

高等学校化学学报 ›› 2021, Vol. 42 ›› Issue (5): 1530.doi: 10.7503/cjcu20200503
收稿日期:2020-07-31
出版日期:2021-05-10
发布日期:2020-10-20
通讯作者:
鲁振达
E-mail:luzhenda@nju.edu.cn
基金资助:
LI Shiheng, WANG Chao, LU Zhenda( )
)
Received:2020-07-31
Online:2021-05-10
Published:2020-10-20
Contact:
LU Zhenda
E-mail:luzhenda@nju.edu.cn
Supported by:摘要:
锂离子二次电池已成为日常生活中不可或缺的一部分, 而现有的锂离子电池并不能完全满足电动汽车领域高能量密度的要求, 发展具有高能量密度的电极材料是解决问题的关键. 硅负极因理论比容量高、 脱嵌锂电位低、 来源广泛等优点而备受关注, 但其巨大的体积变化(约300%)以及低的首次库仑效率阻碍了其商业应用. 预锂化技术可以有效提高首次库仑效率、 实现高性能硅基负极, 本文阐述了预锂化的科学必要性, 介绍了各种预锂化的方法以及优缺点, 最后对硅基负极预锂化应用的挑战和前景进行了展望.
中图分类号:
TrendMD:
李世恒, 王超, 鲁振达. 锂离子电池硅基负极材料的预锂化研究进展. 高等学校化学学报, 2021, 42(5): 1530.
LI Shiheng, WANG Chao, LU Zhenda. Challenges and Recent Progress of Prelithiation for Si-Based Anodes in Lithium-ion Batteries. Chem. J. Chinese Universities, 2021, 42(5): 1530.
| Method | Introduction | Advantages and disadvantages |
|---|---|---|
Stable lithium micron particles(SLMP) | SLMP are directly coated on the silicon electrode or mixed into the silicon anode slurry | Simple operation, controllable degree of prelithiation, but expensive and holes generated in the electrode after cycling |
| Lithium metal additives | Lithium foil is added directly to the silicon electrode | Low cost and simple operation |
| Self?discharge lithiation | Tightly contacting electrode with lithium metal in the presence of electrolyte | No voltage is needed, but it is difficult to precisely control the degree of prelithiation |
| Electrochemical lithiation | Needing temporary battery construction and electric shorting the electrode and lithium metal | Precisely controlling the degree of lithium, but more complicated |
Thermal lithiation | Prelithiating silicon with molten lithium | The degree of prelithiation can be controlled, but the resulting lithium silicon alloy has poor compatibility with conventional solvents |
| Organic?lithium prelithiation | Prelithiating silicon with organic?lithium in solvent | Difficult to control the amount of organic?lithium and requiring an additional solvent cleaning process |
Table 1 A brief introduction and a summary of the advantages and disadvantages of various prelithiation methods
