

Chem. J. Chinese Universities ›› 2016, Vol. 37 ›› Issue (9): 1649.doi: 10.7503/cjcu20160145
• Organic Chemistry • Previous Articles Next Articles
LI Lin1,2, LI Miao3, CHAI Baoshan4, YANG Jichun3, SONG Yuquan3, LIU Changling3,*( )
)
Received:2016-03-11
Online:2016-09-10
Published:2016-08-17
Contact:
LIU Changling
E-mail:liuchangling@vip.163.com
CLC Number:
TrendMD:
LI Lin, LI Miao, CHAI Baoshan, YANG Jichun, SONG Yuquan, LIU Changling. Design, Synthesis and Biological Activity of Novel Substituted Diamides Derivatives Containing Thiophene Ring[J]. Chem. J. Chinese Universities, 2016, 37(9): 1649.
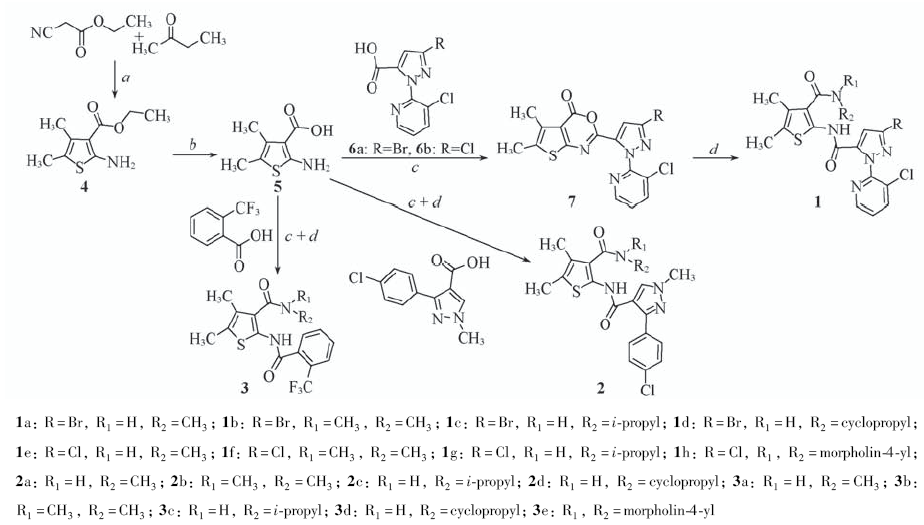
Scheme 1 Synthetic routes of compounds 1—3 Reagents and conditions: a. S, NHEt2, CH3CN, 50 ℃, 1—5 h; b. NaOH, CH3CH2OH/H2O, reflux, 2 h; c. CH3SO2Cl, pyridine, CH3CN, 2 h; d. R1R2NH, CH3CN, 1—6 h.
| Compd. | Yield (%) | m.p./℃ | Elemental analysis(%, calcd.) | IR, | ||
|---|---|---|---|---|---|---|
| C | H | N | ||||
| 1a | 23 | 221—222 | 42.38 (43.56) | 3.44 (3.23) | 15.03 (14.94) | 3460(m, N—H), 2980, 2960(m, C—H), 1620(s, C=O), 1565, 1455(m, aromatic rings), 1450, 1360(m, C—H), 1320(s, C—O), 830, 750, 720(m, Ph-H) |
| 1b | 29 | 238—239 | 45.65 (44.78) | 3.50 (3.55) | 14.34 (14.51) | 3440(m, N—H), 2970, 2940(m, C—H), 1610(s, C=O), 1555, 1440(m, aromatic rings), 1440, 1370(m, C—H), 1310(s, C—O), 810, 770, 730(m, Ph-H) |
| 1c | 21 | 224—225 | 46.524 (45.9) | 3.73 (3.86) | 14.02 (14.10) | 3450(m, N—H), 2980(m, C—H), 1620(s, C=O), 1560, 1440(m, aromatic rings), 1450, 1370(m, C—H), 1330(s, C—O), 820, 760, 700(m, Ph-H) |
| 1d | 25 | 238—239 | 45.43 (46.12) | 3.58 (3.46) | 14.27 (14.15) | 3440(m, N—H), 2950(m, C—H), 1600(s, C=O), 1555, 1435(m, aromatic rings), 1450, 1360(m, C—H), 1320(s, C—O), 800, 770, 720(m, Ph-H) |
| 1e | 32 | 217—218 | 47.36 (48.12) | 3.55 (3.56) | 16.63 (16.51) | 3460(m, N—H), 2975, 2960(m, C—H), 1620(s, C=O), 1560, 1450(m, aromatic rings), 1470, 1370(m, C—H), 1330(s, C—O), 830, 760, 690(m, Ph-H) |
| 1f | 58 | 235—236 | 49.28 (49.32) | 3.89 (3.91) | 15.99 (15.98) | 3440(m, N—H), 2970(m, C—H), 1610(s, C=O), 1565, 1455(m, aromatic rings), 1460, 1350(m, C—H), 1310(s, C—O), 820, 750, 710(m, Ph-H) |
| 1g | 41 | 217—218 | 51.78 (50.45) | 4.16 (4.23) | 15.38 (15.48) | 3430(m, N—H), 2980, 2960(m, C—H), 1630(s, C=O), 1570, 1460(m, aromatic rings), 1470, 1365(m, C—H), 1320(s, C—O), 820, 770, 730(m, Ph-H) |
| 1h | 50 | 257—258 | 51.11 (50.01) | 3.82 (3.99) | 14.50 (14.58) | 3420(m, N—H), 2980, 2960(m, C—H), 1600(s, C=O), 1550, 1440(m, aromatic rings), 1440, 1370(m, C—H), 1300(s, C—O), 800, 760, 740(m, Ph-H) |
| 2a | 25 | 170—171 | 57.84 (56.64) | 4.67 (4.75) | 13.86 (13.91) | 3420(m, N—H), 2980, 2960(m, C—H), 1600(s, C=O), 1550, 1440(m, aromatic rings), 1440, 1370(m, C—H), 1300(s, C—O), 800, 760, 740(m, Ph-H) |
