

Chem. J. Chinese Universities ›› 2014, Vol. 35 ›› Issue (3): 652.doi: 10.7503/cjcu20130708
• Polymer Chemistry • Previous Articles Next Articles
Received:2013-07-25
Online:2014-03-10
Published:2019-08-01
Contact:
YAO Ping
E-mail:yaoping@fudan.edu.cn
Supported by:CLC Number:
TrendMD:
HAO Hequn, YAO Ping. In Vitro Anticancer Effects of Doxorubicin Loaded BSA-dextran Nanoparticles with Different Molecular Weights of Dextran and Different Dextran Conjugation Degrees†[J]. Chem. J. Chinese Universities, 2014, 35(3): 652.
| Sample | Mw(DEX) | n(BSA):n(DEX) in feed | m(BSA):m(DEX) in feed | Average DEX number conjugated to each BSA |
|---|---|---|---|---|
| B2-D1-62k | 62×103 | 2:1 | 1:0.47 | 0.484[ |
| B1-D1-62k | 1:1 | 1:0.94 | ||
| B1-D2-62k | 1:2 | 1:1.88 | ||
| B1-D3-10k | 10×103 | 1:3 | 1:0.45 | 2.4 |
| B1-D4-10k | 1:4 | 1:0.61 | 2.7 | |
| B1-D5-10k | 1:5 | 1:0.76 | 3.1 | |
| B1-D6-5k | 5×103 | 1:6 | 1:0.45 | |
| B1-D7-5k | 1:7 | 1:0.53 | ||
| B1-D8-5k | 1:8 | 1:0.61 | 5.9 | |
| B1-D9-5k | 1:9 | 1:0.68 | 7.5 |
Table 1 BSA-DEX conjugates produced from different molecular weights of dextran and different molar ratios of BSA to dextran
| Sample | Mw(DEX) | n(BSA):n(DEX) in feed | m(BSA):m(DEX) in feed | Average DEX number conjugated to each BSA |
|---|---|---|---|---|
| B2-D1-62k | 62×103 | 2:1 | 1:0.47 | 0.484[ |
| B1-D1-62k | 1:1 | 1:0.94 | ||
| B1-D2-62k | 1:2 | 1:1.88 | ||
| B1-D3-10k | 10×103 | 1:3 | 1:0.45 | 2.4 |
| B1-D4-10k | 1:4 | 1:0.61 | 2.7 | |
| B1-D5-10k | 1:5 | 1:0.76 | 3.1 | |
| B1-D6-5k | 5×103 | 1:6 | 1:0.45 | |
| B1-D7-5k | 1:7 | 1:0.53 | ||
| B1-D8-5k | 1:8 | 1:0.61 | 5.9 | |
| B1-D9-5k | 1:9 | 1:0.68 | 7.5 |
| Sample | Fresh prepared | After 30 d | ||||
|---|---|---|---|---|---|---|
| Dh/nm | PDI | LE(%) | LA(%) | Dh/nm | PDI | |
| DOX/B2-D1-62k | 198±14 | 0.13±0.04 | 92.4±1.9 | 15.7±0.5 | 194±15 | 0.11±0.02 |
| DOX/B1-D1-62kb | 140±6 | 0.15±0.01 | 91.0±1.8 | 11.7±0.2 | — | — |
| DOX/B1-D2-62kb | 108±8 | 0.14±0.02 | 84.8±0.8 | 7.4±0.1 | — | — |