| Method | Introduction | Advantages and disadvantages |
|---|---|---|
Stable lithium micron particles(SLMP) | SLMP are directly coated on the silicon electrode or mixed into the silicon anode slurry | Simple operation, controllable degree of prelithiation, but expensive and holes generated in the electrode after cycling |
| Lithium metal additives | Lithium foil is added directly to the silicon electrode | Low cost and simple operation |
| Self?discharge lithiation | Tightly contacting electrode with lithium metal in the presence of electrolyte | No voltage is needed, but it is difficult to precisely control the degree of prelithiation |
| Electrochemical lithiation | Needing temporary battery construction and electric shorting the electrode and lithium metal | Precisely controlling the degree of lithium, but more complicated |
Thermal lithiation | Prelithiating silicon with molten lithium | The degree of prelithiation can be controlled, but the resulting lithium silicon alloy has poor compatibility with conventional solvents |
| Organic?lithium prelithiation | Prelithiating silicon with organic?lithium in solvent | Difficult to control the amount of organic?lithium and requiring an additional solvent cleaning process |
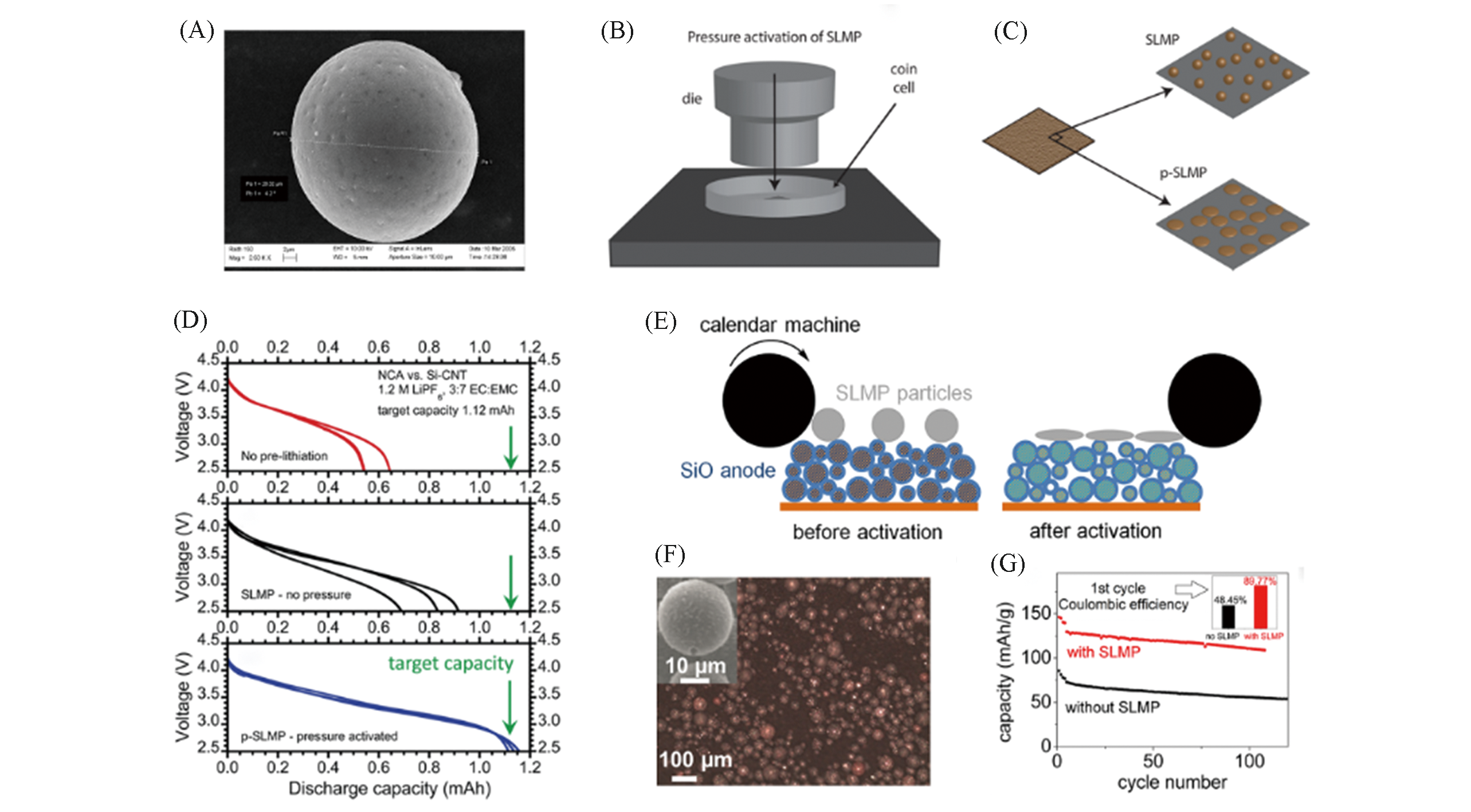
Fig.1 SEM image of SLMP(A)[20], illustration of pressure activated SLMP(B, C), first?cycle charge?discharge curves in different conditions(D)[34], schematics of the utilization of SLMP for the SiO electrode(E), SEM image of SLMP particles loaded on the SiO electrode(F) and full cell performance with or without SLMP additive(G)[36](A) Copyright 2018, MDPI; (B―D) Copyright 2013, American Chemical Society; (E―G) Copyright 2014, American Chemical Society.
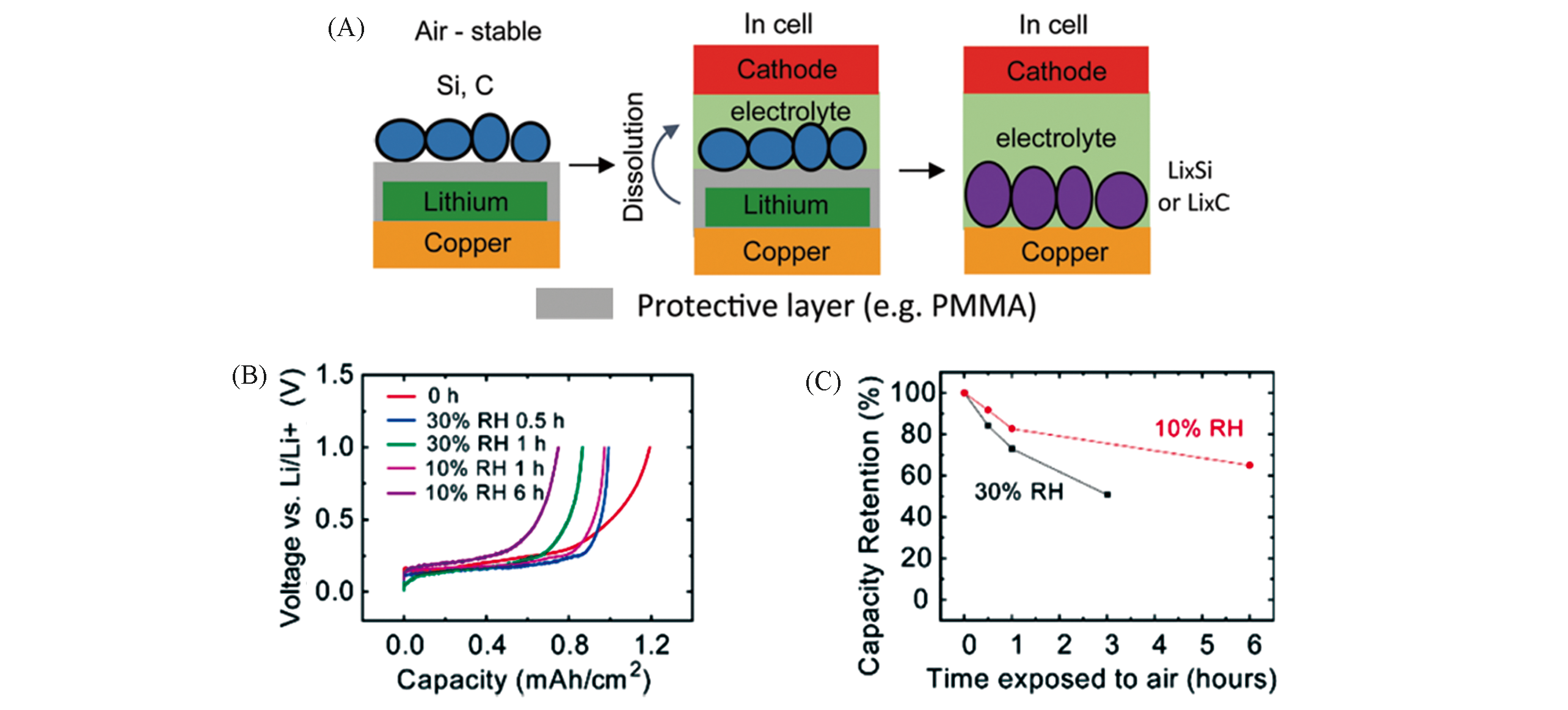
Fig.2 Schematic of the process to prepare ambient?air?stable lithiated anode(A), different curves correspond to exposure to air for various times(B) and amount of active lithium extracted in the first delithiation versus time exposed to air(C)[41]Copyright 2016, American Chemical Society.
