diseases. Chem. J. Chinese Universities, 2022, 43(6): 20220059.
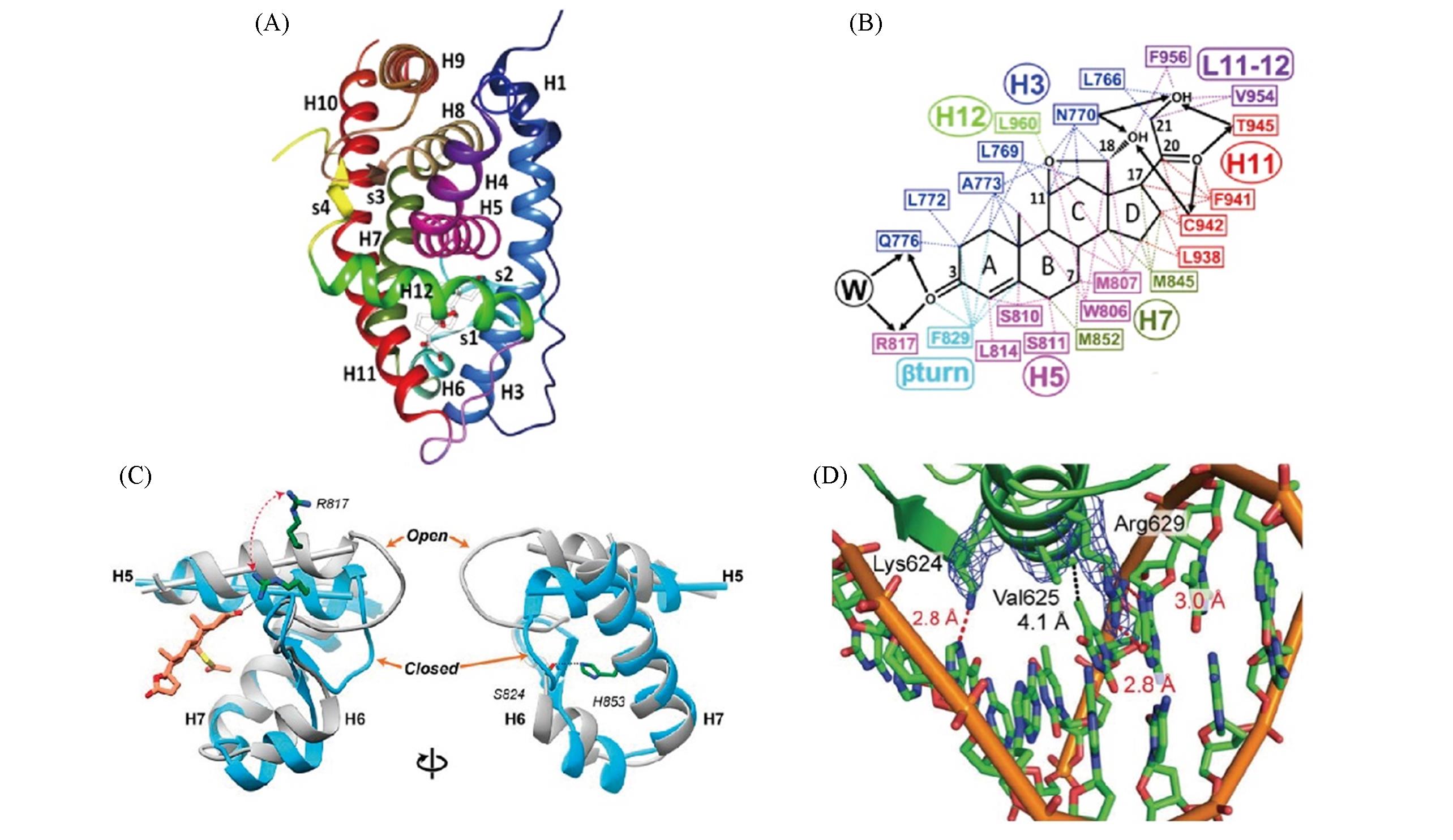
Fig.2 Spatial structures of MR?LBD and MR?DBD(A) Three-dimensional structure of MR-LBD[12]; (B) Amino acid residues of aldosterone[12], Copyright 2012, Elsevier; (C) Relocation of H5 helix of MR-LBD, then MR from antagonism state to agonist state[14], Copyright 2019, PNAS; (D) Schematic diagram of DBD recognition specific DNA sequence[16], Copyright 2014, Plos one.
