

Chem. J. Chinese Universities ›› 2017, Vol. 38 ›› Issue (6): 1099.doi: 10.7503/cjcu20160816
• Polymer Chemistry • Previous Articles
HAN Na*( ), WANG Xiufang, QU Tingsi, QIAN Yongqiang, LU Yahong
), WANG Xiufang, QU Tingsi, QIAN Yongqiang, LU Yahong
Received:2016-11-21
Online:2017-06-10
Published:2017-04-11
Contact:
HAN Na
E-mail:hanna@tjpu.edu.cn
Supported by:CLC Number:
TrendMD:
HAN Na, WANG Xiufang, QU Tingsi, QIAN Yongqiang, LU Yahong. Preparation and Properties of Cellulose Benzoate and Preliminary Exploration About Cellulose Benzoate-g-polyoxyethylene(2) Hexadecyl Ether†[J]. Chem. J. Chinese Universities, 2017, 38(6): 1099.
| Sample | Molar ratio of acyl chloride to AGU | Temperature/℃ | Time/h | DSa | Sample | Molar ratio of acyl chloride to AGU | Temperature/℃ | Time/h | DSa |
|---|---|---|---|---|---|---|---|---|---|
| S-1 | 6∶1b | 50 | 2 | 0.81 | S-9 | 5∶1b | 70 | 2 | 0.76 |
| S-2 | 3∶1b | 60 | 2 | 0.13 | S-10 | 6∶1b | 70 | 2 | 1.05 |
| S-3 | 4∶1b | 60 | 2 | 0.56 | S-11 | 9∶1b | 70 | 2 | 2.98 |
| S-4 | 5∶1b | 60 | 2 | 0.63 | S-12 | 4∶1b | 80 | 2 | 0.69 |
| S-5 | 6∶1b | 60 | 2 | 0.97 | S-13 | 5∶1b | 80 | 2 | 0.80 |
| S-6 | 9∶1b | 60 | 2 | 2.80 | S-14 | 6∶1b | 80 | 2 | 1.82 |
| S-7 | 3∶1b | 70 | 2 | 0.18 | S-15 | 12∶1c | 50 | 2 | 2.05 |
| S-8 | 4∶1b | 70 | 2 | 0.62 |
Table 1 Degrees of substitution and reaction conditions of cellulose and acyl chloride
| Sample | Molar ratio of acyl chloride to AGU | Temperature/℃ | Time/h | DSa | Sample | Molar ratio of acyl chloride to AGU | Temperature/℃ | Time/h | DSa |
|---|---|---|---|---|---|---|---|---|---|
| S-1 | 6∶1b | 50 | 2 | 0.81 | S-9 | 5∶1b | 70 | 2 | 0.76 |
| S-2 | 3∶1b | 60 | 2 | 0.13 | S-10 | 6∶1b | 70 | 2 | 1.05 |
| S-3 | 4∶1b | 60 | 2 | 0.56 | S-11 | 9∶1b | 70 | 2 | 2.98 |
| S-4 | 5∶1b | 60 | 2 | 0.63 | S-12 | 4∶1b | 80 | 2 | 0.69 |
| S-5 | 6∶1b | 60 | 2 | 0.97 | S-13 | 5∶1b | 80 | 2 | 0.80 |
| S-6 | 9∶1b | 60 | 2 | 2.80 | S-14 | 6∶1b | 80 | 2 | 1.82 |
| S-7 | 3∶1b | 70 | 2 | 0.18 | S-15 | 12∶1c | 50 | 2 | 2.05 |
| S-8 | 4∶1b | 70 | 2 | 0.62 |
| Sample | Mass fraction of cellulose(%) | Mass ratio of[Emim]Ac to DMAc | Added way | Mass ratio of BC to AGU | Temperature/℃ | Time/h |
|---|---|---|---|---|---|---|
| SS-1 | 10 | 1∶0 | 6∶1 | 70 | 2 | |