| 2b | 38 | 175—176 | 56.90 (57.62) | 5.21 (5.08) | 13.52 (13.44) | 3440(m, N—H), 2980, 2950(m, C—H), 1620(s, C=O), 1530(m, aromatic rings), 1420, 1350(m, C—H), 1330(s, C—O), 820, 780, 720(m, Ph-H) |
| 2c | 30 | 234—235 | 57.46 (58.53) | 5.44 (5.38) | 13.15 (13.00) | 3430(m, N—H), 2960(m, C—H), 1610(s, C=O), 1540(m, aromatic rings), 1440, 1370(m, C—H), 1300(s, C—O), 800, 760, 740(m, Ph-H) |
| 2d | 40 | 245—246 | 57.69 (58.80) | 5.02 (4.94) | 13.13 (13.06) | 3450(m, N—H), 2980(s, C—H), 1620(s, C=O), 1520(s, aromatic rings), 1450, 1390(m, C—H), 1220, 1200(s, C—O), 840, 760(s, Ph-H) |
| 3a | 52 | 127—128 | 54.63 (53.93) | 4.18 (4.24) | 7.78 (7.86) | 3440(m, N—H), 2970(m, C—H), 1670(s, C=O), 1620, 1560, 1510(s, aromatic rings), 1400, 1310(m, C—H), 1130(s, C—F), 780, 710(m, Ph-H) |
| 3b | 50 | 185—186 | 56.87 (55.13) | 4.57 (4.63) | 7.42 (7.56) | 3430(m, N—H), 2950(m, C—H), 1660(s, C=O), 1610, 1550, 1500(s, aromatic rings), 1410, 1320(m, C—H), 1120(s, C—F), 820, 790, 700(m, Ph-H) |
| 3c | 52 | 147—148 | 55.30 (56.24) | 5.07 (4.98) | 7.35 (7.29) | 3440(m, N—H), 2960(m, C—H), 1670(s, C=O), 1600, 1550(s, aromatic rings), 1420, 1320(m, C—H), 1110(s, C—F), 760, 700(m, Ph-H) |
| 3d | 58 | 151—152 | 56.58 (56.54) | 4.49 (4.48) | 7.34 (7.33) | 3420(m, N—H), 2970(m, C—H), 1650(s, C=O), 1620, 1540(s, aromatic rings), 1430, 1320(m, C—H), 1130(s, C—F), 830, 780, 720(m, Ph-H) |
| 3e | 33 | 187—188 | 55.90 (55.33) | 4.71 (4.64) | 6.68 (6.79) | 3430(m, N—H), 2950(m, C—H), 1660(s, C=O), 1630, 1580, 1520(s, aromatic rings), 1410, 1300(m, C—H), 1120(s, C—F), 800, 770, 710(m, Ph-H) |
Table 1 Yields, melting points, elemental analysis and IR data of compounds 1—3
| Compd. | Yield (%) | m.p./℃ | Elemental analysis(%, calcd.) | IR, | ||
|---|---|---|---|---|---|---|
| C | H | N | ||||
| 1a | 23 | 221—222 | 42.38 (43.56) | 3.44 (3.23) | 15.03 (14.94) | 3460(m, N—H), 2980, 2960(m, C—H), 1620(s, C=O), 1565, 1455(m, aromatic rings), 1450, 1360(m, C—H), 1320(s, C—O), 830, 750, 720(m, Ph-H) |
| 1b | 29 | 238—239 | 45.65 (44.78) | 3.50 (3.55) | 14.34 (14.51) | 3440(m, N—H), 2970, 2940(m, C—H), 1610(s, C=O), 1555, 1440(m, aromatic rings), 1440, 1370(m, C—H), 1310(s, C—O), 810, 770, 730(m, Ph-H) |
| 1c | 21 | 224—225 | 46.524 (45.9) | 3.73 (3.86) | 14.02 (14.10) | 3450(m, N—H), 2980(m, C—H), 1620(s, C=O), 1560, 1440(m, aromatic rings), 1450, 1370(m, C—H), 1330(s, C—O), 820, 760, 700(m, Ph-H) |
| 1d | 25 | 238—239 | 45.43 (46.12) | 3.58 (3.46) | 14.27 (14.15) | 3440(m, N—H), 2950(m, C—H), 1600(s, C=O), 1555, 1435(m, aromatic rings), 1450, 1360(m, C—H), 1320(s, C—O), 800, 770, 720(m, Ph-H) |
| 1e | 32 | 217—218 | 47.36 (48.12) | 3.55 (3.56) | 16.63 (16.51) | 3460(m, N—H), 2975, 2960(m, C—H), 1620(s, C=O), 1560, 1450(m, aromatic rings), 1470, 1370(m, C—H), 1330(s, C—O), 830, 760, 690(m, Ph-H) |
| 1f | 58 | 235—236 | 49.28 (49.32) | 3.89 (3.91) | 15.99 (15.98) | 3440(m, N—H), 2970(m, C—H), 1610(s, C=O), 1565, 1455(m, aromatic rings), 1460, 1350(m, C—H), 1310(s, C—O), 820, 750, 710(m, Ph-H) |
