

Chem. J. Chinese Universities ›› 2021, Vol. 42 ›› Issue (5): 1589.doi: 10.7503/cjcu20200303
• Article • Previous Articles Next Articles
WANG Changyao, WANG Shuai, DUAN Linlin, ZHU Xiaohang, ZHANG Xingmiao, LI Wei( )
)
Received:2020-05-28
Online:2021-05-10
Published:2020-09-24
Contact:
LI Wei
E-mail:weilichem@fudan.edu.cn
Supported by:CLC Number:
TrendMD:
WANG Changyao, WANG Shuai, DUAN Linlin, ZHU Xiaohang, ZHANG Xingmiao, LI Wei. In situ Confinement Growth Strategy for Ordered Mesoporous Carbon Support Ultrasmall MoO3 Nanoparticles[J]. Chem. J. Chinese Universities, 2021, 42(5): 1589.
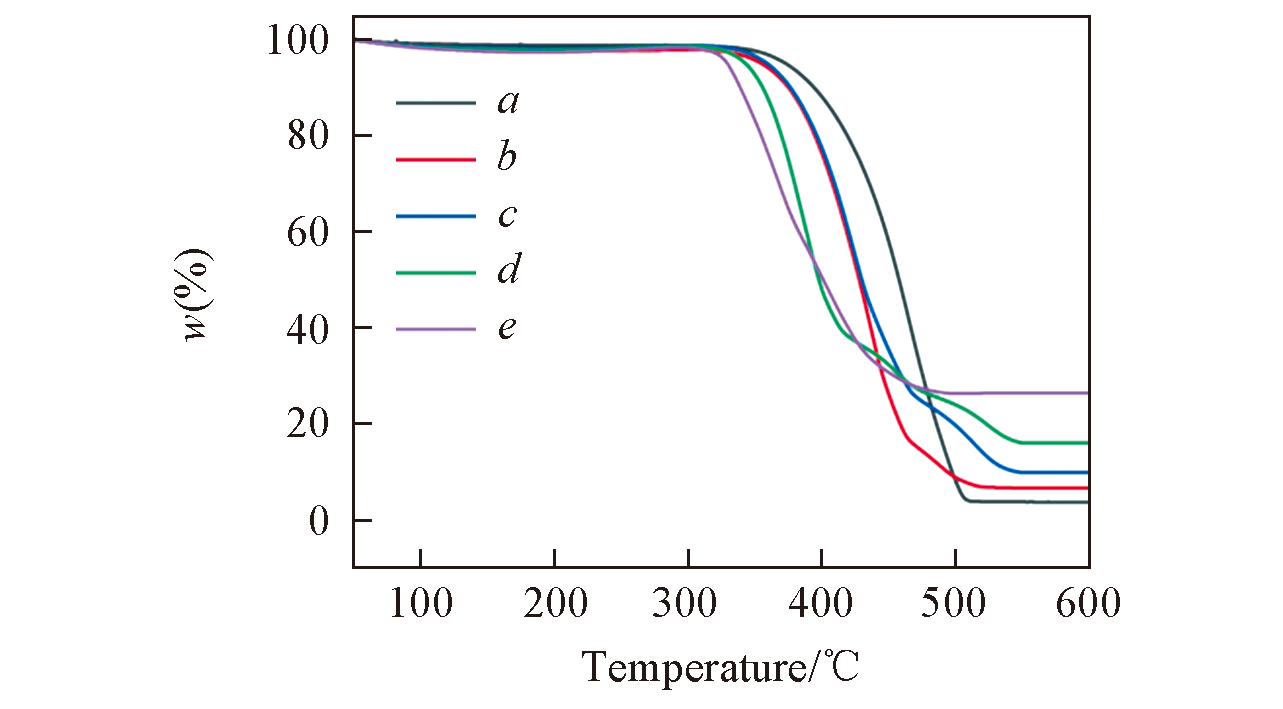
Fig.2 TGA curves of the OMC?US?MoO3 composites with different MoO3 contents obtained after pyrolysis at 600 ℃, respectivelyMass fraction of MoO3(%): a. 4; b. 7; c. 10; d. 16; e. 27.