| DOX/B1-D3-10k | 106±8 | 0.24±0.04 | 98.0±0.2 | 16.9±0.1 | 104±8 | 0.24±0.02 |
| DOX/B1-D4-10kc | 68±8 | 0.24±0.02 | 95.9±0.4 | 14.9±0.1 | 72±10 | 0.22±0.04 |
| DOX/B1-D5-10k | 58±6 | 0.28±0.08 | 95.6±0.6 | 13.7±0.1 | 58±4 | 0.29±0.04 |
| DOX/B1-D6-5k | 198±2 | 0.31±0.01 | 84.3±0.6 | 14.5±0.3 | 208±8 | 0.34±0.02 |
| DOX/B1-D7-5k | 114±2 | 0.25±0.02 | 83.9±0.4 | 14.0±0.2 | 118±6 | 0.27±0.01 |
| DOX/B1-D8-5k | 76±2 | 0.24±0.01 | 82.8±0.3 | 13.3±0.2 | 74±6 | 0.25±0.02 |
| DOX/B1-D9-5k | 66±2 | 0.37±0.01 | 81.1±0.7 | 12.5±0.1 | 70±4 | 0.36±0.03 |
Table 2 Size distribution, loading efficiency(LE), and loading amount(LA) results of DOX loaded nanoparticles before and after 30 d storagea
| Sample | Fresh prepared | After 30 d | ||||
|---|---|---|---|---|---|---|
| Dh/nm | PDI | LE(%) | LA(%) | Dh/nm | PDI | |
| DOX/B2-D1-62k | 198±14 | 0.13±0.04 | 92.4±1.9 | 15.7±0.5 | 194±15 | 0.11±0.02 |
| DOX/B1-D1-62kb | 140±6 | 0.15±0.01 | 91.0±1.8 | 11.7±0.2 | — | — |
| DOX/B1-D2-62kb | 108±8 | 0.14±0.02 | 84.8±0.8 | 7.4±0.1 | — | — |
| DOX/B1-D3-10k | 106±8 | 0.24±0.04 | 98.0±0.2 | 16.9±0.1 | 104±8 | 0.24±0.02 |
| DOX/B1-D4-10kc | 68±8 | 0.24±0.02 | 95.9±0.4 | 14.9±0.1 | 72±10 | 0.22±0.04 |
| DOX/B1-D5-10k | 58±6 | 0.28±0.08 | 95.6±0.6 | 13.7±0.1 | 58±4 | 0.29±0.04 |
| DOX/B1-D6-5k | 198±2 | 0.31±0.01 | 84.3±0.6 | 14.5±0.3 | 208±8 | 0.34±0.02 |
| DOX/B1-D7-5k | 114±2 | 0.25±0.02 | 83.9±0.4 | 14.0±0.2 | 118±6 | 0.27±0.01 |
| DOX/B1-D8-5k | 76±2 | 0.24±0.01 | 82.8±0.3 | 13.3±0.2 | 74±6 | 0.25±0.02 |
| DOX/B1-D9-5k | 66±2 | 0.37±0.01 | 81.1±0.7 | 12.5±0.1 | 70±4 | 0.36±0.03 |

Fig.1 TEM images of DOX/B2-D1-62k(A), DOX/B1-D5-10k(B) and DOX/B1-D9-5k(C) nanoparticles Samples (B) and (C) were negative stained with phosphotungstic acid.
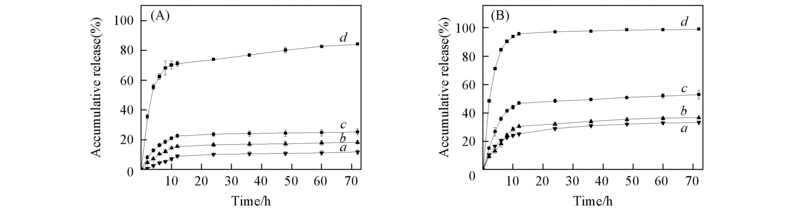
Fig.2 Accumulative release of DOX from DOX/B2-D1-62k(a), DOX/B1-D5-10k(b) and DOX/B1-D9-5k(c) nanoparticles in PBS(A) and 0.1 mol/L pH=5.0 acetate buffer(B) Free DOX solution was assayed as a control(curve d).