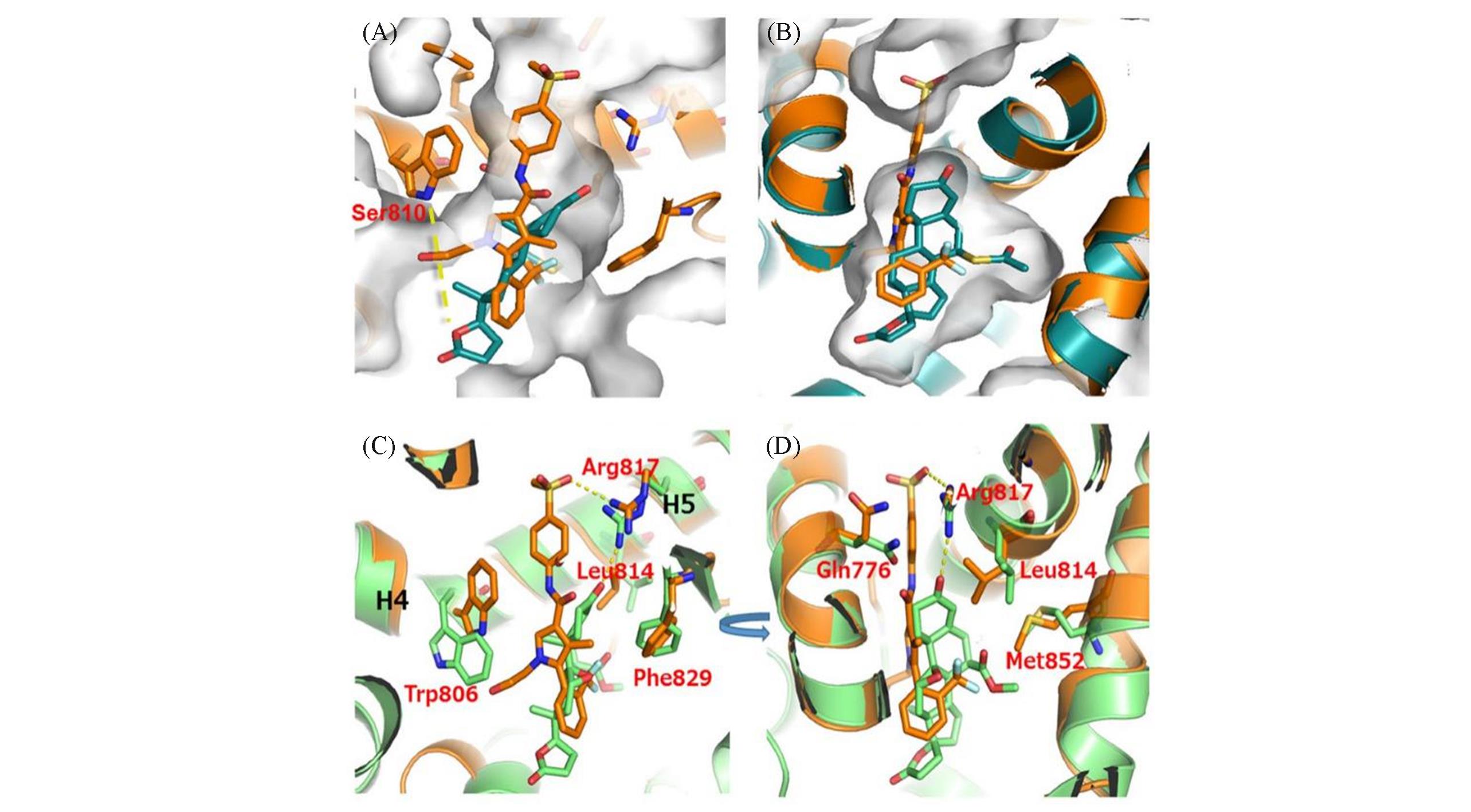
Fig.4 Schematic diagram of the binding sites between mineralocorticoid receptor antagonists and MR?LBD[29](A) Superimposition of MR-LBD/spironolactone (dark green); (B) Superimposition of MR-LBD /esaxerenone(orange);(C) Rearrangement of residues surrounding esaxerenone; (D) Residues from MR-LBD/esaxerenone(orange) and MR-LBD/eplerenone(light green) are shown in the stick model. In the picture, orange: esaxerenone, dark green: spironolactone, light green: eplerenone. Copyright 2020, John Wiley and Sons.