| Sample No. | MoO3 content(%, mass fraction) | SBET/(m2·g-1) | V/(cm3·g-1) | D/nm |
|---|---|---|---|---|
| 1 | 4 | 796 | 0.62 | 4.7 |
| 2 | 7 | 693 | 0.54 | 5.7 |
| 3 | 10 | 652 | 0.49 | 5.5 |
| 4 | 16 | 574 | 0.41 | 5.4 |
| 5 | 27 | 428 | 0.27 | 4.6 |
Table 1 Structural and textural parameters for OMC-US-MoO3 with different content
| Sample No. | MoO3 content(%, mass fraction) | SBET/(m2·g-1) | V/(cm3·g-1) | D/nm |
|---|---|---|---|---|
| 1 | 4 | 796 | 0.62 | 4.7 |
| 2 | 7 | 693 | 0.54 | 5.7 |
| 3 | 10 | 652 | 0.49 | 5.5 |
| 4 | 16 | 574 | 0.41 | 5.4 |
| 5 | 27 | 428 | 0.27 | 4.6 |
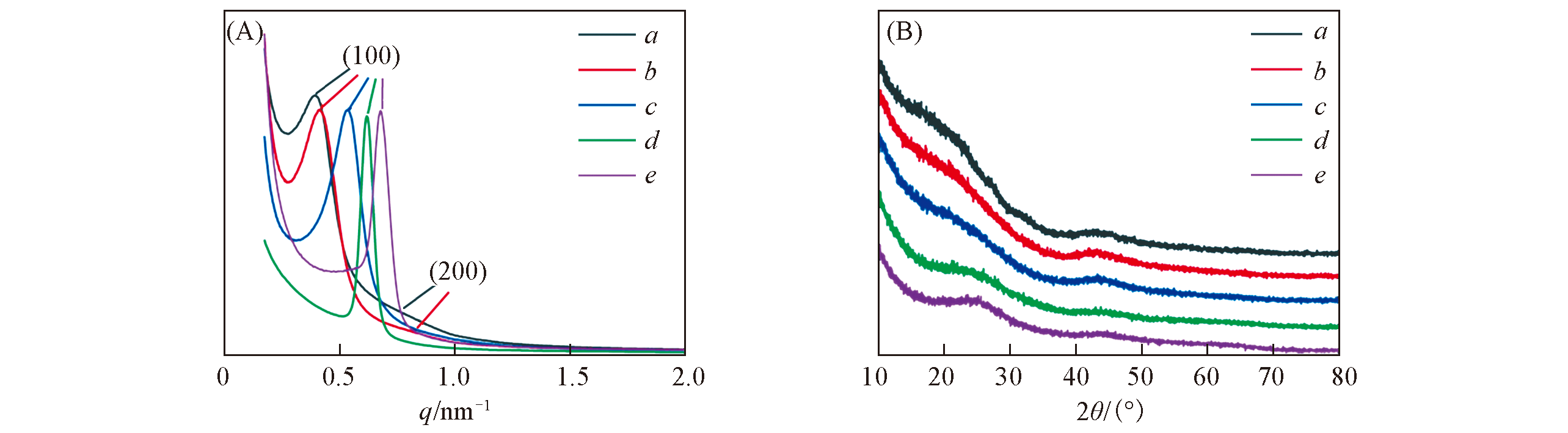
Fig.3 SAXS(A) and WA?XRD(B) patterns of the OMC?US?MoO3 composites with different MoO3 contents obtained after pyrolysis at 600 ℃Mass fraction of MoO3(%): a. 4; b. 7; c. 10; d. 16; e. 27.
