

高等学校化学学报 ›› 2014, Vol. 35 ›› Issue (5): 903.doi: 10.7503/cjcu20130849
于海洋1,3, 汤朝晖1, 宋万通1, 邓明虓2, 陈学思1( )
)
收稿日期:2013-09-02
出版日期:2014-05-10
发布日期:2014-04-17
作者简介:联系人简介: 陈学思, 男, 博士, 研究员, 博士生导师, 主要从事生物可降解高分子研究. E-mail:基金资助:
YU Haiyang1,3, TANG Zhaohui1, SONG Wantong1, DENG Mingxiao2, CHEN Xuesi1,*( )
)
Received:2013-09-02
Online:2014-05-10
Published:2014-04-17
Contact:
CHEN Xuesi
E-mail:xschen@ciac.ac.cn
Supported by:摘要:
综述了肿瘤靶向性高分子纳米载体在抗肿瘤药物的靶向性输送和控制释放方面的研究进展, 并详细介绍了被动肿瘤靶向性、 主动靶向性、 生物可降解性、 pH敏感性、 还原敏感性、 酶敏感性和温度敏感性高分子纳米载体的研究现状, 展望了该研究领域的发展方向.
中图分类号:
TrendMD:
于海洋, 汤朝晖, 宋万通, 邓明虓, 陈学思. 肿瘤靶向性高分子纳米载体研究现状与展望. 高等学校化学学报, 2014, 35(5): 903.
YU Haiyang, TANG Zhaohui, SONG Wantong, DENG Mingxiao, CHEN Xuesi. Current Status and Future Prospects of Polymeric Nanocarrier for Tumor Targeting†. Chem. J. Chinese Universities, 2014, 35(5): 903.
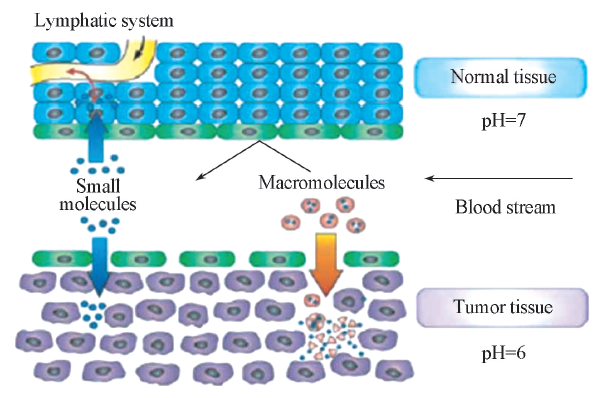
Fig.1 Schematic representation of anatomical diffe-rences between normal and tumor tissues[5]^Hyper-permeable tumor vasculature allows preferential extravasation of the circulating macromolecular drug carriers due to enhanced permeability and retention(EPR) effect.
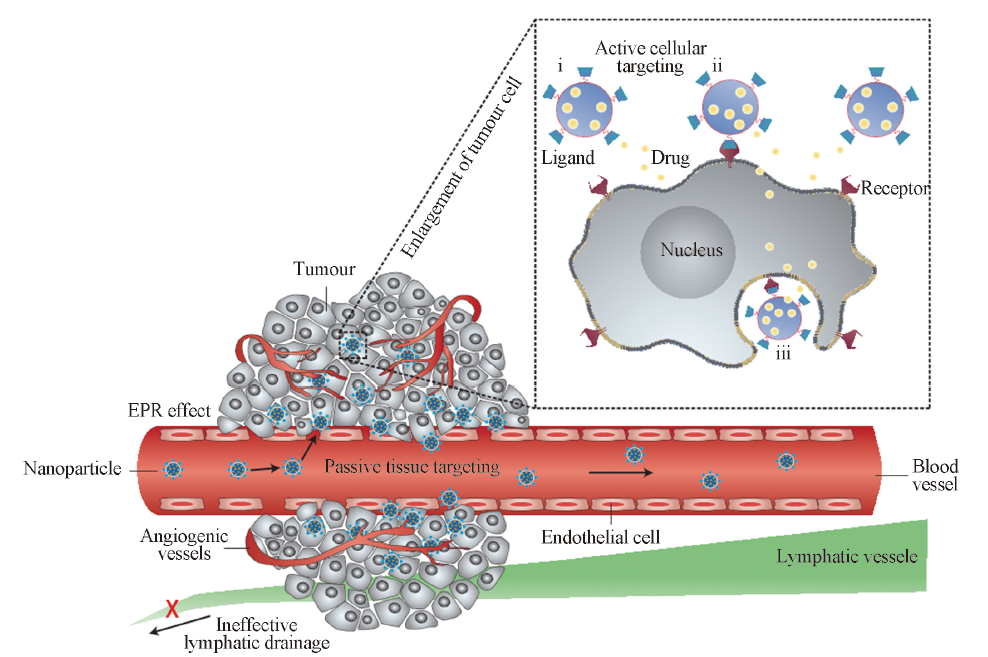
Fig.2 Schematic representation of different mechanisms by which nanocarriers can deliver drugs to tumours[1]^Polymeric nanoparticles are shown as representative nanocarriers(circles). Passive tissue targeting is achieved by extravasation of nanoparticles through increased permeability of the tumour vasculature and ineffective lymphatic drainage(EPR effect). Active cellular targeting(inset) can be achieved by functionalizing the surface of nanoparticles with ligands that promote cell-specific recognition and binding. the nanoparticles can(i) release their contents in close proximity to the target cells;(ii) attach to the membrane of the cell and act as an extracellular sustained-release drug depot; or(iii) internalize into the cell.