| Siprolactone | Eplerenone | Finerenone | |
|---|---|---|---|
| Chemical type | Steroid | Steroid | Non?steroid |
Structural feature | Based on cyclopentane phenanthrene, coupling acetyl sulfhydryl on C?7 | Similar to siprolactone, acetyl sulfhydryl replaced with carbomethoxy group on C?7,9,11?epoxide was added | Based on 1,4?dihydro pyridines |
Half inhibitory concentration (IC50, nmol/L) | 24 | 990 | 18 |
| Oral bioavailability(%) | 80—90 | 69 | N/A |
| Plasma protein binding rate(%) | ? 90 | 50 | N/A |
| Plasma half?life(h) | 12—24 | 3—4 | 2 |
| Active metabolite | Mostly canrenone | N/A | N/A |
| Selectivity to MR | Low | ? 100 fold compared to siprolactone | ? 500 fold compared to siprolactone |
| Metabolic pathway | Liver | Liver | Liver |
| Tissue distribution | Renal concentration six?fold higher than heart | Renal concentration three?fold higher than heart | Equal concentration in kidney and heart |
Table 1 Pharmacokinetic differences between three MR antagonistic compounds[38~40]
| Siprolactone | Eplerenone | Finerenone | |
|---|---|---|---|
| Chemical type | Steroid | Steroid | Non?steroid |
Structural feature | Based on cyclopentane phenanthrene, coupling acetyl sulfhydryl on C?7 | Similar to siprolactone, acetyl sulfhydryl replaced with carbomethoxy group on C?7,9,11?epoxide was added | Based on 1,4?dihydro pyridines |
Half inhibitory concentration (IC50, nmol/L) | 24 | 990 | 18 |
| Oral bioavailability(%) | 80—90 | 69 | N/A |
| Plasma protein binding rate(%) | ? 90 | 50 | N/A |
| Plasma half?life(h) | 12—24 | 3—4 | 2 |
| Active metabolite | Mostly canrenone | N/A | N/A |
| Selectivity to MR | Low | ? 100 fold compared to siprolactone | ? 500 fold compared to siprolactone |
| Metabolic pathway | Liver | Liver | Liver |
| Tissue distribution | Renal concentration six?fold higher than heart | Renal concentration three?fold higher than heart | Equal concentration in kidney and heart |
| 1 | Kolkhof P., Bärfacker L., J. Endocrinol., 2017, 234(1), T125—T140 |
| 2 | Ouvrard⁃Pascaud A., Sainte⁃Marie Y., Bénitah J. P., Perrier R., Soukaseum C., Cat A. N. D., Royer A., Le Quang K., Charpentier F., Demolombe S., Mechta⁃Grigoriou F., Beggah A. T., Maison⁃Blanche P., Oblin M. E., Delcayre C., Fishman G. I., Farman N., Escoubet B., Jaisser F., Circulation, 2005, 111(23), 3025—3033 |
| 3 | Arriza J. L., Weinberger C., Cerelli G., Glaser T. M., Handelin B. L., Housman D. E., Evans R. M., Science, 1987, 237(4812), 268—275 |
| 4 | Ayuzawa N., Fujita T., J. Am. Soc. Nephrol., 2021, 32(2), 279—289 |
| 5 | Cannavo A., Bencivenga L., Liccardo D., Elia A., Marzano F., Gambino G., D'amico M. L., Perna C., Ferrara N., Rengo G., Paolocci N., Oxid. Med. Cell. Longev., 2018, 2018, 1204598 |
| 6 | Gesmundo I., Villanova T., Gargantini E., Arvat E., Ghigo E., Granata R., Frontiers in Endocrinology, 2016, 7, 66 |
| 7 | Otte C., Moritz S., Yassouridis A., Koop M., Madrischewski A., Wiedemann K., Kellnerr M., Neuropsychopharmacology, 2007, 32(1), 232—238 |