| SS-2 | 10 | 1∶0 | 6∶1 | 80 | 2 | |
| SS-3 | 8 | 8∶2 | Mix | 6∶1 | 80 | 2 |
| SS-4 | 8 | 7∶3 | Mix | 6∶1 | 80 | 2 |
| SS-5 | 8 | 6∶4 | Mix | 6∶1 | 80 | 2 |
| SS-6 | 8 | 5∶5 | Mix | 6∶1 | 80 | 2 |
| SS-7 | 8 | 7∶3 | Div | 6∶1 | 80 | 2 |
Table 2 Reaction condition of cellulose and benzoyl chloride*
| Sample | Mass fraction of cellulose(%) | Mass ratio of[Emim]Ac to DMAc | Added way | Mass ratio of BC to AGU | Temperature/℃ | Time/h |
|---|---|---|---|---|---|---|
| SS-1 | 10 | 1∶0 | 6∶1 | 70 | 2 | |
| SS-2 | 10 | 1∶0 | 6∶1 | 80 | 2 | |
| SS-3 | 8 | 8∶2 | Mix | 6∶1 | 80 | 2 |
| SS-4 | 8 | 7∶3 | Mix | 6∶1 | 80 | 2 |
| SS-5 | 8 | 6∶4 | Mix | 6∶1 | 80 | 2 |
| SS-6 | 8 | 5∶5 | Mix | 6∶1 | 80 | 2 |
| SS-7 | 8 | 7∶3 | Div | 6∶1 | 80 | 2 |
| Sample | Molar ratio of BC to AGU | T1/℃ | t1/h | Molar ratio of E2C16 to AGU | T2/℃ | t2/h | Yield(%) |
|---|---|---|---|---|---|---|---|
| D1a | 6∶1 | 70 | 2 | 4∶1 | 90 | 6 | 81.0 |
| D2b | 6∶1 | 70 | 2 | 4∶1 | 90 | 6 | 32.2 |
| D3b | 6∶1 | 70 | 2 | 4∶1 | 70 | 6 | 21.5 |
Table 3 Fabrication conditions and yield of CB-g-E2C16 solid-solid phase change materials
| Sample | Molar ratio of BC to AGU | T1/℃ | t1/h | Molar ratio of E2C16 to AGU | T2/℃ | t2/h | Yield(%) |
|---|---|---|---|---|---|---|---|
| D1a | 6∶1 | 70 | 2 | 4∶1 | 90 | 6 | 81.0 |
| D2b | 6∶1 | 70 | 2 | 4∶1 | 90 | 6 | 32.2 |
| D3b | 6∶1 | 70 | 2 | 4∶1 | 70 | 6 | 21.5 |
| Sample | Solvent | Molar ratio of benzoyl chloride to AGU | Temperature/℃ | DSB | DSA | DS |
|---|---|---|---|---|---|---|
| SS-4 | [Emim]Ac/DMAc/mix | 6∶1 | 80 | 0.09 | 2.04 | 2.13 |
| SS-7 | [Emim]Ac/DMAc/div | 6∶1 | 80 | 0.11 | 2.50 | 2.61 |
| SS-2 | [Emim]Ac | 6∶1 | 80 | 0.15 | 2.71 | 2.86 |
Table 4 Degrees of substitution of CB and CA mixed ester synthesized in [Emim]Ac*
| Sample | Solvent | Molar ratio of benzoyl chloride to AGU | Temperature/℃ | DSB | DSA | DS |
|---|---|---|---|---|---|---|
| SS-4 | [Emim]Ac/DMAc/mix | 6∶1 | 80 | 0.09 | 2.04 | 2.13 |
| SS-7 | [Emim]Ac/DMAc/div | 6∶1 | 80 | 0.11 | 2.50 | 2.61 |
| SS-2 | [Emim]Ac | 6∶1 | 80 | 0.15 | 2.71 | 2.86 |
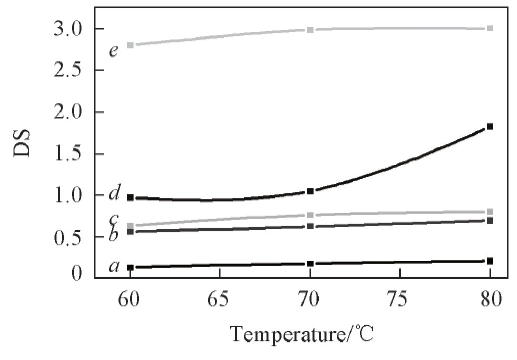
Fig.6 Relation curves of substitution degreeand reaction temperature of CB in [Amim]ClMolar ratio of BC to AGu: a. 3∶1; b. 4∶1; c. 5∶1; d. 6∶1; e. 9∶1.