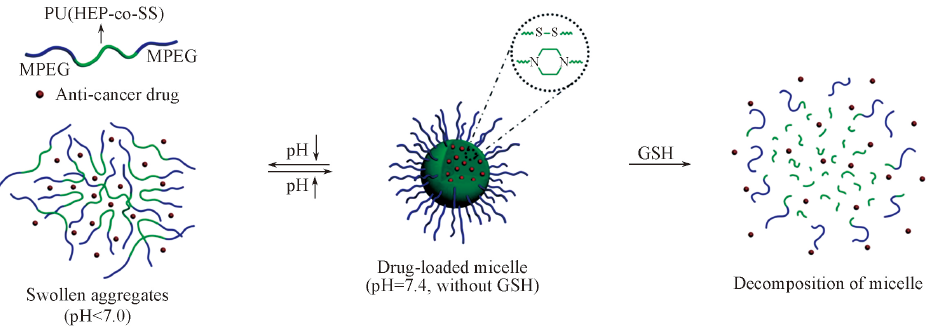
Fig.12 Schematic illustration of the formation and stimuli-dependent structural transitions of the pH- and reduction-responsive polyurethane micelles [98]
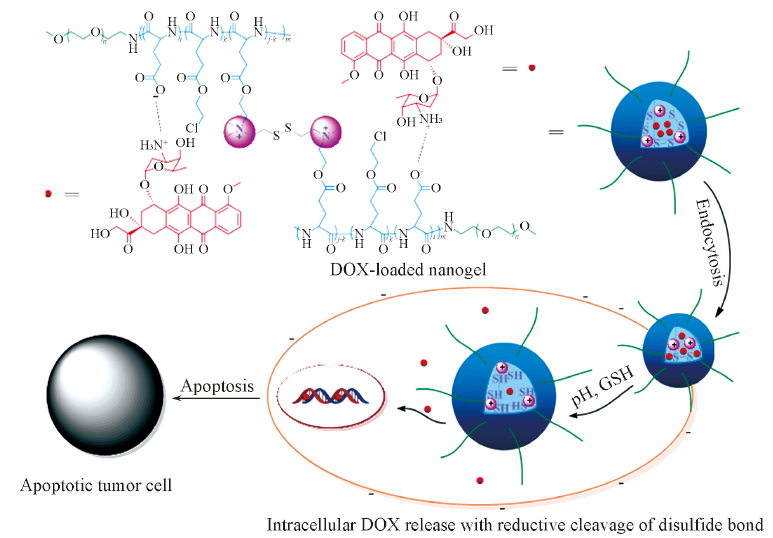
Fig.13 Schematic illustration of improved cellular internalization and efficient intracellular DOX release of DOX-loaded nanogel, and tumor cellular apoptosis[100]
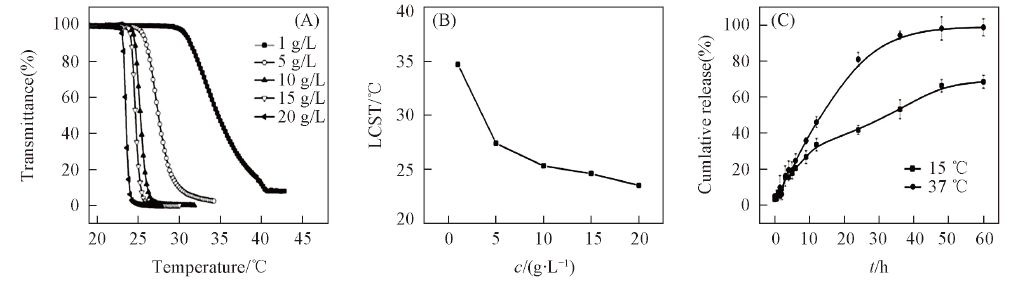
Fig.15 Infiuence of the polymer concentration on the thermosensitive behavior of PPLG112-g-MEO2 in aqueous solution(A), LCST of PPLG112-g-MEO2 solution as a function of its concentration(B) and release profile of doxorubicin from PPLG112-g-MEO2nano-particles(C)[106]
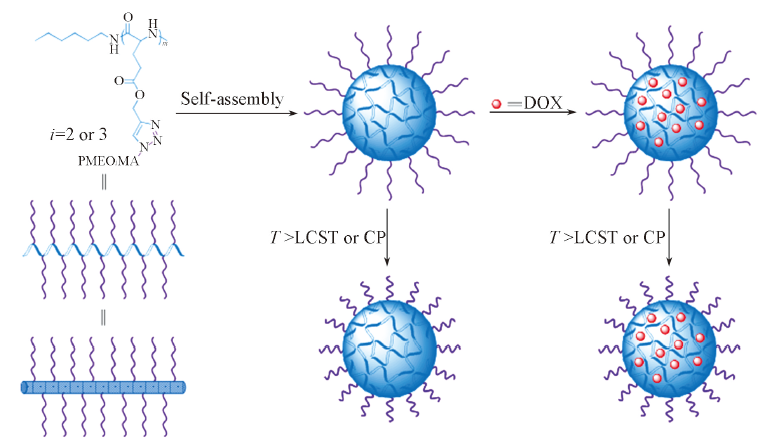
Fig.16 Schematic illustration of self-assembly of “hairy-rod” polypeptides, DOX loading and thermal-responsiveness of micelle and DOX-loaded micelle[107]
| [1] | Peer D., Karp J. M., Hong S., Farokhzad O. C., Margalit R., Langer R., Nature Nanotechnology, 2007, 2, 751—760 |
| [2] | Liao B., Liao Q. J., Progress in Pharmaceutical Sciences, 2012, 36(3), 138—142 |
| (廖斌, 廖清江.药学进展, 2012,36(3), 138—142) | |
| [3] | Chen H. Y., Chen Z. Y., Ma A.X., Journal of North Pharmacy, 2012, 9(11), 51—52 |
| (陈海燕, 陈在余, 马爱霞.北方药学, 2012,9(11), 51—52) | |
| [4] | Taurin S., Nehoff H., Greish K., J. Controlled Release, 2012, 164, 265—275 |
| [5] | Jonathan O. M., Brandon S. B., Nicoletta Q., Michael E., Mauro F., Ennio T., Chinese Science Bulletin, 2012, 57(31), 3961—3971 |
| [6] | Bisht S., Maitra A., Wiley Interdiscip Rev. Nanomed. Nanobiotechnol., 2009, 1, 415—425 |
| [7] | Maeda H., Takeshita J., Kanamaru R., Int. J. Pept. Protein Res., 1979, 14, 81—87 |
| [8] | Maeda H., Ueda M., Morinaga T., Matsumoto T., J. Med. Chem., 1985, 28, 455—461 |
| [9] | Matsumura Y., Maeda H., Cancer Research, 1986, 46, 6387—6392 |
| [10] | Greish K., J. Drug Target., 2007, 15, 457—464 |
| [11] | Gupta M., Agrawal G. P., Vyas S. P., Curr. Mol. Med., 2013, 13, 179—204 |
| [12] | Khandare J., Minko T., Prog. Polym. Sci., 2006, 31, 359—397 |
| [13] | Li C., Adv. Drug Deliver. Rev., 2002, 54, 695—713 |
| [14] | Maeda H., Bharate G. Y., Daruwalla J., Eur. J. Pharm. Biopharm., 2009, 71, 409—419 |
