

Chem. J. Chinese Universities ›› 2018, Vol. 39 ›› Issue (2): 255.doi: 10.7503/cjcu20170586
• Physical Chemistry • Previous Articles Next Articles
Received:2017-08-31
Online:2018-02-10
Published:2017-12-20
Contact:
ZHAO Yilei
E-mail:yileizhao@sjtu.edu.cn
Supported by:CLC Number:
TrendMD:
LI Wenjuan, ZHAO Yilei. Salt Tolerance of T. Versicolor Laccase: Bioinformatics Study and Internal Transportation of Chloride, Dioxygen, and Water†[J]. Chem. J. Chinese Universities, 2018, 39(2): 255.
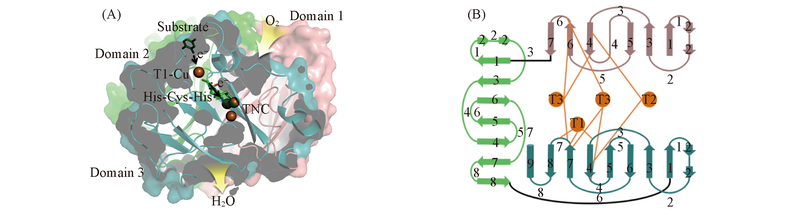
Scheme 1 Graphical representation of laccase structure with 3D surface model(A) and 2D topological diagram(B) Representative structure based on PDB# 1KYA in protein data bank, and the four copper atoms in orange and the three domains in salmon, green and teal, respectively.
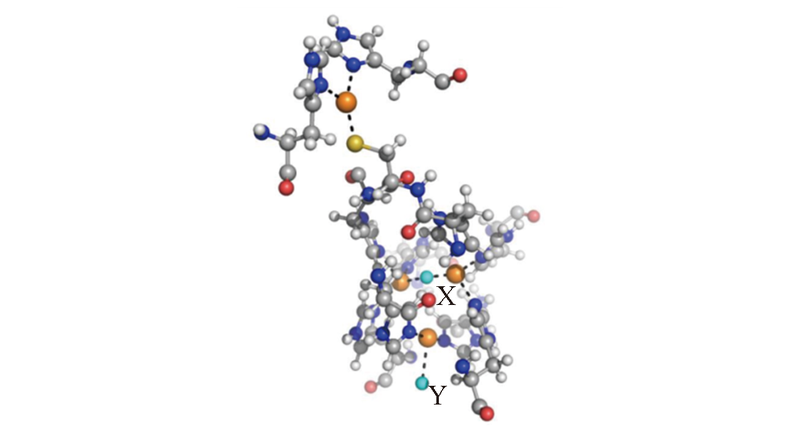
Fig.1 Active sites of single and tri-nuclear copper clustersX and Y were the positions of two oxygen atoms from dioxygen molecule, in which T3-Cu ligand X=OH-, O2 and Cl-, meanwhile T2-Cu ligand Y=OH-, H2O, and Cl-, respectively. The dashed lines denoted coordination to the copper cations.
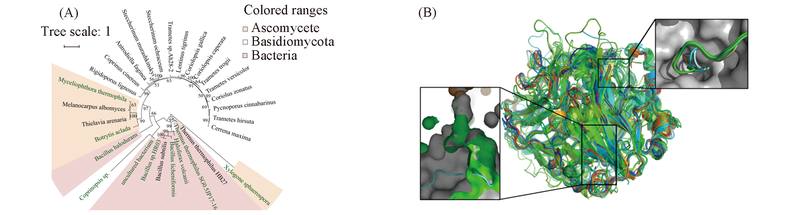
Fig.2 Phylogenetic tree of 29 laccase sequences based on sequence and structure alignment(A) and 26 superposed laccase structures with the POSA program(B) (A) Halotolerant laccases in green color; (B) the insets emphasize the comparison between T. versicolor(gray, from ascomycotas), basidio(green), and bacterial laccases(blue).