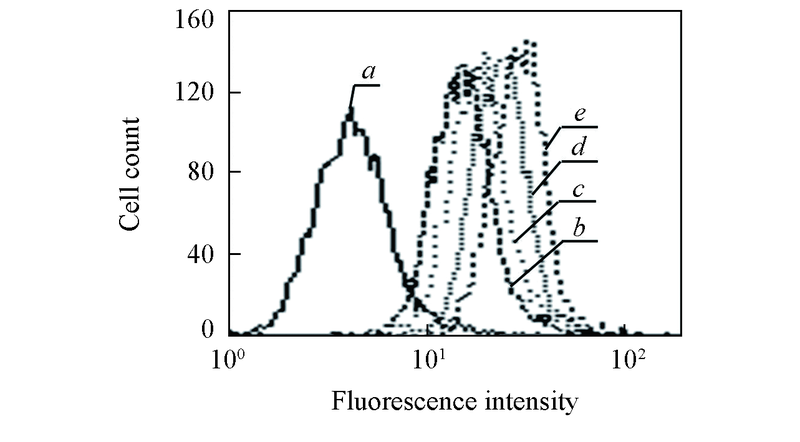
Fig.3 Flow cytometry analysis of the cellular uptake of control(a), free DOX(b), DOX/B2-D1-62k(c), DOX/B1-D5-10k(d) and DOX/B1-D9-5k(e) nanoparticles with a fixed DOX concentration of 2 mg/mL after 2 h incubation at 37 ℃

Fig.4 Confocal fluorescence microscopy images of cellular uptake of free DOX(A, B) and DOX/B1-D9-5k nanoparticles(C, D) with a fixed DOX concentration of 2 μg/mL after 2 h incubation at 37 ℃(A, C) and 4 ℃(B, D)
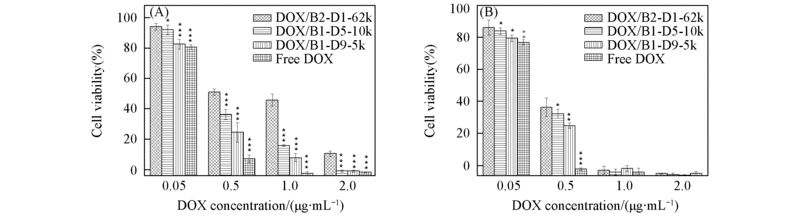
Fig.6 Cell viability against free DOX solution as well as DOX/B2-D1-62k, DOX/B1-D5-10k and DOX/B1-D9-5k nanoparticles after 48 h(A) and 72 h(B) incubation with different DOX concentrations* P>0.05 compared with DOX/B2-D1-62k group; ** P<0.05 compared with DOX/B2-D1-62k group; *** P<0.01 compared with DOX/B2-D1-62k group.
| [1] | Nakanishi T., Fukushima S., Okamoto K., Suzuki M., Matsumura Y., Yokoyama M., Okano T., Sakurai Y., Kataoka K., Journal of Controlled Release,2001, 74(1—3), 295—302 |
| [2] | Minotti G., Menna P., Salvatorelli E.,Cairo G., Gianni L., Pharmacological Reviews,2004, 56(2), 185—229 |
| [3] | Zhang J. C., Ding J. X., Xiao C. S., He C. L., Zhuang X. L., Yang Y. L., Chen X. S., Chem. J. Chinese Universities,2012, 33(12), 2809—2815 |
| (张建成, 丁建勋, 肖春生, 贺超良, 庄秀丽, 杨亚楠, 陈雪思. 高等学校化学学报, 2012, 33(12), 2809—2815) | |
| [4] | Torchilin V. P., European Journal of Pharmaceutics Sciences, 2000, 11(2), S81—S91 |
| [5] | Matsumura Y., Maeda H., Cancer Research,1986, 46, 6387—6392 |
| [6] | Maeda H., Sawa T., Konno T., Journal of Controlled Release,2001, 74, 47—61 |
| [7] | Maeda H., Bharate G.Y., Daruwalla J., European Journal of Pharmaceutics and Biopharmaceutics,2009, 71, 409—419 |
| [8] | Maeda H., Wu J., Sawa T., Matsumura Y., Hori K., Journal of Controlled Release,2000, 65(1/2), 271—284 |
| [9] | Gullotti E., Yeo Y., Molecular Pharmaceatics,2009, 6(4), 1041—1051 |
| [10] | Arnedo A., Espuelas S., Irache J. M., International Journal of Pharmaceutics,2002, 244(1/2), 59—72 |
| [11] | Sahin S., Selek H., Pronchel G., Ercan M. T., Sargon M., Hincal A. A., Kas H. S., Journal of Controlled Release,2002, 82(2/3), 345—358 |
| [12] | Chuo W. H., Tsai T. R., Hsu S. H., Cham T. M., International Journal of Pharmaceutics,1996, 144(2), 241—245 |
| [13] | Zhang Y. D., Pang Y., Lu J., Wei X. Y., Yang W. J., Zhu M. L., Chem. J. Chinese Universities,2010, 31(10), 1999—2004 |
| (张亚东, 庞燕, 陆洁, 魏晓焱, 杨文江, 朱美霖. 高等学校化学学报, 2010, 31(10), 1999—2004) | |
| [14] | Elzoghby A. O., Samy W. M., Elgindy N. A., Journal of Controlled Release,2012, 157(2), 168—182 |
| [15] | Chuang V. T. G., Kragh-Hansen U., Otagiri M., Pharmaceutical. Research,2002, 19(5), 569—577 |
| [16] | Wu F., Wuensc S. A., Azadniv M., Ebrahimkhani M. R., Crispe I. N., Molecular Pharmaceutics,2009, 6(5), 1506—1517 |
| [17] | Kratz F., Journal of Controlled Release, 2008, 132(3), 171—183 |
| [18] | Peters T.Jr., Reed R. G., Albumin: Structure, Biosynthesis, Function, Pergamon Press, 1978, 50, 11—20 |
