

Chem. J. Chinese Universities ›› 2018, Vol. 39 ›› Issue (11): 2492.doi: 10.7503/cjcu20180459
• Physical Chemistry • Previous Articles Next Articles
LIU Pei1,2,3, CHENG Qingqing1,3, CHEN Chi1, ZOU Liangliang1, ZOU Zhiqing1, YANG Hui1,2,*( )
)
Received:2018-06-26
Online:2018-11-10
Published:2018-08-31
Contact:
YANG Hui
E-mail:yangh@sari.ac.cn
Supported by:CLC Number:
TrendMD:
LIU Pei, CHENG Qingqing, CHEN Chi, ZOU Liangliang, ZOU Zhiqing, YANG Hui. Preparation and Oxygen Reduction Reaction Catalytic Performance of Fe, N Co-doped Carbon Nanofibers with Encapsulated Iron Nitride†[J]. Chem. J. Chinese Universities, 2018, 39(11): 2492.

Fig.1 TEM images of the Fe-N-CNF after carbonization and acid-leaching(A) and FexN@Fe-N-C-20 after NH3-treatment for 20 min(B) and high-resolution TEM image of FexN nanoparticle(C)
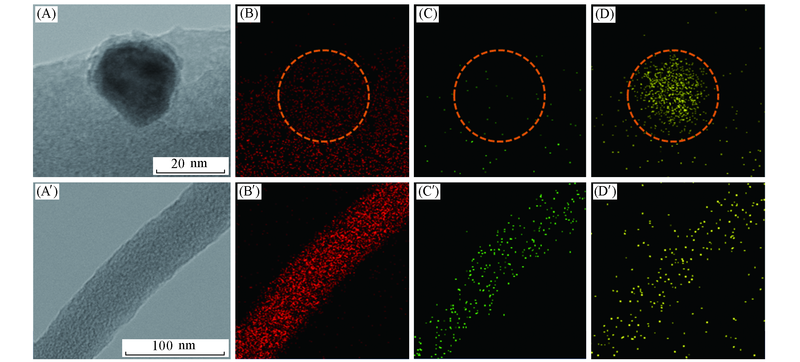
Fig.2 TEM images(A, A') and the elemental EDS-mapping analysis(B—D, B'—D') of C, N and Fe in the regions of FexN nanoparticle(A—D) and carbon nanofiber(A'—D') in FexN@Fe-N-C-20
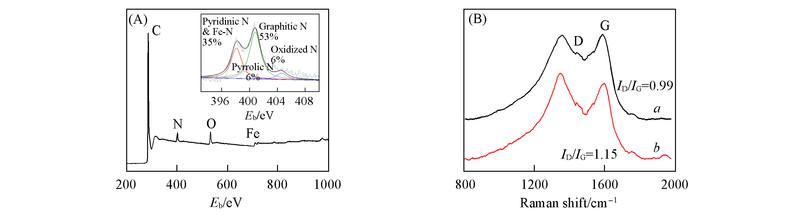
Fig.5 XPS survey spectrum of FexN@Fe-N-C-20(A) and Raman spectra of Fe-N-CNF(a) and FexN@Fe-N-C-20(b)(B)Inset shows the deconvoluted high-resolution N1s XPS spectrum.