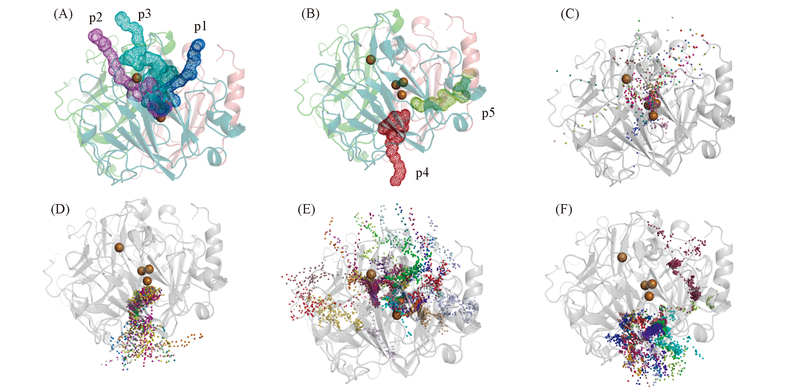
Fig.3 Transportation channels towards T3-Cu(A) and T2-Cu(B), and pathways exhibited with ligand snapshots in the RAMD trajectories of X-Cl-(C), Y-Cl-(D), X-O2(E) and Y-H2O(F) Each trajectory was shown in one color in Fig.3(C—F).
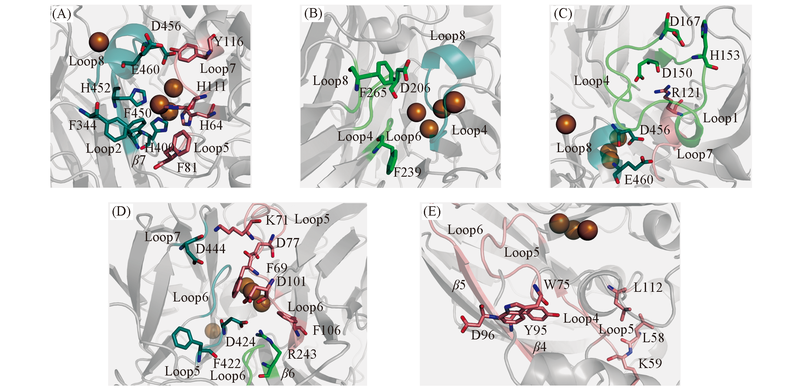
Fig.4 Constructions of tunnels p1(A), p2(B), p3(C), p4(D) and p5(E)The color-coding is same as Fig.1. The important residues surrounding the tunnel are shown as sticks.
| No. | Ligand | 1024 Accel./(kJ·mol-1· nm-1·g-1) | rmin/nm | Number of successful egress | Pathway | Frequency |
|---|---|---|---|---|---|---|
| Ⅰ | X=Cl- | 15.12 | 0.0120 | 18 | p1a | 12/18 |
| p2 | 3/18 | |||||
| p3 | 1/18 | |||||
| p4 | 1/18 | |||||
| Ⅱ | Y=Cl- | 7.56 | 0.0050 | 18 | p4 | 18/18 |
| Ⅱ | X=O2 | 5.29 | 0.0042 | 18 | p1a | 3/18 |
| p1b | 3/18 | |||||
| p1c | 1/18 | |||||
| p2 | 6/18 | |||||
| p3 | 1/18 | |||||
| Others | 4/18 | |||||
| Ⅳ | Y=H2O | 5.29 | 0.0042 | 13 | p4 | 11/13 |
| p5a | 1/13 | |||||
| p5b | 1/13 |
Table 1 Partition of internal transportation calculated with the RAMD simulations
| No. | Ligand | 1024 Accel./(kJ·mol-1· nm-1·g-1) | rmin/nm | Number of successful egress | Pathway | Frequency |
|---|---|---|---|---|---|---|
| Ⅰ | X=Cl- | 15.12 | 0.0120 | 18 | p1a | 12/18 |
| p2 | 3/18 | |||||
| p3 | 1/18 | |||||
| p4 | 1/18 | |||||
| Ⅱ | Y=Cl- | 7.56 | 0.0050 | 18 | p4 | 18/18 |
| Ⅱ | X=O2 | 5.29 | 0.0042 | 18 | p1a | 3/18 |
| p1b | 3/18 | |||||
| p1c | 1/18 | |||||
| p2 | 6/18 | |||||
| p3 | 1/18 | |||||
| Others | 4/18 | |||||
| Ⅳ | Y=H2O | 5.29 | 0.0042 | 13 | p4 | 11/13 |
| p5a | 1/13 | |||||
| p5b | 1/13 |
| [1] | Giardina P., Faraco V., Pezzella C., Piscitelli A., Vanhulle S., Sannia G., Cell Mol. Life Sci., 2010, 67(3), 369—385 |
| [2] | Riva S., Trends Biotechnol., 2006, 24(5), 219—226 |
| [3] | Jiao J., Yang X., Jin L. N., Gao J., Zhou Y., Xiao Y. Z., Zhang Y. J., Chem. J. Chinese Universities, 2016, 37(7), 1320—1327 |
| (焦晶, 杨雪, 金兰娜, 高键, 周洋, 肖亚中, 张应玖. 高等学校化学学报, 2016,37(7), 1320—1327) | |
| [4] | Dwivedi U. N., Singh P., Pandey V. P., Kumar A., J. Mol. Catal. B-Enzym., 2011, 68(2), 117—128 |
| [5] | Hakulinen N., Rouvinen J., Cell Mol. Life Sci., 2015, 72(5), 857—868 |
| [6] | Qi Y. B., Wang X. L., Shi T., Liu S. C., Xu Z. H., Li X. Q., Shi X. L., Xu P., Zhao Y. L., Phys. Chem. Chem. Phys., 2015, 17(44), 29597—29607 |