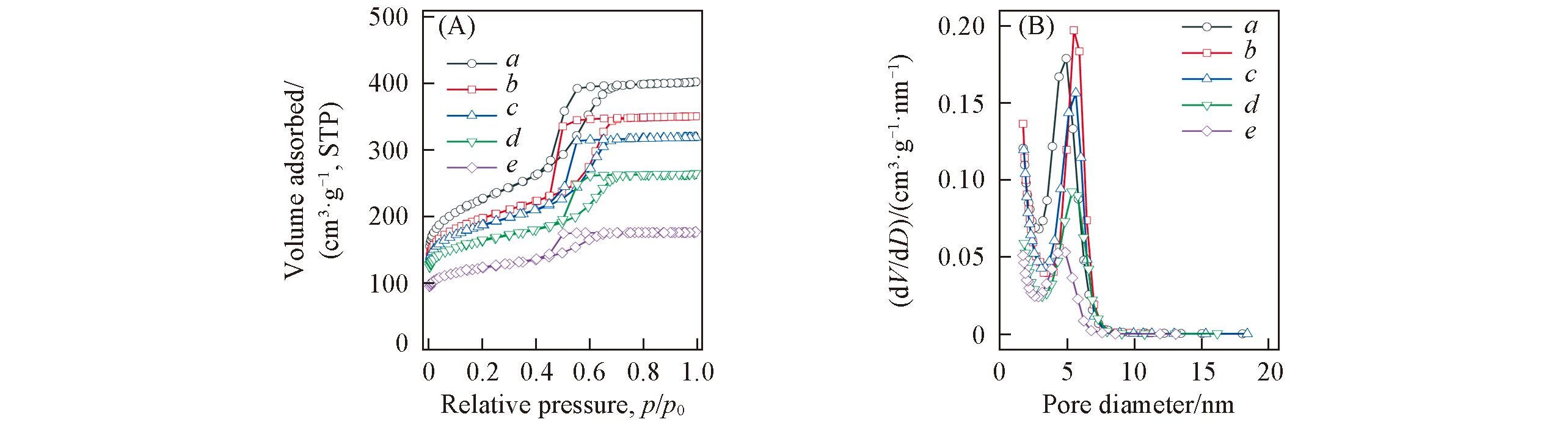
Fig.4 N2 adsorption?desorption isotherms(A) and pore size distributions(B) of the OMC?US?MoO3 composites with different MoO3 contents obtained after pyrolysis at 600 ℃Mass fraction of MoO3(%): a. 4; b. 7; c. 10; d. 16; e. 27.

Fig.5 SEM images of OMC?US?MoO3 composites with different MoO3 contents obtained after pyrolysis at 600 ℃Mass fraction of MoO3(%): (A) 4; (B) 7; (C) 10; (D) 16;(E) 27.

Fig.6 TEM images of OMC?US?MoO3?7 composites obtained after pyrolysis at 600 ℃Viewed along the hexagonal(A) and columnar(B, C) directions and HRTEM image(D) of a representative MoO3 nanoparticle.
| Catalyst | Time/h | Conv.(%) | Epoxide sel.(%) | TOF/h-1 |
|---|---|---|---|---|
| OMC-US-MoO3-7 | 2 | 52 | >99 | 2163 |
| MoO3/C | 2 | 80 | 100 | 53[ |
| MoO3/SiO2 | 6 | 90 | 100 | 72[ |
| Mo-MOFs | 7 | 93 | 99 | 270[ |
| Mo-MCM-41 | 3 | 97 | 95 | 22[ |
| Mo-SBA-15 | 3 | 99 | 93 | 40[ |
| [PiperazinCH2{MoO2(Salen)}]n | 12 | 95 | 98 | 16[ |
| MNP30-Si-inic-Mo | 24 | 46 | 100 | 2[ |
Table 2 Calculating TOF value for epoxidation of cyclooctene and comparing with other catalysts*
| Catalyst | Time/h | Conv.(%) | Epoxide sel.(%) | TOF/h-1 |
|---|---|---|---|---|
| OMC-US-MoO3-7 | 2 | 52 | >99 | 2163 |
| MoO3/C | 2 | 80 | 100 | 53[ |
| MoO3/SiO2 | 6 | 90 | 100 | 72[ |
| Mo-MOFs | 7 | 93 | 99 | 270[ |
| Mo-MCM-41 | 3 | 97 | 95 | 22[ |
| Mo-SBA-15 | 3 | 99 | 93 | 40[ |
| [PiperazinCH2{MoO2(Salen)}]n | 12 | 95 | 98 | 16[ |
| MNP30-Si-inic-Mo | 24 | 46 | 100 | 2[ |
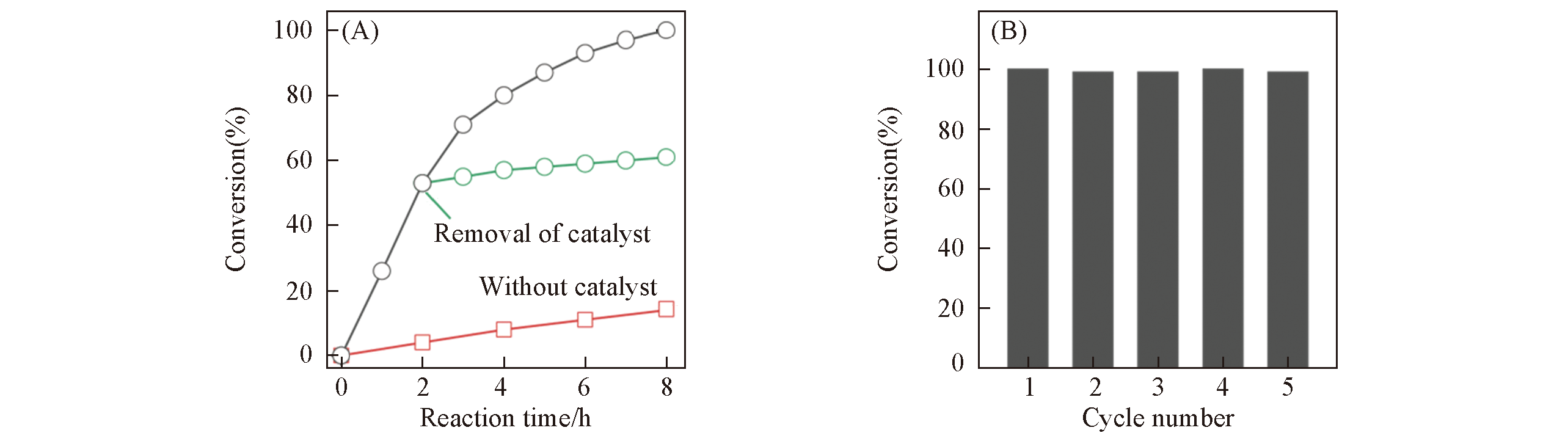
Fig.8 Time course plots of cyclooctene epoxidation(A) and reusability(B) by using OMC?US?MoO3?7 composites as catalystReaction conditions: 40.0 mmol of cyclooctene, 40.0 mmol of 5.5 mol/L TBHP in decane, 10 mg of OMC-US-MoO3-7 catalyst(0.0048 mmol/L of MoO3), 6.0 g of 1,2-dichloroethane as solvent, and 15.0 mmol of chlorobenzene as internal standard. The reaction temperature is 80 ℃.
| 1 | Xi Z., Zhou N., Sun Y., Li K., Science, 2001, 292(5519), 1139—1141 |
| 2 | Kamata K., Yonehara K., Sumida Y., Yamaguchi K., Hikichi S., Mizuno N., Science, 2003, 300(5621), 964—966 |
| 3 | Liu B., Wang P., Lopes A., Jin L., Zhong W., Pei Y., Suib S. L., He J., ACS Catal., 2017, 7(5), 3483—3488 |
| 4 | Bujak P., Bartczak P., Polanski J., J. Catal., 2012, 295, 15—21 |
| 5 | Dou J., Zeng H. C., J. Phys. Chem. C, 2012, 116(14), 7767—7775 |