| 1g | 41 | 217—218 | 51.78 (50.45) | 4.16 (4.23) | 15.38 (15.48) | 3430(m, N—H), 2980, 2960(m, C—H), 1630(s, C=O), 1570, 1460(m, aromatic rings), 1470, 1365(m, C—H), 1320(s, C—O), 820, 770, 730(m, Ph-H) |
| 1h | 50 | 257—258 | 51.11 (50.01) | 3.82 (3.99) | 14.50 (14.58) | 3420(m, N—H), 2980, 2960(m, C—H), 1600(s, C=O), 1550, 1440(m, aromatic rings), 1440, 1370(m, C—H), 1300(s, C—O), 800, 760, 740(m, Ph-H) |
| 2a | 25 | 170—171 | 57.84 (56.64) | 4.67 (4.75) | 13.86 (13.91) | 3420(m, N—H), 2980, 2960(m, C—H), 1600(s, C=O), 1550, 1440(m, aromatic rings), 1440, 1370(m, C—H), 1300(s, C—O), 800, 760, 740(m, Ph-H) |
| 2b | 38 | 175—176 | 56.90 (57.62) | 5.21 (5.08) | 13.52 (13.44) | 3440(m, N—H), 2980, 2950(m, C—H), 1620(s, C=O), 1530(m, aromatic rings), 1420, 1350(m, C—H), 1330(s, C—O), 820, 780, 720(m, Ph-H) |
| 2c | 30 | 234—235 | 57.46 (58.53) | 5.44 (5.38) | 13.15 (13.00) | 3430(m, N—H), 2960(m, C—H), 1610(s, C=O), 1540(m, aromatic rings), 1440, 1370(m, C—H), 1300(s, C—O), 800, 760, 740(m, Ph-H) |
| 2d | 40 | 245—246 | 57.69 (58.80) | 5.02 (4.94) | 13.13 (13.06) | 3450(m, N—H), 2980(s, C—H), 1620(s, C=O), 1520(s, aromatic rings), 1450, 1390(m, C—H), 1220, 1200(s, C—O), 840, 760(s, Ph-H) |
| 3a | 52 | 127—128 | 54.63 (53.93) | 4.18 (4.24) | 7.78 (7.86) | 3440(m, N—H), 2970(m, C—H), 1670(s, C=O), 1620, 1560, 1510(s, aromatic rings), 1400, 1310(m, C—H), 1130(s, C—F), 780, 710(m, Ph-H) |
| 3b | 50 | 185—186 | 56.87 (55.13) | 4.57 (4.63) | 7.42 (7.56) | 3430(m, N—H), 2950(m, C—H), 1660(s, C=O), 1610, 1550, 1500(s, aromatic rings), 1410, 1320(m, C—H), 1120(s, C—F), 820, 790, 700(m, Ph-H) |
| 3c | 52 | 147—148 | 55.30 (56.24) | 5.07 (4.98) | 7.35 (7.29) | 3440(m, N—H), 2960(m, C—H), 1670(s, C=O), 1600, 1550(s, aromatic rings), 1420, 1320(m, C—H), 1110(s, C—F), 760, 700(m, Ph-H) |
| 3d | 58 | 151—152 | 56.58 (56.54) | 4.49 (4.48) | 7.34 (7.33) | 3420(m, N—H), 2970(m, C—H), 1650(s, C=O), 1620, 1540(s, aromatic rings), 1430, 1320(m, C—H), 1130(s, C—F), 830, 780, 720(m, Ph-H) |
| 3e | 33 | 187—188 | 55.90 (55.33) | 4.71 (4.64) | 6.68 (6.79) | 3430(m, N—H), 2950(m, C—H), 1660(s, C=O), 1630, 1580, 1520(s, aromatic rings), 1410, 1300(m, C—H), 1120(s, C—F), 800, 770, 710(m, Ph-H) |
| Compd. | 1H NMR(300 MHz), δ |
|---|---|
| 1a | 13.14(s, 1H, NH), 8.49(dd, J=1.5, 4.5 Hz, 1H, pyridin-6-H), 7.89(dd, J=1.5, 1.8 Hz, 1H, pyridin-4-H), 7.42(dd, J=4.5, 8.4 Hz, 1H, pyridin-5-H), 6.98(s, 1H, pyrazole-H), 6.11(s, 1H, NH), 3.03(d, J=3.0 Hz, 3H, CH3), 2.30(s, 3H, thiophene-5-CH3), 2.25(s, 3H, thiophene-4-CH3) |
| 1b | 11.97(s, 1H, NH), 8.52(dd, J=1.5, 4.8 Hz, 1H, pyridin-6-H), 8.23(dd, J=1.5, 4.8 Hz, 1H, pyridin-4-H), 7.66(dd, J=4.8, 8.4 Hz, 1H, pyridin-5-H), 7.34(s, 1H, pyrazole), 2.87(s, 3H, CH3), 2.79(s, 3H, CH3), 2.21(s, 3H, thiophene-5-CH3), 1.91(s, 3H, thiophene-4-CH3) |