| [15] | Matsumura Y., Kataoka K., Cancer Science, 2009, 100, 572—579 |
| [16] | Torchilin V. P., Cell Mol. Life Sci., 2004, 61, 2549—2559 |
| [17] | Li S. D., Huang L., Molecular Pharmaceutics, 2008, 5, 496—504 |
| [18] | Cabral H., Matsumoto Y., Mizuno K., Chen Q., Murakami M., Kimura M., Terada Y., Kano M. R., Miyazono K., Uesaka M., Nishiyama N., Kataoka K., Nature Nanotechnology, 2011, 6, 815—823 |
| [19] | Liu D., Mori A., Huang L., Biochim. Biophys. Acta Biomembranes, 1992, 1104, 95—101 |
| [20] | Fang C., Shi B., Pei Y. Y., Hong M. H., Wu J., Chen H. Z., Eur. J. Pharm. Sci., 2006, 27, 27—36 |
| [21] | Lee J. S., Ankone M., Pieters E., J. Controlled Release, 2011, 155, 282—288 |
| [22] | Zhang J.S., Liu F., Huang L., Adv. Drug Deliver. Rev., 2005, 57, 689—698 |
| [23] | Owens Iii D. E., Peppas N. A., Int. J. Pharm., 2006, 307, 93—102 |
| [24] | Photos P. J., Bacakova L., Discher B., Bates F. S., Discher D. E., J. Controlled Release, 2003, 90, 323—334 |
| [25] | Zhang L., Cao Z., Li Y., Ella-Menye J. R., Bai T., Jiang S., ACS Nano, 2012, 6, 6681—6686 |
| [26] | Mohanty C., Das M., Kanwar J. R., Sahoo S. K., Current Drug Delivery, 2011, 8, 45—58 |
| [27] | Das M., Mohanty C., Sahoo S. K., Expert Opinion on Drug Delivery, 2009, 6, 285—304 |
| [28] | Jian C. Y., Xiao L. X., Xian W. Z., Hong Y. G., Cheng P. M., Chinese Chemical Letters, 2012, 23(7), 875—878 |
| [29] | Barreto J. A., O'Malley W., Kubeil M., Graham B., Stephan H., Spiccia L., Adv.Mater., 2011, 23, H18—H40 |
| [30] | Zhang H. Z., Zhang Q. Q., Chem. J. Chinese Universities, 2009, 30(6), 1146—1151 |
| (张慧珠, 张其清.高等学校化学学报, 2009,30(6), 1146—1151) | |
| [31] | Chen F., Dong D., Fu Y., Zheng Y. H., Liu S., Chang M. X., Jing X. B., Chem. Res. Chinese Universities, 2012, 28(4), 656—661 |
| [32] | Ding J. X., Xiao C. S., Li Y., Cheng Y. L., Wang N., He C. L., Zhuang X. L., Zhu X., Chen X. S., J. Control Release, 2013, 169, 193—203 |
| [33] | Wagner E., Expert Opin. Biol. Ther., 2007, 7, 587—593 |
| [34] | Mahmud A. Xiong X., Aliabadi H., Lavasanifar A., J. Drug Target., 2007, 15, 553—584 |
| [35] | Han S. F., Duan Y. J., Wang Y. M., Zheng J. N., Wu Y., Cai R., Ou L. L., Kong D. S., Yu Y. T., Chem. J. Chinese Universities, 2008, 29(5), 923—926 |