| 8 | Kolkhof P., Joseph A., Kintscher U., Pharmacol. Res., 2021, 172, 105859 |
| 9 | Garthwaite S. M., Mcmahon E. G., Mol. Cell. Endocrinol., 2004, 217(1/2), 27—31 |
| 10 | Kagawa C. M., Sturtevant F. M., Van Arman C. G., J. Pharmacol. Exp. Ther., 1959, 126(2), 123—130 |
| 11 | Lavery D. N., McEwan I. J., Biochem. J., 2005, 391, 449—464 |
| 12 | Huyet J., Pinon G. M., Fay M. R., Rafestin⁃Oblin M. E., Fagart J., Mol. Cell Endocrinol., 2012, 350(2), 187—195 |
| 13 | Pippal J. B., Fuller P. J., J. Mol. Endocrinol., 2008, 41(5/6), 405—413 |
| 14 | Fuller P. J., Yao Y. Z., Jin R. T., He S. T., Martín⁃Fernandez B., Young M. J., Smith B. J., Proc. Natl. Acad. Sci. USA, 2019, 116(37), 18578—18583 |
| 15 | Nordqvist A., O'mahony G., Fridén⁃Saxin M., Fredenwall M., Hogner A., Granberg K. L., Aagaard A., Bäckström S., Gunnarsson A., Kaminski T., Xue Y. F., Dellsén A., Hansson E., Hansson P., Ivarsson I., Karlsson U., Bamberg K., Hermansson M., Georgsson J., Lindmark B., Edman K., ChemMedChem, 2017, 12(1), 50—65 |
| 16 | Hudson W. H., Youn C., Ortlund E. A., PLoS One, 2014, 9(9), e107000 |
| 17 | Alzamora R., Michea L., Marusic E. T., Hypertension, 2000, 35(5), 1099—1104 |
| 18 | DuPont J. J., McCurley A., Davel A. P., McCarthy J., Bender S. B., Hong K., Yang Y., Yoo J. K., Aronovitz M., Baur W. E., Christou D. D., Hill M. A., Jaffe I. Z., JCI Insight, 2016, 1(14), e88942 |
| 19 | Marzolla V., Armani A., Mammi C., Moss M. E., Pagliarini V., Pontecorvo L., Antelmi A., Fabbri A., Rosano G., Jaffe I. Z., Caprio M., Int. J. Cardiol., 2017, 232, 233—242 |
| 20 | Voelkl J., Alesutan I., Leibrock C. B., Quintanilla⁃Martinez L., Kuhn V., Feger M., Mia S., Ahmed M. S. E., Rosenblatt K. P., Kuro⁃o M., Lang F., J. Clin. Invest., 2013, 123(2), 812—822 |
| 21 | Messaoudi S., Gravez B., Tarjus A., Pelloux V., Ouvrard⁃Pascaud A., Delcayre C., Samuel J., Launay J. M., Sierra⁃Ramos C., de la Rosa D. A., Clément K., Farman N., Jaisser F., Hypertension, 2013, 61(2), 361—367 |
| 22 | Liao X. J., Tang L.D., Liang Y. W., Geng H. W., Xu S. H., Chem. Res. Chinese Universities, 2011, 27(2), 217—220 |
| 23 | Jia L. H., Zhou L., Du W., Zhou L. N., Zhang M. J., Hou B. H., Bao Y., Wang Z., Yin Q. X., Org. Process Res. Dev., 2018, 22(7), 836—845 |
| 24 | Jiang L., Ye W. T., Su W. K., Yu C. M., Chem. Res. Chinese Universities, 2019, 35(1), 21—25 |
| 25 | Kosaka H., Hirayama K., Yoda N., Sasaki K., Kitayama T., Kusaka H., Matsubara M., Eur. J. Pharmacol., 2010, 635(1—3), 49—55 |
| 26 | Martín⁃Martinez M., Pérez⁃Gordillo F. L., de la Rosa D. A., Rodríguez Y., Gerona⁃Navarro G., González⁃Muniz R., Zhou M.M., J. Med. Chem., 2017, 60(7), 2629—2650 |
| 27 | Amazit L., Le Billan F., Kolkhof P., Lamribet K., Viengchareun S., Fay M. R., Khan J. A., Hillisch A., Lombès M., Rafestin⁃Oblin M. E., Fagart J., J. Biol. Chem., 2015, 290(36), 21876—21889 |
| 28 | Kolkhof P., Borden S. A., Mol. Cell. Endocrinol., 2012, 350(2), 310—317 |
| 29 | Takahashi M., Ubukata O., Homma T., Asoh Y., Honzumi M., Hayashi N., Saito K., Tsuruoka H., Aoki K., Hanzawa H., FEBS Letter, 2020, 594(10), 1615—1623 |