| 1c | 13.11(s, 1H, NH), 8.49(dd, J=1.8, 5.1 Hz, 1H, pyridin-6-H), 7.89(dd, J=1.8, 5.1 Hz, 1H, pyridin-4-H), 7.42(dd, J=5.1, 8.1 Hz, 1H, pyridin-5-H), 6.98(s, 1H, pyrazole-H), 5.89(br, 1H, NH), 4.28(m, 1H, CH), 2.27(s, 3H, thiophene-5-CH3), 2.23(s, 3H, thiophene-4-CH3), 1.28(d, J=5.1 Hz, 6H, 2CH3) |
| 1d | 11.91(s, 1H, NH), 8.54(dd, J=1.2, 4.8 Hz, 1H, pyridin-6-H), 8.25(dd, J=1.5, 4.5 Hz, 1H, pyridin-4-H), 7.68(dd, J=4.8, 8.1 Hz, 1H, pyridin-5-H), 7.29(s, 1H, pyrazole), 6.98(s, 1H, NH), 2.81(m, 1H, CH), 2.10(s, 3H, thiophene-5-CH3), 2.06(s, 3H, thiophene-4-CH3), 0.70(m, 2H, CH2), 0.55(m, 2H, CH2) |
| 1e | 13.14(s, 1H, NH), 8.49(dd, J=1.5, 5.1 Hz, 1H, pyridin-6-H), 7.89(dd, J=1.2, 5.1 Hz, 1H, pyridin-4-H), 7.42(dd, J=4.8, 8.7 Hz, 1H, pyridin-5-H), 6.98(s, 1H, pyrazole-H), 6.11(s, 1H, NH), 3.03(d, J=3.3 Hz, 3H, CH3), 2.30(s, 3H, thiophene-5-CH3), 2.25(s, 3H, thiophene-4-CH3) |
| 1f | 10.45(s, 1H, NH), 8.50(dd, J=1.8, 4.5 Hz, 1H, pyridin-6-H), 7.89(dd, J=1.8, 4.8 Hz,1H, pyridin-4-H), 7.42(dd, J=4.5, 8.4 Hz, 1H, pyridin-5-H), 6.99(s, 1H, pyrazole-H), 3.03(s, 6H, 2CH3), 2.23(s, 3H, thiophene-5-CH3), 2.03(s, 3H, thiophene-4-CH3) |
| 1g | 13.11(s, 1H, NH), 8.49(dd, J=1.2, 5.1 Hz, 1H, pyridin-6-H), 7.89(dd, J=1.5, 5.1 Hz, 1H, pyridin-4-H), 7.42(dd, J=5.1, 8.7 Hz, 1H, pyridin-5-H), 6.98(s, 1H, pyrazole-H), 5.89(br, 1H, NH), 4.28(m, 1H, CH), 2.27(s, 3H, thiophene-5-CH3), 2.23(s, 3H, thiophene-4-CH3), 1.28(d, J=6.9 Hz, 6H, 2CH3) |
| 1h | 10.43(s, 1H, NH), 8.49(dd, J=1.5, 4.8 Hz, 1H, pyridin-6-H), 7.91(dd, J=1.2, 4.8 Hz, 1H, pyridin-4-H), 7.43(dd, J=4.8, 8.4 Hz, 1H, pyridin-5-H), 6.96(s, 1H, pyrazole-H), 3.71(m, 4H, morpholine-2,6-2CH2), 3.54(m, 4H, morpholine-3,5-2CH2), 2.22(s, 3H, thiophene-5-CH3), 2.04(s, 3H, thiophene-4-CH3) |
| 2a | 8.33(s, 1H, pyrazole-H), 7.70(s, 1H, NH), 7.65(d, J=6.6 Hz, 2H, Ph-2,6-2H), 7.47(d, J=6.6 Hz, 2H, Ph-3,5-2H), 3.93(s, 3H, pyrazole-CH3), 2.93(s, 3H, CH3), 2.23(s, 3H, thiophene-5-CH3), 1.92(s, 3H, thiophene-4-CH3) |
| 2b | 8.23(s, 1H, pyrazole-H), 7.71(s, 1H, NH), 7.68(d, J=6.9 Hz, 2H, Ph-2,6-2H), 7.47(d, J=6.9 Hz, 2H, Ph-3,5-2H), 3.93(s, 3H, pyrazole-CH3), 2.83(s, 6H, 2CH3), 2.23(s, 3H, thiophene-5-CH3), 1.92(s, 3H, thiophene-4-CH3) |
| 2c | 12.01(s, 1H, NH), 8.31(s, 1H, pyrazole-H), 7.68(d, J=6.3 Hz, 2H, Ph-2,6-2H), 7.47(d, J=6.3 Hz, 2H, Ph-3,5-2H), 3.96(m, 1H, CH), 3.95(s, 3H, pyrazole-CH3), 2.23(s, 3H, thiophene-5-CH3), 2.15(s, 3H, thiophene-4-CH3), 1.13(s, 3H, CH3), 1.11(s, 3H, CH3) |
| 2d | 8.29(s, 1H, pyrazole-H), 7.71(s, 1H, NH), 7.66(d, J=6.6 Hz, 2H, Ph-2,6-2H), 7.43(d, J=6.6 Hz, 2H, Ph-3,5-2H), 3.93(s, 3H, pyrazole-CH3), 2.32(m, 1H, CH), 2.23(s, 3H, thiophene-5-CH3), 1.92(s, 3H, thiophene-4-CH3), 0.83(m, 4H, 2CH2) |
| 3a | 12.40(s, 1H, NH), 7.77—7.58(m, 4H, Ph), 6.05(s, 1H, NH), 2.95(d, J=3.0 Hz, 3H, CH3), 2.32(s, 6H, thiophene-2CH3) |