| [7] | Qi Y. B., Zhu J. R., Sun Y. J., Du Y., Chu J. J., Shi T., Zhao Y. L., Wang X. L., Chem. J. Chinese Universities, 2014, 35(4), 776—783 |
| (齐艳兵, 朱吉人, 孙尧金, 杜芸, 褚建君, 石婷, 赵一雷, 王晓雷. 高等学校化学学报, 2014,35(4), 776—783) | |
| [8] | Jones S. M., Solomon E. I., Cell Mol. Life Sci., 2015, 72(5), 869—883 |
| [9] | Piontek K., Antorini M., Choinowski T., J. Biol. Chem., 2002, 277(40), 37663—37669 |
| [10] | Bento I., Martins L. O., Gato Lopes G., Armenia Carrondo M., Lindley P. F., Dalton. Trans., 2005, 21(21), 3507—3513 |
| [11] | Lyashenko A. V., Bento I., Zaitsev V. N., Zhukhlistova N. E., Zhukova Y. N., Gabdoulkhakov A. G., Morgunova E. Y., Voelter W., Kachalova G. S., Stepanova E. V., Koroleva O. V., Lamzin V. S., Tishkov V. I., Betzel C., Lindley P. F., Mikhailov A. M., J. Biol. Inorg. Chem., 2006, 11(8), 963—973 |
| [12] | Kallio J. P., Rouvinen J., Kruus K., Hakulinen N., Biochemistry-US,2011, 50(21), 4396—4398 |
| [13] | Pietra F., Chem. Biodivers., 2016, 13(11), 1493—1501 |
| [14] | Damas J. M., Baptista A. M., Soares C. M., J. Chem. Theory Comput., 2014, 10(8), 3525—3531 |
| [15] | Farnet A. M., Gil G., Ferre E., Chemosphere,2008, 70(5), 895—900 |
| [16] | Pan T., Sun Y., Wang X. L., Shi T., Zhao Y. L., Chin. Chem. Lett., 2014, 25(7), 983—988 |
| [17] | Vaz-Dominguez C., Campuzano S., Rudiger O., Pita M., Gorbacheva M., Shleev S., Fernandez V. M., De Lacey A. L., Biosens. Bioelectron., 2008, 24(4), 531—537 |
| [18] | Kumar P., Prasad B., Mishra I. M., Chand S., J. Hazard. Mater., 2007, 149(1), 26—34 |
| [19] | Vishnu G., Palanisamy S., Joseph K., J. Clean. Prod., 2008, 16(10), 1081—1089 |
| [20] | Raseda N., Hong S., Kwon O., Ryu K., J. Microbiol. Biotechnol., 2014, 24(12),1673—1678 |
| [21] | Xu F., Biochemistry-US, 1996, 35(23), 7608—7614 |
| [22] | Robles A., Lucas R., Martí Nez-Cañamero M., Omar N. B., Pérez R., Gálvez A., Enzyme Microb. Technol., 2002, 31(4), 516—522 |
| [23] | Ruijssenaars H. J., Hartmans S., Appl. Microbiol. Biotechnol., 2004, 65(2), 177—182 |
| [24] | Jimenez-Juarez N., Roman-Miranda R., Baeza A., Sanchez-Amat A., Vazquez-Duhalt R., Valderrama B., J. Biotechnol., 2005, 117(1), 73—82 |
| [25] | Trovaslet M., Enaud E., Guiavarc’h Y., Corbisier A. M., Vanhulle S., Enzyme Microb. Technol., 2007, 41(3), 368—376 |