| (韩素芳, 段亚军, 王燕铭, 郑骏年, 武艺, 蔡荣, 欧来良, 孔德领, 俞耀庭.高等学校化学学报, 2008,29(5), 923—926) | |
| [36] | Warenius H. M., Galfre G., Bleehen N. M., Milstein C., Eur. J. Cancer Clin. Oncol., 1981, 17, 1009—1015 |
| [37] | Von Mehren M., Adams G. P., Weiner L. M., Annu. Rev. Med., 2003, 54, 343—369 |
| [38] | Weiner L. M., Adams G. P., Oncogene, 2000, 19, 6144—6151 |
| [39] | Albanell J., Baselga J., Drugs of Today, 1999, 35, 931—946 |
| [40] | Ferrara N., Oncology, 2005, 69, 11—16 |
| [41] | Carter P., Nature Reviews Cancer, 2001, 1, 118—129 |
| [42] | White R. R., Sullenger B. A., Rusconi C. P. J., Clin. Invest., 2000, 106, 929—934 |
| [43] | Kolishetti N., Dhar S., Valencia P. M., Lin L. Q., Karnik R., Lippard S. J., Langer R., Farokhzad O. C., Proc. Natl. Acad. Sci. USA, 2010, 107, 17939—17944 |
| [44] | Sanfilippo J. S., Miseljic S., Yang A. R., Doering D. L., Shaheen R. M., Wittliff J. L., Cancer, 1996, 77, 710—716 |
| [45] | Low P. S., Henne W. A., Doorneweerd D. D., Accounts. Chem. Res., 2007, 41, 120—129 |
| [46] | Dong D., Chen F., Fu Y., Wang R., Zheng Y.H., Jing X. B.,Acta Polymerica Sinica, 2012, (8), 915—922 |
| (董丹, 陈凤, 付艳, 王瑞, 郑勇辉, 景遐斌. 高分子学报, 2012, (8), 915—922) | |
| [47] | Garcia-Bennett A., Nees M., Fadeel B., Biochemical Pharmacology, 2011, 81, 976—984 |
| [48] | Nukolova N. V., Oberoi H. S., Cohen S. M., Kabanov A. V., Bronich T. K., Biomaterials, 2011, 32, 5417—5426 |
| [49] | Prost A. C., Menegaux F., Langlois P., Vidal J. M., Koulibaly M., Jost J. L., Duron J. J., Chigot J. P., Vayre P., Aurengo A., Legrand J. C., Rosselin G., Gespach C., Int. J. Oncol., 1998, 13, 871—875 |
| [50] | Yue J., Liu S., Wang R., Hu X.L., Xie Z. G., Huang Y. B., Jing X. B., Molecular Pharmaceutics, 2012, 9, 1919—1931 |
| [51] | Ekblom P., Thesleff I., Lehto V. P., Virtanen I., Int. J. Cancer, 1983, 31, 111—117 |
| [52] | Peer D., Margalit R., Neoplasia, 2004, 6, 343—353 |
| [53] | Eliaz R. E., Szoka F. C., Cancer Research, 2001, 61, 2592—2601 |
| [54] | Folkman J., Bach M., Rowe J. W., Davidoff F., Lambert P., Hirsch C., Goldberg A., Hiatt H. H., Glass J., Henshaw E., New Engl. J. Med., 1971, 285, 1182—1186 |
| [55] | Desgrosellier J. S., Cheresh D. A., Nature Reviews Cancer, 2010, 10, 9—22 |