| 3b | 9.19(s, 1H, NH), 7.75—7.59(m, 4H, Ph), 3.02(s, 6H, 2CH3), 2.32(s, 3H, thiophene-5-CH3), 2.06(s, 3H, thiophene-4-CH3) |
| 3c | 12.31(s, 1H, NH), 7.76—7.58(m, 4H, Ph), 5.85(br, 1H, NH), 4.19(m, 1H, CH), 2.31(s, 6H, thiophene-2CH3), 1.24(d, J=5.1 Hz, 6H, 2CH3) |
| 3d | 12.39(s, 1H, NH), 7.77—7.58(m, 4H, Ph), 6.19(s, 1H, NH), 2.80(m, 1H, CH), 2.31(s, 3H, thiophene-5-CH3), 2.26(s, 3H, thiophene-4-CH3), 0.85(m, 4H, 2CH2) |
| 3e | 9.26(s, 1H, NH), 7.76—7.64(m, 4H, Ph), 3.78—3.46(m, 8H, morpholine), 2.31(s, 3H, thiophene-5-CH3), 2.07(s, 3H, thiophene-4-CH3) |
Table 2 1H NMR data of compounds 1—3*
| Compd. | 1H NMR(300 MHz), δ |
|---|---|
| 1a | 13.14(s, 1H, NH), 8.49(dd, J=1.5, 4.5 Hz, 1H, pyridin-6-H), 7.89(dd, J=1.5, 1.8 Hz, 1H, pyridin-4-H), 7.42(dd, J=4.5, 8.4 Hz, 1H, pyridin-5-H), 6.98(s, 1H, pyrazole-H), 6.11(s, 1H, NH), 3.03(d, J=3.0 Hz, 3H, CH3), 2.30(s, 3H, thiophene-5-CH3), 2.25(s, 3H, thiophene-4-CH3) |
| 1b | 11.97(s, 1H, NH), 8.52(dd, J=1.5, 4.8 Hz, 1H, pyridin-6-H), 8.23(dd, J=1.5, 4.8 Hz, 1H, pyridin-4-H), 7.66(dd, J=4.8, 8.4 Hz, 1H, pyridin-5-H), 7.34(s, 1H, pyrazole), 2.87(s, 3H, CH3), 2.79(s, 3H, CH3), 2.21(s, 3H, thiophene-5-CH3), 1.91(s, 3H, thiophene-4-CH3) |
| 1c | 13.11(s, 1H, NH), 8.49(dd, J=1.8, 5.1 Hz, 1H, pyridin-6-H), 7.89(dd, J=1.8, 5.1 Hz, 1H, pyridin-4-H), 7.42(dd, J=5.1, 8.1 Hz, 1H, pyridin-5-H), 6.98(s, 1H, pyrazole-H), 5.89(br, 1H, NH), 4.28(m, 1H, CH), 2.27(s, 3H, thiophene-5-CH3), 2.23(s, 3H, thiophene-4-CH3), 1.28(d, J=5.1 Hz, 6H, 2CH3) |
| 1d | 11.91(s, 1H, NH), 8.54(dd, J=1.2, 4.8 Hz, 1H, pyridin-6-H), 8.25(dd, J=1.5, 4.5 Hz, 1H, pyridin-4-H), 7.68(dd, J=4.8, 8.1 Hz, 1H, pyridin-5-H), 7.29(s, 1H, pyrazole), 6.98(s, 1H, NH), 2.81(m, 1H, CH), 2.10(s, 3H, thiophene-5-CH3), 2.06(s, 3H, thiophene-4-CH3), 0.70(m, 2H, CH2), 0.55(m, 2H, CH2) |
| 1e | 13.14(s, 1H, NH), 8.49(dd, J=1.5, 5.1 Hz, 1H, pyridin-6-H), 7.89(dd, J=1.2, 5.1 Hz, 1H, pyridin-4-H), 7.42(dd, J=4.8, 8.7 Hz, 1H, pyridin-5-H), 6.98(s, 1H, pyrazole-H), 6.11(s, 1H, NH), 3.03(d, J=3.3 Hz, 3H, CH3), 2.30(s, 3H, thiophene-5-CH3), 2.25(s, 3H, thiophene-4-CH3) |
| 1f | 10.45(s, 1H, NH), 8.50(dd, J=1.8, 4.5 Hz, 1H, pyridin-6-H), 7.89(dd, J=1.8, 4.8 Hz,1H, pyridin-4-H), 7.42(dd, J=4.5, 8.4 Hz, 1H, pyridin-5-H), 6.99(s, 1H, pyrazole-H), 3.03(s, 6H, 2CH3), 2.23(s, 3H, thiophene-5-CH3), 2.03(s, 3H, thiophene-4-CH3) |
| 1g | 13.11(s, 1H, NH), 8.49(dd, J=1.2, 5.1 Hz, 1H, pyridin-6-H), 7.89(dd, J=1.5, 5.1 Hz, 1H, pyridin-4-H), 7.42(dd, J=5.1, 8.7 Hz, 1H, pyridin-5-H), 6.98(s, 1H, pyrazole-H), 5.89(br, 1H, NH), 4.28(m, 1H, CH), 2.27(s, 3H, thiophene-5-CH3), 2.23(s, 3H, thiophene-4-CH3), 1.28(d, J=6.9 Hz, 6H, 2CH3) |
| 1h | 10.43(s, 1H, NH), 8.49(dd, J=1.5, 4.8 Hz, 1H, pyridin-6-H), 7.91(dd, J=1.2, 4.8 Hz, 1H, pyridin-4-H), 7.43(dd, J=4.8, 8.4 Hz, 1H, pyridin-5-H), 6.96(s, 1H, pyrazole-H), 3.71(m, 4H, morpholine-2,6-2CH2), 3.54(m, 4H, morpholine-3,5-2CH2), 2.22(s, 3H, thiophene-5-CH3), 2.04(s, 3H, thiophene-4-CH3) |