| 6 | Xiao L. P., Wang S., Li H., Li Z., Shi Z. J., Xiao L., Sun R. C., Fang Y., Song G., ACS Catal., 2017, 7(11), 7535—7542 |
| 7 | Shetty M., Murugappan K., Green W. H., Román⁃Leshkov Y., ACS Sustainable Chem. Eng., 2017, 5(6), 5293—5301 |
| 8 | Doke D. S., Umbarkar S. B., Gawande M. B., Zboril R., Biradar A. V., ACS Sustainable Chem. Eng., 2016, 5(1), 904—910 |
| 9 | Noh H., Cui Y., Peters A. W., Pahls D. R., Ortuño M. A., Vermeulen N. A., Cramer C. J., Gagliardi L., Hupp J. T., Farha O. K., J. Am. Chem. Soc., 2016, 138(44), 14720—14726 |
| 10 | Luo Z., Miao R., Huan T. D., Mosa I. M., Poyraz A. S., Zhong W., Cloud J. E., Kriz D. A., Thanneeru S., He J., Adv. Energy Mater., 2016, 6(16), 1600528 |
| 11 | Brezesinski T., Wang J., Tolbert S. H., Dunn B., Nat. Mater., 2010, 9(2), 146—151 |
| 12 | Chen L., Jiang H., Jiang H., Zhang H., Guo S., Hu Y., Li C., Adv. Energy Mater., 2017, 7(15), 1602782 |
| 13 | Hosono K., Matsubara I., Murayama N., Woosuck S., Izu N., Chem. Mater., 2005, 17(2), 349—354 |
| 14 | Wang L., Gao P., Zhang G., Chen G., Chen Y., Wang Y., Bao D., Eur. J. Inorg. Chem., 2012, 2012(35), 5831—5836 |
| 15 | Fernandes C. I., Capelli S. C., Vaz P. D., Nunes, C. D., Appl. Catal. A: Gen., 2015, 504, 344—350 |
| 16 | da Palma Carreiro E., Burke A. J., J. Mol. Catal. A: Chem., 2006, 249(1), 123—128 |
| 17 | Chen W., Pei J., He C. T., Wan J., Ren H., Zhu Y., Wang Y., Dong J., Tian S., Cheong W. C., Angew. Chem. Int. Ed., 2017, 56(50), 16086—16090 |
| 18 | Solar J., Derbyshire F., De Beer V., Radovic L. R., J. Catal., 1991, 129(2), 330—342 |
| 19 | Lee S. H., Kim Y. H., Deshpande R., Parilla P. A., Whitney E., Gillaspie D. T., Jones K. M., Mahan A. H., Zhang S., Dillon A. C., Adv. Mater., 2008, 20(19), 3627—3632 |
| 20 | Tavasoli A., Karimi S., Shoja M., Int. J. Ind. Chem., 2013, 4(1), 21 |
| 21 | Wang C., Zhao Y., Zhou L., Liu Y., Zhang W., Zhao Z., Hozzein W. N., Alharbi H. M. S., Li W., Zhao D., J. Mater. Chem. A, 2018, 6(43), 21550—21557 |
| 22 | Wang C., Wan X., Duan L., Zeng P., Liu L., Guo D., Xia Y., Elzatahry A. A., Li W., Zhao D., Angew. Chem. Int. Ed., 2019, 58(44), 15863—15868 |
| 23 | Zhang P., Zhang J., Dai S., Chem. Eur. J., 2017, 23(9), 1986—1998 |
| 24 | Reyes P., Borda G., Gnecco J., Rivas B. L., J. Appl. Polym. Sci., 2004, 93(4), 1602—1608 |