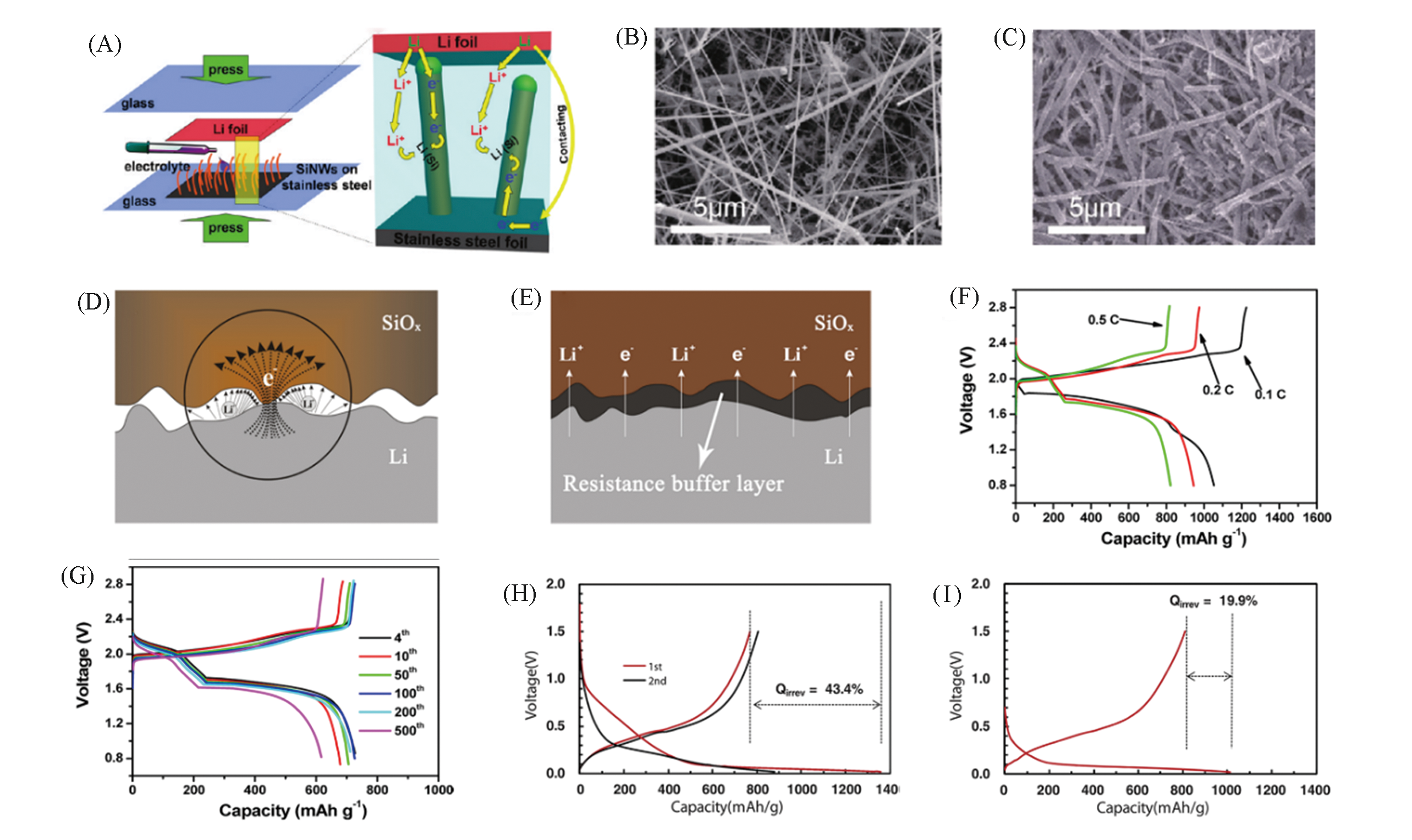
Fig.3 Schematic diagrams showing the prelithiation of SiNWs on stainless steel(SS) foil and the internal electron and Li+ pathways during the prelithiation(A), SEM images of SiNWs before(B) and after 10 min prelithiation(C)[44], illustration of Li?ion and electron transfer in the direct contact prelithiation process(D) and resistance buffer layer(RBL)?regulated prelithiation process(E)[51], voltage profiles of the full cell adopting the lithiated Si?SiOx nanosphere without(F) and with RBL(G)[50], and voltage profiles of Si/C?IWGS(H) and pretreated Si/C?IWGS(I)[45](A―C) Copyright 2011, American Chemical Society; (D), (E) Copyright 2019, American Chemical Society; (F), (G) Copyright 2015, American Chemical Society; (H), (I) Copyright 2014, Wiley?VCH.
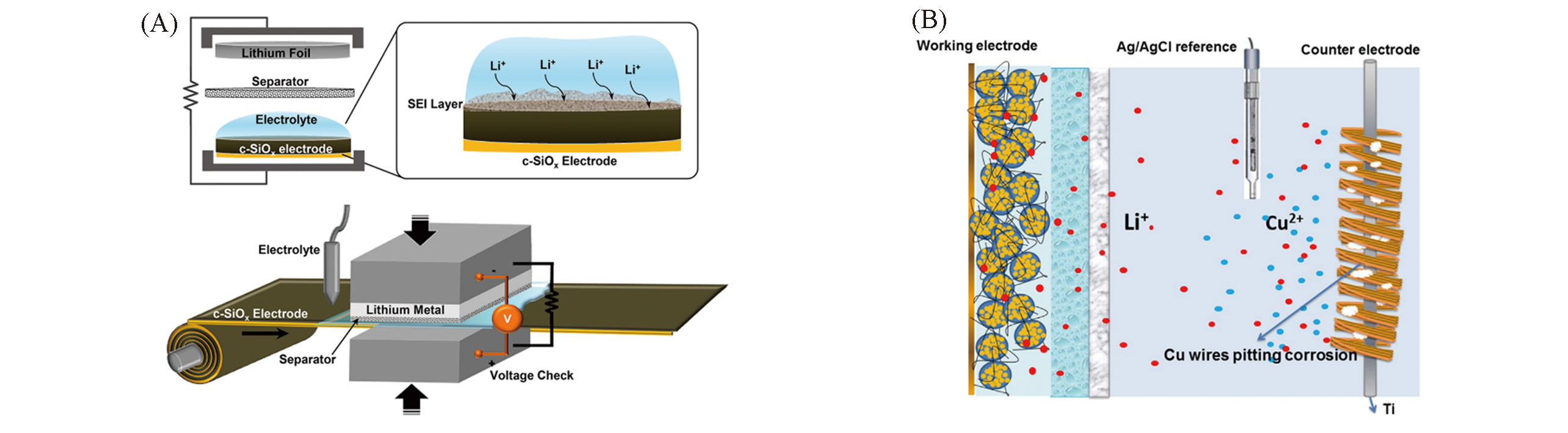
Fig.4 Graphical illustration of prelithiation process of c?SiOx electrode and its scalable roll?to?roll process scheme(A)[52] and schematic diagrams of electrolytic cell with a Cu pitting corrosion type anodic half cell in aqueous electrolyte and a Li?battery type cathodic half cell in gel polymer electrolyte(B)[59](A) Copyright 2016, American Chemical Society; (B) Copyright 2015, Wiley?VCH.