| [19] | Boye J. I., Alli I., Ismail A. A., Journal of Agricultural and Food Chemistry,1996, 44(4), 996—1004 |
| [20] | Tang Q. S., Chen D. Z., Xue W. Q., Xiang J. Y., Gong Y. C., Gong Y. C., Zhang L., Guo C. Q., International Journal of Nanomedicine,2011, 6, 3077—3085 |
| [21] | Passirani C., Barratt G., Devissaguet J. P., Labarre D., Pharmaceutical Research,1998, 15(7), 1046—1050 |
| [22] | Lemarchand C., Gref R., Couvreur P., European Journal of Pharmaceutics and Biopharmaceutics,2004, 58(2), 327—341 |
| [23] | Mehvar R., Journal of Controlled Release, 2000, 69(1), 1—25 |
| [24] | Alhareth K., Vauthier C., Bourasset F., Gueutin C., Ponchel G., Moussa F., European Journal of Pharmaceutics and Biopharmaceutics,2012, 81, 453—457 |
| [25] | Deng W., Li J., Yao P., He F., Huang C., Macromolecular Bioscience,2010, 10(10), 1224—1234 |
| [26] | Hao H. Q., Ma Q. M., Huang C., He F., Yao P., International Journal of Pharmaceutics,2013, 444, 77—84 |
| [27] | Lesniak A., Salvati A., Santos-Martinez M. J., Radomski M. W., Dawson K. A., Åberg C., Journal of the American Chemical Society,2013, 135, 1438—1444 |
| [28] | Fayle S.E., Gerrard J. A., The Maillard Reaction, The Royal Society of Chemistry, Cambridge, 2002, 1—17 |
| [29] | Hodge J. E., Journal of Agricultural and Food Chemistry, 1953, 1(15), 928—943 |
| [30] | Yu S. Y., Gao G., Yao P., Jiang M., Chem. J. Chinese Universities,2008, 29(5), 1027—1032 |
| (喻绍勇, 高杲, 姚萍, 江明. 高等学校化学学报, 2008, 29(5), 1027—1032) | |
| [31] | Deng W., Self-assembly of Complexes Prepared by Protein and Polysaccharides and Its Applications, Fudan University, Shanghai, 2010 |
| (邓伟. 蛋白与多糖复合物的自组装及其应用研究, 上海: 复旦大学, 2010) | |
| [32] | Yang S. C., Ge H. X., Hu Y., Jiang X. Q., Yang C. Z., Journal of Applied Polymer Science,2000, 78(8), 517—526 |
| [33] | Bouzin C., Feron O., Drug Resistance Updates,2007, 10(3), 109—120 |
| [34] | Van Sluis R., Bhujwalla Z. M., Raghunand N., Ballesteros P., Alvarez J., Cerdan S., Galons J. P., Gillies R. J., Magnetic Resonance in Medicine,1999, 41(4), 743—750 |
| [35] | Yang C. F., Sun Y., Zhang L. J., Zhu G. D., Zhang C. Y., Qian Y., Chinese Jounal of Chemistry,2012, 30(9), 1980—1986 |
| [36] | Mao Z. W., Zhang Y. H., Li H. Y., Tong W. J., Gao C. Y., Progress in Chemistry,2013, 25, 1061—1070 |
| [37] | Safi M., Courtois J., Seigneuret M., Conjeaud H., Berret J. F., Biomaterials,2011, 32, 9353—9363 |
| [38] | Qi J. N., Yao P., He F., Yu C. L., Huang C., International Journal of Pharmaceutics,2010, 393, 176—184 |
| [39] | Cai H. X., Yao P., Nanoscale,2013, 5, 2892—2900 |
| [1] | Jinhan Sheng, Qizhen Zheng, Ming Wang. Non-viral delivery of CRISPR/Cas9 Genome Editing [J]. Chem. J. Chinese Universities, 2022, 43(Album-4): 20220344. |
| [2] | WU Yushuai, SHANG Yingxu, JIANG Qiao, DING Baoquan. Research Progress of Controllable Self-assembled DNA Origami Structure as Drug Carrier [J]. Chem. J. Chinese Universities, 2022, 43(8): 20220179. |