| [56] | Danhier F., Breton A. L., Préat V., Molecular Pharmaceutics, 2012, 9, 2961—2973 |
| [57] | Pike D. B., Ghandehari H., Adv. Drug Deliver. Rev., 2010, 62, 167—183 |
| [58] | Jiang X., Sha X., Xin H., Chen L., Gao X., Wang X., Law K., Gu J., Chen Y., Jiang Y., Ren X., Ren Q., Fang X., Biomaterials, 2011, 32, 9457—9469 |
| [59] | Song W. T., Tang Z. H., Zhang D. W., Zhang Y., Yu H. Y., Li M. Q., Lv S. X., Sun H., Deng M. X., Chen X. S., Biomaterials, 2014, 35, 3005—3014 |
| [60] | Zhua Z. S., Xiea C., Liu Q., Zhen X., Zheng X. C., Wu W., Li R. T., Ding Y., Jiang X. Q., Liu B. R., Biomaterials, 2011, 32, 9525—9535 |
| [61] | Ruoslahti E., Adv. Mater., 2012, 24(28), 3747—3756 |
| [62] | Su S. S., Wang H., Liu X. G., Wu Y., Nie G. J., Biomaterials, 2013, 34, 3523—3533 |
| [63] | Sugahara K. N., Teesalu T., Karmali P. P., Kotamraju V. R., Agemy L., Girard O. M., Hanahan D., Mattrey R. F., Ruoslahti E., Cancer Cell, 2009, 16, 510—520 |
| [64] | Sugahara K. N., Teesalu T., Karmali P. P., Kotamraju V. R., Agemy L., Greenwald D. R., Ruoslahti E., Science, 2010, 328, 1031—1035 |
| [65] | Song W. T., Li M. Q., Tang Z. H., Li Q. S., Yang Y., Liu H. Y., Duan T. C., Hong H., Chen X. S., Macromol. Biosci., 2012, 12, 1514—1523 |
| [66] | MacEwan S. R., Callahan D. J., Chilkoti A., Nanomedicine, 2010, 5, 793—806 |
| [67] | Li D., Sun H., Ding J. X., Tang Z. H., Zhang Y., Xu W. G., Zhuang X. L., Chen X. S., Acta Biomater, 2013, 9, 8875—8884 |
| [68] | Kim D.W., Kim S. Y., Kim H. K., Kim S. W., Shin S. W., Kim J. S., Park K., Lee M. Y., Heo D. S., Annals of Oncology, 2007, 18, 2009—2014 |
| [69] | Lee K. S., Chung H. C., Im S. A., Park Y. H., Kim C. S., Kim S. B., Rha S. Y., Lee M. Y., Ro J., Breast. Cancer Res. Tr., 2008, 108, 241—250 |
| [70] | Kim T. Y., Kim D. W., Chung J. Y., Shin S. G., Kim S. C., Heo D. S., Kim N. K., Bang Y. J., Clinical Cancer Research, 2004, 10, 3708—3716 |
| [71] | Nishiyama N., Okazaki S., Cabral H., Miyamoto M., Kato Y., Sugiyama Y., Nishio K., Matsumura Y., Kataoka K., Cancer Research, 2003, 63, 8977—8983 |
| [72] | Uchino H., Matsumura Y., Negishi T., Koizumi F., Hayashi T., Honda T., Nishiyama N., Kataoka K., Naito S., Kakizoe T., Brit. J. Cancer, 2005, 93, 678—687 |