| 25 | Qin M., Liu M., Zhang Q., Li C., Liang S., J. Appl. Polym. Sci., 2013, 128(1), 642—646 |
| 26 | Zhang F., Hu H., Zhong H., Yan N., Chen Q., Dalton Trans., 2014, 43(16), 6041—6049 |
| 27 | Meng Y., Gu D., Zhang F., Shi Y., Yang H., Li Z., Yu C., Tu B., Zhao D., Angew. Chem. Int. Ed., 2005, 44(43), 7053—7059 |
| 28 | Wang S., Zhao Q., Wei H., Wang J. Q., Cho M., Cho H. S., Terasaki O., Wan Y., J. Am. Chem. Soc., 2013, 135(32), 11849—11860 |
| 29 | Chen J., Burger C., Krishnan C. V., Chu B., J. Am. Chem. Soc., 2005, 127(41), 14140—14141 |
| 30 | Liu R., Shi Y., Wan Y., Meng Y., Zhang F., Gu D., Chen Z., Tu B., Zhao D., J. Am. Chem. Soc., 2006, 128(35), 11652—11662 |
| 31 | Dong W., Sun Y., Lee C. W., Hua W., Lu X., Shi Y., Zhang S., Chen J., Zhao D., J. Am. Chem. Soc., 2007, 129(45), 13894—13904 |
| 32 | Liu R., Ren Y., Shi Y., Zhang F., Zhang L., Tu B., Zhao D., Chem. Mater., 2008, 20(3), 1140—1146 |
| 33 | Patnaik S., Swain G., Parida K., Nanoscale, 2018, 10(13), 5950—5964 |
| 34 | Qin P., Fang G., Cheng F., Ke W., Lei H., Wang H., Zhao X., ACS Appl. Mater. Interfaces, 2014, 6(4), 2963—2973 |
| 35 | Ji W., Shen R., Yang R., Yu G., Guo X., Peng L., Ding W., J. Mater. Chem. A, 2014, 2(3), 699—704 |
| 36 | Qin P., Fang G., Ke W., Cheng F., Zheng Q., Wan J., Lei H., Zhao X., J. Mater. Chem. A, 2014, 2(8), 2742—2756 |
| 37 | Chandra P., Doke D. S., Umbarkar S. B., Biradar A. V., J. Mater. Chem. A, 2014, 2(44), 19060—19066 |
| 38 | Bakala P. C., Briot E., Salles L., Brégeault J. M., Appl. Catal. A: Gen., 2006, 300(2), 91—99 |
| 39 | Bagherzadeh M., Zare M., J. Coord. Chem., 2013, 66(16), 2885—2900 |
| 40 | Fernandes C. I., Carvalho M. D., Ferreira L. P., Nunes C. D., Vaz P. D., J. Organomet. Chem., 2014, 760, 2—10 |
| 41 | Muylaert I., Musschoot J., Leus K., Dendooven J., Detavernier C., Van Der Voort P., Eur. J. Inorg. Chem., 2012, 2012(2), 251—260 |
| 42 | El⁃Korso S., Bedrane S., Choukchou⁃Braham A., Bachir R., RSC Adv., 2015, 5(78), 63382—63392 |
| [1] | YANG Ruiqi, YU Xin, LIU Hong. Scientific Study of Photocatalytic Material Based on Sn3O4 [J]. Chem. J. Chinese Universities, 2021, 42(5): 1340. |
| [2] | GE Haoying, DU Jianjun, LONG Saran, SUN Wen, FAN Jiangli, PENG Xiaojun. Surface Functionalized Gold Nanomaterials in Tumor Diagnosis and Treatment [J]. Chem. J. Chinese Universities, 2021, 42(4): 1202. |
