

Chem. J. Chinese Universities ›› 2016, Vol. 37 ›› Issue (10): 1817.doi: 10.7503/cjcu20160360
• Physical Chemistry • Previous Articles Next Articles
LIU Ning, WANG Jiqiong, CHEN Biaohua, LI Yingxia, ZHANG Runduo*( )
)
Received:2016-05-20
Online:2016-10-10
Published:2016-09-23
Contact:
ZHANG Runduo
E-mail:zhangrd@mail.buct.edu.cn
Supported by:CLC Number:
TrendMD:
LIU Ning, WANG Jiqiong, CHEN Biaohua, LI Yingxia, ZHANG Runduo. Study of Eight Membered Ring Zeolitic Catalyst of Cu/SAPO-35 over NH3-SCR†[J]. Chem. J. Chinese Universities, 2016, 37(10): 1817.
| Zeolite | Specific surface area/(m2·g-1) | Pore volume/(cm3·g-1) | Pore diameter/nm | Crystallite size*/nm |
|---|---|---|---|---|
| 0.3Si-SAPO-35 | 398.8 | 0.14 | 4.4 | 1.9 |
| 0.5Si-SAPO-35 | 443.4 | 0.10 | 3.4 | 2.1 |
| 0.7Si-SAPO-35 | 597.4 | 0.09 | 3.8 | 2.2 |
Table 1 N2 adsorption-desorption test results of SAPO-35 samples with different Si/Al molar ratios
| Zeolite | Specific surface area/(m2·g-1) | Pore volume/(cm3·g-1) | Pore diameter/nm | Crystallite size*/nm |
|---|---|---|---|---|
| 0.3Si-SAPO-35 | 398.8 | 0.14 | 4.4 | 1.9 |
| 0.5Si-SAPO-35 | 443.4 | 0.10 | 3.4 | 2.1 |
| 0.7Si-SAPO-35 | 597.4 | 0.09 | 3.8 | 2.2 |
| Zeolite | Molar ratio | Si substitution method | |
|---|---|---|---|
| Al/Si/P | (Si+P)/Al | ||
| 0.3Si-SAPO-35 | 1/0.15/0.83 | 0.98 | SM2 |
| 0.5Si-SAPO-35 | 1/0.25/0.82 | 1.07 | SM2 |
| 0.7Si-SAPO-35 | 1/0.36/0.81 | 1.17 | SM2+SM3 |
Table 2 XRF element analysis data of SAPO-35 samples
| Zeolite | Molar ratio | Si substitution method | |
|---|---|---|---|
| Al/Si/P | (Si+P)/Al | ||
| 0.3Si-SAPO-35 | 1/0.15/0.83 | 0.98 | SM2 |
| 0.5Si-SAPO-35 | 1/0.25/0.82 | 1.07 | SM2 |
| 0.7Si-SAPO-35 | 1/0.36/0.81 | 1.17 | SM2+SM3 |
| Catalyst | Molar ratio of SiO2/Al2O3 | Molar fraction of (Si+P) | Mass fraction of Cu(%) |
|---|---|---|---|
| Cu/0.3Si-SAPO-35 | 0.3 | 0.98 | 1.8 |
| Cu/0.5Si-SAPO-35 | 0.5 | 1.07 | 3.1 |
| Cu/0.7Si-SAPO-35 | 0.7 | 1.17 | 2.5 |
Table 3 XRF element analysis data of SAPO-35 samples
| Catalyst | Molar ratio of SiO2/Al2O3 | Molar fraction of (Si+P) | Mass fraction of Cu(%) |
|---|---|---|---|
| Cu/0.3Si-SAPO-35 | 0.3 | 0.98 | 1.8 |
| Cu/0.5Si-SAPO-35 | 0.5 | 1.07 | 3.1 |
| Cu/0.7Si-SAPO-35 | 0.7 | 1.17 | 2.5 |
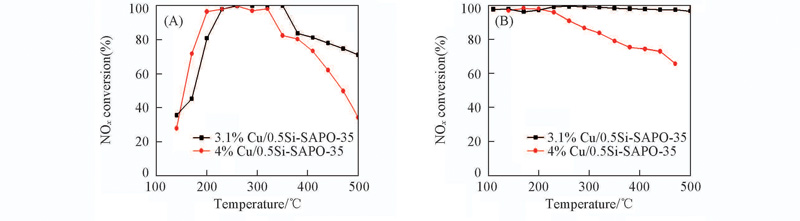
Fig.5 NH3-SCR activities of Cu/SAPO-35 zeolites prepared by ion exchange methodReaction conditions: 1 g/m3 NO, 1 g/m3 NH3, 10%O2 balanced by Ar, GHSV=3×104 h-1.