| 2a | 8.33(s, 1H, pyrazole-H), 7.70(s, 1H, NH), 7.65(d, J=6.6 Hz, 2H, Ph-2,6-2H), 7.47(d, J=6.6 Hz, 2H, Ph-3,5-2H), 3.93(s, 3H, pyrazole-CH3), 2.93(s, 3H, CH3), 2.23(s, 3H, thiophene-5-CH3), 1.92(s, 3H, thiophene-4-CH3) |
| 2b | 8.23(s, 1H, pyrazole-H), 7.71(s, 1H, NH), 7.68(d, J=6.9 Hz, 2H, Ph-2,6-2H), 7.47(d, J=6.9 Hz, 2H, Ph-3,5-2H), 3.93(s, 3H, pyrazole-CH3), 2.83(s, 6H, 2CH3), 2.23(s, 3H, thiophene-5-CH3), 1.92(s, 3H, thiophene-4-CH3) |
| 2c | 12.01(s, 1H, NH), 8.31(s, 1H, pyrazole-H), 7.68(d, J=6.3 Hz, 2H, Ph-2,6-2H), 7.47(d, J=6.3 Hz, 2H, Ph-3,5-2H), 3.96(m, 1H, CH), 3.95(s, 3H, pyrazole-CH3), 2.23(s, 3H, thiophene-5-CH3), 2.15(s, 3H, thiophene-4-CH3), 1.13(s, 3H, CH3), 1.11(s, 3H, CH3) |
| 2d | 8.29(s, 1H, pyrazole-H), 7.71(s, 1H, NH), 7.66(d, J=6.6 Hz, 2H, Ph-2,6-2H), 7.43(d, J=6.6 Hz, 2H, Ph-3,5-2H), 3.93(s, 3H, pyrazole-CH3), 2.32(m, 1H, CH), 2.23(s, 3H, thiophene-5-CH3), 1.92(s, 3H, thiophene-4-CH3), 0.83(m, 4H, 2CH2) |
| 3a | 12.40(s, 1H, NH), 7.77—7.58(m, 4H, Ph), 6.05(s, 1H, NH), 2.95(d, J=3.0 Hz, 3H, CH3), 2.32(s, 6H, thiophene-2CH3) |
| 3b | 9.19(s, 1H, NH), 7.75—7.59(m, 4H, Ph), 3.02(s, 6H, 2CH3), 2.32(s, 3H, thiophene-5-CH3), 2.06(s, 3H, thiophene-4-CH3) |
| 3c | 12.31(s, 1H, NH), 7.76—7.58(m, 4H, Ph), 5.85(br, 1H, NH), 4.19(m, 1H, CH), 2.31(s, 6H, thiophene-2CH3), 1.24(d, J=5.1 Hz, 6H, 2CH3) |
| 3d | 12.39(s, 1H, NH), 7.77—7.58(m, 4H, Ph), 6.19(s, 1H, NH), 2.80(m, 1H, CH), 2.31(s, 3H, thiophene-5-CH3), 2.26(s, 3H, thiophene-4-CH3), 0.85(m, 4H, 2CH2) |
| 3e | 9.26(s, 1H, NH), 7.76—7.64(m, 4H, Ph), 3.78—3.46(m, 8H, morpholine), 2.31(s, 3H, thiophene-5-CH3), 2.07(s, 3H, thiophene-4-CH3) |
| Compd. | Mortality(%) | |
|---|---|---|
| 600 mg/L | 20 mg/L | |
| 1a | 100 | 70 |
| 1b | 100 | 40 |
| 1c | 100 | 50 |
| 1d | 100 | 30 |
| 1e | 100 | 60 |
| 1f | 100 | 50 |
| 1g | 100 | 50 |
| 1h | 100 | 20 |
| Chlomtraniliprole | 100 | 100 |
Table 3 Insecticidal activity against Plutella xylostella of the target compounds 1
| Compd. | Mortality(%) | |
|---|---|---|
| 600 mg/L | 20 mg/L | |
| 1a | 100 | 70 |
| 1b | 100 | 40 |
| 1c | 100 | 50 |
| 1d | 100 | 30 |
| 1e | 100 | 60 |
| 1f | 100 | 50 |
| 1g | 100 | 50 |
| 1h | 100 | 20 |
| Chlomtraniliprole | 100 | 100 |
| [1] | Liu C. L., Chin. J. Pestic., 2005, 44(11), 527 |
| (刘长令. 农药, 2005, 44(11), 527) | |
| [2] | Li Y., Li M., Chai B. S., Chin. J. Pestic., 2006, 45(10), 697—699 |
| (李洋, 李淼, 柴宝山. 农药, 2006, 45(10), 697—699) | |
| [3] | Dong W. L., Xu J. Y., Liu X. H., Zhao W. G., Li Z. M., Chin. J. Pest. Sci., 2008, 10(2), 178—185 |
| (董卫莉, 徐俊英, 刘幸海, 赵卫光, 李正名. 农药学学报, 2008, 10(2), 178—185) | |
| [4] | Liu J. B., Li Y. X., Chen Y. W., Wu C. C., Wan Y. Y., Wei W., Xiong L. X., Zhang X., Yu S. J., Li Z. M., Chem. Res. Chinese Universities,2016, 32(1), 41—48 |