| [3] | SHI Jiangwei, MENG Nannan, GUO Yamei, YU Yifu, ZHANG Bin. Recent Advances of Two-dimensional Materials for Electrocatalytic Hydrogen Evolution [J]. Chem. J. Chinese Universities, 2021, 42(2): 492. |
| [4] | WEI Junyi, GAO Zhihua, HUANG Wei, AI Peipei, YAN Feifei, YOU Xiangxuan. Effect of Structural Ordering on the Performance of Mesoporous Carbon Supported CuCoCe Catalyst in the Synthesis of Higher Alcohols from Syngas† [J]. Chem. J. Chinese Universities, 2018, 39(8): 1741. |
| [5] | . Preparation and Optical Properties of One-dimensional Sub-microstructures of Naphthalene-1,4-dicarbonitrile [J]. Chem. J. Chinese Universities, 2011, 32(7): 1451. |
| [6] | LI Kai-Xiang, LI Xiao-Hong*, SONG Li-Ying, WU Peng. Asymmetric Hydrogenation of α-Ketoesters on Chirally Modified Pt Catalysts Supported on MPC-61 Ordered Mesoporous Carbon [J]. Chem. J. Chinese Universities, 2011, 32(6): 1354. |
| [7] | LI Xia, LIU Gui-Xia*, DONG Xiang-Ting, WANG Jin-Xian. Preparation and Properties of GdF3∶Eu3+ Luminescent Nanomaterials with Special Morphology [J]. Chem. J. Chinese Universities, 2011, 32(1): 23. |
| [8] | SUN Rui-Qin, ZHOU Xu, SUN Lin-Bing, WU Hao, CHUN Yuan*, XU Qin-Hua. Synthesis of High-specific-surface-area Solid Strong Base MgO with Pinewood as Exotemplate [J]. Chem. J. Chinese Universities, 2007, 28(12): 2333. |
| [9] | LI Dao-Rong, SUN Ling-Na, HU Chang-Wen. Influence of NH4Cl on Formation, Structure and Properties of Titania Nanocrystals [J]. Chem. J. Chinese Universities, 2006, 27(5): 797. |
| [10] | ZHANG Xiu-Juan, ZHANG Xiao-Hong, WU Shi-Kang, . Preparation of Pyrene Nanomaterials with Different Mophologies in a Surfactant Solution [J]. Chem. J. Chinese Universities, 2005, 26(7): 1330. |
| [11] | CAO Ya-Li, JIA Dian-Zeng, LIU Lang, FAN Zhao-Tian . Synthesis of Cd(OH)2 Nanorods by Solid-state Chemical Reaction at Room Temperature [J]. Chem. J. Chinese Universities, 2004, 25(9): 1601. |
| [12] | JU Wei-Gang, ZHANG Xiao-Hong, WU Shi-Kang . Preparation of TiO2 Nano-materials Using Gelator Aggregation as Template and Its Properties [J]. Chem. J. Chinese Universities, 2004, 25(12): 2308. |
| [13] | ZHAO Yong-Nan, CHEN Xiang-Ming, LI Xiu-Hong, WANG Yong, WANG Chao-Ying, LI Ming, MAI Zhen-Hong . Solvothermal Synthesis of Nanostructured Cobalt Fiber Under Microemulsion Media [J]. Chem. J. Chinese Universities, 2003, 24(6): 986. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||