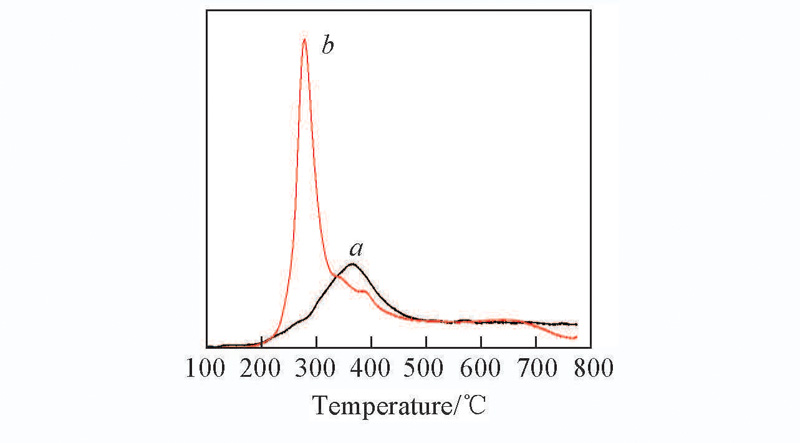
Fig.6 Comparison of H2-TPR test result between the samples prepared by rotating impregnation method and ion exchange methoda. 3.1%Cu/0.5Si-SAPO-35;b. 4%Cu/0.5Si-SAPO-35.
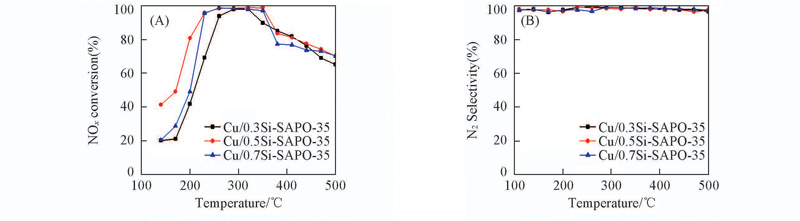
Fig.8 NH3-SCR activities of Cu/SAPO-35 zeolites prepared by ion exchange method with different Si/Al molar ratios Reaction conditions: 1 g/m3 NO, 1 g/m3 NH3, 10%O2 balanced by Ar, GHSV=3×104 h-1.
| Catalyst | H2 Consumption/(mmol·g-1) | H2/Cu* |
|---|---|---|
| Cu/0.3Si-SAPO-35 | 0.185 | 0.65 |
| Cu/0.5Si-SAPO-35 | 0.213 | 0.44 |
| Cu/0.7Si-SAPO-35 | 0.190 | 0.49 |
Table 4 H2-Consumption of Cu/SAPO-35 zeolites calculated by H2-TPR test result
| Catalyst | H2 Consumption/(mmol·g-1) | H2/Cu* |
|---|---|---|
| Cu/0.3Si-SAPO-35 | 0.185 | 0.65 |
| Cu/0.5Si-SAPO-35 | 0.213 | 0.44 |
| Cu/0.7Si-SAPO-35 | 0.190 | 0.49 |
| [1] | Wu B. J., Liu X. Q., Xiao P., Wang S. G., Chem. Res. Chinese Universities, 2008, 24(5), 615—619 |
| [2] | Liu J. D., Huang Z. G., Li Z., Guo Q. Q., Li Q. Y., Chem. J. Chinese Universities, 2014, 35(3), 589—595 |
| (刘建东, 黄张根, 李哲, 郭倩倩, 李巧艳.高等学校化学学报, 2014,35(3), 589—595) | |
| [3] | Chao J. D., He H., Song L. Y., Fang Y. J., Liang Q. M., Zhang G. Z., Qiu W. G., Zhang R., Chem. J. Chinese Univerities, 2015, 36(3), 523—530 |
| (晁晶迪, 何洪, 宋丽云, 房玉娇, 梁全明, 张桂臻, 邱文革, 张然.高等学校化学学报, 2015,36(3), 523—530) | |
| [4] | He L. F., Liu J. D., Huang W., Li Z., Chem. J. Chinese Univerities, 2012, 33(11), 2532—2536 |
| (贺丽芳, 刘建东, 黄伟, 李哲.高等学校化学学报, 2012,33(11), 2532—2536) | |
| [5] | Zhang R., Liu N., Lei Z., Chen B., Chem. Rev., 2016, 116,3658—3721 |
| [6] | Ye Q., Wang L., Yang R. T., Appl. Catal. A, 2012, 427/428, 24—34 |
| [7] | Venkatathri N., Catal. Commun., 2006, 7(10), 773—777 |
| [8] | Pinilla-Herrero I., Gmez-Hortigüelg L., Mrquez-Álvarez C., Sastre E., Microporous Mesoporous Mater., 2016, 219,322—326 |
| [9] | Bates S. A., Verma A. A., Paolucci C., J. Catal., 2014, 312(15), 87—97 |
| [10] | Göltl F., Bulo R. E., Hafner J., J. Phys. Chem. Lett., 2013, 4(14), 2244—2249 |
| [11] | Venkatathri N., Yoo J. W., Appl. Catal. A, 2008, 340(2), 265—270 |
| [12] | Wang L., Li W., Schmieg S. J., Weng D., J. Catal., 2015, 324,98—106 |
| [13] | Zheng J., Zhang W., Liu Z., Huo Q., Zhu K., Zhou X., Yuan W., Microporous Mesoporous Mater., 2016, 225,74—87 |