| [26] | Niladevi K. N., Jacob N., Prema P., Process Biochem., 2008, 43(6), 654—660 |
| [27] | Singh G., Sharma P., Capalash N., J. Gen. Appl. Microbiol., 2009, 55(4), 283—289 |
| [28] | Uthandi S., Saad B., Humbard M. A., Maupin-Furlow J. A., Appl. Environ. Microbiol., 2010, 76(3), 733—743 |
| [29] | Fang Z., Li T., Wang Q., Zhang X., Peng H., Fang W., Hong Y., Ge H., Xiao Y., Appl. Microbiol. Biotechnol., 2011, 89(4), 1103—1110 |
| [30] | Kittl R., Mueangtoom K., Gonaus C., Khazaneh S. T., Sygmund C., Haltrich D., Ludwig R., J. Biotechnol., 2012, 157(2), 304—314 |
| [31] | Qasemian L., Billette C., Guiral D., Alazard E., Moinard M., Farnet A. M., Fungal Biol., 2012, 116(10), 1090—1098 |
| [32] | Lu L., Wang T. N., Xu T. F., Wang J. Y., Wang C. L., Zhao M., Bioresour. Technol., 2013, 134, 81—86 |
| [33] | Brander S., Mikkelsen J. D., Kepp K. P., PLoS One, 2014, 9(6), e99402 |
| [34] | Dabirmanesh B., Khajeh K., Ghazi F., Ranjbar B., Etezad S. M., Int. J. Biol. Macromol., 2015, 79, 822—829 |
| [35] | Liu H., Cheng Y., Du B., Tong C., Liang S., Han S., Zheng S., Lin Y., PLoS One, 2015, 10(3), e0119833 |
| [36] | Imran M., Zhang B., Tang K., Liu J., Chem. Res. Chinese Universities, 2017, 33(1), 87—93 |
| [37] | Tamura K., Stecher G., Peterson D., Filipski A., Kumar S., Mol. Biol. Evol., 2013, 30(12), 2725—2729 |
| [38] | Li Z., Natarajan P., Ye Y., Hrabe T., Godzik A., Nucleic. Acids. Res., 2014, 42(W1), W240—W245 |
| [39] | Dolinsky T. J., Nielsen J. E., Mccammon J. A., Baker N. A., Nucleic. Acids. Res., 2004, 32(W1), W665—W667 |
| [40] | Jorgensen W. L., Chandrasekhar J., Madura J. D., Impey R. W., Klein M. L., J. Chem. Phys., 1983, 79(2), 926—935 |
| [41] | Case D.A., Darden T. A., Cheatham Ⅲ T. E., Simmerling C. L., Wang J., Duke R. E., Luo R., Walker R. C., Zhang W., Merz K. M., Roberts B., Hayik S., Roitberg A., Seabra G., Swails J., Götz A. W., Kolossváry I., Wong K. F., Paesani F., Vanicek J., Wolf R. M., Liu J., Wu X., Brozell S. R., Steinbrecher T., Gohlke H., Cai Q., Ye X., Wang J., Hsieh M. J., Cui G., Roe D. R., Mathews D. H., Seetin M. G., Salomon-Ferrer R., Sagui C., Babin V., Luchko T., Gusarov S., Kovalenko A., Kollman P. A., AMBER, Version 12, University of California,San Francisco, 2012 |