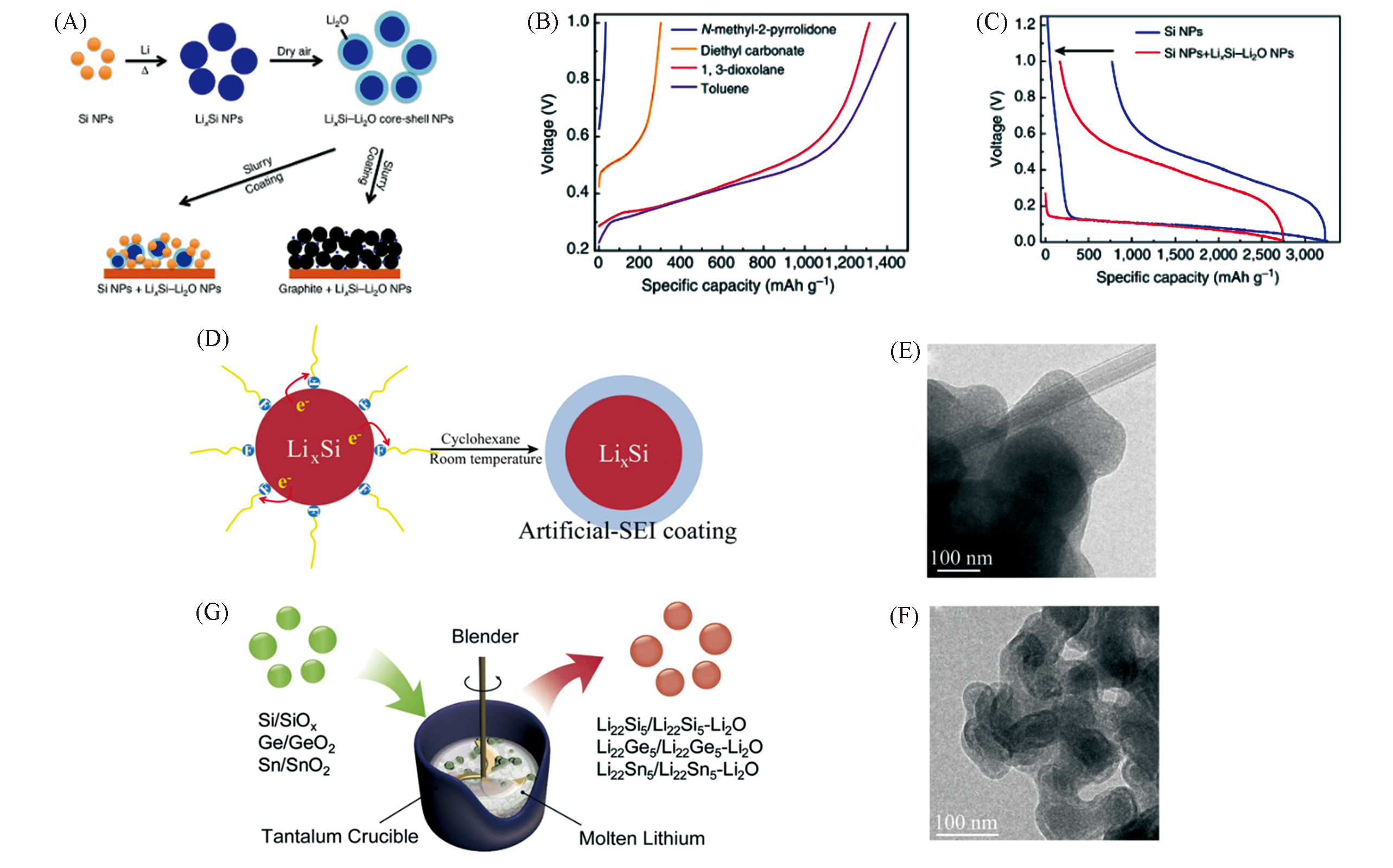
Fig.5 Schematic diagram showing Si NPs react with melted Li to form LixSi NPs(A), first?cycle delithiation capacity of LixSi?Li2O NPs with different solvents(B), first?cycle voltage profiles of Si NPs/LixSi?Li2O and Si NPs(C)[63], schematic diagram of the artificial SEI coating formed on the surface of LixSi NPs(D), TEM images of LixSi NPs before(E) and after coating(F)[60], and schematic diagram showing a one?pot metallurgical process to synthesize Li22Z5 alloys and Li22Z5?Li2O composites(G)[61](A―C) Copyright 2014, Springer Nature; (D―F) Copyright 2015, American Chemical Society; (G) Copyright 2017, Elsevier.