| [73] | Matsumura Y., Hamaguchi T., Ura T., Muro K., Yamada Y., Shimada Y., Shirao K., Okusaka T., Ueno H., Ikeda M., Watanabe N., Brit. J. Cancer, 2004, 91, 1775—1781 |
| [74] | Lv S. X., Li M. Q., Tang Z. H., Song W. T., Sun H., Liu H. Y., Chen X. S., Acta Biomater, 2013, 9, 9330—9342 |
| [75] | Bawa P., Pillay V., Choonara Y. E., du Toit L. C., Biomedical Materials, 2009, 4, 022001 |
| [76] | Guo J. S., Li J. Z., Jing X. B., Chen X. S., Huang Y. B., Chem. Res. Chinese Universities, 2011, 27(2), 329—333 |
| [77] | Lu D. X., Wen X. T., Liang J., Zhang X. D., Gu Z. W., Fan Y. J., Chinese Journal of Polymer Science, 2008, 26, 369—374 |
| [78] | Zhang J. C., Ding J. X., Xiao C. S., He C. L., Zhuang X. L., Yang Y. N., Chen X. S., Chem. J. Chinese Universities, 2012, 33(12), 2809—2815 |
| (张建成, 丁建勋, 肖春生, 贺超良, 庄秀丽, 杨亚楠, 陈学思.高等学校化学学报, 2012,33(12), 2809—2815) | |
| [79] | Qu X.Z., Yang Z. Z., Acta Polymerica Sinica, 2011, (10), 1118—1124(屈小中, 杨振忠. 高分子学报, 2011, (10), 1118—1124) |
| [80] | Yu S.F., Wang Z., Wu G. L., Wang Y. N., Gao H., Ma J. B.,Acta Polymerica Sinica, 2012, (4), 427—432 |
| (于树芳, 王铮, 伍国琳, 王亦农, 高辉, 马建标. 高分子学报, 2012, (4), 427—432) | |
| [81] | Wang Z., Yu S.F., Gu X., Wu G. L., Wang Y. N., Gao H., Ma J. B.,Acta Polymerica Sinica, 2012, (6), 599—605 |
| (王铮, 于树芳, 顾鑫, 伍国琳, 王亦农, 高辉, 马建标. 高分子学报, 2012, (6), 599—605) | |
| [82] | Ko J., Park K., Kim Y. S., Kim M. S., Han J. K., Kim K., Park R. W., Kim I. S., Song H. K., Lee D. S., Kwon I. C., J. Controlled Release, 2007, 123, 109—115 |
| [83] | Du J. Z., Sun T. M., Song W. J., Wu J., Wang J., Angew. Chem. Int. Edit., 2010, 49, 3621—3626 |
| [84] | Ding J. X., He C. L., Xiao C. S., Chen J. J., Zhuang X. L., Chen X. S., Macromolecular Research, 2012, 20, 292—301 |
| [85] | Ding J. X., Zhuang X. L., Xiao C. S., Cheng Y. L., Zhao L., He C. L., Tang Z. H., Chen X. S., J. Mater. Chem., 2011, 21, 11383—11391 |
| [86] | Li M. Q., Song W. T., Tang Z. H., LÜ S. X., Lin L., Sun H., Li Q. S., Yang Y., Hong H., Chen X. S., ACS Applied Materials & Interfaces, 2013, 5, 1781—1792 |
| [87] | Li M. Q., Lv S. X., Tang Z. H., Song W. T., Yu H. Y., Sun H., Liu H. Y., Chen X. S., Macromol. Biosci., 2013, 13, 1150—1162 |