| Sample | DSA | DSB | DMSO | DMF | Acetone |
|---|---|---|---|---|---|
| MCC | - | - | - | ||
| S-2 | 0.13 | + | - | - | |
| S-3 | 0.56 | + | + | - | |
| S-5 | 0.97 | + | + | - | |
| S-10 | 1.05 | + | + | - | |
| S-6 | 2.80 | + | + | + | |
| S-15 | 2.05 | + | + | - | |
| SS-2 | 2.71 | 0.15 | + | + | - |
| SS-4 | 2.04 | 0.09 | + | + | - |
| SS-7 | 2.50 | 0.11 | + | + | - |
Table 5 Solubility of CB, CA and mixed esters in different solutions*
| Sample | DSA | DSB | DMSO | DMF | Acetone |
|---|---|---|---|---|---|
| MCC | - | - | - | ||
| S-2 | 0.13 | + | - | - | |
| S-3 | 0.56 | + | + | - | |
| S-5 | 0.97 | + | + | - | |
| S-10 | 1.05 | + | + | - | |
| S-6 | 2.80 | + | + | + | |
| S-15 | 2.05 | + | + | - | |
| SS-2 | 2.71 | 0.15 | + | + | - |
| SS-4 | 2.04 | 0.09 | + | + | - |
| SS-7 | 2.50 | 0.11 | + | + | - |
| Sample | DSB | DSE | DS | ΔHm/(J·g-1) | Δ | Tcp/℃ | ΔHc/(J·g-1) | Δ | ||
|---|---|---|---|---|---|---|---|---|---|---|
| E2C16 | 100 | 34.4 | 104 | 104 | 23.9 | 95 | 95 | |||
| D1 | 1.05 | 1.24 | 2.29 | 0.52 | 23.9 | 23 | 54 | 15.0 | 24 | 49 |
| D2 | 1.05 | 1.06 | 2.11 | 0.50 | 29.1 | 17 | 52 | 22.8 | 20 | 47 |
| D3 | 1.05 | 1.01 | 2.06 | 0.50 | 12.7 | 12 | 52 | 18.7 | 15 | 47 |
Table 6 DSC data of E2C16 and CB-g-E2C16
| Sample | DSB | DSE | DS | ΔHm/(J·g-1) | Δ | Tcp/℃ | ΔHc/(J·g-1) | Δ | ||
|---|---|---|---|---|---|---|---|---|---|---|
| E2C16 | 100 | 34.4 | 104 | 104 | 23.9 | 95 | 95 | |||
| D1 | 1.05 | 1.24 | 2.29 | 0.52 | 23.9 | 23 | 54 | 15.0 | 24 | 49 |
| D2 | 1.05 | 1.06 | 2.11 | 0.50 | 29.1 | 17 | 52 | 22.8 | 20 | 47 |
| D3 | 1.05 | 1.01 | 2.06 | 0.50 | 12.7 | 12 | 52 | 18.7 | 15 | 47 |
| Sample | Tdo/℃ | Tdp/℃ | Mass loss(%) |
|---|---|---|---|
| Cellulose | 319 | 335 | 85 |
| E2C16 | 246 | 295 | 98 |
| D1 | 281 | 320 | 89 |
| D2 | 294 | 334 | 88 |
| D3 | 282 | 325 | 87 |
Table 7 TG analysis of CB-g-E2C16
| Sample | Tdo/℃ | Tdp/℃ | Mass loss(%) |
|---|---|---|---|
| Cellulose | 319 | 335 | 85 |
| E2C16 | 246 | 295 | 98 |
| D1 | 281 | 320 | 89 |
| D2 | 294 | 334 | 88 |
| D3 | 282 | 325 | 87 |
| [1] | Hebeish A., Guthrie J., The Chemistry and Technology of Cellulosic Copolymers, Springer Science & Business Media, 2012 |
| [2] | Klemm D., Heublein B., Fink H.P., Angew. Chem. Int. Ed., 2005, 44, 3358—3393 |
| [3] | Moon R.J., Martini A., Nairn J., Chem. Soc. Rev., 2011, 40, 3941—3994 |
| [4] | Sun N., Swatloski R.P.,Maxim M. L., Rahman M., Harland A. G., Haque A., Spear S. K., Daly D. T., Rogers R. D., Mater. J.Chem., 2008, 18, 283—290 |