| [14] | Sedighi M., Ghasemi M., Sadeqzadeh M., Hadi M., Powder Technol., 2016, 291,131—139 |
| [15] | Liu Y., Xiao W., Xiao S., Adv. Powder Techonl., 2016, 27,625—630 |
| [16] | Vomscheid R., Briend M., Peltre M. J., J. Phys. Chem., 1994, 98(38), 9614—9618 |
| [17] | Sastre G., Lewis D. W., Catlow C. R. A., J. Mol. Catal. A: Chem., 1997, 119,349—356 |
| [18] | Zhang L., Bates J., Chen D., Nie H., Huang Y., J. Phys. Chem. C, 2011, 115(45), 22309—22319 |
| [19] | Hunger B., Heuchel M., J. Phys. Chem. B, 2002, 106(15), 21—36 |
| [20] | Rodriguez-Gonzalez L., Hermes F., Bertmer M., Appl. Catal. A: Gen., 2007, 328(2), 174—182 |
| [21] | Wang L., Li W., Qi G., J. Catal., 2012, 289(5), 21—29 |
| [22] | Li X., Fu D., Li M., Environ. Sci. Technol., 2014, 48(1), 566—72 |
| [23] | Ye Q., Wang L., Yang R. T., Appl. Catal. A, 2012, s427,24—34 |
| [24] | Richter M., Fait M., Eckelt R., Schneider M., Radnik J., Heidemann D., Fricke R., J. Catal., 2007, 245,11—24 |
| [25] | Xie P., Ma Z., Zhou H., Huang C., Yue Y., Shen W., Xu H., Hua W., Gao Z., Microporous Mesoporous Mater., 2014, 191,112—117 |
| [26] | Prakash A. M., Hartmann M., Kevan L., Cheminform., 1998, 29,308—316 |
| [1] | LI Zhiguang, QI Guodong, XU Jun, DENG Feng. Role of Catalyst Acidity in Glucose Conversion over Sn-Al-β Zeolite as Studied by Solid-state NMR [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220138. |
| [2] | WU Shuaini, ZHU Pengfei, SHI Huaiqi, LI Na, HU Zhaoxia, CHEN Shouwen. Preparation of CoCrx/SAPO-34 Catalyst and Its Catalytic Combustion Performance for 1,2-Dichloroethane [J]. Chem. J. Chinese Universities, 2021, 42(12): 3731. |
| [3] | ZHAO Shufang, HUANG Jun. Study by Solid-state NMR Spectroscopy on the Acidity and Shape-selectivity of Zeolites [J]. Chem. J. Chinese Universities, 2021, 42(1): 165. |
| [4] | ZHANG Guoqiang, SUN Yuchen, SHI Yabo, ZHENG Huayan, LI Zhong, SHANGGUAN Ju, LIU Shoujun, SHI Pengzheng. Surface Properties of Ce1-xMnxO2 Catalyst on the Catalytic Activities for Direct Synthesis of DMC from CO2 and Methanol [J]. Chem. J. Chinese Universities, 2020, 41(9): 2061. |
| [5] | CHEN Wenya, ZHU Li, HE Jun, XIE Yunlong, LU Jiqing, LUO Mengfei. Preparation of Rh/Al2O3-Cr2O3 Monolithic Catalysts for Dichloromethane Combustion† [J]. Chem. J. Chinese Universities, 2017, 38(4): 606. |