| 30 | Couette B., Lombes M., Baulieu E. E., Rafestin⁃Oblin M. E., Biochem J., 1992, 282(Pt 3), 697—702 |
| 31 | Le Billan F., Khan J. A., Lamribet K., Viengchareun S., Bouligand J., Fagart J., Lombès M., Faseb J., 2015, 29(9), 3977—3989 |
| 32 | Rogerson F. M., Yao Y. H., Smith B. J., Fuller P. J., Clin. Exp. Pharmacol. Physiol., 2004, 31(10), 704—709 |
| 33 | Hultman M. L., Krasnoperova N. V., Li S. Z., Du S., Xia C. S., Dietz J. D., Lala D. S., Welsch D. J., Hu X., Mol. Endocrinol., 2005, 19(6), 1460—1473 |
| 34 | Vergin H., Mahr G., Metz R., Eichinger A., Nitsche V., Martens H., Int. J. Clin. Pharmacol. Ther., 1997, 35(8), 334—340 |
| 35 | Cook C. S., Berry L. M., Bible R. H., Hribar J. D., Hajdu E., Liu N. W., Drug Metab. Dispos., 2003, 31(11), 1448—1455 |
| 36 | Bärfacker L., Kuhl A., Hillisch A., Grosser R., Figueroa⁃Pérez S., Heckroth H., Nitsche A., Ergüden J. K., Gielen⁃Haertwig H., Schlemmer K. H., Mittendorf J., Paulsen H., Platzek J., Kolkhof P., ChemMedChem, 2012, 7(8), 1385—1403 |
| 37 | Heinig R., Gerisch M., Engelen A., Nagelschmitz J., Loewen S., Eur. J. Drug Metab. Pharmacokinetics, 2018, 43(6), 715—727 |
| 38 | Kolkhof P., Jaisser F., Kim S. Y., Filippatos G., Nowack C., Pitt B., Handb. Exp. Pharmacol., 2017, 243, 271—305 |
| 39 | Agarwal R., Kolkhof P., Bakris G., Bauersachs J., Haller H., Wada T., Zannad F., Eur. Heart J., 2021, 42(2), 152—161 |
| 40 | Yang J., Young M. J., Curr. Opin. Pharmacol., 2016, 27, 78—85 |
| 41 | Fraccarollo D., Berger S., Galuppo P., Kneitz S., Hein L., Schütz G., Frantz S., Ertl G., Bauersachs J., Circulation, 2011, 123(4), 400—408 |
| 42 | Shen J. Z., Morgan J., Tesch G. H., Rickard A. J., Chrissobolis S., Drummond G. R., Fuller P. J., Young M. J., Endocrinology, 2016, 157(8), 3213—3223 |
| 43 | Li S., The Role of Aldosterone in Small Vesse1 Fibrosis in the Elderly Hypertensive Rats, Shanxi Medical University, Taiyuan, 2019 |
| 李晟. 醛固酮在老年高血压大鼠小血管纤维化中的作用研究, 太原: 山西医科大学, 2019 | |
| 44 | Li S., Liu Y., Song Q. Y., Li L., Zhao X. F., Li H., Wang L., Acad. J. Sec. Mil. Med. Univ., 2020, 41(12), 1414—1419 |
| 李晟, 刘宇, 宋奇颖, 李丽, 赵晓芳, 李晗, 王蕾. 第二军医大学学报, 2020, 41(12), 1414—1419 | |
| 45 | Menon D. P., Qi G. M., Kim S. K., Moss M. E., Penumatsa K. C., Warburton R. R., Toksoz D., Wilson J., Hill N. S., Jaffe I. Z., Preston I. R., Pulm. Circ., 2021, 11(3), 20458940211025240 |
| 46 | Mayyas F. A., Aljohmani A. I., Alzoubi K. H., Curr. Mol. Pharmacol., 2020, 13(3), 206—215 |
| 47 | Liao C. W., Chou C. H., Wu X. M., Chen Z. W., Chen Y. H., Chang Y. Y., Wu V. C., Rose⁃John S., Hung C. S., Lin Y. H., Wu C. H., Ho Y. L., Chang H. W., Lin L. Y., Hu F. C., Liu K. L., Wang S. M., Huang K. H., Chen Y. M., Kuo C. C., Chang C. C., Liao S. C., Yen R. F., Wu K. D., Biochim. Biophys. Acta Mol. Basis Dis., 2020, 1866(3), 165627 |
| 48 | Chen B., Geng J., Gao S. X., Yue W. W., Liu Q., J. Interferon Cytokine Res., 2018, 38(3), 137—144 |
| 49 | Macdonald J. E., Kennedy N., Struthers A. D., Heart, 2004, 90(7), 765—770 |