| [88] | Song W. T., Tang Z. H., Li M. Q., Lv S. X., Yu H. Y., Ma L. L., Zhuang X. L., Huang Y. B., Chen X. S., Macromol. Biosci., 2012, 12, 1375—1383 |
| [89] | Huang Y., Tang Z.H., Zhang X. F., Yu H. Y., Sun H., Pang X., Chen X. S., Biomacromolecules, 2013, 14, 2023—2032 |
| [90] | Guan X. W., Li Y. H., Jiao Z. X., Chen J., Guo Z. P., Tian H. Y. Chen X. S., Acta Biomater, 2013, 9, 7672—7678 |
| [91] | Gao W., Langer R., Farokhzad O. C., Angew. Chem. Int. Ed., 2010, 49, 6567—6571 |
| [92] | Wang Y. C., Li Y., Sun T. M., Wu J., Yang Y. Y., Wang J., Macromol. Rapid. Comm., 2010, 31, 1201—1206 |
| [93] | Xu Y., Meng F., Cheng R., Zhong Z., Macromol. Biosci., 2009, 9, 1254—1261 |
| [94] | Zhang A. P., Zhang Z., Shi F. H., Ding J. X., Xiao C. S., Zhuang X. L., He C. L., Chen L., Chen X. S., Soft Matter, 2013, 9, 2224—2233 |
| [95] | Shi F. H., Ding J. X., Xiao C. S., Zhuang X. L., He C. L., Chen L., Chen X. S., J. Mater. Chem., 2012, 22, 14168—14179 |
| [96] | Ding J. X., Shi F. H., Xiao C. S., Lin L., Chen L., He C. L., Zhuang X. L., Chen X. S., Polymer Chemistry, 2011, 2, 2857—2864 |
| [97] | Ding J. X., Chen J. J., Li D., Xiao C. S., Zhang J. C., He C. L., Zhuang X. L., Chen X. S., J. Mater. Chem. B, 2013, 1, 69—81 |
| [98] | Yu S. J., He C. L., Ding J. X., Cheng Y. L., Song W. T., Zhuang X. L., Chen X. S., Soft Matter, 2013, 9, 2637—2645 |
| [99] | Li M. Q., Tang Z. H., Sun H., Ding J. X., Song W. T., Chen X. S., Polymer Chemistry, 2013, 4, 1199—1207 |
| [100] | Ding J. X., Xu W. G., Zhang Y., Sun D. K., Xiao C. S., Liu D. H., Zhu X. J., Chen X. S., J. Control Release, 2013, 172, 444—455 |
| [101] | Ding J. X., Shi F. H., Li D., Chen L., Zhuang X. L., Chen X. S., Biomaterials Science, 2013, 1, 633—646 |
| [102] | Yu K., Zhang L., Eisenberg A., Langmuir, 1996, 12, 5980—5984 |
| [103] | Zheng X.M., Jiang T., He F.,Acta Polymerica Sinica, 2011, (8), 895—902 |
| (郑晓明, 蒋涛, 贺枫. 高分子学报, 2011, (8), 895—902) | |
| [104] | Kataoka K., Togawa H., Harada A., Yasugi K., Matsumoto T., Katayose S., Macromolecules, 1996, 29, 8556—8557 |
| [105] | Jeong B., Bae Y. H., Kim S. W., J. Controlled Release, 2000, 63, 155—163 |
| [106] | Cheng Y. L., He C. L., Xiao C. S., Ding J. X., Zhuang X. L., Chen X. S., Polymer Chemistry, 2011, 2, 2627—2634 |