| [6] | ZHANG Jian-Cheng, DING Jian-Xun, XIAO Chun-Sheng, HE Chao-Liang, ZHUANG Xiu-Li, YANG Ya-Nan, CHEN Xue-Si. Synthesis and Characterization of Tumor-acidity-sensitive Poly(L-lysine)-doxorubicin Conjugates [J]. Chem. J. Chinese Universities, 2012, 33(12): 2809. |
| [7] | SONG Li-Juan*, PAN Ming-Xue, QIN Yu-Cai, JU Xiu-Fang, DUAN Lin-Hai, CHEN Xiao-Lu. Selective Adsorption Desulfurization Performance and Adsorptive Mechanisms of NiY Zeolites [J]. Chem. J. Chinese Universities, 2011, 32(3): 787. |
| [8] | WANG Tao, WU Guang-Jun, LI Lan-Dong*, GUAN Nai-Jia. Preparation, Characterization and Basic Catalytic Properties of Nitrogen-incorporated FAU Zeolites [J]. Chem. J. Chinese Universities, 2011, 32(3): 743. |
| [9] | WANG Zheng-Yang, YIN Xiao-Ju, YUE Nai-Lin, SUN Fu-Xing, ZHU Guang-Shan*. Preparation and Characterization of X-type Zeolite Membrane Supported by Stainless Steel Net [J]. Chem. J. Chinese Universities, 2011, 32(10): 2250. |
| [10] | SONG Ji-Xia, HAN Dong-Xue*, LI Fei, NIU Li. Preparations of Nano/Micro Structured Polyaniline Membranes for pH Sensing [J]. Chem. J. Chinese Universities, 2010, 31(8): 1688. |
| [11] | FAN Ping*, JIN Zhe, PAN Yi, LIU Guang-Jin. Synthesis and Azo-quinoid Tautomerizm of Azocalixarene [J]. Chem. J. Chinese Universities, 2009, 30(4): 724. |
| [12] | SONG Ke, GUAN Jing-Qi, WU Shu-Jie, XU Hai-Yan, KAN Qiu-Bin*. Synthesis of Mesoporous Aluminosilicates with Enhanced Acidity from Zeolites MCM-56 and MCM-49 [J]. Chem. J. Chinese Universities, 2009, 30(2): 344. |
| [13] | LIN Tao, XU Hai-Di, LI Wei, ZHANG Qiu-Lin, GONG Mao-Chu, CHEN Yao-Qiang*. Preparation of Mn-Fe/ZrO2-TiO2 Monolith Catalyst and Its Properties for Low-Temperature NH3-SCR Reaction [J]. Chem. J. Chinese Universities, 2009, 30(11): 2240. |
| [14] |
LIN Qi-Chun, LIN Wei-Ming, HAO Ji-Ming, LI Jun-Hua, FU Li-Xin.
Effect of Support Acidity on Selective Catalytic Reduction of NO by C3H6 over Cu-based Pillared Clay Catalyst [J]. Chem. J. Chinese Universities, 2006, 27(1): 85. |
| [15] | XU Ling, ZHU Guang-Shan, CHU Bin, SHI Chun-Feng, GAN Mei-Na, XIE Ya-Ru, SHEN Qi-Hui, WANG Run-Wei, QIU Shi-Lun. Synthesis and Characterization of a New Type of Mesoporous SAPO [J]. Chem. J. Chinese Universities, 2005, 26(9): 1585. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||