| Sample | C | N | O | Fe |
|---|---|---|---|---|
| Fe-N-CNF | 85.63 | 3.38 | 10.40 | 0.59 |
| FexN@Fe-N-C-10 | 92.61 | 3.30 | 4.28 | 0.36 |
| FexN@Fe-N-C-20 | 93.09 | 3.25 | 3.32 | 0.34 |
| FexN@Fe-N-C-40 | 95.30 | 2.01 | 2.36 | 0.32 |
Table 1 Atomic fractions(%) of Fe-N-CNF and FexN@Fe-N-C-t(t=10, 20, 40) measured by XPS
| Sample | C | N | O | Fe |
|---|---|---|---|---|
| Fe-N-CNF | 85.63 | 3.38 | 10.40 | 0.59 |
| FexN@Fe-N-C-10 | 92.61 | 3.30 | 4.28 | 0.36 |
| FexN@Fe-N-C-20 | 93.09 | 3.25 | 3.32 | 0.34 |
| FexN@Fe-N-C-40 | 95.30 | 2.01 | 2.36 | 0.32 |
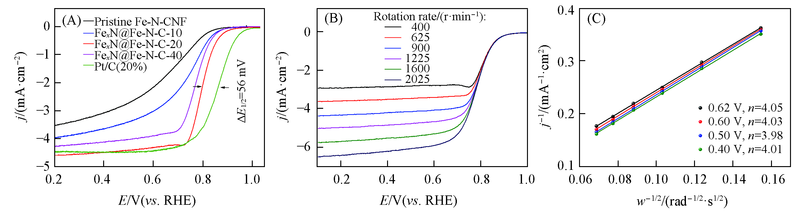
Fig.6 ORR polarization curves of Fe-N-CNF, FexN@Fe-N-C-t(t=10, 20, 40) and Pt/C in O2-saturated 0.1 mol/L HClO4(A), ORR polarization curves of FexN@Fe-N-C-20 at various rotation rates in 0.1 mol/L HClO4(B) and the corresponding K-L plots at various potentials(C)
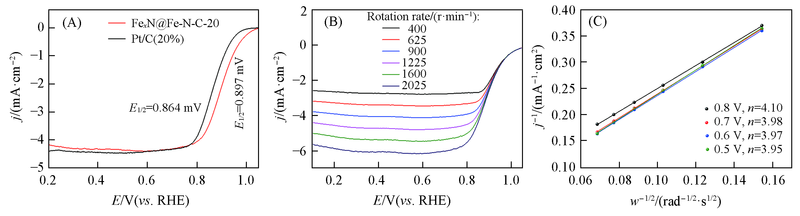
Fig.7 ORR polarization curves of FexN@Fe-N-C-20 and Pt/C in O2-saturated 0.1 mol/L KOH(A), ORR polarization curves of FexN@Fe-N-C-20 at various rotation rates in 0.1 mol/L KOH(B) and the corresponding K-L plots at various potentials(C)
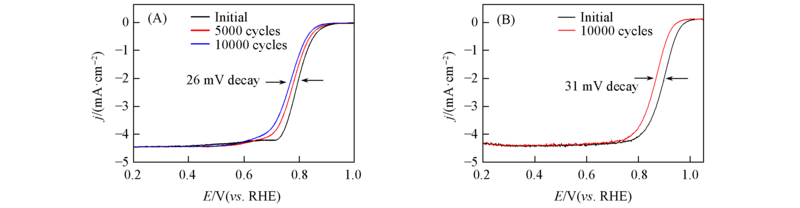
Fig.8 ORR polarization curves of FexN@Fe-N-C-20(A) and Pt/C(B) before and after 10000 cycles of accelerated durability test in O2-saturated 0.1 mol/L HClO4Potential range: 0.6—1.0 V; scan rate: 50 mV/s.
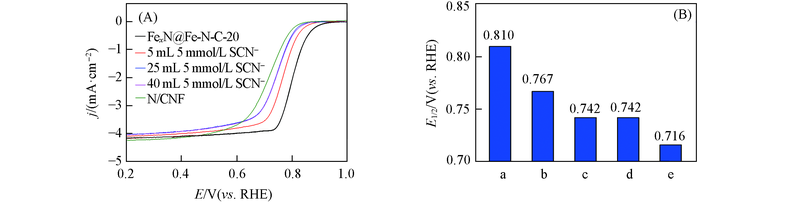
Fig.9 ORR polarization curves of N/CNF and FexN@Fe-N-C-20 with various amount of SCN- in 0.1 mol/L HClO4(A) and the comparison of the corresponding half-wave potentials(B)a. Initial; b. 5 mL SCN-; c. 25 mL SCN-; d. 40 mL SCN-; e. N/CNF.
