

Chem. J. Chinese Universities ›› 2016, Vol. 37 ›› Issue (1): 121.doi: 10.7503/cjcu20150446
• Physical Chemistry • Previous Articles Next Articles
Received:2015-06-08
Online:2016-01-10
Published:2015-12-20
Contact:
CHEN Jiuju
E-mail:jiujuchen09@163.com
Supported by:CLC Number:
TrendMD:
CHEN Jiuju. Theoretical Studies on the of Ambipolar Charge Transport in Terazulene Single Crystal†[J]. Chem. J. Chinese Universities, 2016, 37(1): 121.
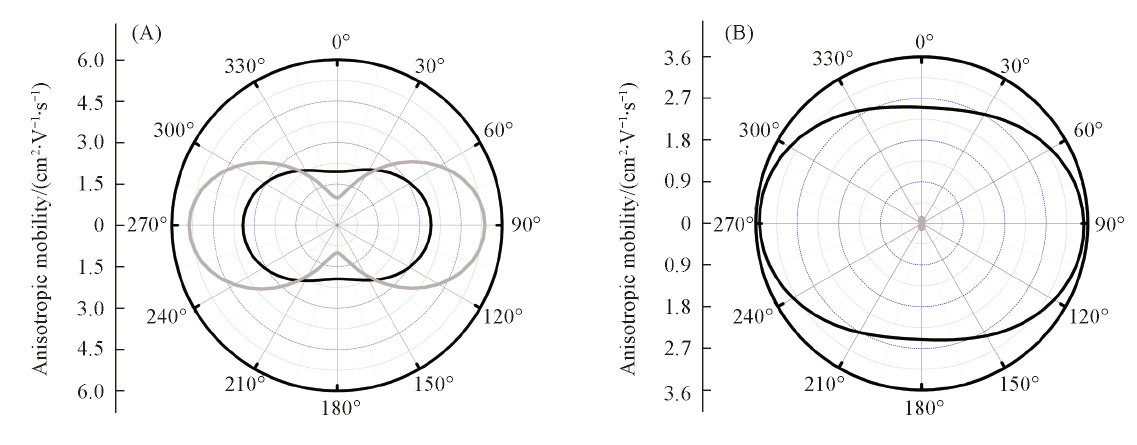
Fig.4 Angular resolution anisotropic mobilities for hole and electron transport in the ab plane of Terazulene(A) and NDT(B) single crystals as a function of α ^Black lines represent the hole mobilities and gray lines represent the electron mobilities.
| Pathway | Terazulene | NDT | |||||||
|---|---|---|---|---|---|---|---|---|---|
| PW91PW91/6-31+G(d,p) | B3LYP/6-31+G(d,p) | PW91PW91/6-31+G(d,p) | B3LYP/6-31+G(d,p) | ||||||
| Vh/meV | Ve/meV | Vh/meV | Ve/meV | Vh/meV | Ve/meV | Vh/meV | Ve/meV | ||
| A1 | -41.0 | 70.0 | -40.0 | 80.4 | -17.0 | 20.5 | -19.3 | 21.8 | |
| A2 | -41.0 | 70.0 | -40.0 | 80.4 | -17.0 | 20.6 | -19.3 | 21.8 | |
| B1 | 47.7 | -23.9 | 47.9 | -27.5 | -57.7 | 5.6 | -63.8 | 8.0 | |
| B2 | 47.7 | -23.9 | 47.9 | -27.5 | -57.7 | 5.6 | -63.8 | 8.0 | |
| C1 | 36.4 | -41.5 | -34.3 | -48.1 | -60.1 | -5.4 | -65.7 | -4.8 | |
| C2 | 36.4 | -41.5 | -34.3 | -48.1 | -60.1 | -5.4 | -65.7 | -4.8 | |
Table 1 Intermolecular transfer integrals of holes(Vh) and electrons(Ve) for the pathways of terazulene and NDT
| Pathway | Terazulene | NDT | |||||||
|---|---|---|---|---|---|---|---|---|---|
| PW91PW91/6-31+G(d,p) | B3LYP/6-31+G(d,p) | PW91PW91/6-31+G(d,p) | B3LYP/6-31+G(d,p) | ||||||
| Vh/meV | Ve/meV | Vh/meV | Ve/meV | Vh/meV | Ve/meV | Vh/meV | Ve/meV | ||
| A1 | -41.0 | 70.0 | -40.0 | 80.4 | -17.0 | 20.5 | -19.3 | 21.8 | |
| A2 | -41.0 | 70.0 | -40.0 | 80.4 | -17.0 | 20.6 | -19.3 | 21.8 | |
| B1 | 47.7 | -23.9 | 47.9 | -27.5 | -57.7 | 5.6 | -63.8 | 8.0 | |
| B2 | 47.7 | -23.9 | 47.9 | -27.5 | -57.7 | 5.6 | -63.8 | 8.0 | |
| C1 | 36.4 | -41.5 | -34.3 | -48.1 | -60.1 | -5.4 | -65.7 | -4.8 | |
| C2 | 36.4 | -41.5 | -34.3 | -48.1 | -60.1 | -5.4 | -65.7 | -4.8 | |
| Species | Average mobility/(cm2·V-1·s-1) | |
|---|---|---|
| Terazulene | Centrosymmetric NDT | |
| Hole | 2.67±0.10 | 3.09±0.09 |
| Electron | 3.08±0.09 | 0.064±0.004 |
Table 2 2D Average mobility of holes and electrons in the ab plane of terazulene and NDT single crystals, respectively