| [42] | Maestre-Reyna M., Liu W. C., Jeng W. Y., Lee C. C., Hsu C. A., Wen T. N., Wang A. H., Shyur L. F., PLoS One, 2015, 10(4), e0120601 |
| [43] | Wang J., Wolf R. M., Caldwell J. W., Kollman P. A., Case D. A., J. Comput. Chem., 2004, 25(9), 1157—1174 |
| [44] | Phillips J. C., Braun R., Wang W., Gumbart J., Tajkhorshid E., Villa E., Chipot C., Skeel R. D., Kale L., Schulten K., J. Comput. Chem., 2005, 26(16), 1781—1802 |
| [45] | Zhou C., Shi T., Zhao Y. L., Wang X. L., Chem. J. Chinese Universities, 2017, 38(10), 1813—1821 |
| (周超, 石婷, 赵一雷, 王晓雷. 高等学校化学学报, 2017,38(10), 1813—1821) | |
| [46] | Li W., Shen J., Liu G., Tang Y., Hoshino T., Proteins,2011, 79(1), 271—281 |
| [47] | Pavlova M., Klvana M., Prokop Z., Chaloupkova R., Banas P., Otyepka M., Wade R. C., Tsuda M., Nagata Y., Damborsky J., Nat. Chem. Biol., 2009, 5(10), 727—733 |
| [48] | Wang T., Duan Y., J. Am. Chem. Soc., 2007, 129(22), 6970—6971 |
| [49] | Kallio J., Auer S., Jänis J., Andberg M., Kruus K., Rouvinen J., Koivula A., Hakulinen N., J. Mol. Biol., 2009, 392(4), 895—909 |
| [50] | Solomon E.I., Augustine A. J., Yoon J.,Dalton. Trans., 2008, (30), 3921—3932 |
| [1] | ZHOU Ning, TANG Xiaohua, CAO Hong, ZHA Fei, LI Chun, XIE Chunyan, XU Mingping, SUN Yige. Preparation, Characterization and Degradation to BPA of Pomegranate-like Gel Microsphere Entrapmented Laccase [J]. Chem. J. Chinese Universities, 2022, 43(2): 20210705. |
| [2] | DUAN Bingyi,WANG Yu,GUO Ningning,WANG Runwei,ZHANG Zongtao,QIU Shilun. Preparation of Yolk-shell Fe3O4@SiO2@PMO Magnetic Microspheres for Laccase Immobilization† [J]. Chem. J. Chinese Universities, 2019, 40(2): 210. |
| [3] | JIAO Jing, YANG Xue, JIN Lanna, GAO Jian, ZHOU Yang, XIAO Yazhong, ZHANG Yingjiu. Conservative and Variability of the Important Functional Sites in a Laccase from Bacillus Subtilis† [J]. Chem. J. Chinese Universities, 2016, 37(7): 1320. |
| [4] | LI Chunyi, HUANG Zhuolie, WANG Chunxiao. Effects of Chemical Modification with Chitooligosaccharides on Catalytic Activity and Enzymological Properties of Laccase from Trametes versicolor† [J]. Chem. J. Chinese Universities, 2015, 36(4): 704. |
| [5] | QI Yanbing, ZHU Jiren, SUN Yaojin, DU Yun, CHU Jianjun, SHI Ting, ZHAO Yilei, WANG Xiaolei. Theoretical Studies of the Binding-affinity and Reactivity Between Laccase and Phenolic Substrates† [J]. Chem. J. Chinese Universities, 2014, 35(4): 776. |