| [3] | LIU Shuwei, JIN Hao, YIN Wanzhong, ZHANG Hao. Gemcitabine/polypyrrole Composite Nanoparticles for Chemo-photothermal Combination Ovarian Cancer Therapy [J]. Chem. J. Chinese Universities, 2022, 43(8): 20220345. |
| [4] | WANG Hong, SAN Khin Nyein Ei, FANG Yun, ZHANG Xinyu, FAN Ye. Pickering Emulsion Stabilization and Interfacial Catalytic Oxidation by Janus Nano-Au [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220105. |
| [5] | WANG Guangqi, BI Yiyang, WANG Jiabo, SHI Hongfei, LIU Qun, ZHANG Yu. Heterostructure Construction of Noble-metal-free Ternary Composite Ni(PO3)2-Ni2P/CdS NPs and Its Visible Light Efficient Catalytic Hydrogen Production [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220050. |
| [6] | JI Fa, LIU Ling, YU Linling, SUN Yan. Effects of Muco-inert and Acid-sensitive Modification on Mucosal Penetration of Nanoparticles [J]. Chem. J. Chinese Universities, 2022, 43(6): 20210837. |
| [7] | SUN Xuefeng, RENAGUL Abdurahman, YANG Tongsheng, YANG Qianting. Synthesis and Luminescence Properties of Cr,In Co-doped Small Size MgGa2O4 Near-infrared Persistent Luminescence Nanoparticles [J]. Chem. J. Chinese Universities, 2022, 43(4): 20210850. |
| [8] | TAO Xingfu, HAN Chenglong, YANG Yang, LIU Kun. Synthesis of Aluminum Nanoparticles@Polymer Core-shell Nanostructures by Surface-initiated Polymerization [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220367. |
| [9] | YANG Zhaohua, CHENG Hongjing, YANG Yi, LIU Hui, DU Feipeng, ZHANG Yunfei. Preparation of Silver-loaded Polyvinyl Alcohol Sponge and Its Interfacial Photothermal Driven Water Evaporation Performance [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220181. |
| [10] | CHU Mingyue, LI Fengbo, GAO Ning, YANG Xin, YU Tingting, MA Huiyuan, YANG Guixin, PANG Haijun. Construction of a Coronal Polyoxometalate-based Composite Film for Determination of Nitrite [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210579. |
| [11] | ZHAO Ying, QIAO Ling, ZHAO Guofeng, CHEN Li. Synthesis and Biological Activity of Lycorine Derivatives Containing Malate Ester [J]. Chem. J. Chinese Universities, 2021, 42(9): 2789. |
| [12] | CHEN Shaoyun, ZHANG Xingying, LIU Ben, TIAN Du, LI Qi, CHEN Fang, HU Chenglong, CHEN Jian. Controllable Growth of Silver Nanoparticles on TiO2 Tetragonal Prism Nanarrays and Its SERS Effect [J]. Chem. J. Chinese Universities, 2021, 42(8): 2381. |
| [13] | WANG Yawen, LI Dong, LIANG Wenkai, SUN Yinghui, JIANG Lin. Multiplex Structures of Plasmonic Metal Nanoparticles and Their Applications [J]. Chem. J. Chinese Universities, 2021, 42(4): 1213. |
| [14] | LIU Zhigang, LI Jiabao, YANG Jian, MA Hao, WANG Chengyin, GUO Xin, WANG Guoxiu. Preparation of a Novel g-C3N4/Sn/N-doped Carbon Composite for Sodium Storage [J]. Chem. J. Chinese Universities, 2021, 42(2): 633. |
| [15] | PAN Jing, XU Minmin, YUAN Yaxian, YAO Jianlin. Rapid Detection of Banned Dyes in Textiles Based on Surface-enhanced Raman Spectroscopy [J]. Chem. J. Chinese Universities, 2021, 42(12): 3716. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||