| Species | Average mobility/(cm2·V-1·s-1) | |
|---|---|---|
| Terazulene | Centrosymmetric NDT | |
| Hole | 2.67±0.10 | 3.09±0.09 |
| Electron | 3.08±0.09 | 0.064±0.004 |
| [1] | Farinola G. M., Ragni R., Chem. Soc. Rev., 2011, 40, 3467—3482 |
| [2] | Mas-Torrent M., Rovira C., Chem. Rev., 2011, 111(8), 4833—4856 |
| [3] | Clarke T. M., Durrant J. R., Chem. Rev., 2010, 110(11), 6736—6767 |
| [4] | Brédas J. L., Beljonne D., Coropceanu V., Cornil J., Chem. Rev., 2004, 104(11), 4971—5003 |
| [5] | Jurchescu O. D., Baas J., Palstra T. T. M., Appl. Phys. Lett., 2004, 84(16), 3061—3063 |
| [6] | Sundar V. C., Zaumseil J., Podzorov V., Menard E., Willett R. L., Someya T., Gershenson M. E., Rogers J. A., Science,2004, 303, 1644—1646 |
| [7] | Shirota Y., Kageyama H., Chem. Rev., 2007, 107(4), 953—1010 |
| [8] | Yamaguchi Y., Ogawa K., Nakayama K., Ohba Y., Katagiri H., J. Am. Chem. Soc., 2013, 135, 19095—19098 |
| [9] | Shinamura S., Osaka I., Miyazaki E., Nakao A., Yamagishi M., Takeya J., Takimiya K., J. Am. Chem. Soc., 2011, 133, 5024—5035 |
| [10] | Cheng Y. C., Silbey R. J., da Silva Filho D. A., Calbert J. P., Cornil J., Brédas J. L., J. Chem. Phys., 2003, 118(8), 3764—3774 |
| [11] | Hutchison G. R., Ratner M. A., Marks T. J., J. Am. Chem. Soc., 2005, 127(7), 2339—2350 |
| [12] | Deng W. Q., Goddard Ⅲ W. A., J. Phys. Chem. B, 2004, 108(25), 8614—8621 |
| [13] | Troisi A., Orlandi G., J. Phys. Chem. A, 2006, 110(11), 4065—4070 |
| [14] | Song Y. B, Di C. A., Yang X. D., Li S. P., Xu W., Liu Y. Q., Yang L. M., Shuai Z. G., Zhang D. Q., Zhu D. B., J. Am. Chem. Soc., 2006, 128(50), 15940—15941 |
| [15] | Nan G. J., Yang X. D., Wang L. J., Shuai Z. G., Zhao Y., Phys. Rev. B, 2009, 79(11), 115203 |
| [16] | Geng H., Peng Q., Wang L. J., Li H. J., Liao Y., Ma Z. Y., Shuai Z. G., Adv. Mater., 2014, 24, 3568—3572 |
| [17] | Marcus R. A., Rev. Mod. Phys., 1993, 65(3), 599—610 |
| [18] | Schein L. B., McGhie A. R., Phys. Rev. B, 1979, 20, 1631—1639 |
| [19] | Norton J. E., Brédas J. L., J. Am. Chem. Soc., 2008, 130(37), 12377—12384 |
| [20] | Malagoli M., Brédas J. L., Chem. Phys. Lett., 2000, 327, 13—17 |
| [21] | Wen S. H., Deng W. Q., Han K. L., Phys. Chem. Chem. Phys., 2010, 12, 9267—9275 |
| [22] | Valeev E. F., Coropceanu V., da Silva Filho D. A., Salman S., Brédas J. L., J. Am. Chem. Soc., 2006, 128(30), 9882—9886 |