| [5] | Zhang X. L., Liu A. L., Zhao Y., Xiong L. X., Li Z. M., Chem. Res. Chinese Universities,2013, 29(6), 1134—1139 |
| [6] | Zhang X. L., Wang B. L., Mao M. Z., Xiong L. X., Yu S. J., Li Z. M., Chem. J. Chinese Universities,2013, 34(1), 96—102 |
| (张秀兰, 王宝雷, 毛明珍, 熊丽霞, 于淑晶, 李正名. 高等学校化学学报,2013, 34(1), 96—102) | |
| [7] | Wu T. T., Pesticidal 5-Amino-4-ethylsulfinyl-1-arylpyrazoles, US 5814652A, 1998-09-29 |
| [8] | Guan A. Y., Liu C. L., Yang X. P., Dekeyser M., Chem. Rev., 2014, 114(14), 7079—7107 |
| [9] | Li M., Liu C. L., Zhang J., Wu Q., Hao S. L., Song Y. Q., Pest Manag. Sci., 2013, 69(5), 635—641 |
| [10] | Li M., Liu C. L., Li L., Yang H., Li Z. N., Zhang H., Li Z. M., Pest Manag. Sci., 2010, 66(1), 107—112 |
| [11] | LiuC. L., Guan A. Y., Yang J. D., Chai B. S., Li M., Li H. C., Yang J. C., Xie Y., J. Agric. Food Chem., 2016, 64(1), 45—51 |
| [12] | Chai B. S., Liu C. L., Li H. C., He X. M., Luo Y. M., Huang G., Zhang H., Chang J. B., Pest Manag. Sci., 2010, 66(11), 1208—1214 |
| [13] | Chai B. S., Liu C. L., Li H. C., Liu S. W., Huang G., Song Y. Q., Chang J. B., Pest Manag. Sci., 2011, 67(9), 1141—1146 |
| [14] | Li H. C., Guan A. Y., Huang G., Liu C. L., Li Z. N., Xie Y., Lan J., Bioorg. Med. Chem., 2016, 24(3), 453—461 |
| [15] | Yang J. C., Li M., Wu Q., Liu C. L., Chang X. H., Bioorg. Med. Chem., 2016, 24(3), 383—390 |
| [16] | Xie Y., Chi H. W., Guan A. Y., Liu C. L., Ma H. J., Cui D. L., Bioorg. Med. Chem., 2016, 24(3), 428—434 |
| [17] | Guan A. Y., Liu C. L., Sun X. F., Xie Y., Bioorg. Med. Chem., 2016, 24(3), 342—353 |
| [18] | Lahm G. P., Selby T. P., Freudenberger J. H., Stevenson T. M., Bioorg. Med. Chem. Lett., 2005, 15(21), 4898—4906 |
| [19] | Coppola G. M., J. Heterocyclic Chem., 1999, 36(3), 563—588 |
| [20] | Lahm G. P., Selby T. P., Stevenson T. M., Arthropodicidal Anthranilamides WO 03015519A1,2003-02-27 |
| [21] | LahmG. P., Novel Anthranilamides Useful for Controlling Invertebrate Pests, WO 2006023783A1, 2006-03-02 |
| [1] | LI Ran, ZHANG Xudong, MU Lidan, SUN Tong, AI Ganggang, SHA Yelong, ZHANG Yuqi, WANG Jijiang. Preparation and Application of Triplethiophene Derivative Functionalized SiO2 Inverse Opal Photonic Crystal Fluorescent Films [J]. Chem. J. Chinese Universities, 2021, 42(9): 2989. |
| [2] | ZHANG Renli, WANG Yao, YU Zhiquan, SUN Zhichao, WANG Anjie, LIU Yingya. Molybdenum Peroxide Anchored on Fluoronated UiO-66 as Catalyst in the Oxidation of Sulfur Containing Compounds [J]. Chem. J. Chinese Universities, 2021, 42(6): 1914. |
| [3] | LI Kangming,CHEN Jia,YI Yangjie,YAN Zhongzhong,YE Jiao,LONG Chuyun,LIU Aiping,HU Aixi,LI Jianming. Design, Synthesis and Insecticidal Activity of 1-(4-Chlorophenyl)-2-cyclopropylpropan-1-one Oxime Ether † [J]. Chem. J. Chinese Universities, 2020, 41(5): 1026. |