| [5] | Zavrel M., Bross D., Funke M., Bioresour. Technol., 2009, 100, 2580—2587 |
| [6] | Ren Q., Wu J., Zhang J., Acta Polymerica Sinica, 2003, 1(3), 448—451 |
| (任强, 武进, 张军. 高分子学报, 2003, 1(3), 448—451) | |
| [7] | Andanson J.M., Bordes E., Devémy J., Green Chemistry, 2014, 16(5), 2528—2538 |
| [8] | Ries M.E., Radhi A., Keating A. S., Biomacromolecules, 2014, 15, 609—617 |
| [9] | Zavrel M., Bross D., Funke M., Bioresour. Technol., 2009, 100, 2580—2587 |
| [10] | Liu L.H., Xue F., Ding E. Y., Polymeric Materials Science and Engineering, 2014, 10, 95—99 |
| (刘丽华, 薛锋, 丁恩勇. 高分子材料科学与工程, 2014, 10, 95—99) | |
| [11] | Le K.A., Rudaz C., Budtova T., Carbohydrate Polymers, 2014, 105, 237—243 |
| [12] | Xiao P., Zhang J., Feng Y., Cellulose, 2014, 21(4), 2369—2378 |
| [13] | Aydin A., Okutan H., Solar Energy Materials and Solar Cells, 2011, 95(10), 2752—2762 |
| [14] | Zhang L., Shi H., Li W., Polymer International, 2014, 63(6), 982—988 |
| [15] | Zhang L., Shi H., Li W., Thermochimica Acta, 2013, 570(9), 1—7 |
| [16] | Han N., Li Z., Zhang X., Cellulose, 2016, 23(3), 1663—1674 |
| [17] | Zhang J., Wu J., Cao Y., Cellulose, 2009, 16(2), 299—308 |
| [18] | Wang R., Fu Y., Qin M., Bio. Resources, 2014, 9(3), 5134—5146 |
| [19] | Goodlett V.W., Dougherty J. T., Patton H. W., Polym. Sci. Part Polym. Chem., 1971, 9, 155—161 |
| [20] | Li X., Zhang Y., Tang J., Journal of Polymer Research, 2016, 23(2), 1—8 |
| [1] | CUI Wei, ZHAO Deyin, BAI Wenxuan, ZHANG Xiaodong, YU Jiang. CO2 Absorption in Composite of Aprotic Solvent and Iron-based Ionic Liquid [J]. Chem. J. Chinese Universities, 2022, 43(8): 20220120. |
| [2] | PENG Kuilin, LI Guilin, JIANG Chongyang, ZENG Shaojuan, ZHANG Xiangping. Research Progress for the Role of Electrolytes in the CO2 Electrochemical Reduction [J]. Chem. J. Chinese Universities, 2022, 43(7): 20220238. |
| [3] | JI Shuangqi, JIN Zhao, GUAN Wenna, PAN Xiangyu, GUAN Tong. Preparation and Chromatographic Performance of Mixed-mode Silica Stationary Phase Modified by Double Cationic Ionic Liquid and Octadecyl Group [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220008. |
| [4] | CHANG Sihui, CHEN Tao, ZHAO Liming, QIU Yongjun. Thermal Degradation Mechanism of Bio-based Polybutylactam Plasticized by Ionic Liquids [J]. Chem. J. Chinese Universities, 2022, 43(11): 20220353. |
| [5] | WAN Ren, SONG Fan, PENG Changjun, LIU Honglai. Group Contribution Method for Infinite Dilution Molar Conductivity of Unconventional Ions in Water [J]. Chem. J. Chinese Universities, 2021, 42(12): 3672. |