| 50 | Hayashi M., Tsutamoto T., Wada A., Tsutsui T., Ishii C., Ohno K., Fujii M., Taniguchi A., Hamatani T., Nozato Y., Kataoka K., Morigami N., Ohnishi M., Kinoshita M., Horie M., Circulation, 2003, 107(20), 2559—2565 |
| 51 | Iraqi W., Rossignol P., Angioi M., Fay R., Nuée J., Ketelslegers J. M., Vincent J., Pitt B., Zannad F., Circulation, 2009, 119(18), 2471—2479 |
| 52 | Stienen S., Rossignol P., Barros A., Girerd N., Pitt B., Zannad F., Ferreira J. P., Clin. Res. Cardiol., 2020, 109(2), 194—204 |
| 53 | Rossignol P., Ménard J., Fay R., Gustafsson F., Pitt B., Zannad F., J. Am. Coll. Cardiol., 2011, 58(19), 1958—1966 |
| 54 | Grune J., Beyhoff N., Smeir E., Chudek R., Blumrich A., Ban Z., Brix S., Betz I. R., Schupp M., Foryst⁃Ludwig A., Klopfleisch R., Stawowy P., Houtman R., Kolkhof P., Kintscher U., Hypertension, 2018, 71(4), 599—608 |
| 55 | Pitt B., Zannad F., Remme W. J., Cody R., Castaigne A., Perez A., Palensky J., Wittes J., N. Engl. J. Med., 1999, 341(10), 709—717 |
| 56 | Rossignol P., Claggett B. L., Liu J. K., Vardeny O., Pitt B., Zannad F., Solomon S., Am. J. Hypertension, 2018, 31(4), 407—414 |
| 57 | Cohen J. B., Schrauben S. J., Zhao L., Basso M. D., Cvijic M. E., Li Z. Y., Yarde M., Wang Z. Q., Bhattacharya P. T., Chirinos D. A., Prenner S., Zamani P., Seiffert D. A., Car B. D., Gordon D. A., Margulies K., Cappola T., Chirinos J. A., JACC Heart Failure, 2020, 8(3), 172—184 |
| 58 | Rossello X., Ariti C., Pocock S. J., Ferreira J. P., Girerd N., Mcmurray J. J. V., Van Veldhuisen D. J., Pitt B., Zannad F., Clin. Res. Cardiol., 2019, 108(5), 477—486 |
| 59 | Montalescot G., Pitt B., de Sa E. L., Hamm C. W., Flather M., Verheugt F., Shi H., Turgonyi E., Orri M., Vincent J., Zannad F., Eur. Heart J., 2014, 35(34), 2295—2302 |
| 60 | Pitt B., Anker S. D., Böhm M., Gheorghiade M., Køber L., Krum H., Maggioni A. P., Ponikowski P., Voors A. A., Zannad F., Nowack C., Kim S. Y., Pieper A., Kimmeskamp⁃Kirschbaum N., Filippatos G., Eur. J. Heart Failure, 2015, 17(2), 224—232 |
| 61 | Filippatos G., Anker S. D., Böhm M., Gheorghiade M., Køber L., Krum H., Maggioni A. P., Ponikowski P., Voors A. A., Zannad F., Kim S. Y., Nowack C., Palombo G., Kolkhof P., Kimmeskamp⁃Kirschbaum N., Pieper A., Pitt B., Eur. Heart J., 2016, 37(27), 2105—2114 |
| 62 | Filippatos G., Anker S. D., Agarwal R., Pitt B., Ruilope L. M., Rossing P., Kolkhof P., Schloemer P., Tornus I., Joseph A., Bakris G. L., Circulation, 2021, 143(6), 540—552 |
| 63 | Diaz⁃Otero J. M., Yen T. C., Fisher C., Bota D., Jackson W. F., Dorrance A. M., Am. J. Physiol. Heart. Circ. Physiol., 2018, 315(5), H1304—H1315 |
| 64 | Armani A., Cinti F., Marzolla V., Morgan J., Cranston G. A., Antelmi A., Carpinelli G., Canese R., Pagotto U., Quarta C., Malorni W., Matarrese P., Marconi M., Fabbri A., Rosano G., Cinti S., Young M. J., Caprio M., FASEB Journal, 2014, 28(8), 3745—3757 |
| [1] | 黄初升, 李秀英, 李瀛, 李裕林. 法呢基苯乙酮及其黄烷酮化合物的全合成[J]. 高等学校化学学报, 1997, 18(11): 1804. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||