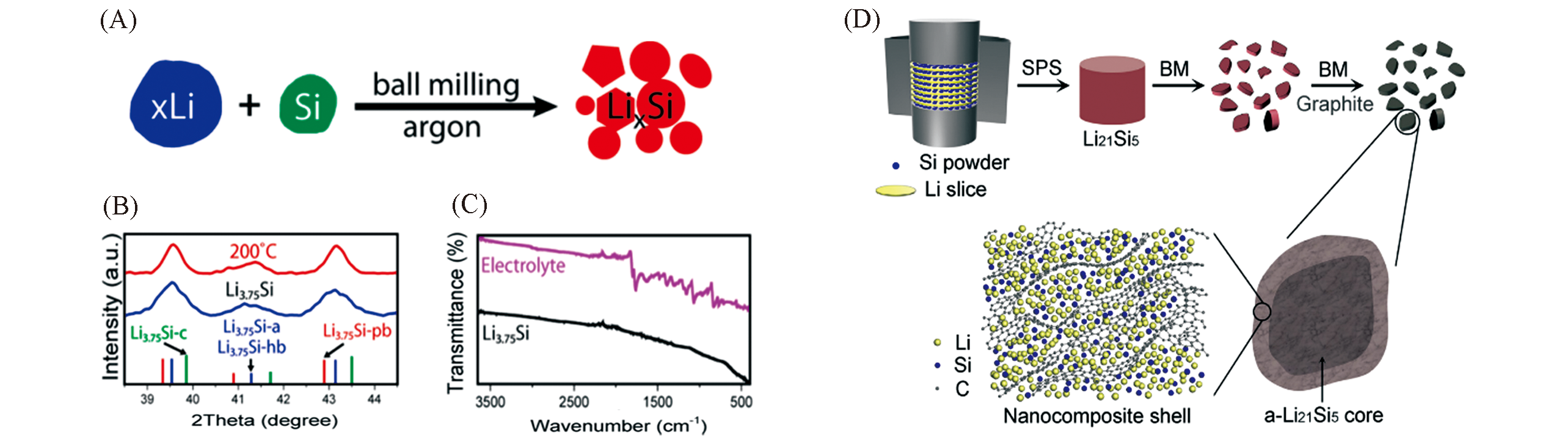
Fig.6 Scheme illustrating the high?energy ball?milling process(A), XRD data(B) and FTIR characte?rization(C)[65] of the high?energy ball?milled products and schematic for design of core?shell structure of a?Li21Si5@C(D)[70](A―C) Copyright 2014, American Chemical Society; (D) Copyright 2018, Elsevier.
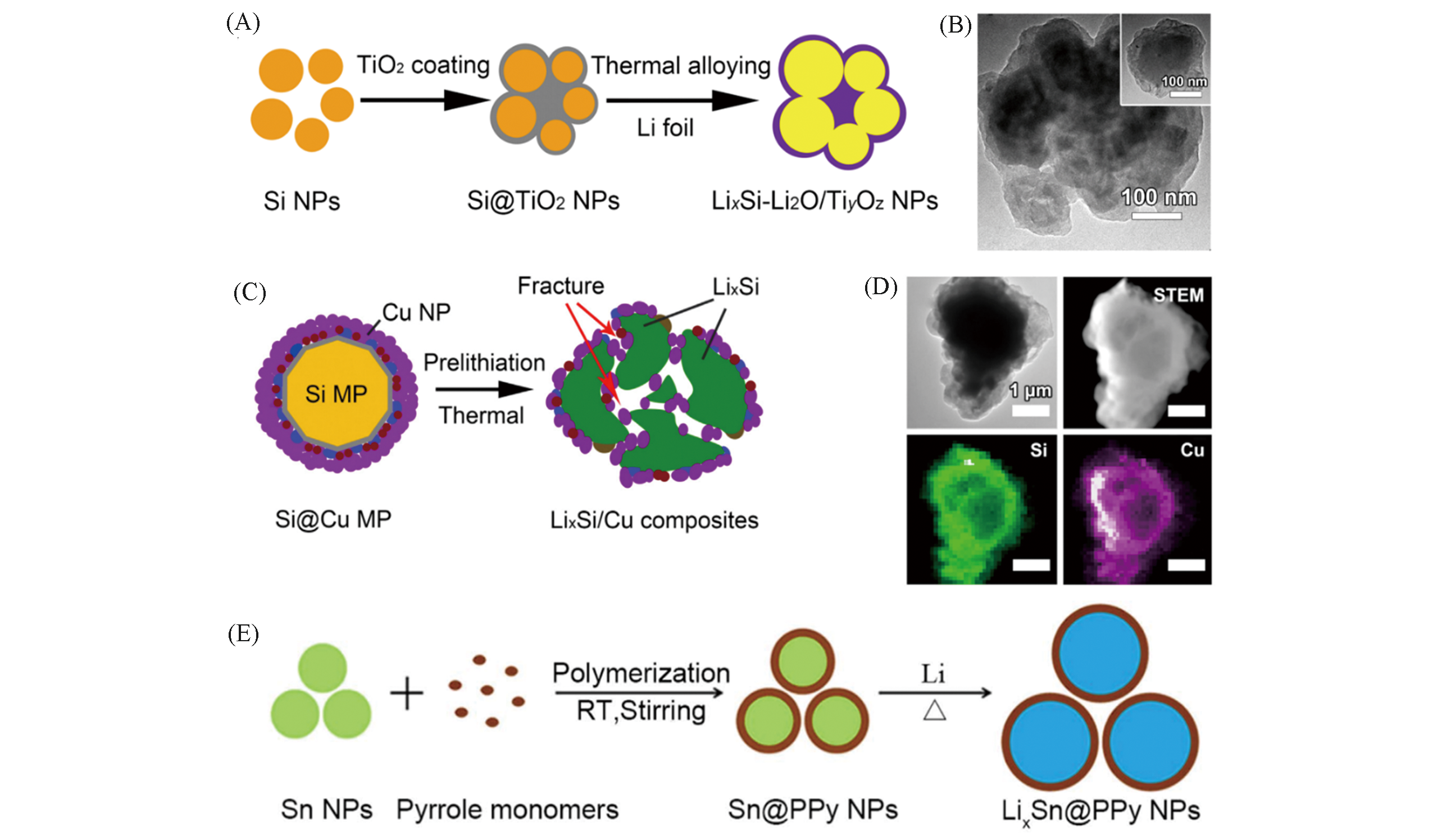
Fig.8 Schematic of the fabrication process for LixSi?Li2O/TiyOz core?shell NPs(A), TEM image of the resultant LixSi?Li2O/TiyOz NPs(B)[74], schematic of the thermal lithiation process for LixSi/Cu alloy composites fabrication(C), EDS elemental mapping analysis of an individual LixSi/Cu composites particle(D)[75] and schematic illustration of the fabrication process of LixSn@PPy NPs(E)[76](A, B) Copyright 2018, American Chemical Society; (C, D) Copyright 2019, Elsevier; (E) Copyright 2019, Elsevier.