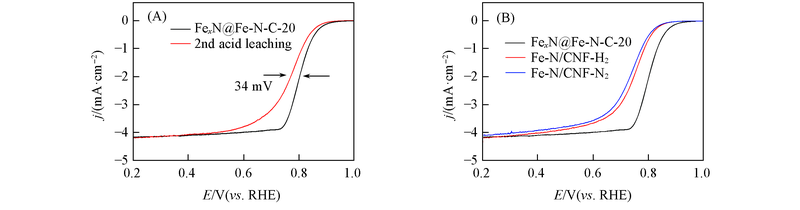
Fig.10 ORR polarization curves of FexN@Fe-N-C-20 before and after second acid-leaching in 0.1 mol/L HClO4(A) and ORR polarization curves of Fe-N/CNF-H2, Fe-N/CNF-N2 and FexN@Fe-N-C-20(B)
| [1] | Yu X. W., Ye S. Y., J. Power Sources, 2007, 172(1), 133—144 |
| [2] | Winter M., Brodd R. J., Chem. Rev., 2004, 104(10), 4245—4270 |
| [3] | Liang Y. Y., Wang H. L., Zhou J. G., Li Y. G., Wang J., Regier T., Dai H. J., J. Am. Chem. Soc., 2012, 134(7), 3517—3523 |
| [4] | Cheng X., Than X. T., Pinault M., Mayne M., Reynaud C., Vigneron J., Etcheberry A., Perez H., Electrochim. Acta, 2014, 135(22), 293—300 |
| [5] | Hirano S., Kim J., Srinivasan S., Electrochim. Acta, 1997, 42(10), 1587—1593 |
| [6] | Antoine O., Bultel Y., Ozil P., Durand R., Electrochim. Acta, 2000, 45(27), 4493—4500 |
| [7] | Tian Z. Q., Jiang S. P., Liang Y. M., Shen P. K., J. Phys. Chem. B, 2006, 110(11), 5343—5350 |
| [8] | Becerik İ., Süzer Ş., Kadırgan F., J. Electroanal. Chem., 1999, 476(2), 171—176 |
| [9] | Lopes T., Antolini E., Colmati F., Gonzalez E. R., J. Power Sources, 2007, 164(1), 111—114 |
| [10] | Wu G., Santandreu A., Kellogg W., Gupta S., Ogoke O., Zhang H., Wang H. L., Dai L. M., Nano Energy, 2016, 29, 83—110 |
| [11] | Watanabe M., Tsurumi K., Mizukami T., Nakamura T., Stonehart P., Cheminform, 1994, 141(10), 2659—2668 |
| [12] | Bashyam R., Zelenay P., Nature, 2006, 443, 63—66 |
| [13] | Parvez K., Yang S., Feng X. L., Müllen K., Synthetic Met., 2015, 210, 123—132 |
| [14] | Yin H., Zhang C., Liu F., Hou Y., Adv. Funct. Mater., 2014, 24(20), 2930—2937 |
| [15] | Lin L., Zhu Q., Xu A. W., J. Am. Chem. Soc., 2014, 136(31), 11027—11033 |
| [16] | Chung H. T., Won J. H., Zelenay P., Nat. Commun., 2013, 4, 1922—1926 |
| [17] | Zhang H. G., Osgood H., Xie X. H., Shao Y. Y., Wu G., Nano Energy, 2017, 31, 331—350 |
| [18] | Kinumoto T., Inaba M., Nakayama Y., Ogata K., Umebayashi R., Tasaka A., Iriyama Y., Abe T., Ogumi Z., J. Power Sources, 2006, 158(2), 1222—1228 |
| [19] | Xiao M. L., Zhu J. B., Feng L. G., Liu C. P., Xing W., Adv. Mater., 2015, 27, 2521—2527 |
| [20] | Cheng Q. Q., Mao K., Ma L. S., Yang L. J., Zou L. L., Zou Z. Q., Hu Z., Yang H., ACS Energy Lett., 2018, 3(5), 1205—1211 |
| [21] | Li Z. H., Shao M. F., Zhou L., Zhang R. K., Zhang C., Wei M., Evans D. G., Duan X., Adv. Mater., 2016, 28(12), 2337—2344 |
| [22] | Artyushkova K., Kiefer B., Halevi B., Knop-Gericke A., Schlogl R., Atanassov P., Chem. Commun., 2013, 49(25), 2539—2541 |
| [23] | Liu Q., Zhang H. Y., Zhong H. W., Zhang S. M., Chen S. L., Electrochim. Acta, 2012, 81, 313—320 |