| [23] | Frisch M.J., Trucks G. W., Schlegel H. B., Scuseria G. E., Robb M. A., Cheeseman J. R., Montgomery J. A., Vreven T., Kudin K. N., Burant J. C., Millam J. M., Iyengar S. S., Tomasi J., Barone V., Mennucci B., Cossi M., Scalmani G., Rega N., Petersson G. A., Nakatsuji H., Hada M., Ehara M., Toyota K., Fukuda R., Hasegawa J., Ishida M., Nakajima T., Honda Y., Kitao O., Nakai H., Klene M., Li X., Knox J. E., Hratchian H. P., Cross J. B., Bakken V., Adamo C., Jaramillo J., Gomperts R., Stratmann R. E., Yazyev O., Austin A. J., Cammi R., Pomelli C., Ochterski J. W., Ayala P. Y., Morokuma K., Voth G. A., Salvador P., Dannenberg J. J., Zakrzewski V. G., Dapprich S., Daniels A. D., Strain M. C., Farkas O., Malick D. K., Rabuck A. D., Raghavachari K., Foresman J. B., Ortiz J. V., Cui Q., Baboul A. G., Clifford S., Cioslowski J., Stefanov B. B., Liu G., Liashenko A., Piskorz P., Komaromi I., Martin R. L., Fox D. J., Keith T., Al-Laham M. A., Peng C. Y., Nanayakkara A., Challacombe M., Gill P. M. W., Johnson B., Chen W., Wong M. W., Gonzalez C., Pople J. A., Gaussian 03, Revision E.01, Gaussian Inc.,Wallingford CT, 2004 |
| [24] | Yang X. D., Wang L. J., Wang C. L., Long W., Shuai Z. G., Chem. Mater., 2008, 20(9), 3205—3211 |
| [1] | LI Kangming, LI Yansai, YI Yangjie, XU Leitao, YE Jiao, OU Xiaoming, LI Jianming, HU Aixi. Design, Synthesis and Biological Activity of 5-Pyrazole Carboxamides † [J]. Chem. J. Chinese Universities, 2020, 41(4): 716. |
| [2] | ZHAO Bangtun, TAO Jingjing, CHEN Xiaoji, FU Huimin, ZHU Weimin. Synthesis, Structure and Electrochemistry of Tetrathiafulvalene Vinylogues Bearing Thienyl and Pyridyl Groups† [J]. Chem. J. Chinese Universities, 2018, 39(7): 1449. |
| [3] | ZHAO Bangtun, MA Shuxiu, TAO Jingjing, ZHU Weimin. Synthesis, Structures and Electrochemical Properties of Pyridine-based Tetrathiafulvalene Derivatives† [J]. Chem. J. Chinese Universities, 2017, 38(2): 193. |
| [4] | MAHMUTJAN Jelil, ABLAT Mamat, ALIMJAN Abaydulla, MAMAT Tursun. Investigation of Topology-distinct Stable Structures for Hydrogen Fluoride Clusters (HF)n(n=2—8) by the Methods of Graph Theoretical Enumerations and Quantum Chemical Calculations† [J]. Chem. J. Chinese Universities, 2016, 37(12): 2275. |
| [5] | XIANG Junfeng, YI Pinggui, YU Xianyong, CHEN Jian, HAO Yanlei, REN Zhiyong. Excited-state Proton Transfer of 2-(2-Hydroxyphenyl)benzothiazole in the Confined Nanocavity† [J]. Chem. J. Chinese Universities, 2015, 36(4): 654. |