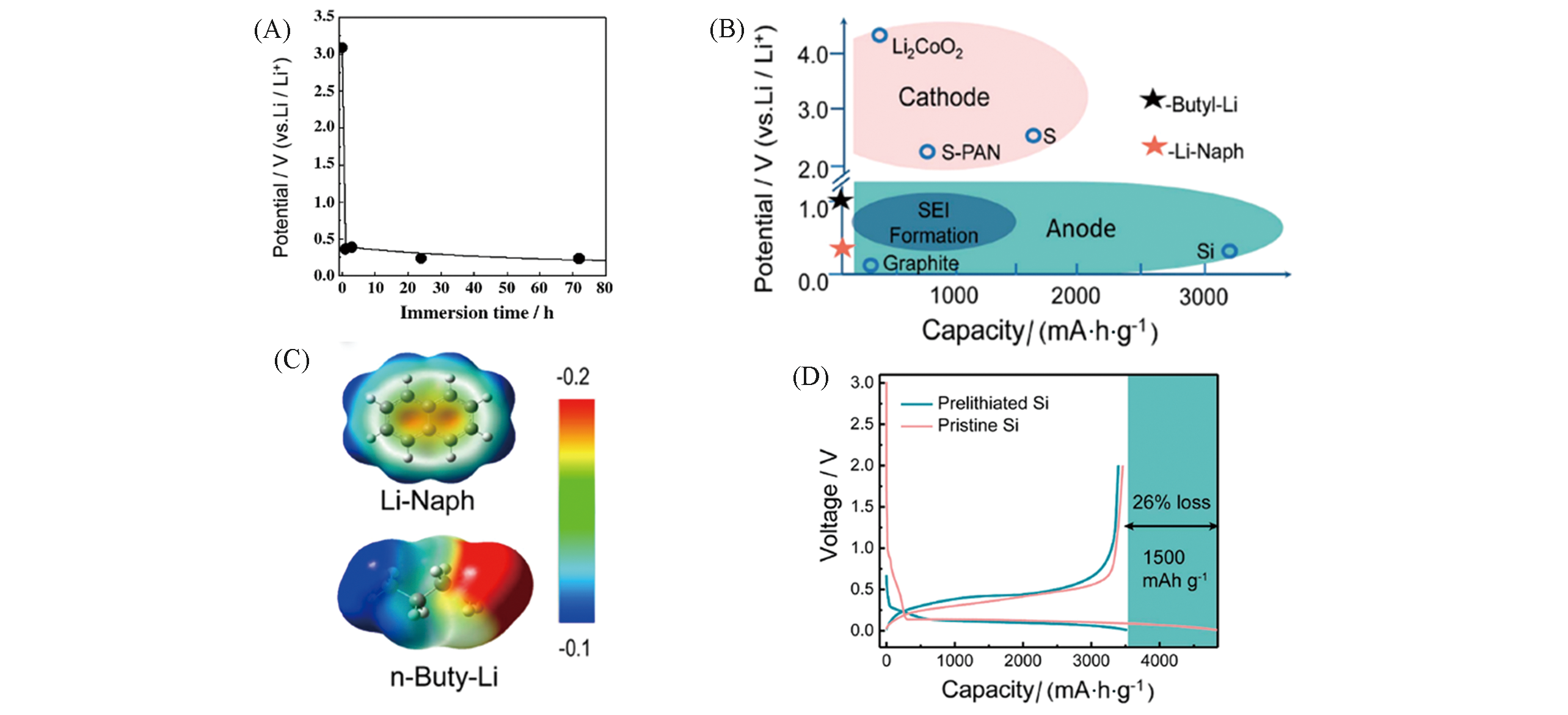
Fig.9 Change in the rest potential of LixSiO electrode(A)[78], potentials of selected materials(B), mapped electrostatic potential(MEP) surfaces of naphthalene anion and n?butyl anion(C) and charge?discharge profiles of nano?Si and prelithiated Si anodes(D)[79](A) Copyright 2005, Elsevier; (B—D) Copyright 2019, American Chemical Society.
| 1 | Cano Z. P., Banham D., Ye S., Hintennach A., Lu J., Fowler M., Chen Z., Nat. Energy, 2018, 3(4), 279—289 |
| 2 | Lu J., Chen Z., Ma Z., Pan F., Curtiss L. A., Amine K., Nat. Nanotechnol., 2016, 11, 1031—1038 |
| 3 | Armand M., Tarascon J. M., Nature, 2008, 451, 652—657 |
| 4 | Aravindan V., Lee Y. S., Madhavi S., Adv. Energy Mater., 2015, 5(13), 1402225 |
| 5 | Chen L., Fan X., Ji X., Chen J., Hou S., Wang C., Joule, 2019, 3(3), 732—744 |
| 6 | Cheng X. B., Zhang R., Zhao C. Z., Zhang Q., Chem. Rev., 2017, 117(15), 10403—10473 |
| 7 | Li L., Raji A. R., Tour J. M., Adv. Mater., 2013, 25(43), 6298—6302 |
| 8 | Liu D., Liu Z. j., Li X., Xie W., Wang Q., Liu Q., Fu Y., He D., Small, 2017, 13(45), 1702000 |
| 9 | Jin Y., Zhu B., Lu Z., Liu N., Zhu J., Adv. Energy Mater., 2017, 7(23), 1700715 |
| 10 | Obrovac M. N., Chevrier V. L., Chem. Rev., 2014, 114(23), 11444—11502 |
| 11 | Zhu B., Wang X., Yao P., Li J., Zhu J., Chem. Sci., 2019, 10(30), 7132—7148 |
| 12 | Liu N., Lu Z., Zhao J., McDowell M. T., Lee H. W., Zhao W., Cui Y., Nat. Nanotechnol., 2014, 9(3), 187—192 |
| 13 | Kwon Y. H., Minnici K., Park J. J., Lee S. R., Zhang G., Takeuchi E. S., Takeuchi K. J., Marschilok A. C., Reichmanis E., J. Am. Chem. Soc., 2018, 140(17), 5666—5669 |
| 14 | Xu Q., Sun J. K., Yu Z. L., Yin Y. X., Xin S., Yu S. H., Guo Y. G., Adv. Mater., 2018, 30(25), 1707430 |
| 15 | Zhang Z., Wang Z. L., Lu X., ACS Nano, 2018, 12(4), 3587—3599 |
| 16 | Chevrier V. L., Liu L., Wohl R., Chandrasoma A., Vega J. A., Eberman K. W., Stegmaier P., Figgemeier E., J. Electrochem. Soc., 2018, 165(5), A1129—A1136 |
| 17 | Verma P., Maire P., Novák P., Electrochim. Acta, 2010, 55(22), 6332—6341 |
| 18 | Tripathi A. M., Su W. N., Hwang B. J., Chem. Soc. Rev., 2018, 47(3), 736—851 |
| 19 | Aravindan V., Lee Y. S., Madhavi S., Adv. Energy Mater., 2017, 7(17), 1602607 |
| 20 | Holtstiege F., Bärmann P., Nölle R., Winter M., Placke T., Batteries, 2018, 4(1), 4 |
| 21 | Ji L., Zheng H., Ismach A., Tan Z., Xun S., Lin E., Battaglia V., Srinivasan V., Zhang Y., Nano Energy, 2012, 1(1), 164—171 |
| 22 | Asadi M., Sayahpour B., Abbasi P., Ngo A. T., Karis K., Jokisaari J. R., Liu C., Narayanan B., Gerard M., Yasaei P., Hu X., Mukherjee A., Lau K. C., Assary R. S., Khalili⁃Araghi F., Klie R. F., Curtiss L. A., Salehi⁃Khojin A., Nature, 2018, 555(7697), 502—506 |
| 23 | Liu X., Huang J. Q., Zhang Q., Mai L., Adv. Mater., 2017, 29(20), 1601759 |
| 24 | Griffith K. J., Wiaderek K. M., Cibin G., Marbella L. E., Grey C. P., Nature, 2018, 559(7715), 556—563 |
| 25 | Hua X., Robert R., Du L. S., Wiaderek K. M., Leskes M., Chapman K. W., Chupas P. J., Grey C. P., J. Phy. Chem. C, 2014, 118(28), 15169—15184 |
| 26 | Jarvis C. R., Lain M. J., Yakovleva M. V., Gao Y., J. Power Sources, 2006, 162(2), 800—802 |