| [24] | Chen C., Zhang X., Zhou Z. Y., Yang X. D., Zhang X. S., Sun S. G., Electrochim. Acta, 2016, 222, 1922—1930 |
| [25] | Deng D. H., Yu L., Chen X. Q., Wang G. X., Jin L., Pan X. L., Deng J., Sun G. Q., Bao X. H., Angew. Chem. Int. Ed., 2013, 52(1), 371—375 |
| [1] | KANG Huan, LI Shang, LIU Chang, GUO Wei, PAN Mu. Synthesis of Ordered Mesoporous Fe-N-C-PANI Catalyst via Self-assembly and Its Oxygen Reduction Reaction Activity in Acid Medium [J]. Chem. J. Chinese Universities, 2017, 38(8): 1423. |
| [2] | LI Weilun, YAO Ying, ZHANG Cunzhong. Applications of Carbon Fiber Ultra-microelectrode and Powder Microelectrode in Exploring Influences of Non-aqueous Solvents and Cathode Materials on ORR and OER† [J]. Chem. J. Chinese Universities, 2017, 38(4): 642. |
| [3] | WU Juan, WU Xue-Yan, WEI Xiao, WANG Kai-Xue, CHEN Jie-Sheng. Synthesis and Supercapacitance Performance of PANI/MCFs Materials [J]. Chem. J. Chinese Universities, 2012, 33(07): 1540. |
| [4] | JIANG Yue, QIN Yuan-Hang, NIU Dong-Fang, ZHANG Xin-Sheng, ZHOU Xing-Gui, SUN Shi-Gang, YUAN Wei-Kang. Effects of Surface Properties and Microstructures of Carbon Nanofibers on Their Electrocatalytic Activity for Oxygen Reduction Reaction [J]. Chem. J. Chinese Universities, 2012, 33(05): 1001. |
| [5] | LI Zhi-Zhou1,2, CUI Xiao-Li2* , ZHENG Jun-Sheng3, WANG Qing-Fei1. Photoelectrochemical Properties of TiO2 Coated on Carbon Nanofibers with Different Microstructure Film Electrodes [J]. Chem. J. Chinese Universities, 2008, 29(6): 1195. |
| [6] | JIN Bao-Duo, GUO Jian-Wei, XIE Xiao-Feng*, WANG Shu-Bo, WANG Jin-Hai. Effect of Operating Condition on Cathodic EIS Parameters in a DMFC [J]. Chem. J. Chinese Universities, 2008, 29(11): 2258. |
| [7] | XU Wei1, WANG Xin2*, JIA Hui3, ZHENG Wei-Tao2, LONG Bei-Hong2. Growth Behavior of Amorphous Fe—N Thin Films Applicability of Conventional Dynamic Scaling Approach [J]. Chem. J. Chinese Universities, 2007, 28(11): 2102. |
| [8] | TANG Tian-Di, CHEN Jiu-Ling, LI Yong-Dan. Effects of Surface Oxidation of Carbon Nanofibers in Mixed Concentrated HNO3-H2SO4 Acid Solution on Dispersion State of Supported Pd-Pt Catalyst Particles [J]. Chem. J. Chinese Universities, 2006, 27(1): 129. |
| [9] | ZHENG Ming-Yuan, CHENG Rui-Hua, CHEN Xiao-Wei, LI Ning, CONG Yu, WANG Xiao-Dong, ZHANG Tao. Preparation of Nano-iron Nitride by Temperature-programmed Reaction from Nano-iron Oxides [J]. Chem. J. Chinese Universities, 2005, 26(4): 623. |
| [10] | YU Li-Yan, ZHANG Qian, CUI Zuo-Lin . Preparation and Characterization of Helical Carbon Nanofibers [J]. Chem. J. Chinese Universities, 2005, 26(1): 5. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||