| [4] | LI Kangming, LI Yansai, YI Yangjie, XU Leitao, YE Jiao, OU Xiaoming, LI Jianming, HU Aixi. Design, Synthesis and Biological Activity of 5-Pyrazole Carboxamides † [J]. Chem. J. Chinese Universities, 2020, 41(4): 716. |
| [5] | WANG Ying, SUN Chuanyin, WANG Runwei, ZHANG Zhendong, ZHANG Daming, ZHANG Zongtao, QIU Shilun. Preparation of Amphiphilic Ti/ZSM-5 Zeolite and Its Catalytic Performance in Oxidative Desulfurization† [J]. Chem. J. Chinese Universities, 2019, 40(6): 1265. |
| [6] |
CHEN Hong,DU Yonghui,ZHANG Xin,LIU Wenyan,ZHOU Xiaoming.
Preparation and Electrochemical Properties of Poly(3-hexylthiophene)-coated Lithium-rich Layered Cathode Material Li1.18Ni0.15Co0.15Mn0.52 |
| [7] | QU Xingxing, DAI Yuyu, LI Weijun, OUYANG Mi, ZHANG Cheng. Synthesis and Electrochromic Properties of Thiophene-carbazol-benzene Derivativies† [J]. Chem. J. Chinese Universities, 2018, 39(8): 1699. |
| [8] | ZHAO Bangtun, TAO Jingjing, CHEN Xiaoji, FU Huimin, ZHU Weimin. Synthesis, Structure and Electrochemistry of Tetrathiafulvalene Vinylogues Bearing Thienyl and Pyridyl Groups† [J]. Chem. J. Chinese Universities, 2018, 39(7): 1449. |
| [9] | WANG Fengyan, YAN Ni, WEI Junji, XIA Huiyun, SONG Lifang, SONG Jiale, GAO Lining, YAN Luke. Synthesis, Characterization and Cell Imaging of Water Soluble Polythiophene Derivative† [J]. Chem. J. Chinese Universities, 2018, 39(11): 2594. |
| [10] | ZHANG Sihang, HE Yongfeng, FU Runfang, JIANG Jie, LI Qingbi, GU Yingchun, CHEN Sheng. Preparation and Electrochromic Properties of Nano Cellulose/Poly(3,4-ethylenedioxythiophene) Composite Films† [J]. Chem. J. Chinese Universities, 2017, 38(6): 1090. |
| [11] | LV Yaokang, LIU Youyou, PAN Yun, LIU Gang, CHEN Jun, GUO Yun, CHU Wenjing, SHEN Lian, ZHANG Cheng. Preparation, Characterisation and Electrochromic Properties of Copolymer Films Based on 3,4-Ethylenedioxythiophene and Pyrrole-3-Carboxylic Acid [J]. Chem. J. Chinese Universities, 2017, 38(3): 484. |
| [12] | FAN Jianxun, JI Lifei, REN Aimin. Theoretical Study on Charge Transport Properties of Copolymers of Diketopyrrolopyrrole and Oligo-thiophene† [J]. Chem. J. Chinese Universities, 2017, 38(11): 2053. |
| [13] | SONG Hua,YU Qi,XU Xiaowei,SONG Hualin,JIANG Nan,LI Feng,CHEN Yanguang. Effect of Yttrium and Citric Acid on the Hydrodesulfurization Performance of Unsupported Nickel Phosphide† [J]. Chem. J. Chinese Universities, 2016, 37(8): 1528. |
| [14] | LIANG Ansheng, LI Junjie, PAN Chengjun, WANG Lei. Preparation and Thermoelectric Properties of Composites Based on Polythiophene Derivatives with Donor-acceptor Structure† [J]. Chem. J. Chinese Universities, 2016, 37(6): 1161. |
| [15] | ZHANG Yan, ZHENG Jing, WANG Juan, GUO Mandong. Preparation and Properties of Doxorubicin Hydrochloride Sensor Based on Molecularly Imprinted Polymer† [J]. Chem. J. Chinese Universities, 2016, 37(5): 860. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||