| 27 | Vaughey J., Liu G., Zhang J. G., Mrs Bull., 2014, 39(5), 429—435 |
| 28 | Li Y., Fitch B., Electrochem. Commun., 2011, 13(7), 664—667 |
| 29 | Wang Z., Fu Y., Zhang Z., Yuan S., Amine K., Battaglia V., Liu G., J. Power Sources, 2014, 260, 57—61 |
| 30 | Ai G., Wang Z., Zhao H., Mao W., Fu Y., Yi R., Gao Y., Battaglia V., Wang D., Lopatin S., Liu G., J. Power Sources, 2016, 309, 33—41 |
| 31 | Jarvis C. R., Lain M. J., Gao Y., Yakovleva M., J. Power Sources, 2005, 146(1/2), 331—334 |
| 32 | Wang L., Fu Y., Battaglia V. S., Liu G., RSC Adv., 2013, 3(35), 15022—15027 |
| 33 | Xiang B., Wang L., Liu G., Minor A. M., J. Electrochem. Soc., 2013, 160(3), A415—A419 |
| 34 | Forney M. W., Ganter M. J., Staub J. W., Ridgley R. D., Landi B. J., Nano Lett., 2013, 13(9), 4158—4163 |
| 35 | Pan Q., Zuo P., Mu T., Du C., Cheng X., Ma Y., Gao Y., Yin G., J. Power Sources, 2017, 347, 170—177 |
| 36 | Zhao H., Wang Z., Lu P., Jiang M., Shi F., Song X., Zheng Z., Zhou X., Fu Y., Abdelbast G., Nano Lett., 2014, 14(11), 6704—6710 |
| 37 | Marinaro M., Weinberger M., Wohlfahrt⁃Mehrens M., Electrochim. Acta, 2016, 206, 99—107 |
| 38 | Pu K., Qu X., Zhang X., Hu J., Gu C., Wu Y., Gao M., Pan H., Liu Y., Adv. Sci., 2019, 6(24), 1901776 |
| 39 | Sun H., He X., Ren J., Li J., Jiang C., Wan C., Electrochim. Acta, 2007, 52(13), 4312—4316 |
| 40 | Ye R., Bell J., Patino D., Ahmed K., Ozkan M., Ozkan C. S., Sci. Rep., 2017, 7(1), 17264 |
| 41 | Cao Z., Xu P., Zhai H., Du S., Mandal J., Dontigny M., Zaghib K., Yang Y., Nano Lett., 2016, 16(11), 7235—7240 |
| 42 | Kulova T. L., Skundin A. M., J. Solid State Electr., 2003, 8(1), 59—65 |
| 43 | Kulova T. L., Skundin A. M., Russ. J. Electrochem., 2010, 46(4), 470—475 |
| 44 | Liu N., Hu L., McDowell M. T., Jackson A., Cui Y., ACS Nano, 2011, 5(8), 6487—6493 |
| 45 | Chae C., Noh H. J., Lee J. K., Scrosati B., Sun Y. K., Adv. Funct. Mater., 2014, 24(20), 3036—3042 |
| 46 | Shi L., Liu Y., Wang W., Wang A., Jin Z., Wu F., Yang Y., J. Alloy. Compd., 2017, 723, 974—982 |
| 47 | Pu X., Yang G., Yu C., Nano Energy, 2014, 9, 318—324 |
| 48 | Kim H. S., Jeong T. G., Kim Y. T., J. Electrochem. Sci. Te., 2016, 7(3), 228—233 |
| 49 | Hassoun J., Jung H. G., Lee D. J., Park J. B., Amine K., Sun Y. K., Scrosati B., Nano Lett., 2012, 12(11), 5775—5779 |
| 50 | Lee S. K., Oh S. M., Park E., Scrosati B., Hassoun J., Park M. S., Kim Y. J., Kim H., Belharouak I., Sun Y. K., Nano Lett., 2015, 15(5), 2863—2868 |
| 51 | Meng Q., Li G., Yue J., Xu Q., Yin Y. X., Guo Y. G., ACS Appl. Mater. Inter., 2019, 11(35), 32062—32068 |
| 52 | Kim H. J., Choi S., Lee S. J., Seo M. W., Lee J. G., Deniz E., Lee Y. J., Kim E. K., Choi J. W., Nano Lett., 2015, 16(1), 282—288 |
| 53 | Krause A., Dörfler S., Piwko M., Wisser F. M., Jaumann T., Ahrens E., Giebeler L., Althues H., Schädlich S., Grothe J., Jeffery A., Grube M., Brückner J., Martin J., Eckert J., Kaskel S., Mikolajick T., Weber W. M., Sci. Rep., 2016, 6(1), 27982 |
| 54 | Fridman K., Sharabi R., Elazari R., Gershinsky G., Markevich E., Salitra G., Aurbach D., Garsuch A., Lampert J., Electrochem. Commun., 2013, 33, 31—34 |