| [6] | WANG Man, WANG Xin, ZHOU Jing, GAO Guohua. Efficient Synthesis of Dimethyl Carbonate via Transesterification of Methanol and Ethylene Carbonate Catalyzed by Poly(ionic liquid)s [J]. Chem. J. Chinese Universities, 2021, 42(12): 3701. |
| [7] | ZHOU Molin, JIANG Xin, YI Ting, YANG Xiangguang, ZHANG Yibo. Improvement of Interface Stability Between Sulfide Solid Electrolyte Li10GeP2S12 and Lithium Metal [J]. Chem. J. Chinese Universities, 2020, 41(8): 1810. |
| [8] | CHENG Shifu,HU Hao,CHEN Bihua,WU Haihong,GAO Guohua,HE Mingyuan. Preparation and Electrochemical Performance of Porous Carbons Prepared from Binary Ionic Liquids † [J]. Chem. J. Chinese Universities, 2020, 41(5): 1048. |
| [9] | GAO Chong,YU Fengli,XIE Congxia,YU Shitao. Baeyer-Villiger Oxidation of Cyclic Ketones Catalyzed by Amino Alcohol Heteropoly Acid Ionic Liquid † [J]. Chem. J. Chinese Universities, 2020, 41(5): 1101. |
| [10] | GAO Naiwei, MA Qiang, HE Yonglin, WANG Yapei. Green Electronic Devices Based on Ionic Liquids † [J]. Chem. J. Chinese Universities, 2020, 41(5): 901. |
| [11] | PIAO Huilan,MA Pinyi,QIN Zucheng,JIANG Yanxiao,SUN Ying,WANG Xinghua,SONG Daqian. Determination of Triazine Herbicides from Fruit Juice Samples Using Effervescence Assisted Microextraction Method Based on Acidic Ionic Liquid Packed Syringe [J]. Chem. J. Chinese Universities, 2020, 41(2): 228. |
| [12] | ZHANG Li,QIAN Mingchao,LIU Xueke,Gao Shuaitao,YU Jiang,XIE Haishen,WANG Hongbin,SUN Fengjiang,SU Xianghong. Dynamic Study of Oxidative Desulfurization by Iron-based Ionic Liquids/NHD † [J]. Chem. J. Chinese Universities, 2020, 41(2): 317. |
| [13] | WANG Nan,YAO Kaisheng,ZHAO Chenchen,LI Tianjin,LU Weiwei. Ionic Liquid-assisted Synthesis of AuPd Nanosponges and Their Catalytic Performance † [J]. Chem. J. Chinese Universities, 2020, 41(1): 62. |
| [14] | LI Chenguang, HUA Er, LIU Tianxia. Tribological Behaviour of Protic Ionic Liquid Composed of 2-Ethylhexylethylenediaminium Cation and Trifluoromethanesulfonate Anion as Liquid Paraffin Additive† [J]. Chem. J. Chinese Universities, 2019, 40(7): 1411. |
| [15] | LIU Xiaozhou, GUAN Xinyu, FANG Qianrong, JIN Yongri. Three-dimensional Covalent Organic Frameworks Synthesized by Room Temperature Ionic Liquid Method† [J]. Chem. J. Chinese Universities, 2019, 40(7): 1341. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||