| [6] | GAO Yunqiao, CHEN Lili, FU Yingqiang, ZHAO Jianwei. Random Walk Simulation of an Asymmetric Obstacle Sieve for Continuous Molecular Separation† [J]. Chem. J. Chinese Universities, 2014, 35(4): 818. |
| [7] | GAO Chenggui, LONG Zhengwen, TAN Xingfeng, LONG Bo, ZHANG Weijun, LONG Chaoyun, QIN Shuijie. Theoretical Investigation on the Reaction Between HRnCCH and X(X=H2O, NH3) in Gas Phase† [J]. Chem. J. Chinese Universities, 2014, 35(2): 344. |
| [8] | QIHE Ri-Ma, ZHAO Xiao-Guang, ZHANG Yun-Hong, YUAN Hui, XU Guang-Tong. Investigation of Selectivity over HDS Catalysts by in situ IR Spectra of Adsorbed CO and Molecular Simulation Calculation [J]. Chem. J. Chinese Universities, 2012, 33(02): 383. |
| [9] | FENG Jin-Ling, ZHANG Jian-Guo*, WANG Kun, ZHANG Tong-Lai*. Crystal Structure and Theoretical Studies of 3,6-Dihydrazino-1,2,4,5-tetrazine [J]. Chem. J. Chinese Universities, 2011, 32(7): 1519. |
| [10] | BAI Feng-Ying, LI Xiao-Tian*, ZHU Guang-Shan, XING Yong-Heng , ZENG Xiao-Qing,... Synthesis, Structure and Molecular Configuration of a Series of Ionic Salt Compounds of Pyrophosphate Ester of Thiamine [J]. Chem. J. Chinese Universities, 2009, 30(10): 1919. |
| [11] | LI Xiao-Li, DUAN Ran, ZHANG Ying-Hui, RUAN Wen-Juan, ZHU Zhi-Ang. Studies on Molecular Recognition of Chiral Asymmetry Salen Ni to Guests Imidazole and Diamine Derivatives [J]. Chem. J. Chinese Universities, 2006, 27(9): 1689. |
| [12] | DU Jin-Tang, LI Yan-Mei, CHEN Zhong-Zhou, TAN Bo, JIANG Yang, ZHONG Ru-Gang, ZHAO Yu-Fen . Theoretical Studies on N-Phosphorylized Amino Acids Properties by Quantum Chemical Calculation [J]. Chem. J. Chinese Universities, 2005, 26(3): 540. |
| [13] | YANG Guo-Bo, ZHOU Yin-Hua, TIAN Wen-Jing, ZHOU Xin, PAN Qing-Jiang, REN Ai-Min. Quantum Chemical Study on the Energy Level Structure of Thiophene Oligomers [J]. Chem. J. Chinese Universities, 2004, 25(6): 1104. |
| [14] | QIU Yong-Qing, YU Hai-Ying, SU Zhong-Min, ZHAO Liang, WANG Rong-Shun, ZHU Yu-Lan, . Theoretical Studies on Electronic Spectrum and Third-order Nonlinear Optical Properties of Centrosymmetric Pyrazine Derivatives [J]. Chem. J. Chinese Universities, 2004, 25(12): 2335. |
| [15] | RUAN Wen-Juan, ZHANG Yu-Ling, ZHAO Xiao-Jing, JIANG Dong-Qing, ZHU Zhi-Ang . Studies on Chiral Molecular Recognition of Salen Zn with Amino Acid Esters [J]. Chem. J. Chinese Universities, 2003, 24(9): 1657. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||