| 55 | Elazari R., Salitra G., Gershinsky G., Garsuch A., Panchenko A., Aurbach D., Electrochem. Commun., 2012, 14(1), 21—24 |
| 56 | Elazari R., Salitra G., Gershinsky G., Garsuch A., Panchenko A., Aurbach D., J. Electrochem. Soc., 2012, 159(9), A1440—A1445 |
| 57 | Derrien G., Hassoun J., Panero S., Scrosati B., Adv. Mater., 2007, 19(17), 2336—2340 |
| 58 | Wan Y., Wang L., Chen Y., Xu X., Wang Y., Teng C., Zhou D., Chen Z., J. Alloy. Compd., 2018, 740, 830—835 |
| 59 | Zhou H., Wang X., Chen D., ChemSusChem, 2015, 8(16), 2737—2744 |
| 60 | Zhao J., Lu Z., Wang H., Liu W., Lee H. W., Yan K., Zhuo D., Lin D., Liu N., Cui Y., J. Am. Chem. Soc., 2015, 137(26), 8372—8375 |
| 61 | Zhao J., Sun J., Pei A., Zhou G., Yan K., Liu Y., Lin D., Cui Y., Energy Storage Mater., 2018, 10, 275—281 |
| 62 | Iwamura S., Nishihara H., Ono Y., Morito H., Yamane H., Nara H., Osaka T., Kyotani T., Sci. Rep., 2015, 5, 8085 |
| 63 | Zhao J., Lu Z., Liu N., Lee H. W., McDowell M. T., Cui Y., Nat. Commun., 2014, 5(1), 5088 |
| 64 | Zhao J., Lee H. W., Sun J., Yan K., Liu Y., Liu W., Lu Z., Lin D., Zhou G., Cui Y., P. Natl. A. Sci., 2016, 113(27), 7408—7413 |
| 65 | Cloud J. E., Wang Y., Li X., Yoder T. S., Yang Y., Yang Y., Inorg. Chem., 2014, 53(20), 11289—11297 |
| 66 | Cloud J. E., Wang Y., Yoder T. S., Taylor L. W., Yang Y., Angew. Chem. In. Ed., 2014, 53(52), 14527—14532 |
| 67 | Li X., Kersey⁃Bronec F. E., Ke J., Cloud J. E., Wang Y., Ngo C., Pylypenko S., Yang Y., ACS Appl. Mater. Inter., 2017, 9(19), 16071—16080 |
| 68 | Domi Y., Usui H., Iwanari D., Sakaguchi H., J. Electrochem. Soc., 2017, 164(7), A1651—A1654 |
| 69 | Chang S., Moon J., Cho K., Cho M., Comp. Mater. Sci., 2015, 98, 99—104 |
| 70 | Hou C., Song X., Yang K., Wang H., Liu X., Nie Z., Mater. Today Energy, 2018, 7, 122—128 |
| 71 | Fan X., Shao J., Xiao X., Wang X., Li S., Ge H., Chen L., Nano Energy, 2014, 9, 196—203 |
| 72 | Hou C., Yang K., Tang F., Wang H., Liu X., Song X., Pro. Nat. Sci. Mater., 2019, 29(3), 310—315 |
| 73 | Zhao J., Zhou G., Yan K., Xie J., Li Y., Liao L., Jin Y., Liu K., Hsu P. C., Wang J., Cheng H. M., Cui Y., Nat. Nanotechnol., 2017, 12(10), 993—999 |
| 74 | Wang C., Han Y., Li S., Chen T., Yu J., Lu Z., ACS Appl. Mater. Inter., 2018, 10(15), 12750—12758 |
| 75 | Wang C., Yu J., Li S., Lu Z., Chem. Eng. J., 2019, 370, 1019—1026 |
| 76 | Li S., Wang C., Yu J., Han Y., Lu Z., Energy Storage Mater., 2019, 20, 7—13 |
| 77 | Scott M., Whitehead A., Owen J., J. Electrochem. Soc., 1998, 145(5), 1506—1510 |
| 78 | Tabuchi T., Yasuda H., Yamachi M., J. Power Sources, 2005, 146(1/2), 507—509 |
| 79 | Shen Y., Zhang J., Pu Y., Wang H., Wang B., Qian J., Cao Y., Zhong F., Ai X., Yang H., ACS Energy Lett., 2019, 4(7), 1717—1724 |
| 80 | Wang G., Li F., Liu D., Zheng D., Luo Y., Qu D., Ding T., Qu D., ACS Appl. Mater. Inter., 2019, 11(9), 8699—8703 |
| 81 | Alaboina P. K., Cho J. S., Uddin M. J., Cho S. J., Electrochim. Acta, 2017, 258, 623—630 |
| [1] | 李辉阳, 朱思颖, 李莎, 张桥保, 赵金保, 张力. 锂离子电池硅氧化物负极首次库伦效率的影响因素与提升策略[J]. 高等学校化学学报, 2021, 42(8): 2342. |
| [2] | 刘铁峰, 张奔, 盛欧微, 佴建威, 王垚, 刘育京, 陶新永. 硅负极黏结剂的研究进展[J]. 高等学校化学学报, 2021, 42(5): 1446. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||