| [6] | WANG Miao-Miao, LI Qun-Yan, WEI Qi, NIE Zuo-Ren. Studies on Laccase Immobilization of Mesoporous SiO2/Fe3O4 Hollow Microspheres [J]. Chem. J. Chinese Universities, 2013, 34(2): 299. |
| [7] | ZHOU Hai-Feng, YANG Dong-Jie, WU Xiao-Lei, QIU Xue-Qing. Structure and Adsorption Characterization of Sodium Lignosulfonate by Laccase Modification [J]. Chem. J. Chinese Universities, 2013, 34(1): 218. |
| [8] | LAN Jia-Ning, NA Wei, WEI Qi*, Li Qun-Yan, WANG Wei, NIE Zuo-Ren. Immobilization of Laccase on Epoxy Group-functionalized Periodic Mesoporous Organosilicas [J]. Chem. J. Chinese Universities, 2010, 31(8): 1579. |
| [9] | ZHANG Bao-Hua, WANG Hai-Shui, XU Lu. Codes of DNA Primary Sequences and the Similarity Calculation [J]. Chem. J. Chinese Universities, 2006, 27(12): 2277. |
| [10] | TU Chu-Qiao, LIANG Hong, WANG Guang-Hui . Catalyzed Oxidation by Rhus Vernicifera Laccase(ⅹⅢ) Effects of Fe2+Ion on the Catalytic Oxidation by Laccase [J]. Chem. J. Chinese Universities, 1999, 20(2): 243. |
| [11] | WU Xing-Guo, CAI Ru-Xiu, HUANG Hou-Ping, WANG Peng. The Kinetic Simulation Study of Laccase Catalyzed Formation and Decay of Semiquinone Radicals [J]. Chem. J. Chinese Universities, 1998, 19(8): 1223. |
| [12] | WANG Tian-Zhi, WU Ding-Quan, WAN Hong-Went, QU Song-Sheng, DU Yu-Min. Studies on the Reaction Between Laccase and 3,4,5-Trihydroxybenzoic Acid by Microcalorimetry [J]. Chem. J. Chinese Universities, 1997, 18(5): 786. |
| [13] | XIONG Ya, TAN Zhi-Qun, LIU Yi, PAN Zheng-Jun, WU Ding-Quan, KANG Li-Shan, QU Song-Sheng. Studies on the Metabolism of Mitochondria and Sub-mitochondria and the Effect of Laccase on It by Microcalorimetry [J]. Chem. J. Chinese Universities, 1997, 18(5): 753. |
| [14] | XIONG Ya, WU Ding-Quan, ZHOU Guang-Ming, DU Yu-Min, QU Song-Sheng. The Microcalorimetric Studies on the Inhibition Kinetics of Enzyme-catalyzed Reaction──Fluoride Ion Inhibiting Laccase-catalyzed Oxidation Reaction of o-Phenylenediamine [J]. Chem. J. Chinese Universities, 1996, 17(3): 460. |
| [15] | DU Yu-Min, ZHANG Jin-Mei. Studies on the Oxidation Performance and Reaction Product of Rhus Laccase in Reverse Micelles [J]. Chem. J. Chinese Universities, 1995, 16(5): 715. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||