| [107] | Ding J. X., Zhao L., Li D., Xiao C. S., Zhuang X. L., Chen X. S., Polymer Chemistry, 2013, 4, 3345—3356 |
| [108] | Cheng Y. L., He C. L., Ding J. X., Xiao C. S., Zhuang X. L., Chen X. S., Biomaterials, 2013, 34, 10338—10347 |
| [109] | Roy R., Yang J., Moses M. A., Journal of Clinical Oncology, 2009, 27, 5287—5297 |
| [110] | Kratz F., Drevs J., Bing G., Stockmar C., Scheuermann K., Lazar P., Unger C., Bioorg. Med. Chem.Lett., 2001, 11, 2001—2006 |
| [111] | Bae M., Cho S., Song J., Lee G. Y., Kim K., Yang J., Cho K., Kim S. Y., Byun Y., Drug. Exp. Clin. Res., 2003, 29, 15—23 |
| [112] | Wong C., Stylianopoulos T., Cui J. A., Martin J., Chauhan V. P., Jiang W., Popovic Z., Jain R. K., Bawendi M. G., Fukumura D., Proc. Natl. Acad. Sci. USA, 2011, 108, 2426—2431 |
| [1] | 赵宇, 曹琬晴, 刘阳. 聚合物纳米药物载体的研究进展[J]. 高等学校化学学报, 2020, 41(5): 909. |
| [2] | 管清香, 纪丹阳, 孙波, 乔瑾, 何彤, 张广远, 虞振静, 尹建元, 杨薇. 叶酸修饰硬脂酸接枝白芨共聚物的合成及作为抗肿瘤药物载体的研究[J]. 高等学校化学学报, 2018, 39(8): 1815. |
| [3] | 简宇航, 颜世峰, 李星, 黄亚南, 尹静波. 基于β-环糊精接枝聚L-谷氨酸星型聚合物的可注射水凝胶的制备与表征[J]. 高等学校化学学报, 2017, 38(8): 1489. |
| [4] | 韩海玲, 金顺子, 苗壮, 陈平, 王占峰, 谢志刚. 嗜神经病毒衍生肽RVG29介导的靶向纳米载药胶束的合成及抗肿瘤活性[J]. 高等学校化学学报, 2016, 37(4): 661. |
| [5] | 朱华, 李一林, 赵传科, 解清华, 刘菲, 韩雪迪, 高静, 夏传琴, 沈琳, 杨志. 64Cu-NOTA-Herceptin的设计、 活性测定及肿瘤靶向分子显像研究[J]. 高等学校化学学报, 2016, 37(12): 2132. |
| [6] | 曾莉, 胡俊, 魏俊超. 聚乳酸-乙醇酸/纳米氧化锌复合电纺纤维装载亲疏水药物的控释及体外细胞毒性[J]. 高等学校化学学报, 2014, 35(8): 1788. |
| [7] | 徐洁, 赵满, 侯万国. MgFe2O4@(TF-LDHs)磁性纳米复合体的制备与表征[J]. 高等学校化学学报, 2012, 33(12): 2750. |
| [8] | 赵明礼, 赵玉涛, 张召, 高洁, 屈文, 徐锦梅, 刘宏民. 抗肿瘤药物卡培他滨的合成新方法[J]. 高等学校化学学报, 2012, 33(08): 1733. |
| [9] | 赵满, 徐洁, 侯万国. Fe3 O4@(TF-LDHs)纳米复合体的制备及药物缓释性能[J]. 高等学校化学学报, 2012, 33(07): 1572. |
| [10] | 陈阳娟 钟世安 师琼. 可降解光交联聚酸酐与聚酰胺-甲基丙烯酰胺凝胶的合成及药物控释[J]. 高等学校化学学报, 2011, 32(5): 1194. |
| [11] | 张慧珠, 张其清. 叶酸靶向乙酰普鲁兰纳米粒的制备及其靶向作用[J]. 高等学校化学学报, 2009, 30(6): 1146. |
| [12] | 刘洪玲,李军 . 肿瘤靶向PEI包覆磁性纳米凝胶的光化学制备及表征[J]. 高等学校化学学报, 2008, 29(8): 1703. |
| [13] | 韩素芳,段亚君,王燕铭,郑骏年,武艺,蔡荣,欧来良,孔德领,俞耀庭 . 基于单克隆抗体G250修饰的肿瘤细胞靶向基因载体研究[J]. 高等学校化学学报, 2008, 29(5): 923. |
| [14] | 李耀武, 周有骏, 朱驹, 郑灿辉, 张珉, 盛春泉, 陈军, 吕加国 . β微管蛋白中紫杉醇(Taxol)结合腔的性质分析[J]. 高等学校化学学报, 2006, 27(11): 2084. |
| [15] | 张雪飞, 胡俊丽, 陈学思, 景遐斌. 载有胰岛素的可生物降解微球的制备与表征[J]. 高等学校化学学报, 2005, 26(3): 554. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||